Note on Accessibility: Persons using mobile devices may find some tables are not fully accessible. Note that you can view tables on a larger screen or in the PDF version of the report/monograph. If you need additional assistance, email us or use our contact form and identify the tables for which access is required. We will assist you in accessing the content. NIH has helpful information on accessibility.
Technical Report 602
NTP Technical Report on the Toxicology and Carcinogenesis Studies of an Isomeric Mixture of Tris(chloropropyl) Phosphate Administered in Feed to Sprague Dawley (Hsd:Sprague Dawley SD) Rats and B6C3F1/N Mice

Abstract
Tris(chloropropyl) phosphate (TCPP) is used as a flame retardant in textiles, furniture foam, and other related products. In addition, it is manufactured for use in construction materials, electronic products, paints, coatings, and adhesives. Several flame retardants, including structurally similar organohalogen compounds, have been removed from products in commerce due to toxicity concerns, and TCPP has been proposed as a replacement flame retardant for use in these products. An anticipated increase in use of TCPP has generated concerns for increased human exposure through oral, dermal, and inhalation routes; however, publicly available toxicity data are scarce. The U.S. Consumer Product Safety Commission therefore requested that the National Toxicology Program (NTP) form a research program on TCPP to conduct subchronic and chronic exposure studies in rats and mice for hazard identification and characterization information. Because TCPP is commercially available as an isomeric mixture, the NTP studies tested a commercial TCPP product containing four isomers commonly found in other commercial mixtures of TCPP: tris(1-chloro-2-propyl) phosphate (TCIPP; CASRN 13674-84-5), bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (CASRN 76025-08-6), bis(2-chloropropyl) 2-chloroisopropyl phosphate (CASRN 76649-15-5), and tris(2-chloropropyl) phosphate (CASRN 6145-73-9). Following procurement of TCPP, the percent purity of the four isomers was determined prior to conducting hazard characterization studies.
In the subchronic toxicity studies of TCPP in male and female Sprague Dawley (Hsd:Sprague Dawley SD) rats and B6C3F1/N mice, animals were exposed via dosed feed for 3 months. In rats, perinatal TCPP exposure of time-mated females from gestation day (GD) 6 through postnatal day (PND) 21 (weaning) preceded the subchronic exposure. Exposure concentrations for these studies were selected based on palatability studies conducted as part of the NTP research program on TCPP and on industry reports. Pregnant rats (20 dams) were exposed to 0, 2,500, 5,000 (8 dams only), 10,000, or 20,000 (8 dams only) ppm TCPP throughout gestation and lactation. Groups of 10 rats/sex/exposure concentration continued on study after weaning and were fed diets containing the same respective TCPP concentrations as their respective dam for 3 months. Mice (10/sex/exposure concentration) were exposed to 0, 1,250, 2,500, 5,000, 10,000, or 20,000 ppm TCPP for 3 months. Toxicity was evaluated by assessing survival, clinical observations, body weight, and feed consumption in all rats (including during the perinatal exposure period) and mice for 3 months. At study termination, additional toxicity parameters—including organ weight, hematology and clinical chemistry (rats only), sperm motility (males), genetic toxicity, and histopathology—were evaluated in rats and mice. The results of the 3-month studies were used to design and select exposure concentrations for the 2-year studies in rats and mice.
For the chronic toxicity studies, time-mated female rats were provided dosed feed beginning on GD 6 through lactation. On PND 28, offspring (50/sex/group) continued on the study and were provided dosed feed containing the same TCPP concentration as their respective dam for 2 years. In mice, groups of 50 mice per sex, aged 5 to 6 weeks at study start, were provided dosed feed containing TCPP for 2 years. At study termination, toxicity (e.g., survival, body weights) and the incidence of neoplasms and chemical-related histopathological changes were evaluated in rats and mice.
Three-month Study in Rats
In the perinatal portion of the 3-month study, pregnant rats exposed to 40,000 ppm were humanely euthanized due to overt toxicity early in gestation. With the exception of sporadic decreases in maternal body weight and feed consumption during gestation and lactation (approximately 10%–20% lower than control rats), no other toxicologically relevant findings were reported for dams. TCPP exposure also had no effects on littering parameters at concentrations ≤20,000 ppm, and offspring survived through lactation. Offspring in the 20,000 ppm TCPP group did exhibit a time-dependent decrease in weight gain during lactation. Male offspring in the 20,000 ppm group failed to thrive after weaning and were removed from the subchronic portion of the study on day 5; females in this exposure group were kept on study.
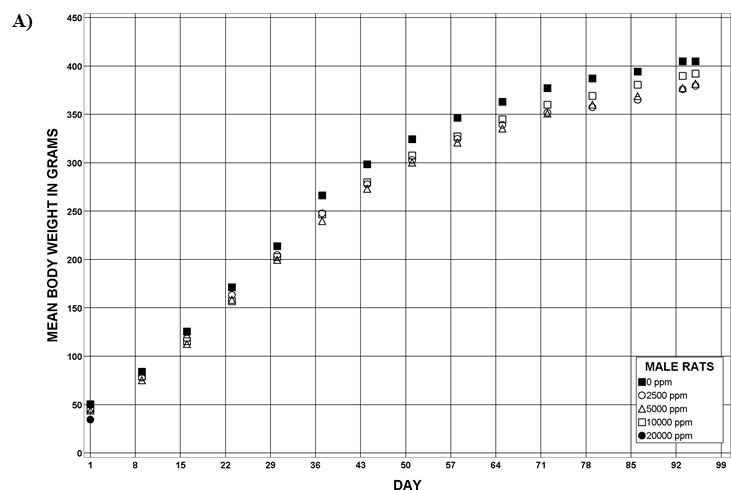
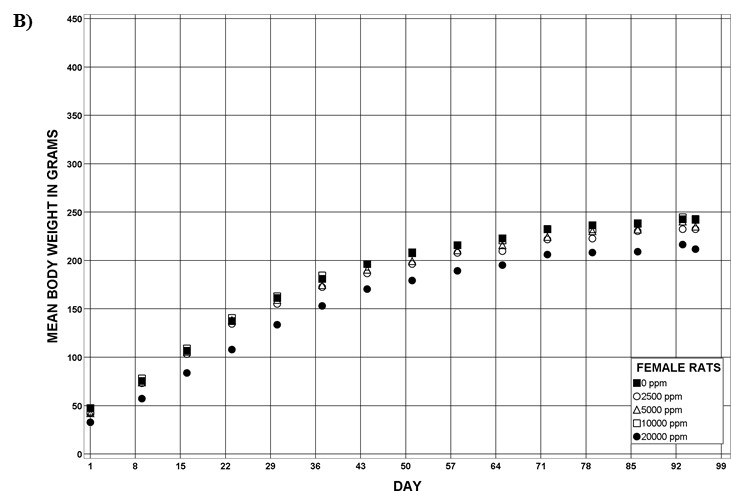
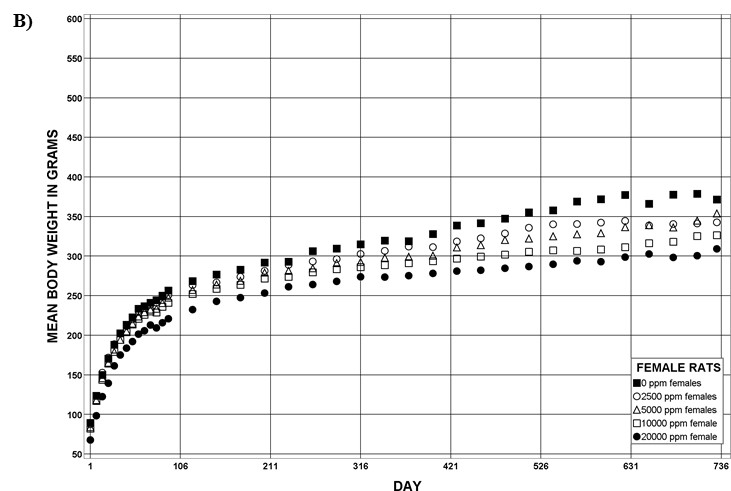
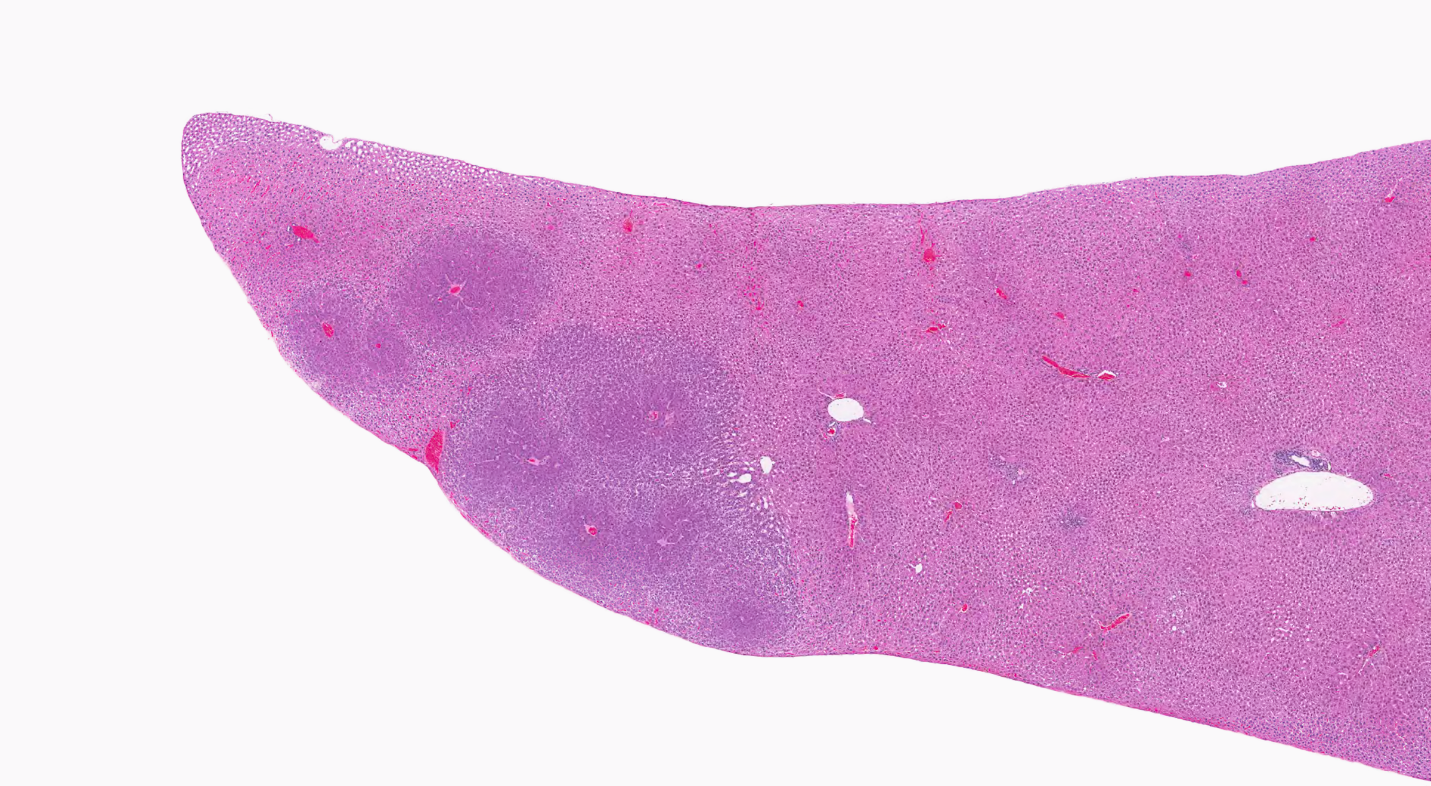
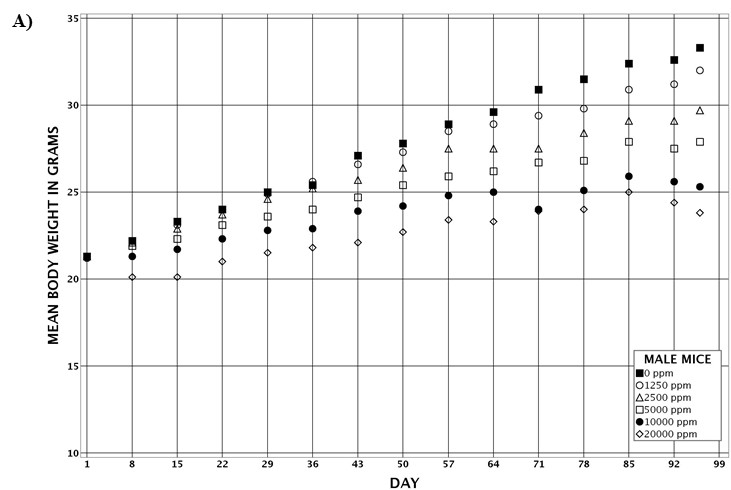
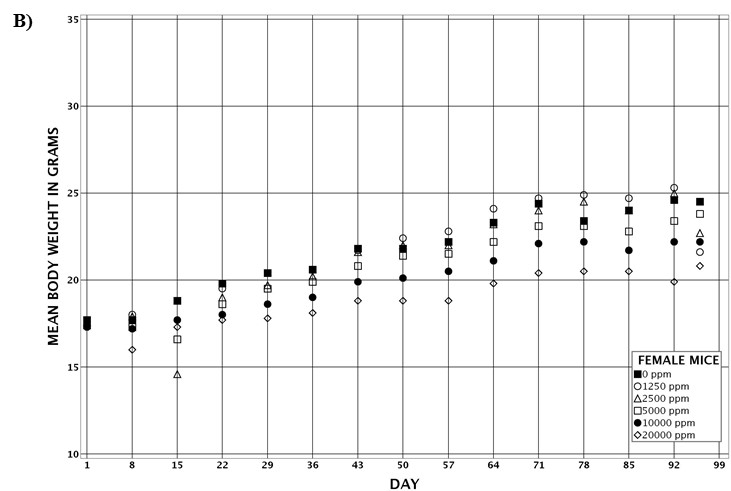
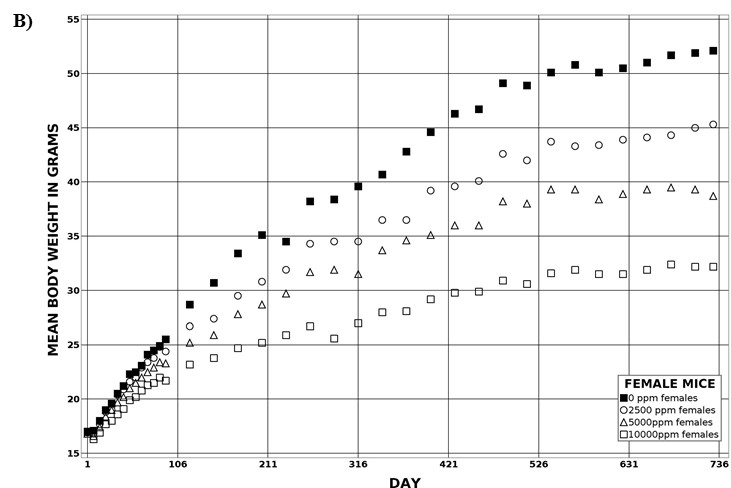
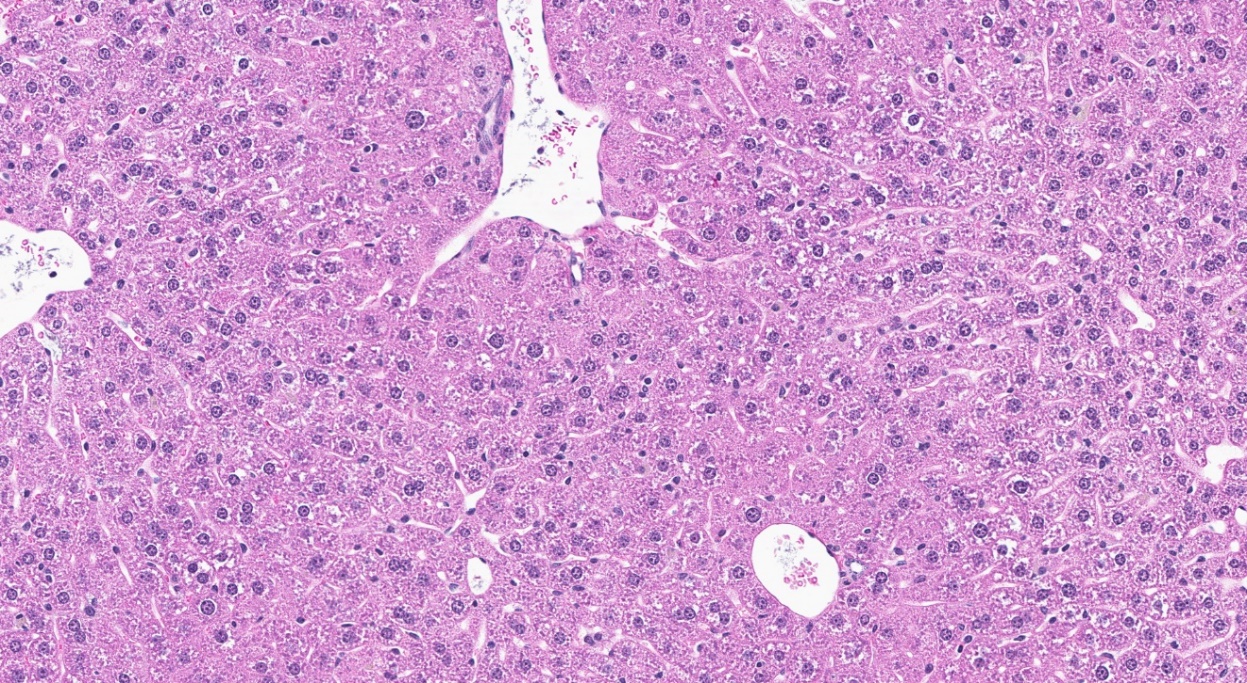
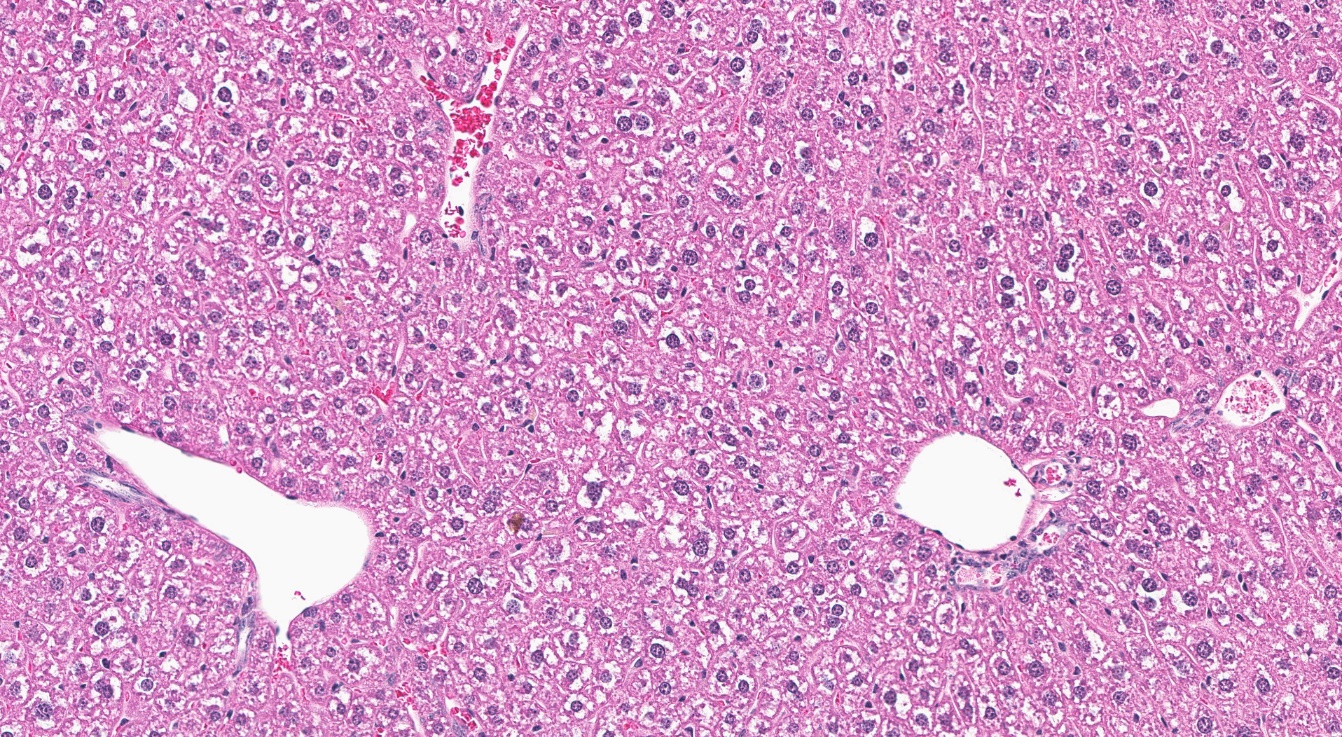
For the remainder of the 3-month study, male and female rats survived and displayed no clinical signs of toxicity when exposed to 10,000 ppm TCPP or lower concentrations. Females in the 20,000 ppm TCPP group had a mean body weight that was 12% lower than that of control females, which corresponded with a similar decrease in feed consumption (18%) by study termination. No biologically relevant alterations in hematological parameters were observed in either sex. Serum cholesterol concentrations were significantly increased in both sexes. TCPP did elicit exposure concentration-related effects (i.e., significantly increased organ weights and microscopic changes) in the liver and thymus of rats. In the liver, bile duct hyperplasia was observed in the highest exposure groups for both sexes. Increases in thymus weight were correlated with significantly larger thymic cortices in all males exposed to TCPP and in females in the 10,000 ppm group.
