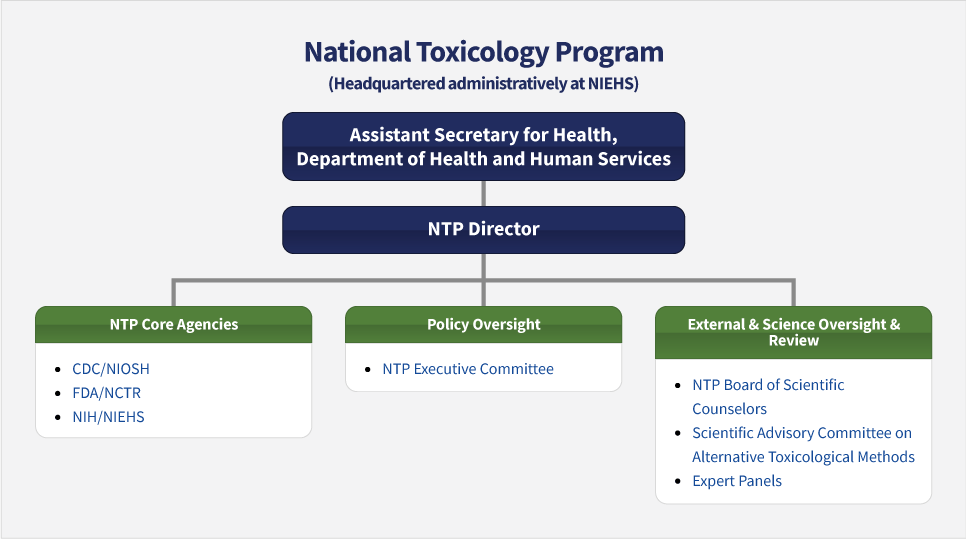
Organization
NTP is an interagency program composed of, and supported by, three government agencies within the Department of Health and Human Services:
- The National Center for Toxicological Research (NCTR) of the Food and Drug Administration (FDA)
- The National Institute of Environmental Health Sciences (NIEHS) of the National Institutes of Health (NIH)
- The National Institute for Occupational Safety and Health (NIOSH) of the Centers for Disease Control and Prevention (CDC)
The NIEHS Director also serves as the director of NTP. The Director reports to the Assistant Secretary for Health, and receives regular input from various external groups. Kyle M. Walsh, Ph.D., is the NIEHS and NTP Director.
What is the relationship between NTP and the Division of Translational Toxicology?
NTP is the National Toxicology Program; its activities are supported by the Division of Translational Toxicology (DTT), an intramural research division located at NIEHS in Research Triangle Park, North Carolina. You can learn more about DTT here.