Two-year Study in Rats
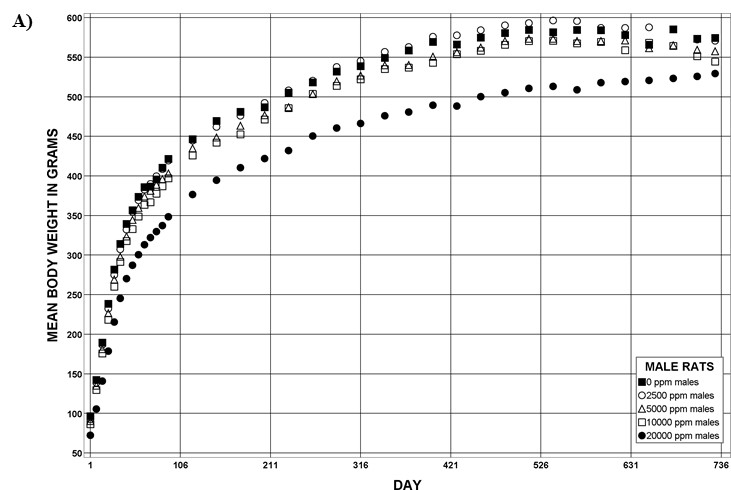
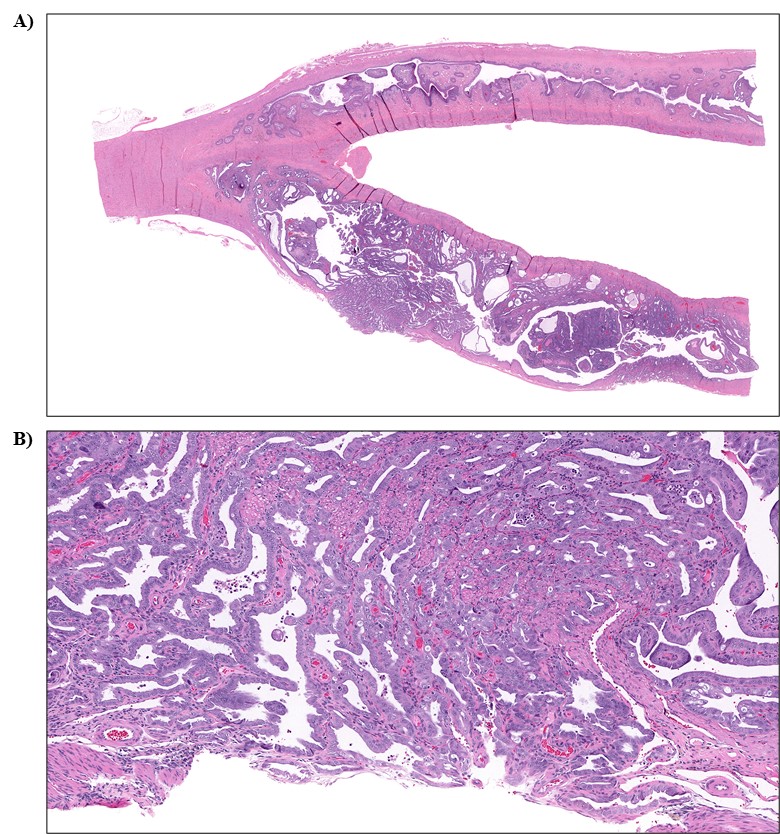
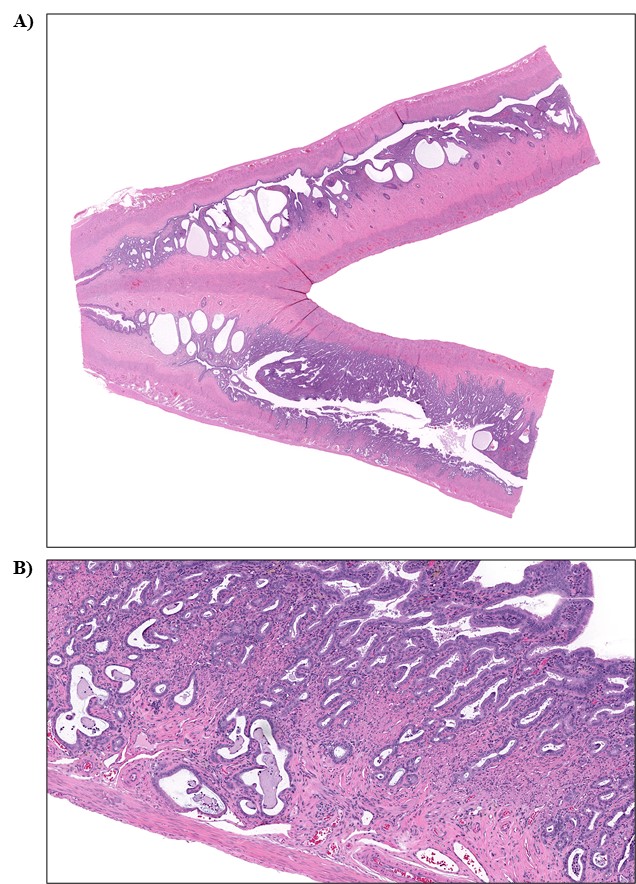
The effect of chronic TCPP exposure was evaluated in rats, beginning in utero and through adulthood, following feed administration at target concentrations of 0, 2,500, 5,000, 10,000, or 20,000 ppm TCPP. TCPP exposure to dams had no toxicologically relevant effects on maternal measurements during gestation or lactation with the exception of a slightly lower mean body weight and feed consumption in the 20,000 ppm group over this perinatal period. An exposure concentration-related decrease in mean body weight relative to control animals was observed in male and female offspring in the 20,000 ppm TCPP group during lactation. At the end of the 2-year study, mean body weights of males and females in the 20,000 ppm group were 8% and 17% lower, respectively, than those of the control groups. Histopathological evaluations identified a positive trend for incidences of hepatocellular adenoma or carcinoma (combined) in male rats. Accompanying significant nonneoplastic lesions included hyperplasia of the bile duct and an increase in basophilic, eosinophilic, mixed-cell foci, and pigment in the liver of males exposed to 20,000 ppm TCPP. A nonsignificant increase in the incidence of hepatocellular adenomas was observed in females exposed to 2,500, 10,000, and 20,000 ppm TCPP, and a spectrum of nonneoplastic lesions, similar to those in male rats, was observed. Histopathological evaluations also identified a positive trend for incidences of uterine adenoma or adenocarcinoma (combined) in female rats, although this was not significant at any exposure concentration.
Three-month Study in Mice
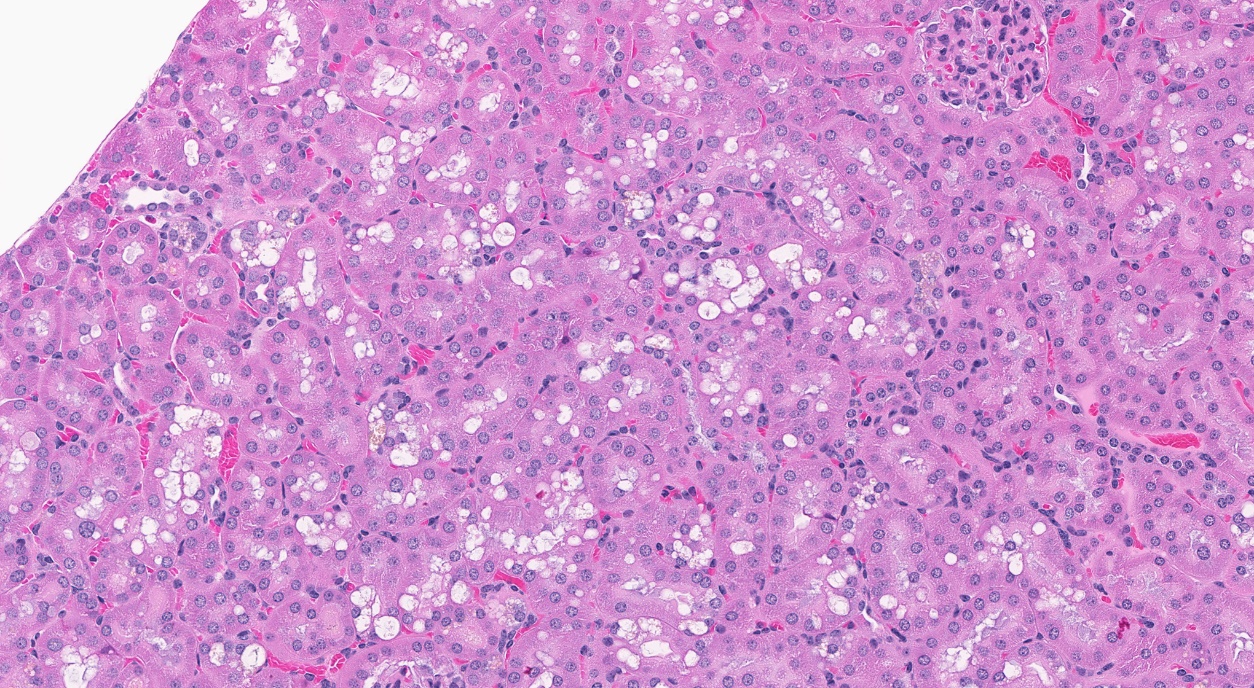
In the 3-month TCPP study with mice, mortality and clinical signs of toxicity were not observed across the exposure groups ranging from 1,250 to 20,000 ppm. TCPP-exposed male and female mice gained less weight than control mice; however, this response was only concentration related in the males. At the end of the subchronic study, TCPP exposure was associated with a spectrum of organ weight changes and microscopic changes in both sexes. Significantly increased liver weights were observed alongside a significant increase in the incidences of hepatocellular hypertrophy in males and females exposed to 5,000, 10,000, and 20,000 ppm TCPP. A significant increase in the incidences of cytoplasmic alteration was observed in the renal tubules of male mice exposed to 2,500, 5,000, 10,000, and 20,000 ppm TCPP. This observation was not evident in females and was not correlated with an observed decrease in kidney weights of both sexes.
Two-year Study in Mice
The TCPP exposure groups were different for male and female mice exposed chronically. Exposure concentrations of TCPP in feed were 0, 1,250 (males only), 2,500, 5,000, or 10,000 (females only) ppm. An exposure concentration-related decrease in mean body weights was recorded in males and females relative to their respective control groups; however, survival, clinical observations, and feed consumption measurements were not suggestive of overt toxicity. Lower mean body weight was interpreted as a failure to gain weight. Similar to rats, mice also had a significant increase in liver neoplasms. Male mice had a significant increase in the incidence of hepatocellular carcinoma across all TCPP-exposed groups, but the incidences were similar among these groups. Significant increases in the incidence of hepatocellular adenoma, hepatocellular carcinoma, and hepatocellular adenoma or carcinoma (combined) were also noted in the 10,000 ppm TCPP-exposed female mice relative to the control females. Exposure-related nonneoplastic lesions were not observed in male mice. However, a significant increase in cytoplasmic alteration of hepatocytes was observed in nearly all females of the 10,000 ppm group. Additionally, a significant increase in eosinophilic foci was recorded in all female TCPP-exposed groups.
Genetic Toxicology
In two independent studies, TCPP was not mutagenic in any of several strains of bacteria in tests conducted with and without rat or hamster liver S9 fraction. In the in vivo rodent peripheral blood micronucleus assay, no increases in micronucleated erythrocytes were observed in male or female Sprague Dawley rats administered TCPP via dosed feed. Results of the in vivo micronucleus assay in B6C3F1/N female mice were also judged to be negative. In male mice, a small but significant increase in micronucleated mature erythrocytes, accompanied by a small increase in the micronucleated immature erythrocyte population, resulted in an equivocal call. In both rats and mice, the percentage of immature erythrocytes increased in a dose-related manner, suggesting a stimulation of erythropoiesis.
Conclusions
Under the conditions of these 2-year feed studies, there was some evidence of carcinogenic activity of tris(chloropropyl) phosphate (TCPP) in male Hsd:Sprague Dawley SD rats based on the increased incidence of hepatocellular adenoma or carcinoma (combined). There was some evidence of carcinogenic activity of TCPP in female Hsd:Sprague Dawley SD rats based on the increased incidence of uterine adenoma or adenocarcinoma (combined). The marginal increase in the incidence of hepatocellular adenoma in female rats may have been related to exposure.
There was some evidence of carcinogenic activity of TCPP in male B6C3F1/N mice based on the increased incidence of hepatocellular carcinoma. There was clear evidence of carcinogenic activity of TCPP in female B6C3F1/N mice based on the increased incidences of hepatocellular adenoma, hepatocellular carcinoma, and hepatocellular adenoma or carcinoma (combined).
In the 2-year studies, exposure to TCPP resulted in increased incidences of nonneoplastic lesions in the liver of male and female rats and in female mice, and in the kidney of male mice.
Synonyms: tris(chloropropyl) phosphate and TCPP
tris(1-chloro-2-propyl) phosphate: 2-propanol, 1 chloro-, 2,2’,2”-phosphate; 2 propanol, 1-chloro-, phosphate (3:1); TCIPP; tris(2-chloroisopropyl) phosphate; tris(2-chloro-1-methylethyl) phosphate; tris(1-chloropropan-2-yl) phosphate
bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate: bis(2-chloro isopropyl) 2-chloropropyl phosphate; bis(1-chloropropan-2-yl) 2-chloropropyl phosphate; bis(1-chloro-2-propyl) 2-chloro-1-propyl phosphate; phosphoric acid, bis(2-chloro-1-methylethyl) 2-chloropropyl ester
bis(2-chloropropyl) 2-chloroisopropyl phosphate: (2-chloro-1-methylethyl) bis(2-chloropropyl) phosphate; 1-chloropropan-2-yl bis(2-chloropropyl) phosphate; bis(2-chloropropyl) 2-chloro-1-methylethyl phosphate; bis(2-chloro-1-propyl) 1-chloro-2-propyl phosphate; phosphoric acid 2-chloro-1-methylethyl bis(2-chloropropyl) ester
tris(2-chloropropyl) phosphate: 2-chloro-1-propanol phosphate (3:1); 1-propanol, 2-chloro-, phosphate (3:1); tris(beta-chloropropyl) phosphate; tris-(2-chloropropyl) phosphate; tris(2-chloro-1-propyl) phosphate
Trade names: Amgard TMCP, Antiblaze 80, Antiblaze TMCP, AP 33, Fyrol PCF, Hostaflam OP 820
Summary of the Perinatal and Two-year Carcinogenesis and Genetic Toxicology Studies of Tris(chloropropyl) Phosphate
| Male Sprague Dawley Rats | Female Sprague Dawley Rats | Male B6C3F1/N Mice | Female B6C3F1/N Mice | |
|---|---|---|---|---|
| Concentrations in feed | 0, 2,500, 5,000, 10,000, or 20,000 ppm | 0, 2,500, 5,000, 10,000, or 20,000 ppm | 0, 1,250, 2,500, or 5,000 ppm | 0, 2,500, 5,000, or 10,000 ppm |
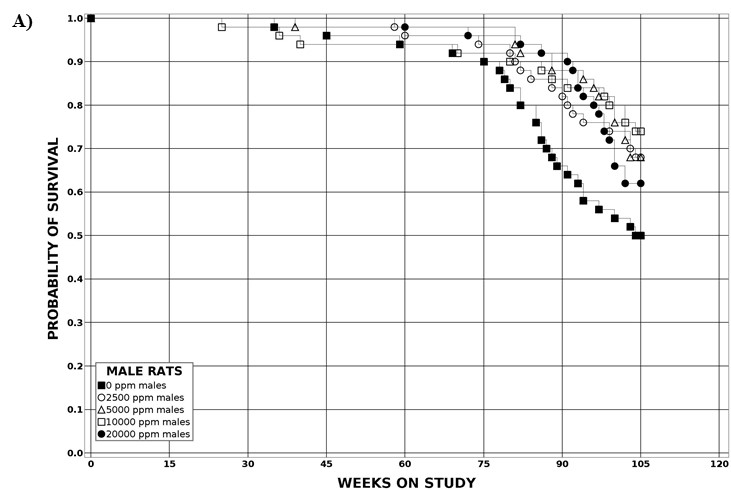
| Survival rates | 25/50, 34/50, 34/50, 37/50, 31/50 | 22/50, 31/50, 33/50, 34/50, 33/50 | 38/50, 44/50, 42/50, 43/50 | 46/50, 43/50, 45/50, 46/50 |
| Body weights | 20,000 ppm group: 7.9% lower than the control group | 20,000 ppm group: 16.8% lower than the control group | 5,000 ppm group: 17.9% lower than the control group | 10,000 ppm group: 38.2% lower than the control group |
| Nonneoplastic effects | Liver: basophilic focus (1/50, 1/50, 2/50, 9/50, 11/49); eosinophilic focus (3/50, 5/50, 3/50, 5/50, 13/49); mixed-cell focus (1/50, 2/50, 4/50, 1/50, 8/49); pigment (0/50, 0/50, 0/50, 1/50, 22/49); bile duct, hyperplasia (12/50, 23/50, 17/50, 19/50, 29/49) | Liver: basophilic focus (5/50, 7/50, 9/50, 8/50, 10/50); eosinophilic focus (5/50, 2/50, 13/50, 18/50, 25/50); mixed-cell focus (2/50, 0/50, 1/50, 1/50, 7/50); pigment (0/50, 0/50, 0/50, 3/50, 23/50); bile duct, cyst (1/50, 6/50, 12/50, 19/50, 21/50); bile duct, hyperplasia (7/50, 21/50, 24/50, 29/50, 11/50) | Liver: basophilic focus (4/50, 4/50, 3/50, 2/50); eosinophilic focus (13/50, 16/50, 9/50, 15/50) Kidney: renal tubule, cytoplasmic alteration (0/49, 28/50, 40/50, 48/50) | Liver: eosinophilic focus (1/50, 7/50, 13/50, 16/50); hepatocellular, cytoplasmic alteration (0/50, 0/50, 2/50, 48/50) |
| Neoplastic effects | Liver: hepatocellular adenoma or carcinoma (combined) (1/50, 0/50, 1/50, 7/50, 6/49) | Uterus: adenoma or adenocarcinoma (combined) (3/50, 4/50, 6/50, 8/49, 9/50) | Liver: hepatocellular carcinoma (includes multiple) (5/50, 14/50, 17/50, 14/50) | Liver: hepatocellular adenoma (includes multiple) (11/50, 5/50, 13/50, 23/50); hepatocellular carcinoma (includes multiple) (1/50, 2/50, 5/50, 10/50); hepatocellular adenoma or carcinoma (combined) (12/50, 7/50, 16/50, 29/50) |
| Equivocal findings | None | Liver: hepatocellular adenoma (1/50, 3/50, 0/50, 3/50, 3/50) | None | None |
| Level of evidence of carcinogenic activity | Some evidence | Some evidence | Some evidence | Clear evidence |
| Genetic toxicology Bacterial mutagenicity: Negative in Salmonella typhimurium strains TA97, TA98, TA100, TA1535, and TA1537 with and without rat or hamster S9; negative in Escherichia coli WP2 uvrA (pKM101) with or without rat S9 Micronucleated Erythrocytes (In Vivo) Rat peripheral blood: Negative in male and female rats for via dosed feed for up to 3 months Mouse peripheral blood: Equivocal in male and negative in female mice exposed via dosed feed for up to 3 months | ||||
Introduction
Chemical and Physical Properties
Tris(chloropropyl) phosphate (TCPP) is a clear, colorless, liquid mixture. TCPP has a molar mass of 327.56 g/mol and a relative density of 1.3 g/cm3. Estimated/predicted ranges of physical properties for this mixture include: boiling point, 283°C–365°C; vapor pressure, 5.25e-5–3.74e-3 mm Hg; and water solubility, 1.58e-4–3.63e-3 mol/L. The experimental log P (octanol:water partition coefficient) of TCPP was determined to be 2.59 and the predicted range is 1.53–2.89.2-7
Production, Use, and Human Exposure
TCPP is produced as an isomeric mixture in a closed system by the reaction of phosphorus oxychloride and propylene oxide to generate a combination of four isomers.5 The most abundant isomer in commercial products is tris(1-chloro-2-propyl) phosphate (TCIPP; 50%–85%) (Table 1). Additional isomers include bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (15%–40%), bis(2-chloropropyl) 2-chloroisopropyl phosphate (<15%), and tris(2-chloropropyl) phosphate (<1%). Variations in manufacturing methods result in commercial formulations that contain different ratios of the four isomers. The TCPP isomeric mixture and commercial products are commonly referred to by the CASRN 13674-84-5, which is the CASRN of the major isomer, TCIPP.4
The United States manufactured approximately 43 million pounds of TCPP in 2014.2 TCPP is used as a flame retardant in textiles, furniture (flexible polyurethane foam), and other related products. In addition, it is manufactured for use in construction materials (rigid polyurethane foam), electronic products, paints, coatings, and adhesives.3 TCPP has been proposed as a substitute for brominated flame retardants and as a replacement for other chlorinated flame retardants such as tris(2-chloroethyl) phosphate.8,9
It is beyond the scope of this report to summarize all publications and reports on TCPP in various environmental media; however, reports from other agencies thoroughly review TCPP in food, ambient and indoor air, dust, soil and sediment, drinking water, and other media. Environmental fate and transport of TCPP were recently summarized by the U.S. Environmental Protection Agency (EPA) Design for the Environment Branch4 and Health Canada.11 TCPP is expected to migrate to air and dust after release from industrial sites and wastewater. It is not expected to be highly mobile in soil or water.12 Monitoring studies suggest that TCPP is associated with particles in air, which can increase its persistence in the environment.11 Limited data from aquatic biota also suggest that TCPP is not bioaccumulative.11
The EPA Office of Pollution Prevention and Toxics suggests that exposure to TCPP is likely to occur through inhalation of vapors or particulates and via dermal exposure during the manufacturing or use of consumer products containing TCPP.3 Because TCPP is considered ubiquitous in the environment, consumers could be exposed by inhalation of vapors or particulates, direct skin contact, and incidental ingestion. Exposures can occur in offices, homes, and other indoor environments from using consumer products such as upholstered furniture that contains TCPP. Children are considered more susceptible to ingestion of TCPP because of increased object-to-mouth behaviors.13 Human exposure to TCPP can be measured using biomarkers of TCPP (both the parent compound and its metabolites). Human biomonitoring studies of TCPP have used urine, serum or plasma, breast milk, hair, placenta, and nails, although urine is the most frequently used matrix. TCPP metabolites measured in urine samples include bis(1-chloro-2-propyl) phosphate (BCIPP or BCPP) and bis(1-chloro-2-propyl) 1-hydroxy-2-propyl phosphate (BCIPHIPP).14,15 TCPP, BCPP, and BCIPHIPP have been detected in urine samples in study populations from North America,16-22 Europe,23 Asia,24-26 and Australia.14,27 Among these studies, urine has been collected from populations of children, adolescents, mother-child pairs, and adults, and in a variety of settings (e.g., general population, hospital-based, occupational, school/daycare). Concentrations of individual biomarkers vary by population characteristics,18,28 including age, sex, and other sociodemographic characteristics including race/ethnicity, and socioeconomic status. The highest reported urinary concentration of BCPP (1,620 µg BCPP/g creatinine) was in a sample from male spray polyurethane foam workers.16
Regulatory Status
TCPP is listed on the EPA Toxic Substances Control Act (TSCA) Inventory. Currently, no regulations restrict production or use of TCPP in the United States, but in 2015, EPA announced plans to further assess the risk to consumers, the general population, and aquatic organisms following exposure to TCPP and similar chemicals.3 The U.S. Consumer Product Safety Commission (CPSC) is assessing the risks to consumers’ health and safety from the use of additive, nonpolymeric organohalogen flame retardants, as a class of chemicals, in the following products: (1) durable infant or toddler products, children's toys, childcare articles, or other children's products (other than children's car seats); (2) upholstered furniture sold for use in residences; (3) mattresses and mattress pads; and (4) plastic casings surrounding electronics. TCPP is one of the chemicals under investigation. Final recommendations from this assessment have not been released.
A similar evaluation of organohalogen flame retardants is under investigation by the European Chemicals Agency (ECHA) and other European agencies. TCPP is registered under REACH (Registration, Evaluation, Authorisation, and Restriction of Chemicals).29 A 2008 European Union Risk Assessment Report for TCPP indicated no unacceptable risks for workers, consumers, or the general population apart from effects on fertility and developmental toxicity related to dermal exposure to workers manufacturing TCPP.6 A 2018 screening report by ECHA identified a risk for children from exposure to tris(2-chloroethyl) phosphate (TCEP), TCPP, and tris(1,3-dichloro-2-propyl) phosphate (TDCPP) childcare articles and residential upholstered furniture. This report recommended that a restriction proposal be prepared.29
Absorption, Distribution, Metabolism, and Excretion
Experimental Animals
Absorption, distribution, metabolism, and excretion (ADME) data are summarized in the European Union Risk Assessment Report,6 the Health Canada Screening Assessment Report,11 and the EPA Design for the Environment Report.4 These reports, whose findings were based on limited animal studies, indicate that TCPP is readily absorbed and excreted. The literature contains no studies on the toxicokinetics of TCPP in animals.
Briefly, TCPP is readily absorbed and excreted by male Wistar rats following gavage administration of 50 µmol [14C]TCPP/kg body weight.30 Approximately 98% of the administered dose was recovered during the 168 hours after dosing. Of the administered dose, 67%, 22%, and 7.7% TCPP was recovered in urine, feces, and expired air, respectively, within 48 hours. TCPP was rapidly distributed to tissues, with tissue to blood ratios highest in the liver and kidney followed by lung, spleen, and adipose during the first 12 hours after administration. The elimination half-life in blood, based on total radioactivity, was estimated to be approximately 59 hours. Biliary excretion studies showed that approximately 45% of the administered dose was excreted in bile within 48 hours and that TCPP excreted in feces is likely from biliary excretion.
Humans
The literature contains no studies on the ADME of TCPP in humans. TCPP metabolism was investigated in vitro with human liver microsomes by Van den Eede et al.15 Incubation of microsomes with TCPP resulted in several Phase I metabolites including BCPP, a major metabolite; BCIPHIPP; bis(1-chloro-2-propyl) 1-carboxy-2-propyl phosphate; and 1-chloro-2-propyl,1-hydroxy-2-propyl phosphate. No Phase II metabolites were detected.
Toxicity
Toxicity data on TCPP, both published and unpublished, have been summarized in reports by the National Industrial Chemicals Notification and Assessment Scheme,31 EPA Design for the Environment Branch,4 World Health Organization,9 Screening Information Dataset (SIDS),10 European Union,6 Agency for Toxic Substances and Disease Registry,7 National Academy of Sciences,32 and Health Canada.11 It is beyond the scope of this report to discuss all available toxicity data for TCPP; as such, a brief summary of information from the aforementioned reports is provided here with a focus on published peer-reviewed literature.
Experimental Animals
Reported acute oral median lethal dose (LD50) values for TCPP were above 500 mg TCPP/kg body weight (mg/kg) in male rats and 632 mg/kg in female rats of multiple strains, with the majority of values less than 2,000 mg/kg.5,6,12 Common clinical observations in acute studies included ataxia, hunched posture, lethargy, labored respiration, increased salivation, body tremors, and piloerection. Macroscopic signs of toxicity included hemorrhagic lungs and dark liver and kidneys. The acute dermal LD50 values for Sprague Dawley rats and New Zealand albino rabbits are reported to be >2,000 mg/kg and the inhalation median lethal concentration (LC50) in Sprague Dawley rats is >4.6 mg/L.5,6,12 The EPA Design for the Environment Branch assigned a low hazard to TCPP for acute toxicity.4
In a 13-week toxicity study, Fyrol PCF (i.e., TCPP) administered in feed to Sprague Dawley rats (20/sex/concentration) at 800 to 20,000 ppm had no effect on survival, clinical observations, hematology, clinical chemistry, or urinalysis parameters compared to the control animals.33 Body weights were decreased (approximately 2% compared to control animals) at the highest exposure concentration in male and female rats. Significantly increased absolute and relative liver weights were noted in all exposed male rats and in the two highest exposure groups of female rats (7,500 and 20,000 ppm). Relative kidney weights were increased in male rats in the 7,500 and 20,000 ppm groups compared to the control group. Histopathological evaluation revealed minor changes in the liver, kidney, and thyroid gland, which were most prevalent in the two highest exposure concentration groups. These data informed the EPA Design for the Environment Branch’s decision to assign a moderate hazard to TCPP for repeat-dose toxicity.4
Humans
The literature contains no studies on the toxicity of TCPP in humans.
Data Mining
In Tox21 (Toxicology in the 21st Century program) in vitro screening assays, TCPP showed activity in 10 endpoints, 7 of which were related to xenobiotic homeostasis, including activation of the pregnane X receptor (PXR) signaling pathway and the constitutive androstane receptor (CAR) pathway, although to a lesser degree than for reference chemicals tested (Appendix E). In a Division of Translational Toxicology (DTT)-sponsored 5-day toxicogenomic study, oral administration of TCPP to male rats resulted in the upregulation of biomarker gene expression in the liver and kidney of male rats (Appendix F). In the rat liver, TCPP activates CAR, PXR, and PPARα pathways, and to a lesser extent may activate the Nrf2 pathway.
Reproductive and Developmental Toxicity
Experimental Animals
Summaries of the results of a two-generation reproduction study in Wistar rats exposed to TCPP are presented in various hazard and risk assessment reports.4,6,11 Rats (28/sex/concentration) received dosed feed formulated for approximate TCPP doses of 0, 100, 333, or 1,000 mg/kg/day over two generations. Animals were administered feed containing TCPP 10 weeks before mating, during mating, throughout gestation and lactation, and until study termination. No exposure concentration-related clinical observations or mortality were reported in either parental generation. TCPP exposure did not affect precoital time, mating index, fecundity index, fertility index, duration of gestation, or postimplantation loss. The mean number of pups delivered was lower in the F0 and F1 generations in the 333 and 1,000 mg/kg/day groups compared to the control group. The reported lowest-observed-adverse-effect level (LOAEL) for F0 females in these studies was 99 mg/kg/day due to a significant decrease in uterus weights and effects on the estrous cycle. Effects such as decreased body and absolute seminal vesicle weights were observed in F0 males exposed to 293 mg/kg/day. F1 males and females had LOAELs of 85 and 99 mg/kg/day, respectively. This assignment was based on a significant decrease in kidney weights in males and pituitary weights in females.
Two separate studies suggest that TCPP is not a developmental toxicant in rodents. In 1982, Kawasaki et al.34 reported results from a developmental toxicity study in which Wistar rats were fed a diet containing 0%, 0.01%, 0.1%, or 1% TCPP from gestation day (GD) 0 through GD 20 (n = 11 to 14). Daily TCPP intake for the exposed groups was estimated to be 6, 70, or 625 mg/kg/day TCPP, respectively. Following TCPP exposure, no significant effects were observed on maternal toxicity, littering endpoints, or fetal survival. The only identified fetal abnormality was an exposure concentration-related increase in the incidences of cervical ribs and absent 13th ribs. In 2020, NTP reported results from a prenatal developmental toxicity of TCPP.35 In these studies, time-mated female Sprague Dawley (Hsd:Sprague Dawley SD) rats received TCPP by gavage on GD 6 (expected implantation) to the day before expected parturition (GD 20). The test lot of TCPP was also used in the current NTP 3-month subchronic toxicity studies in rats and mice. Rats were administered 0, 162.5, 325, or 650 mg/kg/day of TCPP following evidence of maternal toxicity at 1,000 mg/kg/day in a dose range-finding study. Under the conditions of the study, there was no evidence of developmental toxicity observed. Unlike the Kawasaki study, NTP found no biologically relevant exposure-related malformations during external, visceral, or skeletal fetal exams of rats exposed to TCPP.
Toxicity studies evaluating the developmental and neurodevelopmental toxicity of TCPP are also available in the embryonic zebrafish model.32 Overall, the data from these studies suggest that TCPP is not teratogenic or overtly toxic compared with other flame retardants.36-39
Humans
Associations among flame-retardant exposure and effects on human reproductive or developmental toxicity have been suggested from epidemiological studies.40-44 However, a strong association with adverse effects on reproductive or developmental toxicity in humans has not yet been determined for TCPP or its metabolites.
Carcinogenicity
Experimental Animals
The literature contains no studies on the carcinogenicity of TCPP in experimental animal models. Health Canada summarized recent activities to assess the carcinogenic potential of TCPP through read-across approaches with the structurally similar flame retardant, TCEP.11,45 The evidence suggested that TCPP may be carcinogenic in rodents.
Humans
Only two observational epidemiology studies were identified that examined measured TCPP metabolites and papillary thyroid cancer, both with null findings. A population-based case-control study of 200 U.S. women found no association between either urinary concentration of BCIPP or BCIPHIPP and papillary thyroid cancer.46 A hospital-based case-control study of 140 U.S. women found no association between TCIPP measured in household dust and papillary thyroid cancer.47
Genetic Toxicity
Few peer-reviewed publications reporting on the genetic toxicity of TCPP were identified in the literature. Results from all reported bacterial mutation assays, in which TCPP was tested in several different strains with and without induced male rat or hamster liver S9, were negative.48-50 Negative results also were reported for TCPP in an in vitro rat hepatocyte DNA repair test51 and in an in vitro comet assay for DNA damage in hamster V79 cells that was conducted with and without induced rat liver S9.48 The literature contains no studies on the genetic toxicity of TCPP in in vivo models.
Study Rationale
CPSC nominated TCPP because its use as a flame retardant for flexible polyurethane foam in home furnishings and construction materials is expected to increase.52 Exposure of consumers to TCPP via oral, dermal, and inhalation routes was also expected to increase, and, at the time of nomination, publicly available toxicity data from long-term exposure were considered limited. CPSC therefore requested subchronic and chronic oral studies in rats and/or mice. Exposure through feed was selected to mimic intermittent human exposure through accidental ingestion from dust, food, and water sources. Exposure to the isomeric mixture, rather than an individual isomer of TCPP, was also chosen to best represent human exposure.
Materials and Methods
Procurement and Characterization of Tris(chloropropyl) Phosphate
An isomeric mixture of tris(chloropropyl) phosphate (TCPP) was obtained from Albemarle (Orangeburg, SC) in two lots (101 and 134). Lot 101 was used in the 3-month rat and mouse studies. Lot 134 and a portion of lot 101 were blended to form lot M072911NP, which was used in the 2-year rat and mouse studies. Identity, purity, and stability analyses were conducted by the analytical chemistry laboratory at MRIGlobal (Kansas City, MO) for the study laboratory at Battelle (Columbus, OH). Reports on analyses performed in support of the TCPP studies are on file at the National Institute of Environmental Health Sciences (NIEHS).
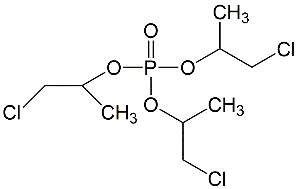
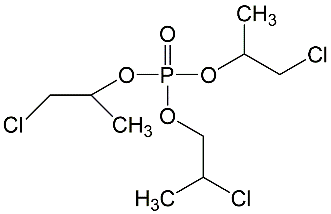
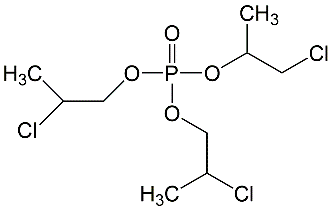
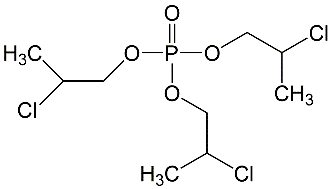
All lots appeared as clear, colorless, oily liquids. Chemical identities of homogenized lot 101 and mixed lot M072911NP used in these studies were confirmed using proton (1H) and carbon-13 (13C) nuclear magnetic resonance (NMR) spectroscopy, infrared (IR) spectroscopy, ultraviolet-visible (UV/Vis) spectroscopy, elemental analysis, and gas chromatography (GC) with mass spectrometry (MS). Four major isomeric components were identified as tris(1-chloro-2-propyl) phosphate (isomer 1, CASRN 13674-84-5), bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (isomer 2, CASRN 76025-08-6), bis(2-chloropropyl) 2-chloroisopropyl phosphate (isomer 3, CASRN 76649-15-5), and tris(2-chloropropyl) phosphate (isomer 4, CASRN 6145-73-9) (Table 2).
Multiple physical properties were also measured to characterize TCPP: acid number and ester value, octanol/water partition coefficients (log P) of isomers 1 and 2, density, and an extinction coefficient, Єmax, at 280 nm. Moisture content of lots 101 and M072911NP was determined by Karl Fischer titration, and the purity profiles were measured using gas chromatography (GC) with flame ionization detection (FID). Accelerated stability studies were conducted using GC/FID, and the stability of the bulk chemical was confirmed for at least 2 weeks when stored at temperatures up to 60°C. The purities of lots 101 and M072911NP were reevaluated before, during, and after the studies; all purity reanalyses determined the test articles as statistically similar to a frozen reference sample of the same lots. Purity results are described in Appendix A.
Preparation and Analysis of Dose Formulations
Dose formulations were prepared approximately monthly by mixing TCPP with NIH-07 or NTP-2000 feed (Appendix A). The rat studies used dose formulations of 0, 2,500, 5,000, 10,000, and 20,000 ppm, whereas the mouse studies used dose formulations of 0, 1,250, 2,500, 5,000, 10,000, and 20,000 ppm. The 3-month rat study included an additional formulation at 40,000 ppm.
Before the 3-month studies, the study laboratory conducted homogeneity studies of the 2,500 and 40,000 ppm dose formulations in 25 kg NIH-07 batch sizes and the 1,250 and 40,000 ppm dose formulations in 25 kg NTP-2000 batch sizes using GC/FID. Additional homogeneity studies of the 2,500 and 20,000 ppm dose formulations in 65 kg NIH-07 batch sizes and the 1,250 and 20,000 ppm dose formulations in 84 kg NTP-2000 batch sizes were performed before the 2-year studies by the study laboratory using GC/FID. All formulations were determined to be homogenous and of appropriate concentration.
Stability studies of TCPP in NIH-07 and NTP-2000 feed prepared at 3,000 ppm were performed by the analytical laboratory MRIGlobal (Kansas City, MO) using GC/FID. It was concluded that TCPP formulations could be stored up to 42 days frozen with ≤4.4% loss of TCPP. Dose formulations for the 3-month and 2-year studies were stored frozen (−15ºC to −30ºC) in sealed containers protected from light and were used within 42 days after preparation.
Periodic analyses of the dose formulations of TCPP were conducted by the study laboratory to determine purity (Table A-4, Table A-5, Table A-6, Table A-7). All preadministration dose formulations were within 10% of target concentrations. In the 3-month rat study, all samples were within 10% of the target concentrations except for four samples of collected residual feed from the feeders (−10.1% to −17.3%). In the 3-month mouse study, all samples were within 10% of the target concentrations except for three samples collected from the feeders and one sample from the storage bucket (−10.9% to −14.9%). In the 2-year rat study, all samples were within 10% of the target concentrations except for two samples from the storage barrel (−16.0% and −10.3%). All samples in the 2-year mouse study were within 10% of the target concentrations.
Animal Source
Time-mated (F0) female Sprague Dawley (Hsd:Sprague Dawley SD) rats were obtained from Envigo (formerly Harlan Laboratories, Inc., Indianapolis, IN). Male and female B6C3F1/N mice were obtained from the National Toxicology Program (NTP) colony maintained by Taconic Biosciences, Inc. (Germantown, NY).
Animal Welfare
Animal care and use are in accordance with the Public Health Service Policy on Humane Care and Use of Animals. All animal studies were conducted in an animal facility accredited by AAALAC International. Studies were approved by the Battelle (Columbus, OH) Animal Care and Use Committee and conducted in accordance with all relevant National Institutes of Health (NIH) and NTP animal care and use policies and applicable federal, state, and local regulations and guidelines.
Three-month Studies
Exposure Concentration Selection Rationale
Exposure concentrations for the 3-month studies were selected based on palatability studies in adult Wistar Han rats and B6C3F1 mice (data not shown). Male and female rats and mice were provided NTP-2000 rodent diet containing TCPP at target concentrations of 0, 30,000, 40,000, or 50,000 ppm for a maximum of 14 consecutive days (n = 5/sex). In rats, 40,000 ppm was considered the maximum tolerated dose; at this exposure concentration, one female was euthanized, and one male died. Animals in this exposure group also had body weight reductions accompanied by lower feed consumption. Clinical signs—such as dehydration, thinness, rough hair coat, discolored urine, and hyperactivity—were dependent on exposure concentration. Mice were more sensitive than rats to TCPP in the palatability studies. At the lowest exposure concentration of 30,000 ppm, male and female mice experienced clinical signs of toxicity including thinness, dehydration, and rough hair coat throughout the 14-day exposure period. Higher concentrations were not well tolerated, and mice exhibited lower mean body weight and feed consumption. These data informed the decision to provide TCPP to rats in feed at concentrations of 0, 2,500, 5,000, 10,000, 20,000, or 40,000 ppm and to mice at concentrations of 0, 1,250, 2,500, 5,000, 10,000, or 20,000 ppm.
Study Design for Rats
F0 female Sprague Dawley rats were 11 to 12 weeks old upon receipt. Evidence of mating is defined as gestation day (GD) 1; F0 females were received on GD 2 and held for 4 days. They were randomly assigned to one of six exposure groups on GD 5. Randomization was stratified by body weight that produced similar group mean body weights using PATH/TOX SYSTEM software (Xybion Medical Systems Corporation, Lawrenceville, NJ).
F0 females were quarantined for 39 days after receipt. Ten nonmated females received with the time-mated females were designated for disease monitoring 2 days after arrival; samples were collected for serological analyses, and the rats were euthanized, necropsied, and examined for the presence of disease or parasites. The health of the F1 animals was monitored during the study according to the protocols of the NTP Sentinel Animal Program (Appendix C). All test results were negative.
Beginning on GD 6, groups of 20 (0, 2,500, 10,000, and 40,000 ppm) or 8 (5,000 and 20,000 ppm) F0 time-mated females were fed diets containing 0, 2,500, 5,000, 10,000, 20,000, or 40,000 ppm TCPP throughout gestation and lactation. Groups of 10 F1 rats/sex/exposure concentration continued on study after weaning and were fed diets containing the same respective TCPP concentrations for 3 months.
F0 female rats were housed individually during gestation and with their respective litters during lactation. Water and dosed feed were available ad libitum. F0 females were weighed on GDs 5, 6, 9, 12, 15, 18, and 21 and on lactation days (LDs) 1, 4, 7, 14, and 21. During gestation, feed consumption was continuously measured over 3-day intervals from GD 6 through GD 21 (GDs 6–9, 9–12, 12–15, 15–18, and 18–21). The day of parturition was considered to be postnatal day (PND) 0. On apparent GD 27, all time-mated females that failed to deliver were euthanized and the uteri were examined and stained for evidence of implantation. Total litter weight and litter weight by sex were collected on PND 1. Individual F1 pups were weighed on PNDs 4, 7, 14, and 21. Clinical observations and survival were evaluated throughout lactation. During lactation, feed consumption was continuously measured over 3-day intervals from LD 1 through LD 21 (LDs 1–4, 4–7, 7–10, 10–14, 14–17, and 17–21).
Select dams and their litters were removed on GD 18 and LD 4 to quantify TCPP plasma and tissue concentrations in the 0, 2,500, and 10,000 ppm groups. On GD 18, blood was collected from the retroorbital site of randomly selected dams (n = 5/exposure group). Blood samples were collected in tubes containing ethylenediaminetetraacetic acid (EDTA) and centrifuged, and the plasma was harvested. Amniotic fluid was collected and pooled by litter. Dams’ fetuses were collected, pooled by litter, and flash frozen in liquid nitrogen. On LD 4, randomly selected dams (n = 5/exposure group) from the 0, 2,500 and 10,000 ppm groups were selected for biological sampling. Plasma was collected in the same manner as on GD 18. Up to two randomly selected pups were collected on PND 4 from each dam and flash frozen in liquid nitrogen. All samples were stored frozen at approximately −20°C before shipment to MRIGlobal (Kansas City, MO) for analysis.
F1 litters were standardized on PND 4 to eight pups per litter, with at least two pups of each sex and a preference for four males and four females each. Litters that did not meet the minimum of eight pups (or if they had fewer than two pups of either sex) were removed from the study. On the day the last litter reached PND 19, pups were randomly assigned to the 3-month study. For all exposure concentrations, two pups per sex from five randomly selected litters per exposure group were chosen for the 3-month study. After assignments to the 3-month study were complete, five pups per sex from the remaining vehicle control pups were randomly selected as the study termination sentinel animals. On the day the last litter reached PND 21, dams were removed, and the pups were weaned. Weaning marked the beginning of the 3-month study.
After weaning, F1 rats were housed five per cage. Water and dosed feed were available ad libitum. Feed consumption was measured weekly for 3 months. Cages were changed weekly though PND 4, then changed twice weekly. Racks were changed and rotated at least every 2 weeks. Further details of animal maintenance are given in Table 3.
Two diets were used in the rat studies: (1) NIH-07 during the perinatal phase and (2) NTP-2000 during the postnatal phase. The NIH-07 diet is a higher protein diet that supports reproduction and lactation in rodents, whereas the NTP-2000 diet is a lower protein diet that decreases the incidence of chronic nephropathy in adult rats. Information on feed composition and contaminants for both diets is provided in Appendix B.
Study Design for Mice
Male and female B6C3F1/N mice were 4 to 5 weeks old upon receipt and were quarantined for 15 (females) or 16 (males) days before study start. Mice were randomly assigned to one of six exposure groups (n = 10 mice/sex/group). Randomization was stratified by body weight that produced similar group mean weights using PATH/TOX SYSTEM software (Xybion Medical Systems Corporation, Lawrenceville, NJ). Mice were fed diets containing 0, 1,250, 2,500, 5,000, 10,000, or 20,000 ppm TCPP for 3 months.
Five male and five female mice were randomly selected for parasite evaluation and gross observation of disease. The health of the mice was monitored during the study according to the protocols of the NTP Sentinel Animal Program (Appendix C). All test results were negative.
Mice were housed individually (males) or up to five (females) per cage. Water and dosed feed were available ad libitum. Feed consumption was measured weekly for 3 months. Cages were changed at least once weekly (males) or twice weekly (females) and rotated every 2 weeks. Racks were changed and rotated every 2 weeks. Further details of animal maintenance are given in Table 3. Information on feed composition and contaminants is given in Appendix B.
Clinical Examinations and Pathology
In the 3-month studies in rats and mice, animals were observed twice daily for signs of morbidity and moribundity and were weighed before dosed feed exposure on study day 1, weekly for 3 months, and at study termination. Clinical observations were recorded on study day 1, weekly for 3 months, and at study termination. Feed consumption was determined weekly throughout the studies.
Blood was collected from the retroorbital plexus (rats) or sinus (mice) at the end of the 3-month studies for hematology, clinical chemistry (rats only), and micronuclei determination. Animals were anesthetized with a carbon dioxide/oxygen mixture and bled in a random order. Blood was collected in tubes containing EDTA (for hematology and micronuclei determination) or serum separator tubes (for clinical chemistry). Hematology parameters were analyzed using an Advia 120 system (Bayer Diagnostics Division, Tarrytown, NY). Clinical chemistry parameters were analyzed using the Roche cobas c501 Chemistry Analyzer (Roche Diagnostics, Indianapolis, IN). The parameters measured are listed in Table 3. Samples for erythrocyte micronuclei determination were stored at 2°C–8°C immediately after collection and shipped that day to Integrated Laboratory Systems, LLC (ILS, Durham, NC) for analysis.
At the end of the 3-month studies, samples were collected for sperm motility and vaginal cytology evaluations from F1 male and female rats in the 0, 2,500 (males only), 5,000, 10,000, and 20,000 (females only) ppm groups and male and female mice in the 0, 5,000, 10,000, and 20,000 ppm groups. The parameters evaluated are listed in Table 3. Due to low cellularity and poor quality of samples, estrous cyclicity could not be determined for female rats. Estrous cyclicity was evaluated in female mice; however, missing values in the data set precluded conclusive interpretations regarding effects of the administered TCPP. Male animals were evaluated for sperm count and motility. The left testis and left epididymis were isolated and weighed. The tail of the epididymis (cauda epididymis) was then removed from the epididymal body (corpus epididymis) and weighed. Test yolk (rats) or modified Tyrode’s buffer (mice) was applied to slides, and a small incision was made at the distal border of the cauda epididymis. The sperm that effluxed from the incision were dispersed in the buffer on the slides, and the numbers of motile and nonmotile spermatozoa were counted for five fields per slide by two observers. After completion of sperm motility estimates, each left cauda epididymis was placed in buffered saline solution. Caudae were finely minced, and the tissue was incubated in the saline solution and then heat fixed at 65°C. Sperm density was determined microscopically with the aid of a hemocytometer. To quantify spermatogenesis, the testicular spermatid head count was determined by removing the tunica albuginea and homogenizing the left testis in phosphate-buffered saline containing 10% dimethyl sulfoxide. Homogenization-resistant spermatid nuclei were counted with a hemocytometer.
Necropsies were performed on all rats and mice at the end of the 3-month studies. Organ weights were recorded for the liver, thymus, right kidney, right testis, heart, and lungs. At necropsy, all organs and tissues were examined for grossly visible lesions and all major tissues were fixed and preserved in 10% neutral buffered formalin except for eyes, which were first fixed in Davidson’s solution, and testes, vaginal tunics, and epididymides, which were first fixed in modified Davidson’s solution. Tissues were processed and trimmed, embedded in paraffin, sectioned at a thickness of approximately 5 μm, and stained with hematoxylin and eosin (H&E) for microscopic examination. Complete histopathological examinations were performed by the study laboratory pathologist on all organs with gross lesions and on all tissues collected from the 0 and 20,000 ppm rats and mice. Due to overt toxicity of TCPP in the male rats at 20,000 ppm, all protocol-required tissues were examined in the 10,000 ppm group, as well. In rats, the liver, bone marrow, thymus, spleen, and lymph nodes were identified as target organs and examined to a no-effect level. In mice, the liver and kidney were identified as target organs and examined to a no-effect level. Tissues examined microscopically are listed in Table 3.
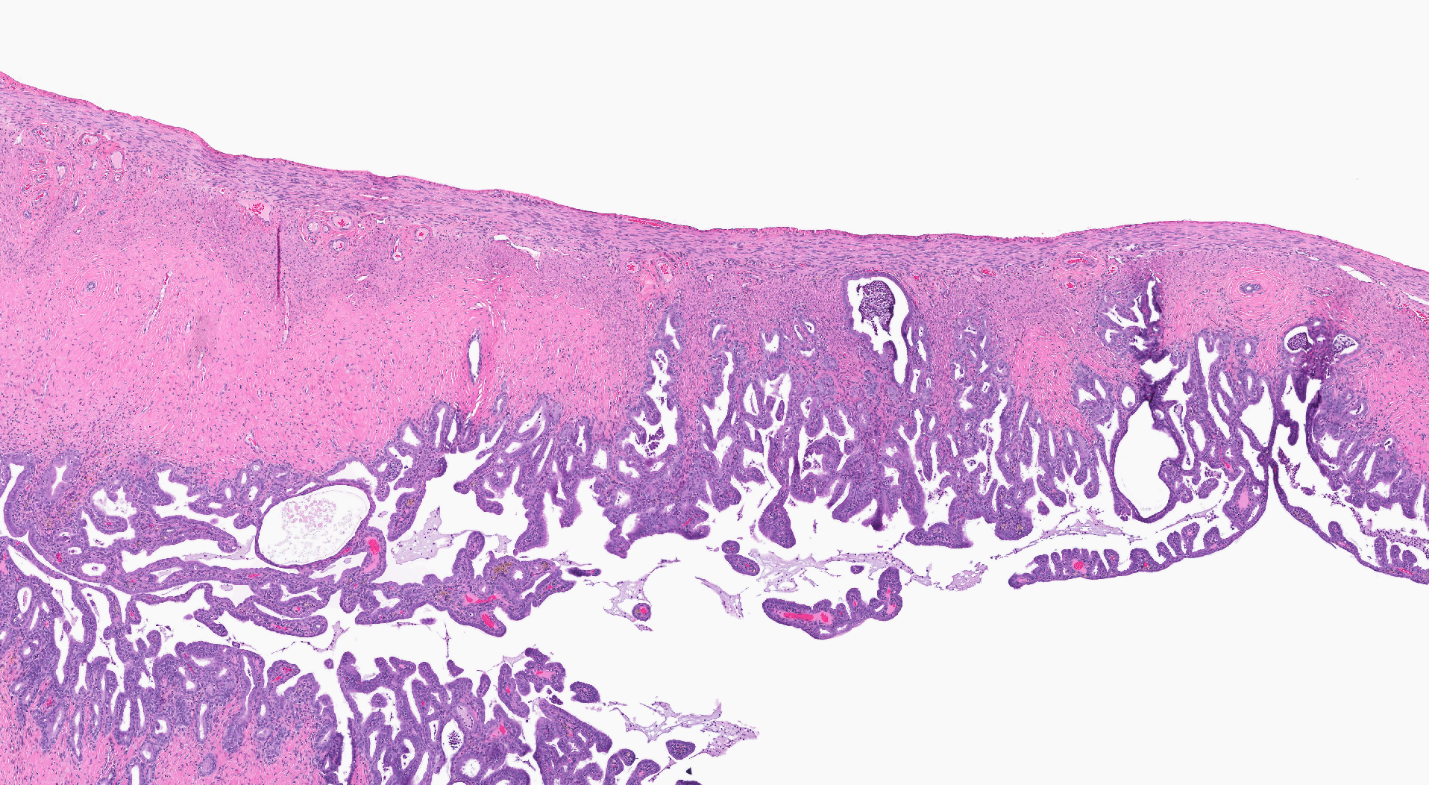
Morphometric analysis was performed on H&E-stained rat thymus sections from all exposure groups to better characterize the increase in thymus weights within male and female rats exposed to TCPP. Slides were scanned at 40× using the Aperio Scanscope XT Digital Slide Scanner (Leica Biosystems, Buffalo Grove, IL) and viewed using ImageScope software (v. 10.2.0.2352, Leica Biosystems). Image analysis was performed on the entire thymus section using the Definiens Tissue Studio software (v. 3.6.1, Carlsbad, CA). Specific regions of interest were selected across multiple thymic samples using the Composer algorithm, training the algorithm to identify the cortical (dark) and medullary (light) regions. The algorithm was then applied across all prepared thymic tissue sections to quantitate the percentage of dark and light staining areas. The total thymic area was quantified for each sample for relative comparison of the cortical and medullary areas.
After a review of the laboratory reports and selected histopathology slides by a quality assessment (QA) pathologist, the findings and reviewed slides were submitted to a Pathology Peer Review Group (PPR) coordinator for a second independent review. Any inconsistencies in the diagnoses made by the study laboratory and QA pathologists were resolved by the pathology peer-review process. Final diagnoses for reviewed lesions represent a consensus of the PPR or a consensus between the study laboratory pathologists, Division of Translational Toxicology (DTT) pathologist, QA pathologist, and the PPR coordinator. Details of these review procedures have been described, in part, by Maronpot and Boorman53 and Boorman et al.54
Two-year Studies
Study Design for Rats
F0 female Sprague Dawley rats were 11 to 15 weeks old upon receipt. Evidence of mating is defined as GD 1; F0 females were received on GD 2 and held for 4 days. F0 females were randomly assigned to one of five exposure groups on GD 5. Randomization was stratified by body weight that produced similar group mean weights using PATH/TOX SYSTEM software (Xybion Medical Systems Corporation, Lawrenceville, NJ).
F0 females were quarantined for 11 days after receipt. Ten nonmated females received with the time-mated females were designated for disease monitoring 4 days after arrival; samples were collected for serological analyses, and the rats were euthanized, necropsied, and examined for the presence of disease or parasites. The health of the F1 rats was monitored during the study according to the protocols of the NTP Sentinel Animal Program (Appendix C). Pinworms (Syphacia spp.) were diagnosed in sentinel animals during routine health monitoring evaluations. Infected animals did not display clinical signs, and no pathological lesions were noted in relation to the presence of the pinworms. Following this finding, DTT, in coordination with the testing laboratory, developed and implemented a successful plan of pinworm containment and eradication, without the use of medication. The NTP Sentinel Animal Program required the testing laboratories to actively monitor animals to ensure the continued exclusion of pinworms from all studies going forward. All other test results were negative.
Beginning on GD 6, groups of 38 F0 time-mated female rats were fed diets containing 0, 2,500, 5,000, 10,000, or 20,000 ppm TCPP throughout gestation and lactation. Groups of 50 F1 rats/sex/exposure concentration continued on study after weaning and were fed diets containing the same respective TCPP concentrations for 2 years.
F0 female rats were housed individually during gestation and with their respective litters during lactation. Water and dosed feed were available ad libitum. F0 female body weights were recorded on GDs 5, 6, 9, 12, 15, 18, and 21 and on LDs 1, 4, 7, 10, 14, 17, 21, 24, and 28. During gestation, feed consumption was continuously measured over 3-day intervals from GD 6 through GD 21 (GDs 6–9, 9–12, 12–15, 15–18, and 18–21). The day of parturition was considered to be LD 0. On apparent GD 25, all time-mated female rats that failed to deliver were euthanized and the uteri were examined and stained for evidence of implantation. Total litter weight and litter weights by sex were collected on PND 1. Individual F1 pup weights were recorded on PNDs 4, 7, 10, 14, 17, 21, 24, and 28. Clinical observations and survival were evaluated throughout lactation. During lactation, feed consumption was measured over 3-day intervals from LD 1 through LD 21 (LDs 1–4, 4–7, 7–10, 10–14, 14–17, 17–21, 21–24, and 24–28).
Select dams and their litters were removed on LD 28 to quantify tris(1-chloro-2-propyl) phosphate plasma concentrations. On LD 28, blood was collected from the retroorbital sinus of randomly selected dams (n = 5/exposure group) and pups (n = 1/sex/exposure group as available). Blood samples were collected in tubes containing tripotassium (K3) EDTA and centrifuged, and the plasma was harvested. All samples were stored frozen at approximately −20°C before shipment to MRIGlobal (Kansas City, MO) for analysis.
F1 litters were standardized on PND 4 to eight pups per litter, with at least two pups of each sex and a preference for four males and four females each. Litters that did not meet the minimum of eight pups (or had fewer than two pups of either sex) were removed from the study. For continuation of exposure after weaning, two males and two females per litter were randomly selected from 30 (0, 2,500, 5,000, and 20,000 ppm) and 28 (10,000 ppm) litters. Before weaning, on the day the last litter reached PND 26, 25 litters per exposure group were randomly selected and pups (generally two/sex/litter) were randomly assigned to the 2-year study. On the day the last litter reached PND 28, dams were removed from the cages, and the pups were weaned. Weaning marked the beginning of the 2-year study.
After weaning, F1 rats were housed two (males) or up to four (females) per cage. Water and dosed feed were available ad libitum. Feed consumption was measured weekly for the first 3 months, then for one 7-day period every 4 weeks thereafter, and at study termination. Cages were changed at least once weekly through PND 4, then changed at least twice weekly. Racks were changed and rotated every 2 weeks. Further details of animal maintenance are given in Table 3.
Two diets were used in the rat studies: (1) NIH-07 during the perinatal phase and (2) NTP-2000 during the postnatal phase. The NIH-07 diet is a higher protein diet that supports reproduction and lactation in rodents, whereas the NTP-2000 diet is a lower protein diet that decreases the incidence of chronic nephropathy in adult rats. Information on feed composition and contaminants for both diets is provided in Appendix B.
Study Design for Mice
Male and female B6C3F1/N mice were approximately 3 to 4 weeks old upon receipt and were quarantined for 11 (females) or 12 (males) days before study start. Mice were randomly assigned to one of four exposure groups (n = 50 mice/sex/exposure group). Randomization was stratified by body weight that produced similar group mean weights using PATH/TOX SYSTEM software (Xybion Medical Systems Corporation, Lawrenceville, NJ). Mice were fed diets containing 0, 1,250 (males only), 2,500, 5,000, or 10,000 (females only) ppm TCPP for 2 years.
Five male and five female mice were randomly selected for parasite evaluation and gross observation of disease. The health of the mice was monitored during the study according to the protocols of the NTP Sentinel Animal Program (Appendix C). All test results were negative.
Mice were housed individually (males) or up to four (females) per cage. Water and dosed feed were available ad libitum. Feed consumption was measured weekly for the first 3 months, then for one 7-day period every 4 weeks thereafter, and at study termination. Cages were changed at least once weekly (males) or twice weekly (females) and rotated every 2 weeks. Racks were changed and rotated every 2 weeks. Further details of animal maintenance are given in Table 3. Information on feed composition and contaminants is given in Appendix B.
Clinical Examinations and Pathology
In the 2-year studies in rats and mice, animals were observed twice daily for signs of morbidity and moribundity and were weighed before dosed feed exposure on study day 1, weekly for the first 13 weeks, every 4 weeks thereafter, and at study termination. Clinical observations were recorded every 4 weeks and at study termination.
At the 3-, 6-, 12-, and 18-month interim evaluations, blood was collected from up to 10 predesignated F1 rats/sex/exposure group and up to 5 predesignated mice/sex/exposure group for determination of TCPP concentrations. Blood samples were collected from the retroorbital plexus into tubes containing K3 EDTA as the anticoagulant (rats) or from the retroorbital sinus into tubes containing EDTA as the anticoagulant (mice). All rats continued on study after blood collection. Following blood collection at each interval, mice were euthanized via carbon dioxide inhalation and disposed of properly without further evaluation. Plasma was isolated from the blood via centrifugation and maintained frozen on dry ice or in liquid nitrogen. Frozen samples were either stored in a freezer set at −85°C to −60°C or shipped immediately following collection. Samples were shipped to MRIGlobal (Kansas City, MO) for analysis.
Complete necropsies and microscopic examinations were performed on all F1 rats and all mice. At necropsy, all organs and tissues were examined for grossly visible lesions and all major tissues were fixed and preserved in 10% neutral buffered formalin (NBF) except for eyes, testes, vaginal tunics, and epididymides, which were first fixed in Davidson’s solution or modified Davidson’s solution. Mouse liver tumors >5 mm in diameter were dissected in half and one half was collected in 10% NBF and the other half was snap-frozen in liquid nitrogen and stored at −80°C until processed for a molecular pathology study (Appendix G). Tissues were processed and trimmed, embedded in paraffin, sectioned at a thickness of approximately 5 μm, and stained with H&E for microscopic examination. For all paired organs (e.g., adrenal gland, kidney, ovary), samples from each organ were examined. In F1 rats, the uterus, cervix, vagina, and ovaries were mounted on cardstock before placement in fixative. Uterine horns were bisected at their midpoint, and one transverse section was taken from the midpoint of each horn for histopathological evaluation. The uterine body and the two free portions of each uterine horn were also examined. Tissues examined microscopically are listed in Table 3.
Microscopic evaluations were completed by the study laboratory pathologist, and the pathology data were entered into the Toxicology Data Management System. The report, slides, paraffin blocks, residual wet tissues, and pathology data were sent to the NTP Archives for inventory, slide/block match, wet tissue audit, and storage. The slides, individual animal data records, and pathology tables were evaluated by a QA pathologist at a pathology laboratory independent of the study laboratory. The individual animal records and tables were compared for accuracy, the slide and tissue counts were verified, and the histotechnique was evaluated. For the 2-year studies, a QA pathologist evaluated slides from all tumors and all potential target organs, which included the liver of rats and mice; the kidney of rats and male mice; the adrenal gland of rats and mice; the heart, pituitary gland, and thyroid gland of rats; the testis of male rats; the uterus of female rats; and the skin, lymph node, and lung of mice.
The DTT pathologist reviewed and addressed the diagnostic discrepancies between the QA and the laboratory pathologists. The QA pathologist also served as the Pathology Working Group (PWG) coordinator, and in consultation with the DTT pathologist, selected tissues for further review by the PWG. Representative histopathology slides containing examples of lesions related to chemical administration, examples of disagreements in diagnoses between the laboratory and QA pathologists, or lesions of general interest were presented by the coordinator to the PWG for review. The PWG consisted of the DTT pathologist and other pathologists experienced in rodent toxicologic pathology. This group examined the tissues without any knowledge of exposure groups. When the PWG consensus diagnosis differed from that of the laboratory pathologist, the diagnosis was changed. Final diagnoses for reviewed lesions represent a consensus between the laboratory pathologists, reviewing pathologists, DTT pathologist, and the PWG. Details of these review procedures have been described, in part, by Maronpot and Boorman53 and Boorman et al.54 For subsequent analyses of the pathology data, the decision of whether to evaluate the diagnosed lesions for each tissue type separately or combined was generally based on the guidelines of Brix et al.55
Statistical Methods
For all analyses, p values ≤0.05 were considered statistically significant. Statistical significance is one component of the “weight-of-evidence” approach to evaluate carcinogenicity (described in the Explanation of Levels of Evidence of Carcinogenic Activity section).
Survival Analyses
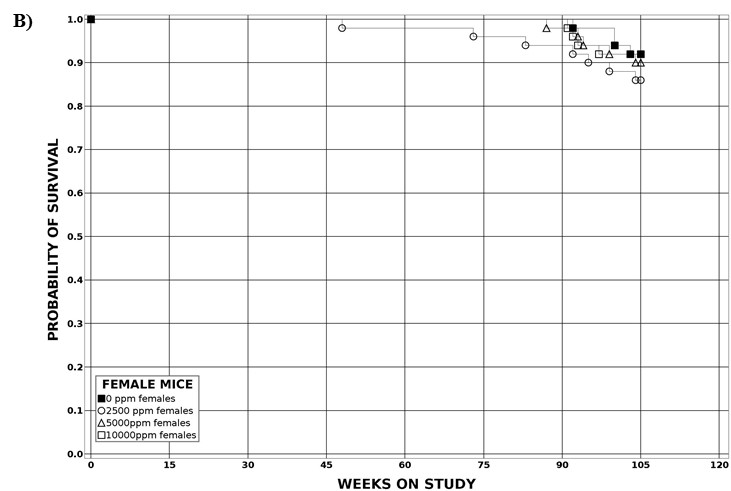
The probability of survival was estimated by the product-limit procedure of Kaplan and Meier57 and is presented graphically. Animals surviving to the end of the observation period are treated as censored observations, as are animals dying from unnatural causes within the observation period. Animals dying from natural causes are included in analyses and are treated as uncensored observations. For the 2-year mouse study, exposure concentration-related trends are identified with Tarone’s life-table test,58 and pairwise exposure concentration-related effects are assessed using Cox’s method.59 For the rat perinatal and 2-year study, exposure concentration-related trends and pairwise exposure concentration-related effects on survival are assessed using a Cox proportional hazards model59 with a random litter effect. All reported p values for the survival analyses are two-sided.
Calculation of Incidence
The incidences of neoplasms or nonneoplastic lesions are presented as the numbers of animals bearing such lesions at a specific anatomic site. For calculation of incidence rates, the denominator for most neoplasms and all nonneoplastic lesions is the number of animals for which the site was examined microscopically. When macroscopic examination was required to detect neoplasms in certain tissues (e.g., mesentery, pleura, peripheral nerve, skeletal muscle, tongue, tooth, and Zymbal’s gland) before microscopic evaluation, however, the denominator consists of the number of animals that had a gross abnormality. When neoplasms had multiple potential sites of occurrence (e.g., leukemia or lymphoma), the denominator consists of the number of animals on which a necropsy was performed. Additional study data also give the survival-adjusted neoplasm rate for each group and each site-specific neoplasm. This survival-adjusted rate (based on the Poly-3 method described below) accounts for differential mortality by assigning a reduced risk of neoplasm, proportional to the third power of the fraction of time on study, only to site-specific, lesion-free animals that do not reach terminal euthanasia.
Analysis of Neoplasm and Nonneoplastic Lesion Incidence
Statistical analyses of neoplasm and nonneoplastic lesion incidence for the 2-year studies considered two features of the data. Some animals did not survive the entire 2 years of the study, so survival differences between groups had to be considered. In addition, for the rat perinatal and 2-year study, up to two animals per sex were randomly selected from each litter to participate in the study. The statistical analysis of lesion incidence used the Poly-3 test to account for survival differences, with a Rao-Scott adjustment for litter effects, as described below.
The Poly-k test60-62 was used to assess neoplasm and nonneoplastic lesion prevalence. This test is a survival-adjusted quantal-response procedure that modifies the Cochran-Armitage linear trend test to account for survival differences. More specifically, this method modifies the denominator in the quantal estimate of lesion incidence to approximate more closely the total number of animal years at risk. For analysis of a given site, each animal is assigned a risk weight. This value is 1 if the animal had a lesion at that site or if it survived until terminal euthanasia; if the animal died before terminal euthanasia and did not have a lesion at that site, its risk weight is the fraction of the entire study time that it survived, raised to the kth power.
This method yields a lesion prevalence rate that depends only on the choice of a shape parameter for a Weibull hazard function describing cumulative lesion incidence over time.60 Unless otherwise specified, a value of k = 3 was used in the analysis of site-specific lesions. This value was recommended by Bailer and Portier60 after an evaluation of neoplasm onset time distributions for a variety of site-specific neoplasms in control Fischer 344 rats and B6C3F1 mice.63 Bailer and Portier60 showed that the Poly-3 test gave valid results if the true value of k was anywhere in the range of 1 to 5. A further advantage of the Poly-3 method is that it does not require lesion lethality assumptions. Variation introduced by the use of risk weights, which reflect differential mortality, was accommodated by adjusting the variance of the Poly-3 statistic as recommended by Bieler and Williams.64 Poly-3 tests used the continuity correction described by Nam.65
Littermates tend to be more like each other than like fetuses/pups in other litters. Failure to account for correlation within litters leads to underestimates of variance in statistical tests, resulting in higher probabilities of Type I errors (“false positives”). Because up to two pups/sex/litter were present in the rat perinatal and 2-year study, the Poly-3 test was modified to accommodate litter effects using the Rao-Scott approach.66 The Rao-Scott approach accounts for litter effects by estimating the ratio of the variance in the presence of litter effects to the variance in the absence of litter effects. This ratio is then used to adjust the sample size downward to yield the estimated variance in the presence of litter effects. The Rao-Scott approach was implemented in the Poly-3 test as recommended by Fung et al.67 formula ₸RS2.
Tests of significance included pairwise comparisons of each exposed group with control groups and a test for an overall exposure concentration-related trend. Reported p values are one-sided. The significance of a lower incidence or negative trend in lesions is approximated as 1−p with the letter N added (e.g., p = 0.99 is presented as p = 0.01N). For the rat perinatal and 2-year study, Rao-Scott-adjusted Poly-3 tests were used for trend and pairwise comparisons to the control group. For neoplasms and nonneoplastic lesions observed without litter structure (e.g., the mouse 2-year study), Poly-3 tests that included the continuity correction, but without adjustment for potential litter effects, were used. For the rat perinatal and 3-month study, Rao-Scott-adjusted Cochran-Armitage tests without the Poly-3 adjustment for survival were used.
To evaluate incidence rates by litter in the rat perinatal and 2-year study, the proportions of litters affected by each lesion type were tested among groups. Cochran-Armitage trend tests and Fisher’s exact test68 were used to test for trends and pairwise differences from the control group, respectively.
Analysis of Continuous Variables
Before statistical analysis, outliers identified using the Dixon and Massey test69 for small samples (n < 20) and Tukey’s outer fences method70 for large samples (n ≥ 20) were examined by DTT personnel, and biologically implausible values (likely due to experimental error) were eliminated from the analysis.
For the mouse 3-month study, organ and body weight measurements, which historically have approximately normal distributions, were analyzed with the parametric multiple comparison procedures of Dunnett71 and Williams.72,73 Hematology and sperm count data were analyzed using the nonparametric multiple comparison methods of Shirley74 [as modified by Williams75] and Dunn76 given that these endpoints typically have skewed distributions. For all quantitative endpoints, the Jonckheere test77 was used to assess the significance of the exposure concentration-related trends and to determine at the 0.01 level of significance, whether a trend-sensitive test (the Williams or Shirley test) was more appropriate for pairwise comparisons than a test that does not assume a monotonic exposure concentration-related trend (the Dunnett or Dunn test).
For the rat perinatal studies, dam gestational and lactational feed consumption, litter sizes, pup survival, and proportion of male pups per litter were analyzed using the nonparametric Shirley or Dunn tests described above. Organ weights, terminal body weights, clinical chemistry, and hematology data were measured on two pups/sex/litter in the rat perinatal studies; more than two pups/sex/litter were possible in preweaning body weight measurements. The analysis of the clinical chemistry and hematology data took litter effects into account using a bootstrapped Jonckheere test for trend and a Datta-Satten78 modification to the Wilcoxon test for pairwise comparisons, with a Hommel79 adjustment for multiple comparisons. The analyses of pup body weights, organ weights, and body weights adjusted for litter size (described below) of these animals took litter effects into account using a mixed model with litter as a random effect. To adjust for multiple comparisons a Dunnett-Hsu adjustment was used.80 Dam body weights during gestation and lactation were analyzed with the parametric multiple comparison procedures of Dunnett71 or Williams,72,73 depending on whether the Jonckheere test indicated the use of a trend-sensitive test. P values for these analyses are two-sided. For the image analysis of thymus size in the rat perinatal and 3-month study, pairwise comparisons with the control group were performed using one-sided Dunnett tests.
Analysis of Gestational and Fertility Indices
Cochran-Armitage trend tests were used to test the significance of trends in gestational and fertility indices across exposure groups in the rat perinatal studies. Fisher’s exact test was used to conduct pairwise comparisons of each exposed group with the control group. P values for these analyses are two-sided.
Body Weight Adjustments
Preweaning pup body weights in the rat perinatal studies were adjusted for live litter size as follows: A linear model was fit to body weights as a function of exposure and litter size. The estimated coefficient of litter size was then used to adjust each pup body weight based on the difference between its litter size and the mean litter size. Prestandardization PND 4 body weights were adjusted for PND 1 litter size, and body weights measured between PND 4 poststandardization and PND 28 were adjusted for PND 4 poststandardization litter size. After adjustment, mean body weights were analyzed with a linear mixed model with a random litter effect.
Historical Control Data
The concurrent control group is the most valid comparison to the exposed groups and is the only control group analyzed statistically in NTP bioassays. Historical control data are often helpful in interpreting potential exposure concentration-related effects, however, particularly for uncommon or rare neoplasm types. For meaningful comparisons, the conditions for studies in the historical control data must be generally similar. Significant factors affecting the background incidence of neoplasms at a variety of sites are diet, sex, strain/stock, and route of exposure. The NTP historical control database contains all 2-year studies for each species, sex, and strain/stock with histopathology findings in control animals completed within the most recent 5-year period,81-83 including the concurrent control for comparison across multiple technical reports. In general, the historical control data for a given study includes studies using the same route of administration, and the overall incidence of neoplasms in controls for all routes of administration are included for comparison, including the current study.
Quality Assurance Methods
The 3-month and 2-year studies were conducted in compliance with U.S. Food and Drug Administration Good Laboratory Practice Regulations.84 In addition, the 3-month and 2-year study reports were audited retrospectively by an independent QA contractor against study records submitted to the NTP Archives. Separate audits covered completeness and accuracy of the pathology data, pathology specimens, final pathology tables, and a draft of this NTP Technical Report. Audit procedures and findings are presented in the reports and are on file at NIEHS. The audit findings were reviewed and assessed by DTT staff, and all comments were resolved or otherwise addressed during the preparation of this Technical Report.
Genetic Toxicology
The genetic toxicity of TCPP was assessed by testing whether the chemical induces mutations in various strains of Salmonella typhimurium and Escherichia coli or increases the frequency of micronucleated erythrocytes in rat and mouse peripheral blood. The protocol for these studies and the results are given in Appendix D.
The genetic toxicity studies have evolved from an earlier effort to develop a comprehensive database permitting a critical anticipation of a chemical’s carcinogenicity in experimental animals based on numerous considerations, including the relationship between the molecular structure of the chemical and its observed effects in short-term in vitro and in vivo genetic toxicity tests (structure-activity relationships). The short-term tests were developed originally to clarify proposed mechanisms of chemical-induced DNA damage, given the relationship between electrophilicity and mutagenicity,85 and the somatic mutation theory of cancer.86,87 Not all cancers, however, arise through genotoxic mechanisms.
Bacterial Mutagenicity
DNA reactivity combined with Salmonella mutagenicity is highly correlated with induction of carcinogenicity in multiple species/sexes of rodents and at multiple tissue sites.88 A positive response in the Salmonella test was shown to be the most predictive in vitro indicator for rodent carcinogenicity (89% of the Salmonella mutagens are rodent carcinogens).89,90 Additionally, no battery of tests that included the Salmonella test improved predictivity over the Salmonella test alone. Other tests, however, can provide useful information on the types of DNA and chromosomal damage induced by the chemical under investigation.
Peripheral Blood Micronucleus Test
Micronuclei (literally “small nuclei” or Howell-Jolly bodies) are biomarkers of induced structural or numerical chromosomal alterations and are formed when acentric fragments or whole chromosomes fail to incorporate into either of two daughter nuclei during cell division.91,92 Acute in vivo bone marrow chromosome aberration and micronucleus tests appear to be less predictive of carcinogenicity than the Salmonella test.93,94 However, clearly positive results in long-term peripheral blood micronucleus tests have high predictivity for rodent carcinogenicity; a weak response in one sex only or negative results in both sexes in this assay do not correlate well with either negative or positive results in rodent carcinogenicity studies.95 Because of the theoretical and observed associations between induced genetic damage and adverse effects in somatic and germ cells, determination of in vivo genetic effects is important to overall understanding of the risks associated with exposure to a particular chemical.
Results
Data Availability
All study data were evaluated. Data relevant for evaluating toxicological findings are presented here. All study data are available in the National Toxicology Program (NTP) Chemical Effects in Biological Systems (CEBS) database: https://doi.org/10.22427/NTP-DATA-TR-602.96
Rats
Three-month Study (Perinatal Phase)
Maternal toxicity was observed in dams exposed to 40,000 ppm tris(chloropropyl) phosphate (TCPP). One dam was moribund and euthanized on gestation day (GD) 11; on GD 13, all remaining 40,000 ppm dams were either found dead or euthanized moribund (data not shown). Therefore, this exposure group will not be displayed in tables for lactation or postweaning data. The mean body weight of dams in this group was approximately 20% lower than that of the control group on GD 9 and GD 12, and feed consumption was significantly decreased by 31% from GD 6 through GD 9 and was lower by 24% from GD 9 through GD 12 compared to the control group (Appendix H).
All dams in the control group and remaining TCPP-exposed groups (2,500, 5,000, 10,000, and 20,000 ppm) survived during gestation (Table 4). The percentage of dams delivering was variable across TCPP-exposed groups. There was no clear exposure concentration response, and the number of dams in each group was different, so the biological significance of this finding is unclear. Maternal survival was not affected in the control group or TCPP-exposed groups during the lactation phase (Table 5).
Mean body weight gain and feed consumption by dams in the 20,000 ppm TCPP group fluctuated during gestation. Body weight gain was significantly decreased during GD 6–9 and then significantly increased during GD 12–15. Overall, mean body weight gain during gestation (GD 6–21) was within 10% of that of the control dams (Table 5). Likewise, dams in the 20,000 ppm group had a terminal mean body weight on lactation day (LD) 21 that was only 6% lower than that of the control group. Mean body weight gains in TCPP-exposed groups ≤10,000 ppm were largely within 10% of, and not statistically different from, the control group (Table 5). During several gestation intervals (GD 6–9, 12–15, and 15–18), feed consumption trended higher with increasing exposure concentration (Table 6). Feed consumption during GD intervals 12–15 and 6–21 was significantly increased in the 20,000 ppm TCPP group compared to the control group (Table 6). Chemical intake during gestation (GD 6 through GD 21) was estimated to be 186, 379, 802, and 1,756 mg TCPP/kg body weight/day (mg/kg/day) for the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively. In general, the estimated TCPP intake was proportional to the twofold increase in the respective TCPP exposure concentrations in feed.
There were no significant differences in mean body weight of TCPP-exposed dams compared to control animals during lactation, with values within 10% of those of the control group. However, maternal body weight gain was significantly decreased in the 20,000 ppm TCPP group over the course of lactation (LD 1 through LD 21); weight gains by dams exposed to ≤10,000 ppm TCPP were similar to the control group despite the fact that values fluctuated over time (Table 5). Feed consumption during lactation also sporadically changed over time with the greatest decrease (compared to control animals) of 22% in the 20,000 ppm group over the LD 10–14 interval (Table 6). Chemical intake during LD 1–14 was estimated to be 366, 743, 1,430, and 2,974 mg/kg/day for the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively. Chemical intake from LD 14 through LD 21 was not calculated because the entire litter ate the feed and an accurate assessment could not be made. Overall, the estimated TCPP intake was proportional to the twofold increase in the respective TCPP exposure concentrations in feed.
At postnatal day (PND) 1, there were no effects of TCPP exposure on litter size, litter weight, or sex distribution (Table 7, Table 8). Pup survival was also not affected during lactation. Male and female pup mean body weights in the 20,000 ppm group were significantly decreased compared to those of the control groups from PND 4 through PND 21 (Table 8), which was interpreted as lower body weight gains compared to control pups. Male pup mean body weights were significantly decreased by 15%, 19%, 20%, and 31% relative to that of the control group at PNDs 4, 7, 14, and 21, respectively (Table 8). Female pup mean body weights were significantly decreased by 14%, 18%, 19%, and 27% relative to that of the control group at PNDs 4, 7, 14, and 21, respectively (Table 8).
Three-month Study (Postweaning Phase)
Male rats in the 20,000 ppm group did not survive to study termination (Table 9). Seven males in this group were euthanized moribund after displaying thinness, lethargy, paleness, and small size on study days 4 and 5, and one male was found dead on study day 5. Additionally, the mean feed consumption by this group was 14% of the control group (Appendix H). The remaining males in the 20,000 ppm group were removed from the study on study day 5. All females in the 20,000 ppm group and all animals in the remaining groups (males and females) survived until study termination with no clinical signs of toxicity (Table 9, Table 10; Appendix H).
Mean body weights of the remaining male rats exposed to TCPP were within 10% of the control group by study termination (Table 9). The mean body weight of the 20,000 ppm females was 31% lower than that of the control group on study day 1, but only 12% lower than that of the control group by study termination (Table 10). Group mean body weights of the 2,500, 5,000, and 10,000 ppm male and female rats were approximately 7%–13% lower than those of the respective control groups at the beginning of the postweaning period (Table 9, Table 10; Figure 1); however, weight gain was sufficient enough such that mean body weights of male and female rats in these groups were within 6% of those of the respective control groups by study termination (Table 9, Table 10; Appendix H).
In general, feed consumption by male and female rats in TCPP-exposed groups was lower (males = 13%–86%; females = 5%–47%) than that of the control groups during the first week of the study (Table 11, Table 12; Appendix H). Feed consumption values fluctuated over the course of the study but were within approximately 10% of the control groups at study termination with the exception of female rats in the 20,000 ppm group, whose consumption was 18% lower than that of the female control group at study termination. For males, average daily TCPP intakes based on weekly average feed ingestion and body weight were estimated to be 223, 431, and 911 mg/kg/day for males in the 2,500, 5,000, and 10,000 ppm groups, respectively; for females, they were estimated to be 236, 458, 906, and 1,890 mg/kg/day in the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively (Appendix H). Table 11 and Table 12 highlight feed consumption (i.e., feed [g/day]) and estimated TCPP intake (i.e., dose [mg/kg/day]) at select time points. Overall, the increase in TCPP intake across groups was of similar proportion to the increase in exposure concentration for both sexes.
At study termination, significant exposure concentration-related decreases in alanine aminotransferase (ALT) and alkaline phosphatase (ALP) activities were observed in both male and female rats (Table 13). Cholesterol concentrations were significantly increased in the 10,000 ppm male and the 10,000 and 20,000 ppm female rats (Table 13). All other statistically significant clinical pathology changes were minimal or inconsistent and not considered due to TCPP exposure (Appendix H).
Exposure-related organ weight changes were observed in the liver and thymus (Table 14). Male rats exposed to 10,000 ppm TCPP had significant increases in absolute (15%) and relative liver weights compared to the control group. Liver weights were within 10% of the control group in male rats exposed to ≤5,000 ppm TCPP. A similar pattern was observed in female rats with absolute liver weights 13% and 19% higher in female rats exposed to 10,000 and 20,000 ppm TCPP, respectively. Relative liver weights in female rats were also increased in a significant exposure concentration-related manner (Table 14).
Absolute and relative thymus weights of male and female rats were higher in TCPP-exposed groups (Table 14). Absolute thymus weights were 21%–35% higher in all TCPP-exposed male rats with statistical significance achieved in the 5,000 and 10,000 ppm groups. Similarly, relative thymus weights were significantly increased in all TCPP-exposed male rats when compared to those of the control group. TCPP-exposed female rats exhibited a 14%–37% increase in absolute thymus weights; the differences were statistically significant in the 10,000 ppm group. At the highest exposure concentration of 20,000 ppm, absolute female thymus weights were 22% higher than those of the control group but not statistically significant. Relative thymus weights were higher in all TCPP-exposed female groups with statistical significance in groups exposed to ≥5,000 ppm.
Alterations in kidney, lung, and heart weights were observed in both male and female rats exposed to TCPP (Appendix H). These changes were not considered to be related to exposure and did not have a histological correlate upon tissue examination.
There were no changes in weights of the testis or epididymis, or alterations in sperm parameters attributable to TCPP exposure (Appendix H). Observed changes—although at times statistically significant—were of small magnitude, were not related to exposure concentration, did not exhibit histopathological correlates, and were consistent with normal biological variability.
Histopathology
This section describes the statistically significant or biologically noteworthy changes in the incidence of nonneoplastic lesions of the liver and thymus.
Liver: In male and female rats, there was minimal bile duct hyperplasia in the 10,000 and 20,000 ppm (female only) groups (Table 15). This lesion is characterized by increased numbers (2–5) of biliary ductules of variable sizes, lined by well-differentiated low cuboidal epithelial cells in the portal areas. The occasional presence of inflammatory cells, predominantly lymphocytes, and macrophages and/or fibroblasts surrounded by scant collagen, expanded the portal areas. There were no apparent histological correlates to the increases in absolute and relative liver weights in male or female rats.
Thymus: In male and female rats, there were significantly increased relative thymus weights in all exposed groups with the exception of the female 2,500 ppm group (Table 14). Given that this was an unusual finding within a subchronic rodent study, additional morphometry analysis was conducted with hematoxylin and eosin (H&E)-stained sections to characterize the histological changes. The increases in thymus weights manifested microscopically in male rats with significantly increased total area of the thymus and expanded thymic cortices in all exposed groups compared to those of the control groups as demonstrated by morphometry. In female rats, only the 10,000 ppm group showed significantly increased thymic area with expanded cortices and medullae (Table 16; Appendix H). These histological changes correlated with thymus weights (Table 14).
Exposure Concentration Selection Rationale for Two-year Study in Rats
During gestation, maternal toxicity was observed in dams exposed to 40,000 ppm TCPP, which resulted in removal of that group from study. In the lactation period, dams and offspring exposed to ≤20,000 ppm TCPP did not show significant signs of toxicity. Postweaning (PND 21), male rats exposed to 20,000 ppm TCPP were removed on study day 5 because of the presence of adverse clinical signs and reductions in mean body weight and feed consumption. Males exposed to ≤10,000 ppm TCPP and all female rats exposed to TCPP survived to study termination with no clinical signs of toxicity or nonneoplastic lesions of concern for chronic exposure. In parallel with this study, findings from additional NTP research suggested that PND 21 was potentially too early for weaning of Sprague Dawley rats and a global decision to extend the weaning period to PND 28 was made to reduce stress on offspring and increase survival. Concentrations for the 2-year study were selected to be 0, 2,500, 5,000, 10,000, or 20,000 ppm TCPP in feed. The extended weaning period and results from the 3-month study were the primary rationale for a top exposure concentration of 20,000 ppm TCPP.
Two-year Study (Perinatal Phase)
No exposure-related effects were observed on the pregnancy status, maternal survival, or number of dams that littered (Table 17).
There were exposure-related effects on maternal body weights during gestation with a significant decrease of up to 9% in the 20,000 ppm group at GD 9 compared to the control group, but the magnitude of the effect subsided as gestation progressed (Table 18). During lactation, a significant decrease was observed in mean body weights of up to 12% in the 20,000 ppm group compared to that of the control group, with the magnitude of the effect decreasing after the LD 14 time point (likely coinciding with the reduced feed consumption that resulted from F1 pups switching from nursing to consuming feed).
Feed consumption (g/animal/day) by TCPP-exposed dams during gestation and lactation was largely within 10% of control animals (Table 19); slight but significant increases or decreases in feed consumption were observed sporadically during this exposure period. Feed spillage was evident across all exposure groups at multiple times during both gestation and lactation. When spillage occurred, data were not included for a specific dam and time point, which is reflected by the number of dams in Table 19. Chemical intake during gestation (GD 6–21) was estimated to be 183, 368, 749, and 1,544 mg/kg/day for the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively. Chemical intake during the LD 1–14 interval was estimated to be 389, 763, 1,606, and 3,839 mg/kg/day for the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively (Table 19). Chemical intake from LD 14 through LD 28 was not calculated because the entire litter ate the feed, and an accurate assessment could not be made. The consumed dose at successive exposure concentrations was of similar proportion to the increase in exposure concentration for both sexes.
Total and live litter sizes and survival of the F1 rats during lactation were not affected by TCPP exposure (Table 20). An exposure concentration-related decrease in male and female pup mean body weights was observed during lactation; this effect increased over time and plateaued between PND 21 and 28 (Table 21). In the highest exposure group of 20,000 ppm, mean body weights were significantly decreased by 12%–31% in male and 11%–30% in female rats from PND 7 through PND 28 (Table 21). These changes were attributed to lower body weight gains rather than to body weight loss during postnatal exposure.
Two-year Study (Postweaning Phase)
Survival of F1 male and female rats was not adversely affected by exposure to TCPP. At study termination, survival rates for male and female rats exposed to TCPP exceeded that of their respective control group (Table 22; Figure 2). There were no exposure-related clinical observations noted during the 2-year study (Appendix H).
At the start of the 2-year exposure phase, mean body weights of male and female rats in the 20,000 ppm group were approximately 75% of control group values (Table 23, Table 24; Figure 3). Over the course of the study, this gap decreased for male rats and the terminal mean body weight of the 20,000 ppm group was within 8% of that of the control group. Female rats in the 20,000 ppm group also recovered, to some degree, but their mean body weight over the course of the study fluctuated between 79% and 89% of that of the control group; their terminal mean body weight was 17% lower than that of the control group (Table 24; Figure 3). The lower weights were interpreted to be the result of lower body weight gains rather than body weight loss during the exposure period. Although mean body weights for male and female rats fluctuated over the course of the study in the lower exposed groups, mean body weights remained within 10% of the control groups during the majority of time intervals. Overall, mean body weights of female rats were more affected by TCPP exposure than those of male rats.
In general, feed consumption by male and female rats in the 20,000 ppm TCPP-exposed groups was slightly lower (15% for males; 4% for females) than that of the respective control groups during the first week of the study (Table 25, Table 26; Appendix H). At study termination (week 102), feed consumption by TCPP-exposed male and female rats in the 20,000 ppm group was approximately 17% and 27% lower, respectively, than that of the control groups (Table 25, Table 26). All other TCPP-exposed male and female rats (2,500, 5,000, and 10,000 ppm groups) were comparable (between 1% and 13%) to that of the control groups at study termination (Table 25, Table 26). Daily TCPP intakes, based on weekly body weight and feed consumption averages (Appendix H), were estimated to be 141, 294, 626, and 1,155 mg/kg/day for male rats in the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively, over the 2-year period. For female rats, TCPP intakes, based on weekly body weight and feed consumption averages (Appendix H), were estimated to be 156, 323, 674, and 1,295 mg/kg/day in the 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively, over the 2-year period. Table 25 and Table 26 highlight feed consumption (i.e., feed [g/day]) and estimated TCPP intake (i.e., dose [mg/kg/day]) at select time points. In general, the estimated TCPP intake was proportional to the twofold increase in the respective TCPP concentration in dosed feed for both male and female rats. Further, the doses of TCPP consumed by male rats relative to female rats, in mg/kg/day, were similar for a given concentration of TCPP in feed.
Histopathology
This section describes the statistically significant or biologically noteworthy changes in the incidence of neoplasms and/or nonneoplastic lesions of the liver, uterus, small intestine, testis, adrenal cortex, kidney, and ovary.
Liver: In male rats in the 10,000 and 20,000 ppm groups, the incidences of hepatocellular adenomas were higher than in control rats. In addition, the incidences of hepatocellular carcinomas also were higher in the 5,000, 10,000, and 20,000 ppm groups. The higher incidences of hepatocellular adenoma or carcinoma (combined) showed a positive trend with exposure concentration but lacked significance in pairwise tests when compared to the control group (Table 27). Nonneoplastic lesions, such as altered hepatic foci (basophilic, eosinophilic, and mixed cell), exhibited positive trends with increasing exposure concentration. In particular, the increased incidences in the 10,000 (basophilic only) and 20,000 ppm groups were significant by pairwise comparisons with the control group. There were higher incidences of biliary hyperplasia in all exposed groups (significantly increased at 2,500 and 20,000 ppm) compared to the control group, but the severity grade was minimal (Table 27). In female rats, the incidences of hepatocellular neoplasms were lower than in male rats. There were slightly higher incidences of hepatocellular adenomas in the 2,500, 10,000, and 20,000 ppm females than in the control group, but these incidences were not significant (Table 27). There were no incidences of hepatocellular carcinomas in any female exposure group. The incidences of altered hepatic foci (basophilic, eosinophilic, and mixed cell) increased with exposure concentration, but pairwise significance was achieved only with eosinophilic foci in the 10,000 and 20,000 ppm groups. There were higher incidences of biliary hyperplasia, which were statistically significant by pairwise comparison, in the 2,500, 5,000, and 10,000 ppm exposed females, but the severity grade was minimal. Interestingly, in female rats, there were also significantly increased incidences of bile duct cysts in the top three exposed groups compared to the control group. In addition, in both male and female rats, there was a significantly increased incidence of pigment in the 20,000 ppm group, but severity was minimal (Table 27).
Microscopically, hepatocellular adenomas were well-circumscribed expansile masses comprising irregular plates of hepatocyte cords with eosinophilic to basophilic cytoplasm, and occasionally vacuolated cytoplasm, with minimal to mild compression of the adjacent parenchyma (Figure 4). Central veins and portal areas were occasionally entrapped in hepatocellular neoplasms. Hepatocellular carcinomas were large and invasive and were composed of nodules of neoplastic hepatocytes arranged in solid, glandular, or trabecular patterns (Figure 5). These neoplasms were also associated with increased cellular atypia accompanied by a few mitoses. Occasionally, there were areas of necrosis and hemorrhage within these masses. Hepatic foci are tinctorially discrete areas of hepatocytes that blend imperceptibly into the surrounding hepatic parenchyma with no evidence of compression or invasion. In addition, the presence of pale-gold color in the cytoplasm of hepatocytes was diagnosed as pigment. Biliary hyperplasia was of minimal severity and was characterized by increased numbers (3–5) of biliary ductules of variable sizes, lined by well-differentiated low cuboidal epithelial cells in the portal areas. The bile duct cysts observed in female rats were characterized by dilated cystic spaces lined by low columnar epithelial cells. The increased incidences of minimal biliary hyperplasia and bile duct cysts are probably not related to the hepatocellular changes.
Uterus: In female rats, incidences of uterine adenocarcinomas across all exposed groups were slightly higher than that of the control group but did not reach statistical significance in a pairwise test or trend analysis (Table 28). Single incidences of adenomas were observed in the 10,000 and 20,000 ppm female groups, and a significant trend was observed for the combined incidence of uterine adenomas and adenocarcinomas. Remarkably, there was a doubling in the 5,000 and 10,000 ppm groups and a tripling in the 20,000 ppm group of the adenoma and adenocarcinoma (combined) incidences. There were slightly higher incidences of atypical hyperplasia and cystic endometrial hyperplasia in the exposed groups compared to the control group, but they did not reach statistical significance. There was also a significant increase in the incidence of uterine polyps in the 2,500 ppm group and a higher, but not statistically significant, incidence in the 20,000 ppm group. There were single incidences of squamous cell carcinomas in the 10,000 and 20,000 ppm groups and a single incidence of squamous cell papilloma in the 20,000 ppm group. Incidences of squamous metaplasia in the uterus did not reach statistical significance in a pairwise test or trend analysis.
Uterine adenocarcinomas were characterized by poorly circumscribed proliferations of cuboidal to columnar epithelial cells that displayed pleomorphism and atypia (Figure 6A). The cells were arranged in papillary or glandular structures, and there was effacement of the normal parenchyma and invasion into the underlying musculature and serosa (Figure 6B). Atypical endometrial hyperplasia was characterized by clusters of enlarged glands lined by thickened, disorganized epithelium with epithelial cells displaying loss of nuclear polarity, karyomegaly, mitoses, and cellular pleomorphism (Figure 7A). Occasionally, these proliferating cells abutted or invaded into the myometrium (Figure 7B). Cystic endometrial hyperplasia was characterized by an increased number of endometrial glandular epithelial cells surrounding large open spaces (cystic); the increased endometrial epithelial cells did not lose nuclear polarity or exhibit atypia or mitoses. Adenomyosis, characterized by extension of the normal endometrial glands into myometrial layers, was occasionally observed (Figure 8). Stromal polyps were exophytic nodules that projected into the uterine lumen and were characterized by broad stalks of endometrial stroma that were covered by normal-appearing endometrial surface epithelium. The endometrial stroma contained blood vessels and occasional endometrial glands.
Squamous cell carcinomas were characterized by poorly circumscribed proliferation of polygonal epithelial cells arranged in nests, cords, and papillary structures that occupied the uterine lumen and extended deep into the myometrium and onto the serosal surface. The neoplastic cells exhibited keratinization multifocally, cellular atypia, and mitotic figures. There was marked desmoplasia surrounding nests of squamous cells, large areas of keratinization, areas of necrosis, hemorrhage, and inflammation within the tumors. Squamous cell papilloma is characterized by exophytic proliferation of squamous cells in the form of nests, cords, and papillary fronds surrounded by stroma but the endometrium or myometrium was not invaded. The tumor mass had a broad base, multifocal keratinized squamous cells, areas of necrosis, and hemorrhage. Squamous metaplasia is characterized by multifocal replacement of the columnar to cuboidal endometrial epithelium with stratified squamous epithelial cells with varying degree of keratinization.
In addition to the neoplastic lesions discussed above, there were nonsignificant increases in neoplastic lesions in the small intestine, testis, adrenal gland, kidney, and ovary (Appendix H).
Small Intestine: In male rats, there were slightly higher incidences of adenocarcinoma in the small intestine in one rat in the 5,000 ppm group and in three rats in the 20,000 ppm group; these increases were not statistically significant in comparison to the control group (Appendix H). These neoplasms are uncommon in rats, with an approximate 2% incidence in NTP historical control animals for all routes of exposure. They are characterized by neoplastic glandular epithelium with nuclear atypia, loss of polarity, and invasion into the underlying muscular tunics. There were no incidences of these neoplasms in female rats in this study.
Testis: In male rats, there were slightly higher incidences of interstitial cell adenoma (also known as Leydig cell neoplasm) in the testis of three rats each in the 2,500, 10,000, and 20,000 ppm TCPP groups; these increases were not statistically significant in comparison to the control group (Appendix H). These neoplasms are not uncommon in rats with approximately 0%–6% and 0%–14% incidences in NTP historical control animals from experiments with exposure via feed versus all routes of exposure, respectively. These neoplasms are characterized by circumscribed masses larger than three normal seminiferous tubules that slightly compress the surrounding seminiferous tubules and are composed of uniform polyhedral neoplastic interstitial cells with abundant finely granular eosinophilic or vacuolated cytoplasm and centrally located nuclei with prominent nucleoli.
Adrenal Cortex: In female rats, there were slightly higher incidences of adenoma in the adrenal cortex in two rats each in the 10,000 and 20,000 ppm TCPP groups; these increases were not statistically significant in comparison to the control group (Appendix H). These neoplasms are uncommon in rats with an approximate 2% incidence in NTP historical control animals from all routes of exposure. These neoplasms are characterized by discrete expansile masses compressing the adjacent cortical tissue and are composed of cords, trabeculae, or solid clusters of enlarged polyhedral cells with eosinophilic, vacuolated cytoplasm, and may be accompanied by atypia and occasional mitosis. In male rats, there were single incidences in the 2,500 and 10,000 ppm groups.
Kidney: In female rats, there were renal tubule adenomas in the kidneys of two individuals in the 20,000 ppm group; the increased incidence was not statistically significant in comparison to the control group (Appendix H). These neoplasms are very rare in rats with <1% incidence in NTP historical control animals for all routes of exposure and 0% incidence in feed studies. These neoplasms are characterized by discrete expansile masses compressing the adjacent renal tubules and are composed of solid clusters of enlarged polyhedral cells with amphiphilic cytoplasm and may be accompanied by atypia and occasional mitosis. In male rats, there was a single incidence of renal tubule carcinoma in the 5,000 ppm TCPP group.
Ovary: In female rats, there were higher incidences of malignant granulosa cell neoplasms in the 2,500 ppm (one) and 5,000 ppm (three) groups and benign granulosa cell neoplasms in the 20,000 ppm group (two) (Appendix H). These neoplasms are relatively uncommon and have <2% incidence in NTP historical control animals from feed as well as from all routes of exposure. The benign granulosa cell neoplasms are composed of a discrete nodule larger than a corpus luteum and comprise >70% granulosa cell follicle-like nests and nodules intermixed with other sex cord/stromal cells. The granulosa cells are small round to fusiform, basophilic with scant eosinophilic cytoplasm, and nuclei with stippled chromatic and nucleoli. In the benign neoplasm, there is some compression and in the malignant variant, there is invasion into the adjacent parenchyma.
Other Tissues: In addition to the neoplastic findings, there were higher incidences of some nonneoplastic lesions including focal granulomatous inflammation in the lung (all exposure groups) and hyperplasia of the prostatic epithelium (5,000 and 20,000 ppm groups) in male rats and suppurative inflammation in the kidney (0 and 20,000 ppm groups) in female rats (Appendix H). However, the biological or toxicological significance of these lesions could not be determined.
Mice
Three-month Study
All male and female mice survived to study termination, and no clinical signs of toxicity were observed in TCPP-exposed groups (Table 29, Table 30; Appendix H). TCPP-exposed males gained less weight than did animals in the control group in an exposure concentration-dependent manner throughout the study. By study termination, mean body weights of male mice in the 2,500, 5,000, 10,000, and 20,000 ppm groups were approximately 11%, 16%, 24%, and 29% lower, respectively, than that of the control group (Table 29; Figure 9). Mean body weights for males exposed to 1,250 ppm remained within 10% of the control group throughout the study. Female mice also gained less weight than did animals in the control group, but this response was not exposure concentration dependent. By study termination, mean body weights of female mice in the 1,250, 2,500, 5,000, 10,000, and 20,000 ppm groups were approximately 12%, 7%, 3%, 9%, and 15% lower, respectively, than that of the control group (Table 30; Figure 9).
Feed consumption by male and female mice was typically within 10% of that of control animals (Table 31, Table 32; Appendix H). At times, consumption by mice exposed to ≥5,000 ppm TCPP was approximately 10% higher than by control groups. TCPP intakes, based on weekly averaging (Appendix H), were estimated to be 225, 473, 1,050, 2,509, and 4,446 mg/kg/day for male mice in the 1,250, 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively. For female mice, TCPP intakes, based on weekly averaging (Appendix H), were estimated to be 204, 442, 924, 1,841, and 3,645 mg/kg/day in the 1,250, 2,500, 5,000, 10,000, and 20,000 ppm groups, respectively. Table 31 and Table 32 highlight feed consumption (i.e., feed [g/day]) and estimated TCPP intake (i.e., dose [mg/kg/day]) at select time points. In general, the increase in TCPP intake across groups was of similar proportion to the increase in exposure concentration across groups for both sexes.
At study termination, the white blood cell count and lymphocyte count were significantly decreased in 20,000 ppm male mice (Appendix H). These decreases are consistent with chronic stress of exposure, which is supported by the moderate decreases in mean body weight observed in this exposed group.97 All other significant hematology changes were minimal or inconsistent and not considered to be due to TCPP exposure (Appendix H).
TCPP exposure was associated with organ weight changes in the liver and kidney (Table 33). In the liver, the only significant increases in absolute weight were observed in male and female mice in the 20,000 ppm group; liver weight was increased by approximately 39% and 25% in male and female mice, respectively, at study termination. Relative liver weights were significantly increased in the 2,500, 5,000, 10,000, and 20,000 ppm male and female groups (Table 33). These liver weight changes were associated with microscopic changes (described below).
Absolute kidney weights decreased with increasing exposure concentration, with the greatest difference from the control mice reported for the 20,000 ppm groups: a 22% significant decrease in males and 13% significant decrease in females (Table 33). Relative kidney weights were generally higher in male and female mice exposed to TCPP. Some changes were significantly different from the control groups; however, they were not considered related to exposure concentration. Although microscopic changes were observed in the male kidney (described below), there is no direct link with organ weight changes.
Compared to the control groups, mean absolute heart weights in male mice exposed to 10,000 and 20,000 ppm were significantly decreased by 15%; mean absolute heart weights were significantly decreased by 19% in female mice in the 20,000 ppm group. Mean relative heart weights were significantly increased in male mice exposed to ≥5,000 ppm TCPP, but this effect did not occur in female mice. Changes in absolute or relative heart weight did not correspond with microscopic changes (Appendix H). The biological and toxicological significance of this change is unclear. Absolute testis weights in mice were significantly decreased by 8% in the 20,000 ppm group and relative testis weights were significantly increased in ≥2,500 ppm TCPP dosed groups (Appendix H). The weights of the epididymis and cauda epididymis in the 20,000 ppm group were significantly decreased by 15% and 14%, respectively. No exposure-related changes were observed in counts (spermatid or sperm), density (spermatid or sperm), or sperm motility (Appendix H). These observations are consistent with the absence of microscopic changes, and, therefore, changes in testicular and epididymal weights are considered a consequence of exposure-related lower body weight of these mice.
Histopathology
This section describes the statistically significant or biologically noteworthy changes in the incidence of nonneoplastic lesions of the liver and kidney.
Liver: Relative liver weights were significantly increased in all ≥2,500 ppm groups of male and female mice (Table 33). Microscopically, these increases in liver weights were associated with an increase in the incidences of hepatocellular hypertrophy; these increases were significant in the 5,000, 10,000, and 20,000 ppm male and female groups (Table 34). Depending on the severity of the lesion, the hepatocyte hypertrophy was distributed within the centrilobular to panlobular regions. In severe cases, the centrilobular hepatocytes contained intensely eosinophilic intracytoplasmic granular material. In less severe cases, these cytoplasmic tinctorial differences were not apparent.
Kidney: There were significant increases in the incidences of cytoplasmic alteration in renal tubules of male mice exposed to 2,500, 5,000, 10,000, and 20,000 ppm (Table 34). Control male mice contained multiple, irregular, cytoplasmic vacuoles within the cortical renal tubular epithelial cells, whereas control female mice did not contain these vacuoles. Cytoplasmic alteration within the renal tubules of male mice was characterized by lack of cytoplasmic vacuoles in the tubular epithelial cells in the cortex.
Exposure Concentration Selection Rationale for Two-year Study in Mice
For the 3-month exposures in mice, there was no effect at 20,000 ppm on survival or clinical signs of toxicity. However, male mice in the 10,000 and 20,000 ppm groups had lower mean body weights compared to control males, which exhibited >20% difference, whereas the female mean body weight was 15% lower in the 20,000 ppm group compared to that of the respective control group. Histopathological evidence in the liver suggested that male mice displayed a slightly higher severity than female mice to TCPP exposure. Informed by these data, 0, 1,250, 2,500, and 5,000 ppm TCPP were selected for male mice and 0, 2,500, 5,000, and 10,000 ppm TCPP were chosen for female mice in the 2-year feed study.
Two-year Study
Survival of male and female mice was not adversely affected by exposure to TCPP (Table 35; Figure 10), and there were no exposure-related clinical observations noted during study (Appendix H).
Exposure concentration-related decreases in mean body weights relative to the control groups were observed in both male and female mice, although the response was greater in females. Mean body weights of male mice in the 1,250 and 2,500 ppm groups remained within 10% of the control group value over the 2-year exposure period (Table 36; Figure 11). The 5,000 ppm male mice maintained mean body weights within 10% of the control group value for approximately the first 2 months of exposure. After this time, mean body weights fluctuated between 12% and 21% of the control group value and the mean body weights of this group were 18% lower than that of the control group at study termination. Lower, exposure concentration-dependent group mean body weight gains were apparent for female mice in all TCPP-exposed groups. At study termination, mean body weights were 87%, 74%, and 62% of the control group in the 2,500, 5,000, and 10,000 ppm TCPP-exposed groups, respectively (Table 37; Figure 11). The lower mean body weights observed in mice were interpreted as the result of lower body weight gains rather than body weight loss during the exposure period. Despite the exposed female mice having lower weight gain compared to the control group, these animals displayed no adverse clinical observations or notable decreases in feed consumption (Appendix H).
Feed consumption by all TCPP-exposed male and female mice was comparable (within 2%–6%) with that of control animals at study termination (week 102; Table 38, Table 39; Appendix H). TCPP intake, based on weekly averaging (Appendix H), was estimated to be 160, 330, and 711 mg/kg/day for male mice in the 1,250, 2,500, and 5,000 ppm groups, respectively. For female mice, TCPP intake, based on weekly averaging (Appendix H), was estimated to be 329, 673, and 1,491 mg/kg/day in the 2,500, 5,000, and 10,000 ppm groups, respectively. Table 38 and Table 39 highlight feed consumption (i.e., feed [g/day]) and estimated TCPP intake (i.e., dose [mg/kg/day]) at select time points. Similar to rats, the estimated TCPP intake was proportional to the twofold increase in TCPP concentration in dosed feed for both male and female mice. Moreover, the doses of TCPP consumed by males relative to females, in mg/kg/day, were similar for a given concentration of TCPP-exposed feed.
Histopathology
This section describes the statistically significant or biologically noteworthy changes in the incidence of neoplasms and/or nonneoplastic lesions of the liver, Harderian gland, kidney, lymph node, and spleen in mice.
Liver: In male mice, there was a significant increase in the incidences of hepatocellular carcinomas in all exposed groups compared to the control group, whereas the incidences of hepatocellular adenomas remained similar across all groups including the control group (Table 40). In addition, there were slight increases in the incidence of tumor multiplicities in female mice for hepatocellular adenomas at 5,000 and 10,000 ppm, hepatocellular carcinomas at 2,500, 5,000, and 10,000 ppm, and in male mice for hepatocellular carcinomas at 1,250, 2,500, and 5,000 ppm. In male mice, there was one hepatoblastoma in the control group and one in the 5,000 ppm group. In addition, there was one hepatocholangiocarcinoma in each of the 2,500 and 5,000 ppm male groups. The incidences of nonneoplastic lesions, such as altered hepatic foci (basophilic and eosinophilic), were similar in all groups of male mice, including the control group. In female mice, there was a positive trend and significant increase at 10,000 ppm in the incidences of hepatocellular adenomas and carcinomas when compared to the control group. The incidences of nonneoplastic lesions, such as eosinophilic foci, were significantly increased in an exposure concentration-dependent manner in all exposed female mice. In addition, the 10,000 ppm group exhibited a significant increase in cytoplasmic alteration within the hepatocytes. Interestingly, this change was negligible to absent in the lower TCPP-exposed groups (Table 40).
Microscopically, hepatocellular adenomas were well-circumscribed expansile masses comprising irregular plates of hepatocyte cords with eosinophilic to basophilic cytoplasm, and occasionally vacuolated cytoplasm, with minimal to mild compression of the adjacent parenchyma. Portal triads were generally absent but occasionally were entrapped in these neoplasm masses. Hepatocellular carcinomas were large and invasive and were composed of nodules of hepatocytes arranged in solid, glandular, or trabecular patterns (Figure 12). These neoplasms in both male and female mice were also associated with increased cellular atypia accompanied by a few mitoses. Occasionally, there were areas of necrosis and hemorrhage within these masses. The hepatoblastomas are characterized by dark, basophilic masses usually accompanied by either hepatocellular adenomas or hepatocellular carcinomas. These neoplasms were composed of primitive hepatic progenitor cells, which are elongated with scant eosinophilic cytoplasm and fusiform darkly basophilic nucleoli. The hepatocholangiocarcinomas were characterized by neoplastic hepatocytes intermixed with neoplastic biliary profiles, which invade the adjacent hepatic parenchyma and can occasionally metastasize to distant organs. Hepatic foci are tinctorially discrete areas of hepatocytes that blend imperceptibly into the surrounding hepatic parenchyma with no evidence of compression or invasion (Figure 13). The cytoplasmic alterations observed in the 20,000 ppm female mice were characterized by bright eosinophilic granular cytoplasm within the hepatocytes and lack of ground glass (glycogen) cytoplasm (Figure 14). The cytoplasm of hepatocytes from control rodents with access to feed and water ad libitum are filled with glycogen and have a ground glass appearance with clear spaces (an artifact due to tissue processing) (Figure 15).
Harderian Gland: In addition to the neoplastic lesions discussed above, increases in the incidence of neoplastic lesions in the Harderian gland of male mice were observed. In male mice, there was a significant increase in the incidence of Harderian gland adenomas in the 2,500 ppm group along with higher, but nonsignificant incidences in the other exposed groups compared to the control group (Appendix H). Incidences of Harderian gland neoplasms (adenoma or carcinoma) were found in all groups of male mice, both exposed and control; however, there were no significant differences when these neoplasms were combined. There were incidences of hyperplasia within the Harderian gland in all exposed and control male groups (Appendix H). There were no increases of Harderian gland neoplasms in female mice exposed to TCPP compared to the control group (Appendix H).
Harderian gland carcinomas were characterized by neoplastic cuboidal to columnar acinar cells arranged in tubular, acinar, or solid areas, often invading the adjacent glandular epithelium. The tumor cells had varying degrees of cellular atypia and mitoses. Harderian gland adenomas were composed of neoplastic acinar epithelial cells arranged in a papillary, cystic, acinar, or combination of these morphologies and formed well demarcated nodules that compressed the surrounding glandular tissue. These neoplastic cells were usually tall and columnar with finely stippled/vacuolated eosinophilic cytoplasm and had occasional loss of nuclear polarity and atypia. Harderian gland hyperplasia was characterized by proliferative glandular epithelial cells with retained architecture of acini and did not compress or distort the adjacent glandular tissue.
In addition to the neoplasms discussed above, there were increased incidences of some nonneoplastic lesions, including cytoplasmic alteration of renal tubules in male mice, hyperplasia of lymphocytes within the mandibular lymph node in female mice, and extramedullary hematopoiesis within the spleen of female mice (Appendix H).
Kidney: There were significant increases in the incidences of cytoplasmic alteration in all exposed male mice in an exposure concentration-dependent manner (Table 41). These changes were significant by trend analysis and pairwise tests. Cytoplasmic alteration in the renal tubules of male mice was characterized by the loss of cytoplasmic vacuoles within the normal proximal renal tubules, and the resultant tubules appeared similar to that of female renal tubules, which did not have these intracytoplasmic vacuoles (Figure 16, Figure 17).
Lymph Node: For lymphocyte hyperplasia within the mandibular lymph node, there was a positive trend as well as a significantly increased incidence by pairwise test in the 10,000 ppm female mice. Lymphocyte hyperplasia was characterized by increased numbers of lymphocytes that were mature, small, and present in their respective compartments (follicular, parafollicular, or medullary cords, and intravascular); the architecture of the lymph node was generally preserved, and all the compartments were apparent within the section. These increased numbers of lymphocytes may represent an immune response. This lesion was not present in male mice (Appendix H).
Spleen: There was a significant increase in the incidence of extramedullary hematopoiesis within the spleen in the 5,000 ppm female mice. This lesion was characterized by an increase in a mixture of progenitor blood cells belonging to myeloid, erythroid, and megakaryocytic lineages. Extramedullary hematopoiesis is a normal physiological response seen in the spleen of all mice, but an increase above a certain threshold compared to the control group merits a diagnosis. In some cases, the observed increases might have been related to neoplasia or chronic toxicity, but no definitive associations could be made in this study. An increase in the incidence of this lesion was not observed in male mice (Appendix H).
Genetic Toxicology
Data from all NTP genetic toxicity tests with TCPP are available in the NTP Chemical Effects in Biological Systems database: https://doi.org/10.22427/NTP-DATA-TR-602.96
Bacterial Mutation Studies
TCPP did not induce mutations, with or without the addition of exogenous metabolic activation enzymes supplied by induced rat or hamster liver S9 in various concentrations, in any of several strains of bacteria (Salmonella typhimurium and Escherichia coli), in two bacterial mutagenicity studies (Appendix D; Table D-1, Table D-2). In the first study, the highest dose tested was limited by toxicity to 1,000 µg/plate; in the second study, although toxicity was again observed in some trials at 1,000 µg/plate, higher doses (ranging up to 6,000 µg/plate) could be tested, particularly in the E. coli strain (Appendix D).
In Vivo Peripheral Blood Micronucleus Test
Micronuclei are biomarkers of chromosomal changes, either in chromosome number or structure (breaks). In the in vivo peripheral blood micronucleus test, TCPP exposure for 3 months via dosed feed did not result in an increase in micronucleated immature erythrocytes (polychromatic erythrocytes [PCEs]) in male or female Sprague Dawley rats (Table D-3). However, the percentage of PCEs among total erythrocytes was increased in an exposure concentration-related manner in both sexes, suggesting a stimulation of erythropoiesis in the rats exposed to TCPP. In female B6C3F1/N mice, exposure to TCPP for 3 months via dosed feed did not result in an increase in micronucleated PCEs or mature erythrocytes (normochromatic erythrocytes [NCEs]; Table D-4). In male mice, the values for micronucleated red blood cells were within the laboratory historical control 95% confidence interval and the absolute increase in micronucleated NCEs and PCEs was quite small, amounting to an increase over control values of approximately 17% or 23%, respectively. Due to the questionable biological significance of the observed increase in micronucleated cells, the results in male mice were judged to be equivocal (Table D-4). In both male and female mice, the percentage of PCEs among total erythrocytes was increased in an exposure concentration-related manner, similar to what was observed in the rat samples, suggesting that exposure to TCPP also produced a stimulation of erythropoiesis in mice.
Discussion
Tris(chloropropyl) phosphate (TCPP) is a high-production flame-retardant isomeric mixture used in textiles, furniture (flexible polyurethane foam), construction materials (rigid polyurethane foam), electronic products, paints, coatings, and adhesives.98 TCPP has been proposed as a substitute for brominated flame retardants and as a replacement for other chlorinated flame retardants, such as tris(2-chloroethyl) phosphate (TCEP) and tris(1,3-dichloro-2-propyl) phosphate (TDCPP), which have been identified as carcinogenic32,99,100 and which also pose developmental, reproductive, and neurotoxicity risks.29,32,101 Given the potential for increased use and exposure and the lack of publicly available toxicity data, the U.S. Consumer Product Safety Commission (CPSC) nominated TCPP for toxicological testing by the National Toxicology Program (NTP). The purpose of this report is to summarize any hazards associated with subchronic (3-month) and chronic (2-year) TCPP exposure in rats and mice so that others may compare and contrast toxicity profiles of TCPP exposure with other structurally similar flame retardants. For these NTP studies, a commercial TCPP product containing four isomers commonly found in other commercial mixtures of TCPP was procured, characterized, and utilized. These four isomers included tris(1-chloro-2-propyl) phosphate (TCIPP; CASRN 13674-84-5), bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (CASRN 76025-08-6), bis(2-chloropropyl) 2-chloroisopropyl phosphate (CASRN 76649-15-5), and tris(2-chloropropyl) phosphate (CASRN 6145-73-9).
The NTP research program on TCPP studied subchronic toxicity in male and female rats and mice exposed via dosed feed for 3 months at 0, 1,250 (mice only), 2,500, 5,000, 10,000, 20,000, or 40,000 (rats only) ppm. In rats, perinatal exposure from gestation day (GD) 6 through postnatal day (PND) 21 (weaning) preceded the subchronic exposure to provide insight to TCPP toxicity following early-life exposures. Feed exposure to TCPP was chosen to mimic intermittent ingestion, which is a common exposure route for humans.102,103 Literature available for exposure concentration selection in rodents was primarily from industry summaries. Reports indicated that acute toxicity of TCPP was low4 and feed studies with adult CD Sprague Dawley rats suggested that TCPP was well tolerated at dietary concentrations of 800–20,000 ppm after 13-weeks of exposure.4,12,33 Given the limited knowledge of TCPP effects on rodents during perinatal exposure and that a different rat strain was to be used, the exposure concentration selection was based on the reported lowest-observed-adverse-effect levels (LOAELs) and maximal tolerated doses. The NTP research program on TCPP also included palatability studies in adult rats and mice to better estimate acceptable exposure concentrations.
In the perinatal portion of the 3-month study, pregnant rats exposed to 40,000 ppm were euthanized humanely due to overt toxicity early in gestation. TCPP exposure did not adversely affect the survival of dams exposed to ≤20,000 ppm in feed, and signs of clinical toxicity were not observed in these animals throughout gestation or lactation. Although decreases in dam mean body weight and feed consumption appear to be related to TCPP exposure concentration, the overall effects were minimal with the majority of mean values for these parameters staying within 10%–20% of control values during the perinatal phase of TCPP exposure. TCPP exposure also had no toxicologically significant effects on littering parameters, such as percent dam delivery, litter size, or sex ratio, suggesting that pregnant rats tolerated TCPP exposures in feed at concentrations ≤20,000 ppm. These outcomes are similar to an industry summary report of a two-generation reproductive study. In this experiment, F0 male and female Wistar rats had lower body weights and feed consumption when exposed to approximately 300 and 1,000 mg TCPP/ kg body weight/day (mg/kg/day; through feed). At these doses, no effects were observed on littering parameters or reproductive function.4 In the current NTP subchronic study, TCPP-exposed offspring survived through lactation with no signs of clinical toxicity in either sex. However, offspring in the 20,000 ppm TCPP group did exhibit a time-dependent decrease in body weight gain from PND 4 through PND 21. At weaning, male and female offspring weighed approximately 30% less than control animals, whereas body weights were unaffected in groups exposed to lower TCPP concentrations. After weaning, male rats in the 20,000 ppm group were euthanized moribund by study day 5 (i.e., PND 26) of the 3-month study, while the female rats in this exposure group were kept on study. This observation informed NTP’s determination that PND 21 was potentially too early for weaning of Sprague Dawley rat offspring and the subsequent decision to extend the weaning period to PND 28 for the 2-year study to reduce stress on offspring and to increase survival.
For the remainder of the 3-month study, male and female rats survived and displayed no signs of clinical toxicity. Although female rats exposed to 20,000 ppm TCPP had mean body weights approximately 30% lower than those of control rats at the beginning of the subchronic study, this gap did not persist, and mean body weights were within 12% of control animals by study termination. Lower mean body weights in this group corresponded with a similar decrease in feed consumption. Male and female rats exposed to ≤10,000 ppm TCPP had mean body weights and feed consumption similar to those of control animals throughout the subchronic exposure period.
At the end of the 3-month study, TCPP exposure resulted in various perturbations to the liver of male and female rats. Absolute and relative liver weights were significantly increased at exposure concentrations of 10,000 (males only) and 20,000 ppm TCPP (females only); however, the magnitude of the increase was small and ranged from 10% to 20% for absolute liver weights. Minimal bile duct hyperplasia was observed in the top exposure groups (≥10,000 ppm) of both sexes and similar minimal changes were also observed in the 2-year study. Finally, several liver-related serum clinical chemistry parameters (alkaline phosphatase [ALP], alanine aminotransferase [ALT], and cholesterol) were altered in an exposure concentration-related manner for both sexes. Both ALT and ALP activities were significantly decreased. The mechanism for the decreased ALT and ALP activities was unknown but may have indicated decreased hepatocellular enzyme production or release. In particular, decreases in ALT have been associated with perturbations of gluconeogenesis or administration of substances that inhibit the cofactor pyridoxal phosphate, whereas decreases in feed consumption can cause decreases in ALP (i.e., the ALP intestinal isoenzyme).104,105 Although these liver enzyme changes were considered biologically relevant, there is no known toxicological significance of decreases in serum liver enzyme activity and the causes of these decreases are unknown. The mild but significant increases in cholesterol concentrations suggest an alteration in lipid metabolism. The liver has been shown to be a target organ of TCPP in previous studies, including those conducted as part of the NTP research program. In the NTP study investigating the effects of TCPP on prenatal developmental toxicity in Sprague Dawley rats, dose-related increases in absolute liver weight (9%, 16%, and 26% at 162.5, 325, and 650 mg/kg/day, respectively) were measured in dams exposed to TCPP by oral gavage from GD 6 to 20.35 In a different NTP study focusing on transcriptomic responses to chemical exposure, male Sprague Dawley rats that were exposed to TCPP by oral gavage (18–2,000 mg/kg/day) for 5 days had significantly increased (approximately 33%) absolute and relative liver weights in a dose-related manner.106 The liver is also reported to be affected by TCPP exposure in various industry summary reports. For example, in the previously mentioned studies in Sprague Dawley (CD-1) rats, an increase in liver weights occurred in the absence of histopathological changes after 14 days of repeat exposure to TCPP (4,200–16,600 ppm in feed).4,6 In a separate study with CD Sprague Dawley rats exposed to TCPP for 13 weeks in feed, absolute and relative liver weights were significantly increased in all male (800–20,000 ppm TCPP) and female (7,500–20,000 ppm TCPP) rats. Corresponding mild periportal hepatocellular swelling was noted in some animals at 20,000 ppm; no changes in liver histopathology were seen at other concentrations.4,12,33
An unusual finding in the current 3-month rat study of TCPP was an exposure concentration-related increase in absolute and relative thymus weight and size of both sexes. This increase in the thymic weights in TCPP-exposed animals correlated histologically with enlarged thymic cortex and medulla in the exposed rats compared to the control animals. The process of thymus involution in rats begins soon after birth with a gradual decrease in mitoses and an increase in apoptosis.107 The rate of thymic involution increases as the rats become sexually mature and increased apoptosis in the thymic cortex leads to decreased thickness. Morphometry on rat thymic tissues in this study revealed that the exposed animals exhibited enlarged cortical thickness in an exposure-dependent manner compared to control animals. Surprisingly, there were no corresponding increases in the lymphocyte populations in other lymphoid organs at the end of the subchronic exposure, and there were no lesions in the lymphoid organs at the end of the 2-year exposure. Overall, these data suggest that the increased thymic weights compared to the control animals at 3 months were likely due to a delay in thymus involution rather than due to proliferation of thymic lymphocytes. These delays in thymic involution due to perinatal chemical exposures have not been reported in the literature. To learn more, a second study was initiated to investigate potential immunological consequences of perinatal exposure to TCPP.1
In the 3-month TCPP studies with mice, mortality and clinical signs of toxicity were not observed in males or females exposed to 1,250, 2,500, 5,000, 10,000, or 20,000 ppm TCPP. TCPP-exposed male mice gained less weight compared to the control group in an exposure concentration-related manner. Female mice also gained less weight compared to the control group, but this response was not exposure concentration dependent. By study termination, male and female mice in the 20,000 ppm TCPP group had mean body weights that were 29% (males) and 15% (females) lower than the respective control groups. Despite lower mean body weights, feed consumption remained within 10% of control animals for the duration of the study.
Similar to rats, TCPP exposure was associated with changes in the liver of mice. Absolute liver weight was significantly increased by 39% and 25% in male and female mice in the 20,000 ppm TCPP group. Relative liver weights were also significantly increased in male and female mice exposed to ≥2,500 ppm TCPP. Liver weight increases were associated with a statistically significant increase in the incidences of hepatocellular hypertrophy in male and female mice exposed to ≥5,000 ppm TCPP. As discussed earlier in this report, significantly increased absolute and relative liver weights were also noted in rats of both sexes exposed to TCPP, but no histological correlate was noted. Typically, increased liver weights with or without corresponding hepatocellular hypertrophy are considered an adaptive change due to cytochrome P450 (CYP) enzyme induction resulting from exposure to various nuclear receptor activators. TCPP appears to be a weak activator of constitutive androstane receptor (CAR), pregnane X receptor (PXR), and peroxisome proliferator-activated receptor alpha (PPARα) in various in vitro assays as well as in a 5-day rat liver toxicogenomic study (Appendix E, Appendix F). In addition, TCPP has been shown to activate CYP enzymes at the gene expression level and protein levels across several species, including the rat, chicken, and fish model systems.106,108-110 Repeated chronic exposures to these nuclear receptor activators may result in the progression of hypertrophy to hyperplasia and potentially to preneoplasia and neoplasia; although this progression was not observed in rats, hypertrophy was observed in mice.
Subchronic exposure to TCPP also resulted in a decrease of absolute kidney weights in an exposure concentration-dependent manner, with the mice exposed at 20,000 ppm showing significant decreases relative to the control mice with a 22% decrease for males and a 12% decrease for females. There was a positive trend for relative kidney weights in male TCPP-exposed groups, but the response was not statistically significant by pairwise comparison. A histopathological correlate to the changes in kidney weights was not observed. However, in male mice exposed to 2,500, 5,000, 10,000, or 20,000 ppm, there was a significant increase in the incidences of cytoplasmic alteration in the outer cortical renal tubular epithelium. Cytoplasmic alteration in the renal tubular epithelium of male mice is defined as the loss of cytoplasmic vacuoles (autophagic vacuoles, lysosomes); normal female renal tubular epithelium does not exhibit similar vacuoles, suggesting that it is a sexually dimorphic feature. The toxicologic significance of this lesion is not clearly understood. Further studies are needed to better understand the toxicological significance of cytoplasmic alterations in male renal tubular epithelium.
Despite some unique differences in toxicity across rats and mice, the subchronic studies suggest that the liver is the likely target of TCPP exposure. Minimal sex differences were observed in toxicological endpoints for rats and mice. Delayed thymic involution in rat offspring may have been related to perinatal exposure to TCPP; however, more studies are needed to determine the toxicological significance of this finding. Overall, these data aided in the design and selection of exposure concentrations for the chronic studies in rats and mice.
TCPP was well tolerated in rats exposed through feed for 2 years. Maternal endpoints evaluated in time-mated rats exposed to 2,500–20,000 ppm TCPP were similar to those observed in the subchronic studies. As anticipated, there were no toxicologically significant exposure concentration-related effects on maternal survival, clinical observations, or reproductive performance. Mean body weights and feed consumption by dams during the perinatal period were similar to those of the subchronic study perinatal period. For dams, mean body weights and body weight gains fluctuated during the gestation and lactational periods, but the overall change (GD 6–21 and lactation day [LD] 1–28) in TCPP-exposed groups was consistent with control animals. Likewise, the dam feed consumption and estimated chemical intake was similar among exposure groups in both the subchronic and chronic studies of TCPP. Rat offspring in the chronic study experienced a similar pattern of lower mean body weights throughout lactation (up to 31%). The extension of the weaning time from PND 21 to PND 28 for the chronic study likely prevented premature mortality of male offspring in the 20,000 ppm group. As a result, there were no toxicologically significant effects on survival, clinical observations, feed consumption, or mean body weight for either sex by the end of the 2-year exposure period.
Lower concentrations of TCPP were used in the 2-year study of mice (i.e., males = 1,250, 2,500, and 5,000 ppm and females = 2,500, 5,000, and 10,000 ppm). Survival was not affected, and there were no clinical signs of toxicity, suggesting TCPP was well tolerated at these exposure concentrations. Given the subchronic study results, mean body weights of mice were anticipated to be ≤10% lower than that of control mice following chronic exposure. This pattern held true until approximately 2–3 months on study. Subsequently, the difference in mean body weights of male and female mice exposed to TCPP grew larger over time. By study termination, male mice in the highest exposure group of 5,000 ppm TCPP weighed 18% less, on average, than the control group. Interestingly, mean body weights of females at study termination were lower than that of the control group by 13%, 26%, and 38% in the 2,500, 5,000, and 10,000 ppm TCPP-exposed groups, respectively. Although unexpected, female mice showed no signs of clinical toxicity or significant decrease in feed consumption.
Carcinogenicity was observed in the liver of male rats and male and female mice after a 2-year oral exposure to TCPP, although the magnitude of response was greater in mice compared to rats. The relationship of the marginal response in female rats to TCPP exposure was considered uncertain. Compared to B6C3F1/N mice, incidences of hepatocellular neoplasms are rare in Sprague Dawley rats.111 In male rats, a positive trend (p = 0.013) in the combined incidences of hepatocellular adenomas and carcinomas was observed. The incidence of these neoplasms in the exposed groups was not statistically significant by pairwise comparison to the control group, but the incidences in the 10,000 and 20,000 ppm groups did exceed the historical control ranges (in feed studies or by all routes of exposures) for these neoplasms in male rats. On the basis of the collective study data, it was determined that there was some evidence of carcinogenicity in male rats following chronic exposure to TCPP. Additionally, a statistically significant exposure concentration-related increase in the incidences of various potential preneoplastic lesions, such as hepatic foci, was observed in male rats. The neoplasm response in the liver of female rats was not as robust. Hepatocellular carcinomas were not observed in female rats, but there were slightly higher incidences of hepatocellular adenomas in the lowest and highest exposure groups and two females in the 20,000 ppm group had multiple adenomas. The adenoma increases in female rats did not reach statistical significance for either the trend or pairwise comparisons and were within historical control ranges (in feed studies or by all routes of exposures). Similar to males, there were statistically significant exposure concentration-related increases in the incidences of various potential preneoplastic lesions, such as hepatic foci, in female rats. Having compared the strength of evidence in male rats and male and female mice, it was determined that the carcinogenic response in female rat livers may have been related to exposure.
In male mice, the incidences of hepatocellular carcinomas were significantly increased by pairwise comparison to the control group in all TCPP-exposed groups, although the incidence rates in TCPP groups were within the historical control range. It is noteworthy that the incidence of hepatocellular carcinoma in control male mice in this study was at the lower end of NTP’s historical range for this neoplasm (i.e., 10%–20% for feed studies; 10%–36% by all exposure routes). These data demonstrate that TCPP elicited some evidence of carcinogenic activity in the liver of male mice following 2 years of exposure.
Female mice exhibited a positive trend in the incidences of hepatocellular adenomas, carcinomas, and adenomas or carcinomas (combined) with the incidence in the highest exposure group of 10,000 ppm significantly different from the control group by pairwise test. Additionally, the incidence of hepatocellular adenomas, carcinomas, and adenomas or carcinomas (combined) in female mice exposed to either 5,000 or 10,000 ppm was higher than historical control values in other feed studies. There was a statistically significant increase in the incidences of potential preneoplastic changes such as hepatic foci, which was not observed in male mice. This study demonstrated that TCPP had clear evidence of carcinogenic activity in female mice based on the increased incidence of liver neoplasms.
Additional neoplasms of toxicological significance were noted in female rats. A positive trend was observed in the incidences of uterine adenoma or adenocarcinoma (combined). Although the incidence for these combined neoplasms in TCPP-exposed female rats was not statistically significant by pairwise comparison to concurrent study control animals, there was an exposure-related doubling and tripling of the incidence. In addition, the combined incidence of uterine adenoma or adenocarcinoma did exceed historical control ranges by feed or all exposure routes in the 5,000, 10,000, and 20,000 ppm groups. These data suggest that TCPP contributed to the formation of rat uterine lesions in this study and represent some evidence of carcinogenic activity. Furthermore, solitary incidences of squamous cell carcinoma in the 10,000 and 20,000 ppm groups as well as the higher incidences of potential preneoplastic lesions, such as atypical endometrial hyperplasia, support the observation of the uterus being a neoplasm target organ after chronic TCPP exposure. In general, proliferative lesions in the uterus are more common in the Hsd:Sprague Dawley SD rats than in mice. Not surprisingly, the uterus was not a target organ in the mouse study. In summary, the toxicological or carcinogenic mode of action needs further mechanistic evaluation to completely understand the potential for endocrine disruption due to TCPP exposure.
The mechanisms for the observed carcinogenic response in the liver in these studies are not well understood. The findings from NTP’s bacterial mutagenicity studies and in vivo micronucleus assessments, as well as reports in the literature suggest that TCPP has little or no genotoxic potential, and therefore the development of neoplasms is likely due to a nongenotoxic mode of action. To learn more about potential mechanisms of TCPP toxicity and carcinogenicity, data were integrated from three separate investigations. First, the biological activity of TCPP measured in various Toxicology in the 21st Century (Tox21) high-throughput screening assays suggest it is a weak activator of the constitutive androstane receptor (CAR) and pregnane X receptor (PXR) compared to the prototype CAR and/or PXR activators (Appendix E). In addition, an NTP toxicogenomic study also suggests that TCPP activates several biomarker hepatic transcripts related to CAR, PXR, and peroxisome proliferator-activated receptor alpha (PPARα) signaling pathways following a 5-day exposure in rats (Appendix F). A third data stream comes from a multiomics examination of male mouse hepatocellular carcinomas arising spontaneously or following chronic exposure to TCPP (i.e., tumor analysis from this 2-year study). This investigation indicated no significant differences in the overall mutation burden in animals developing hepatocellular carcinomas spontaneously or after chronic exposure to TCPP, supporting the hypothesis that TCPP may not function through a genotoxic mode of action. The mutation signatures of mouse hepatocellular carcinomas arising spontaneously or due to chronic exposure to TCPP are comparable, but the TCPP-exposed group contained additional mutation spectra and cancer driver genes, suggesting that TCPP is likely associated with a nongenotoxic mechanism and may function as a tumor promoter (Appendix G). Future mechanistic evaluations regarding the role of specific TCPP isomers or metabolites may provide additional clarity to the tumor promotor hypothesis.
To provide additional context to the toxicological data, an assessment of internal exposure in rats and mice at 3, 6, 12, and 18 months was included in the NTP chronic study. The major isomer of TCPP, tris(1-chloro-2-propyl) phosphate (TCIPP), was quantified in plasma. The data are published in Collins et al.,56 and are briefly summarized here. In male and female rats exposed to 2,500–20,000 ppm TCPP, TCIPP plasma concentrations ranged from 3.43 to 78.4 ng/mL and increased with exposure concentration at all time points but was not often in proportion with exposure concentration. TCIPP concentrations were variable within and between time points for a given exposure concentration, which made it difficult to discern whether there was sex difference in rats. TCIPP concentrations in mouse plasma were somewhat higher than in rats and ranged from 6.6 to 1,180 ng/mL when exposed to 1,250–5,000 ppm (males) or 2,500–10,000 ppm (females). At a similar exposure concentration of TCPP in feed (i.e., 5,000 ppm), the estimated TCPP intake was approximately 250–800 mg/kg/day by rats versus approximately 575–1,500 mg/kg/day by mice. The estimated intake of TCPP, based on feed consumption data, may partly explain why plasma TCIPP levels were up to three times higher in mice. Moreover, higher internal exposure in mice, compared to rats, may help explain or distinguish the evidence of carcinogenicity observed in mice on this study. Similar to rats, TCIPP concentrations in mice increased with exposure concentration at all time points, but this increase was not always in proportion with exposure concentration. Sex differences were noted in mice, with males having consistently higher plasma concentrations than females. This difference may result from slightly higher feed consumption and estimated TCPP uptake in male mice compared to females. TCPP did not bioaccumulate in rats or mice over the course of the study. This finding is consistent with other reports demonstrating that TCPP and other chlorinated organophosphate flame retardants exhibit lower bioaccumulation or bioconcentration compared with other classes of flame retardants.102,112 It is noteworthy that low concentrations of TCIPP were observed in some rat and mouse plasma from control groups. Sample preparation and analysis were attributed as the source of these low concentrations because of the ubiquitous presence of TCPP, which was determined by measuring a metabolite of TCPP—bis(2-chloroisopropyl) 1-carboxyethyl phosphate (BCPCP) —in plasma from limited control and TCPP-exposed animals and utilizing the ratio of BCPCP:TCIPP.56 Overall, internal concentrations of TCIPP were estimated for comparison to TCPP exposure concentrations that were associated with an increase in the incidence of neoplasms in rats and mice.
The measurement of TCPP isomers or their metabolites in biological matrices is not often reported in a consistent manner, which complicates attempts to associate human exposure with rodent exposure levels in toxicity studies. Plasma TCIPP concentrations were not available for humans and hence using the rodent TCIPP data to provide context for human exposures is not feasible at this time. It is clear that more consistent internal exposure data are required to assess the relevance of animal toxicity data to human exposures. Because TCPP is cleared rapidly and metabolized extensively to BCPCP, BCPCP might be a more suitable biomarker of TCPP exposure for consideration in biomonitoring studies. Future studies are suggested to further characterize this possibility. Despite this limitation, these chronic toxicological assessments in rats and mice are expected to assist with several national and international efforts to evaluate the comparative toxicity of TCPP and structurally similar flame retardants TCEP and TDCPP. Organizations such as CPSC, the European Chemicals Agency, and Health Canada have published guidance for or noted their intent to restrict the use of this class of chemicals on the basis of their toxicity profiles.11,29,32,113
References
1. National Toxicology Program (NTP). Testing status of tris(Chloropropyl) phosphate (TCPP) M20263. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2016. https://ntp.niehs.nih.gov/go/ts-m20263
2. U.S. Environmental Protection Agency (USEPA). CompTox Chemicals Dashboard. Washington, DC: U.S. Environmental Protection Agency; 2021. https://comptox.epa.gov/dashboard
3. U.S. Environmental Protection Agency (USEPA). TSCA work plan chemical problem formulation and initial assessment: Chlorinated phosphate ester cluster flame retardants. Washington, DC: U.S. Environmental Protection Agency, Office of Chemical Safety and Pollution Prevention, Office of Pollution Prevention and Toxics; 2015. EPA Document No. EPA-740-R1-5001. https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=P100N3WF.txt
4. U.S. Environmental Protection Agency (USEPA). Flame retardants used in flexible polyurethane foam: An alternatives assessment update. Washington, DC: U.S. Environmental Protection Agency; 2015. EPA Document No. EPA-744-R-15-002. https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=P100MXUQ.txt
5. National Research Council (NRC). Toxicological risks of selected flame-retardant chemicals. Washington, DC: National Academies Press; 2000. https://www.ncbi.nlm.nih.gov/books/NBK225647/
6. European Union (EU). Tris(2 chloro-1 methylethyl) phosphate (TCPP), (CAS No. 13674-84-5, EINECS No. 237-158-7) risk assessment. Luxembourg: Office for Official Publications of the European Communities; 2008.
7. Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological profile for phosphate ester flame retardants. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry; 2012. https://www.atsdr.cdc.gov/toxprofiles/tp202.pdf
8. Wilczynski SL, Killinger JM, Zwicker GM, Freudenthal RI. Fyrol FR-2 fertility study in male rabbits. [abstract]. Toxicologist. 1983; 3(1):22.
9. World Health Organization (WHO). Flame retardants: Tris(chloropropyl) phosphate and tris(2-chloroethyl) phosphate. Geneva, Switzerland: World Health Organization; 1998. Environmental Health Criteria 209. https://apps.who.int/iris/handle/10665/42148
10. U.S. Environmental Protection Agency (USEPA). Substance Registry Service (SRS). Washington, DC: U.S. Environmental Protection Agency; 2016. https://sor.epa.gov/sor_internet/registry/substreg/LandingPage.do
11. Health Canada. Updated draft screening assessment: Certain organic flame retardants substance grouping. 2-Propanol, 1-chloro-, phosphate (3:1) (TCPP) chemical abstracts service registry number 13674-84-5, 2-propanol, 1,3-dichloro-, phosphate (3:1) (TDCPP) chemical abstracts service registry number 13674-87-8. Ottowa, ON: Environment and Climate Change Canada, Health Canada; 2020.
12. Organisation for Economic Co-operation and Development (OECD). Screening Information Dataset (SIDS) initial assessment profile: Tris(1-chloro-2-propyl)phosphate, CAS No: 13674-84-5. Geneva, Switzerland: UNEP Chemicals; 2000.
13. U.S. Environmental Protection Agency (USEPA). Reducing your child’s exposure to flame retardant chemicals. Washington, DC: U.S. Environmental Protection Agency; 2016. EPA Document No. EPA-740-16-001. https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/reducing-your-childs-exposure-flame-retardant-chemicals
14. Van den Eede N, Heffernan AL, Aylward LL, Hobson P, Neels H, Mueller JF, Covaci A. Age as a determinant of phosphate flame retardant exposure of the Australian population and identification of novel urinary PFR metabolites. Environ Int. 2015; 74:1-8. DOI: 10.1016/j.envint.2014.09.005 PubMed: 25277340
15. Van den Eede N, Maho W, Erratico C, Neels H, Covaci A. First insights in the metabolism of phosphate flame retardants and plasticizers using human liver fractions. Toxicol Lett. 2013; 223(1):9-15. DOI: 10.1016/j.toxlet.2013.08.012 PubMed: 23994729
16. Estill CF, Slone J, Mayer AC, Phillips K, Lu J, Chen IC, Christianson A, Streicher R, Guardia MJ, Jayatilaka N, et al. Assessment of spray polyurethane foam worker exposure to organophosphate flame retardants through measures in air, hand wipes, and urine. J Occup Environ Hyg. 2019; 16(7):477-488. DOI: 10.1080/15459624.2019.1609004 PubMed: 31112485
17. Gibson EA, Stapleton HM, Calero L, Holmes D, Burke K, Martinez R, Cortes B, Nematollahi A, Evans D, Herbstman JB. Flame retardant exposure assessment: Findings from a behavioral intervention study. J Expo Sci Environ Epidemiol. 2019; 29(1):33-48. DOI: 10.1038/s41370-018-0049-6 PubMed: 29950671
18. Ospina M, Jayatilaka NK, Wong LY, Restrepo P, Calafat AM. Exposure to organophosphate flame retardant chemicals in the U.S. general population: Data from the 2013-2014 National Health and Nutrition Examination Survey. Environ Int. 2018; 110:32-41. DOI: 10.1016/j.envint.2017.10.001 PubMed: 29102155
19. Bello A, Carignan CC, Xue Y, Stapleton HM, Bello D. Exposure to organophosphate flame retardants in spray polyurethane foam applicators: Role of dermal exposure. Environ Int. 2018; 113:55-65. DOI: 10.1016/j.envint.2018.01.020 PubMed: 29421408
20. Hoffman K, Lorenzo A, Butt CM, Adair L, Herring AH, Stapleton HM, Daniels JL. Predictors of urinary flame retardant concentration among pregnant women. Environ Int. 2017; 98:96-101. DOI: 10.1016/j.envint.2016.10.007 PubMed: 27745946
21. Hammel SC, Hoffman K, Webster TF, Anderson KA, Stapleton HM. Measuring personal exposure to organophosphate flame retardants using silicone wristbands and hand wipes. Environ Sci Technol. 2016; 50(8):4483-4491. DOI: 10.1021/acs.est.6b00030 PubMed: 26975559
22. Kosarac I, Kubwabo C, Foster WG. Quantitative determination of nine urinary metabolites of organophosphate flame retardants using solid phase extraction and ultra performance liquid chromatography coupled to tandem mass spectrometry (UPLC-MS/MS). J Chromatogr B Analyt Technol Biomed Life Sci. 2016; 1014:24-30. DOI: 10.1016/j.jchromb.2016.01.035 PubMed: 26869296
23. Fromme H, Lahrz T, Kraft M, Fembacher L, Mach C, Dietrich S, Burkardt R, Völkel W, Göen T. Organophosphate flame retardants and plasticizers in the air and dust in German daycare centers and human biomonitoring in visiting children (LUPE 3). Environ Int. 2014; 71:158-163. DOI: 10.1016/j.envint.2014.06.016 PubMed: 25033099
24. Shi F, Liang K, Liu R, Dong Q, He Z, Xu J, Liu J. Elevated occupational exposure to chlorinated phosphate esters at a construction materials manufacturing plant. Environ Int. 2020; 139:105653. DOI: 10.1016/j.envint.2020.105653 PubMed: 32361061
25. Ding J, Deng T, Ye X, Covaci A, Liu J, Yang F. Urinary metabolites of organophosphate esters and implications for exposure pathways in adolescents from Eastern China. Sci Total Environ. 2019; 695:133894. DOI: 10.1016/j.scitotenv.2019.133894 PubMed: 31425989
26. Araki A, Bastiaensen M, Ait Bamai Y, Van den Eede N, Kawai T, Tsuboi T, Ketema RM, Covaci A, Kishi R. Associations between allergic symptoms and phosphate flame retardants in dust and their urinary metabolites among school children. Environ Int. 2018; 119:438-446. DOI: 10.1016/j.envint.2018.07.018 PubMed: 30031263
27. He C, Wang X, Tang S, Thai P, Li Z, Baduel C, Mueller JF. Concentrations of organophosphate esters and their specific metabolites in food in southeast Queensland, Australia: Is dietary exposure an important pathway of organophosphate esters and their metabolites? Environ Sci Technol. 2018; 52(21):12765-12773. DOI: 10.1021/acs.est.8b03043 PubMed: 30303374
28. Hammel SC, Stapleton HM, Eichner B, Hoffman K. Reconsidering an appropriate urinary biomarker for flame retardant tris(1-chloro-2-propyl) phosphate (TCIPP) exposure in children. Environ Sci Technol Lett. 2021; 8(1):80-85. DOI: 10.1021/acs.estlett.0c00794
29. European Chemicals Agency (ECHA). Screening report: An assessment of whether the use of TCEP, TCPP, and TCDP in articles should be restricted. Helsinki, Finland: European Chemicals Agency; 2018. https://echa.europa.eu/documents/10162/13641/screening_report_tcep_tcpp_td-cp_en.pdf/e0960aa7-f703-499c-24ff-fba627060698
30. Minegishi K, Kurebayashi H, Nambaru S, Morimoto K, Takahashi T, Yamaha T. Comparative studies on absorption, distribution, and excretion of flame retardants halogenated alkyl phosphate in rats. Eisei Kagaku. 1988; 34(2):102-114. DOI: 10.1248/jhs1956.34.102
31. National Industrial Chemicals Notification Assessment Scheme (NICNAS). Inventory Multi-tiered Assessment and Prioritisation (IMAP): 2-Propanol, 1-chloro-, 2,2',2”-phosphate: Human health tier II assessment. Sydney, Australia: Australian Government, Department of Health, National Industrial Chemicals Notification and Assessment Scheme; 2016. https://www.industrialchemicals.gov.au/sites/default/files/2-Propanol%2C%201-chloro-%2C%202%2C2%27%2C2%27%27-phosphate_Human%20health%20tier%20II%20assessment.pdf
32. National Academies of Sciences Engineering and Medicine (NASEM). A class approach to hazard assessment of organohalogen flame retardants. Washington, DC: National Academies Press; 2019. https://nap.nationalacademies.org/catalog/25412/a-class-approach-to-hazard-assessment-of-organohalogen-flame-retardants
33. Freudenthal RI, Henrich RT. A subchronic toxicity study of Fyrol PCF in Sprague-Dawley rats. Int J Toxicol. 1999; 18(3):173-176. DOI: 10.1080/109158199225468
34. Kawasaki H, Murai T, Kanoh S. Studies on the toxicity of insecticides and food additives in pregnant rats. V. Fetal toxicity of tris-(chloropropyl) phosphate (TCPP). [In Japanese]. Oyo Yakuri. 1982; 24:697-702.
35. National Toxicology Program (NTP). Developmental and reproductive toxicity technical report on the prenatal development studies of tris(chloropropyl) phosphate (CASRN 13674-84-5) in Sprague Dawley (Hsd:Sprague Dawley SD) rats (gavage studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2020. NTP DART No. 1. https://ntp.niehs.nih.gov/go/dart01abs PubMed: 32716615
36. Dishaw LV, Macaulay LJ, Roberts SC, Stapleton HM. Exposures, mechanisms, and impacts of endocrine-active flame retardants. Curr Opin Pharmacol. 2014; 19:125-133. DOI: 10.1016/j.coph.2014.09.018 PubMed: 25306433
37. Du Z, Wang G, Gao S, Wang Z. Aryl organophosphate flame retardants induced cardiotoxicity during zebrafish embryogenesis: By disturbing expression of the transcriptional regulators. Aquat Toxicol. 2015; 161:25-32. DOI: 10.1016/j.aquatox.2015.01.027 PubMed: 25661707
38. McGee SP, Cooper EM, Stapleton HM, Volz DC. Early zebrafish embryogenesis is susceptible to developmental TDCPP exposure. Environ Health Perspect. 2012; 120(11):1585-1591. DOI: 10.1289/ehp.1205316 PubMed: 23017583
39. Noyes PD, Haggard DE, Gonnerman GD, Tanguay RL. Advanced morphological - behavioral test platform reveals neurodevelopmental defects in embryonic zebrafish exposed to comprehensive suite of halogenated and organophosphate flame retardants. Toxicol Sci. 2015; 145(1):177-195. DOI: 10.1093/toxsci/kfv044 PubMed: 25711236
40. Meeker JD, Stapleton HM. House dust concentrations of organophosphate flame retardants in relation to hormone levels and semen quality parameters. Environ Health Perspect. 2010; 118(3):318-323. DOI: 10.1289/ehp.0901332 PubMed: 20194068
41. Ingle ME, Mínguez-Alarcón L, Carignan CC, Butt CM, Stapleton HM, Williams PL, Ford JB, Hauser R, Meeker JD. The association between urinary concentrations of phosphorous-containing flame retardant metabolites and semen parameters among men from a fertility clinic. Int J Hyg Environ Health. 2018; 221(5):809-815. DOI: 10.1016/j.ijheh.2018.05.001 PubMed: 29739653
42. Carignan CC, Mínguez-Alarcón L, Butt CM, Williams PL, Meeker JD, Stapleton HM, Toth TL, Ford JB, Hauser R. EARTH Study Team. Urinary concentrations of organophosphate flame retardant metabolites and pregnancy outcomes among women undergoing in vitro fertilization. Environ Health Perspect. 2017; 125(8):087018. DOI: 10.1289/EHP1021 PubMed: 28858831
43. Hoffman K, Stapleton HM, Lorenzo A, Butt CM, Adair L, Herring AH, Daniels JL. Prenatal exposure to organophosphates and associations with birthweight and gestational length. Environ Int. 2018; 116:248-254. DOI: 10.1016/j.envint.2018.04.016 PubMed: 29698901
44. Doherty BT, Hoffman K, Keil AP, Engel SM, Stapleton HM, Goldman BD, Olshan AF, Daniels JL. Prenatal exposure to organophosphate esters and behavioral development in young children in the Pregnancy, Infection, and Nutrition Study. Neurotoxicology. 2019; 73:150-160. DOI: 10.1016/j.neuro.2019.03.007 PubMed: 30951742
45. Health Canada. Screening assessment for the challenge: Ethanol, 2-chloro-, phosphate (3:1) (tris(2-chloroethyl) phosphate [TCEP]) chemical abstracts service registry number 115-96-8. Environment Canada, Health Canada; 2009.
46. Deziel NC, Yi H, Stapleton HM, Huang H, Zhao N, Zhang Y. A case-control study of exposure to organophosphate flame retardants and risk of thyroid cancer in women. BMC Cancer. 2018; 18(1):637. DOI: 10.1186/s12885-018-4553-9 PubMed: 29871608
47. Hoffman K, Lorenzo A, Butt CM, Hammel SC, Henderson BB, Roman SA, Scheri RP, Stapleton HM, Sosa JA. Exposure to flame retardant chemicals and occurrence and severity of papillary thyroid cancer: A case-control study. Environ Int. 2017; 107:235-242. DOI: 10.1016/j.envint.2017.06.021 PubMed: 28772138
48. Föllmann W, Wober J. Investigation of cytotoxic, genotoxic, mutagenic, and estrogenic effects of the flame retardants tris-(2-chloroethyl)-phosphate (TCEP) and tris-(2-chloropropyl)-phosphate (TCPP) in vitro. Toxicol Lett. 2006; 161(2):124-134. DOI: 10.1016/j.toxlet.2005.08.008 PubMed: 16154716
49. Nakamura A, Tateno N, Kojima S, Kaniwa MA, Kawamura T. The mutagenicity of halogenated alkanols and their phosphoric acid esters for Salmonella typhimurium. Mutat Res. 1979; 66(4):373-380. DOI: 10.1016/0165-1218(79)90048-X PubMed: 379633
50. Zeiger E, Anderson B, Haworth S, Lawlor T, Mortelmans K. Salmonella mutagenicity tests: V. Results from the testing of 311 chemicals. Environ Mol Mutagen. 1992; 19 Suppl 21:2-141. DOI: 10.1002/em.2850190603 PubMed: 1541260
51. Williams GM, Mori H, McQueen CA. Structure-activity relationships in the rat hepatocyte DNA-repair test for 300 chemicals. Mutat Res. 1989; 221(3):263-286. DOI: 10.1016/0165-1110(89)90039-0 PubMed: 2682231
52. National Toxicology Program (NTP). Nomination of FR chemicals for NTP testing. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2005.
53. Maronpot RR, Boorman GA. Interpretation of rodent hepatocellular proliferative alterations and hepatocellular tumors in chemical safety assessment. Toxicol Pathol. 1982; 10(2):71-78. DOI: 10.1177/019262338201000210 PubMed: 28094716
54. Boorman GA, Haseman JK, Waters MD, Hardisty JF, Sills RC. Quality review procedures necessary for rodent pathology databases and toxicogenomic studies: The National Toxicology Program experience. Toxicol Pathol. 2002; 30(1):88-92. DOI: 10.1080/01926230252824752 PubMed: 11890481
55. Brix AE, Hardisty JF, McConnell EE. Combining neoplasms for evaluation of rodent carcinogenesis studies. In: Hsu CH, Stedeford T, editors. Cancer Risk Assessment: Chemical Carcinogenesis, Hazard Evaluation, and Risk Quantification. Hoboken, NJ: Wiley; 2010. p. 699-715. DOI: 10.1002/9780470622728.ch28
56. Collins B, Slade D, Aillon K, Stout M, Betz L, Waidyanatha S, Ryan K. Plasma concentrations of tris(1-chloro-2-propyl) phosphate and a metabolite bis(2-chloroisopropyl) 1-carboxyethyl phosphate in Sprague-Dawley rats and B6C3F1/N mice from a chronic study of tris(chloropropyl) phosphate via feed. Toxicol Rep. 2022; 9:690-698. DOI: 10.1016/j.toxrep.2022.03.025 PubMed: 35433273
57. Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958; 53(282):457-481. DOI: 10.1080/01621459.1958.10501452
58. Tarone RE. Tests for trend in life table analysis. Biometrika. 1975; 62(3):679-690. DOI: 10.1093/biomet/62.3.679
59. Cox DR. Regression models and life‐tables. J R Stat Soc Series B Stat Methodol. 1972; 34(2):187-202.
60. Bailer AJ, Portier CJ. Effects of treatment-induced mortality and tumor-induced mortality on tests for carcinogenicity in small samples. Biometrics. 1988; 44(2):417-431. DOI: 10.2307/2531856 PubMed: 3390507
61. Piegorsch WW, Bailer AJ. Statistics for environmental biology and toxicology: Section 6.3.2. London, UK: Chapman and Hall; 1997.
62. Portier CJ, Bailer AJ. Testing for increased carcinogenicity using a survival-adjusted quantal response test. Fundam Appl Toxicol. 1989; 12(4):731-737. DOI: 10.1016/0272-0590(89)90004-3 PubMed: 2744275
63. Portier CJ, Hedges JC, Hoel DG. Age-specific models of mortality and tumor onset for historical control animals in the National Toxicology Program’s carcinogenicity experiments. Cancer Res. 1986; 46(9):4372-4378. PubMed: 3731095
64. Bieler GS, Williams RL. Ratio estimates, the delta method, and quantal response tests for increased carcinogenicity. Biometrics. 1993; 49(3):793-801. DOI: 10.2307/2532200 PubMed: 8241374
65. Nam JM. A simple approximation for calculating sample sizes for detecting linear trend in proportions. Biometrics. 1987; 43(3):701-705. DOI: 10.2307/2532006 PubMed: 3663825
66. Rao JNK, Scott AJ. A simple method for the analysis of clustered binary data. Biometrics. 1992; 48(2):577-585. DOI: 10.2307/2532311 PubMed: 1637980
67. Fung KY, Krewski D, Rao JNK, Scott AJ. Tests for trend in developmental toxicity experiments with correlated binary data. Risk Anal. 1994; 14(4):639-648. DOI: 10.1111/j.1539-6924.1994.tb00277.x PubMed: 7972964
68. Gart JJ, Chu KC, Tarone RE. Statistical issues in interpretation of chronic bioassay tests for carcinogenicity. J Natl Cancer Inst. 1979; 62(4):957-974. DOI: 10.1093/jnci/62.4.957 PubMed: 285297
69. Dixon WJ, Massey FJ. Introduction to statistical analysis. 2nd ed. New York, NY: McGraw Hill; 1957.
70. Tukey JW. Easy summaries--numerical and graphical.Exploratory Data Analysis. Reading, MA: Addison-Wesley; 1977. p. 27-56.
71. Dunnett CW. A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc. 1955; 50(272):1096-1121. DOI: 10.1080/01621459.1955.10501294
72. Williams DA. A test for differences between treatment means when several dose levels are compared with a zero dose control. Biometrics. 1971; 27(1):103-117. DOI: 10.2307/2528930 PubMed: 5547548
73. Williams DA. The comparison of several dose levels with a zero dose control. Biometrics. 1972; 28(2):519-531. DOI: 10.2307/2556164 PubMed: 5037867
74. Shirley E. A non-parametric equivalent of Williams’ test for contrasting increasing dose levels of a treatment. Biometrics. 1977; 33(2):386-389. DOI: 10.2307/2529789 PubMed: 884197
75. Williams DA. A note on Shirley’s nonparametric test for comparing several dose levels with a zero-dose control. Biometrics. 1986; 42(1):183-186. DOI: 10.2307/2531254 PubMed: 3719054
76. Dunn OJ. Multiple comparisons using rank sums. Technometrics. 1964; 6(3):241-252. DOI: 10.1080/00401706.1964.10490181
77. Jonckheere AR. A distribution-free k-sample test against ordered alternatives. Biometrika. 1954; 41(1-2):133-145. DOI: 10.1093/biomet/41.1-2.133
78. Datta S, Satten GA. Rank-sum tests for clustered data. J Am Stat Assoc. 2005; 100(471):908-915. DOI: 10.1198/016214504000001583
79. Hommel G. A stagewise rejective multiple test procedure based on a modified Bonferroni test. Biometrika. 1988; 75(2):383-386. DOI: 10.1093/biomet/75.2.383
80. Hsu JC. The factor analytic approach to simultaneous inference in the general linear model. J Comput Graph Stat. 1992; 1(2):151-168. DOI: 10.1080/10618600.1992.10477011
81. Haseman JK. Value of historical controls in the interpretation of rodent tumor data. Drug Inf J. 1992; 26(2):191-200. DOI: 10.1177/009286159202600210
82. Haseman JK. Data analysis: Statistical analysis and use of historical control data. Regul Toxicol Pharmacol. 1995; 21(1):52-59, discussion 81-86. DOI: 10.1006/rtph.1995.1009 PubMed: 7784636
83. Haseman JK, Rao GN. Effects of corn oil, time-related changes, and inter-laboratory variability on tumor occurrence in control Fischer 344 (F344/N) rats. Toxicol Pathol. 1992; 20(1):52-60. DOI: 10.1177/019262339202000107 PubMed: 1411131
84. U.S. Food and Drug Administration (FDA). 21 CFR Part 58. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=58
85. Miller JA, Miller EC. Ultimate chemical carcinogens as reactive mutagenic electrophiles. In: Hiatt HH, Watson JD, Winsten JA, editors. Origins of Human Cancer. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory; 1977. p. 605-627.
86. Crawford BD. Perspectives on the somatic mutation model of carcinogenesis. In: Flamm WG, Lorentzen RJ, editors. Mechanisms and Toxicity of Chemical Carcinogens and Mutagens. Princeton, NJ: Princeton Scientific Publishing; 1985. p. 13-59.
87. Straus DS. Somatic mutation, cellular differentiation, and cancer causation. J Natl Cancer Inst. 1981; 67(2):233-241. DOI: 10.1093/jnci/67.2.233 PubMed: 7021938
88. Ashby J, Tennant RW. Definitive relationships among chemical structure, carcinogenicity and mutagenicity for 301 chemicals tested by the U.S. NTP. Mutat Res. 1991; 257(3):229-306. DOI: 10.1016/0165-1110(91)90003-E PubMed: 1707500
89. Tennant RW, Margolin BH, Shelby MD, Zeiger E, Haseman JK, Spalding J, Caspary W, Resnick M, Stasiewicz S, Anderson B, et al. Prediction of chemical carcinogenicity in rodents from in vitro genetic toxicity assays. Science. 1987; 236(4804):933-941. DOI: 10.1126/science.3554512 PubMed: 3554512
90. Zeiger E, Haseman JK, Shelby MD, Margolin BH, Tennant RW, Holden HE. Evaluation of four in vitro genetic toxicity tests for predicting rodent carcinogenicity: Confirmation of earlier results with 41 additional chemicals. Environ Mol Mutagen. 1990; 16 Suppl 18:1-14. DOI: 10.1002/em.2850160502 PubMed: 2091921
91. Heddle JA, Hite M, Kirkhart B, Mavournin K, MacGregor JT, Newell GW, Salamone MF. The induction of micronuclei as a measure of genotoxicity: A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutat Res. 1983; 123(1):61-118. DOI: 10.1016/0165-1110(83)90047-7 PubMed: 6888413
92. Schmid W. The micronucleus test. Mutat Res. 1975; 31(1):9-15. DOI: 10.1016/0165-1161(75)90058-8 PubMed: 48190
93. Shelby MD, Erexson GL, Hook GJ, Tice RR. Evaluation of a three-exposure mouse bone marrow micronucleus protocol: Results with 49 chemicals. Environ Mol Mutagen. 1993; 21(2):160-179. DOI: 10.1002/em.2850210210 PubMed: 8444144
94. Shelby MD, Witt KL. Comparison of results from mouse bone marrow chromosome aberration and micronucleus tests. Environ Mol Mutagen. 1995; 25(4):302-313. DOI: 10.1002/em.2850250407 PubMed: 7607185
95. Witt KL, Knapton A, Wehr CM, Hook GJ, Mirsalis J, Shelby MD, MacGregor JT. Micronucleated erythrocyte frequency in peripheral blood of B6C3F(1) mice from short-term, prechronic, and chronic studies of the NTP carcinogenesis bioassay program. Environ Mol Mutagen. 2000; 36(3):163-194. DOI: 10.1002/1098-2280(2000)36:3<163::AID-EM1>3.0.CO;2-P PubMed: 11044899
96. National Toxicology Program (NTP). TR-602: Pathology tables, survival and growth curves from NTP short-term, long-term, genetic toxicology, and other information studies. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2020. DOI: 10.22427/NTP-DATA-TR-602
97. Everds NE, Snyder PW, Bailey KL, Bolon B, Creasy DM, Foley GL, Rosol TJ, Sellers T. Interpreting stress responses during routine toxicity studies: A review of the biology, impact, and assessment. Toxicol Pathol. 2013; 41(4):560-614. DOI: 10.1177/0192623312466452 PubMed: 23475558
98. Marklund A, Andersson B, Haglund P. Screening of organophosphorus compounds and their distribution in various indoor environments. Chemosphere. 2003; 53(9):1137-1146. DOI: 10.1016/S0045-6535(03)00666-0 PubMed: 14512118
99. California Environmental Protection Agency (CalEPA). Chemicals known to the state to cause cancer or reproductive toxicity, December 29, 2017 (Proposition 65 list). Sacramento, CA: Office of Environmental Health Hazard Assessment; 2017. https://oehha.ca.gov/proposition-65/proposition-65-list
100. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of tris(2-chloroethyl) phosphate (CAS No. 115-96-8) in F344/N rats and B6C3F1 mice (gavage studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 1991. NTP Technical Report No. 391. https://ntp.niehs.nih.gov/go/tr391abs PubMed: 12637968
101. Dishaw LV, Powers CM, Ryde IT, Roberts SC, Seidler FJ, Slotkin TA, Stapleton HM. Is the PentaBDE replacement, tris (1,3-dichloro-2-propyl) phosphate (TDCPP), a developmental neurotoxicant? Studies in PC12 cells. Toxicol Appl Pharmacol. 2011; 256(3):281-289. DOI: 10.1016/j.taap.2011.01.005 PubMed: 21255595
102. Hou R, Xu Y, Wang Z. Review of OPFRs in animals and humans: Absorption, bioaccumulation, metabolism, and internal exposure research. Chemosphere. 2016; 153:78-90. DOI: 10.1016/j.chemosphere.2016.03.003 PubMed: 27010170
103. Wang X, Zhu Q, Yan X, Wang Y, Liao C, Jiang G. A review of organophosphate flame retardants and plasticizers in the environment: Analysis, occurrence and risk assessment. Sci Total Environ. 2020; 731:139071. DOI: 10.1016/j.scitotenv.2020.139071 PubMed: 32438088
104. Hall RL. Clinical pathology of laboratory animals. In: Gad SC, editor. Animal Models in Toxicology. 2nd ed. Boca Raton, FL: Taylor & Francis; 2007. p. 787-830.
105. Waner T, Nyska A. The influence of fasting on blood glucose, triglycerides, cholesterol, and alkaline phosphatase in rats. Vet Clin Pathol. 1994; 23(3):78-80. DOI: 10.1111/j.1939-165X.1994.tb00683.x PubMed: 12666020
106. Gwinn WM, Auerbach SS, Parham F, Stout MD, Waidyanatha S, Mutlu E, Collins B, Paules RS, Merrick BA, Ferguson S, et al. Evaluation of 5-day in vivo rat liver and kidney with high-throughput transcriptomics for estimating benchmark doses of apical outcomes. Toxicol Sci. 2020; 176(2):343-354. DOI: 10.1093/toxsci/kfaa081 PubMed: 32492150
107. Quaglino D, Capri M, Bergamini G, Euclidi E, Zecca L, Franceschi C, Ronchetti IP. Age-dependent remodeling of rat thymus. Morphological and cytofluorimetric analysis from birth up to one year of age. Eur J Cell Biol. 1998; 76(2):156-166. DOI: 10.1016/S0171-9335(98)80029-0 PubMed: 9696356
108. Ji C, Lu Z, Xu L, Li F, Cong M, Shan X, Wu H. Global responses to tris(1-chloro-2-propyl)phosphate (TCPP) in rockfish Sebastes schlegeli using integrated proteomic and metabolomic approach. Sci Total Environ. 2020; 724:138307. DOI: 10.1016/j.scitotenv.2020.138307 PubMed: 32272412
109. Porter E, Crump D, Egloff C, Chiu S, Kennedy SW. Use of an avian hepatocyte assay and the avian Toxchip Polymerse chain reaction array for testing prioritization of 16 organic flame retardants. Environ Toxicol Chem. 2014; 33(3):573-582. DOI: 10.1002/etc.2469 PubMed: 24273086
110. Farhat A, Crump D, Chiu S, Williams KL, Letcher RJ, Gauthier LT, Kennedy SW. In ovo effects of two organophosphate flame retardants—TCPP and TDCPP—on pipping success, development, mRNA expression, and thyroid hormone levels in chicken embryos. Toxicol Sci. 2013; 134(1):92-102. DOI: 10.1093/toxsci/kft100 PubMed: 23629516
111. National Toxicology Program (NTP). Historical controls. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2021. https://ntp.niehs.nih.gov/data/controls/index.html
112. van der Veen I, de Boer J. Phosphorus flame retardants: Properties, production, environmental occurrence, toxicity and analysis. Chemosphere. 2012; 88(10):1119-1153. DOI: 10.1016/j.chemosphere.2012.03.067 PubMed: 22537891
113. Consumer Product Safety Commission (CPSC). Guidance document on hazardous additive, non-polymeric organohalogen flame retardants in certain consumer products. Fed Regist. 2017; 82(Sep 28):45268-45269.
114. National Institute of Standards and Technology (NIST). NIST/EPA/NIH mass spectral library (NIST 08) and NIST mass spectral search program (version 2.0d). Gaithersburg, MD: U.S. Department of Commerce, National Institute of Standards and Technology; 2008.
115. Witt KL, Livanos E, Kissling GE, Torous DK, Caspary W, Tice RR, Recio L. Comparison of flow cytometry- and microscopy-based methods for measuring micronucleated reticulocyte frequencies in rodents treated with nongenotoxic and genotoxic chemicals. Mutat Res. 2008; 649(1-2):101-113. DOI: 10.1016/j.mrgentox.2007.08.004 PubMed: 17869571
116. Dertinger SD, Camphausen K, MacGregor JT, Bishop ME, Torous DK, Avlasevich S, Cairns S, Tometsko CR, Menard C, Muanza T, et al. Three‐color labeling method for flow cytometric measurement of cytogenetic damage in rodent and human blood. Environ Mol Mutagen. 2004; 44(5):427-435. DOI: 10.1002/em.20075 PubMed: 15517570
117. Kissling GE, Dertinger SD, Hayashi M, MacGregor JT. Sensitivity of the erythrocyte micronucleus assay: Dependence on number of cells scored and inter-animal variability. Mutat Res. 2007; 634(1-2):235-240. DOI: 10.1016/j.mrgentox.2007.07.010 PubMed: 17851117
118. Hsieh JH, Sedykh A, Huang R, Xia M, Tice RR. A data analysis pipeline accounting for artifacts in Tox21 quantitative high-throughput screening assays. J Biomol Screen. 2015; 20(7):887-897. DOI: 10.1177/1087057115581317 PubMed: 25904095
119. Sedykh A. CurveP method for rendering high-throughput screening dose-response data into digital fingerprints. In: Zhu H, Xia M, editors. High-Throughput Screening Assays in Toxicology. 16th ed. New York, NY: Humana Press; 2016. p. 135-141. DOI: 10.1007/978-1-4939-6346-1_14
120. Hubbard TD, Hsieh JH, Rider CV, Sipes NS, Sedykh A, Collins BJ, Auerbach SS, Xia M, Huang R, Walker NJ, et al. Using Tox21 high-throughput screening assays for the evaluation of botanical and dietary supplements. Appl In Vitro Toxicol. 2019; 5(1):10-25. DOI: 10.1089/aivt.2018.0020 PubMed: 30944845
121. Judson RS, Thomas RS, Baker N, Simha A, Howey XM, Marable C, Kleinstreuer NC, Houck KA. Workflow for defining reference chemicals for assessing performance of in vitro assays. ALTEX. 2019; 36(2):261-276. DOI: 10.14573/altex.1809281 PubMed: 30570668
122. Mav D, Shah RR, Howard BE, Auerbach SS, Bushel PR, Collins JB, Gerhold DL, Judson RS, Karmaus AL, Maull EA, et al. A hybrid gene selection approach to create the S1500+ targeted gene sets for use in high-throughput transcriptomics. PLoS One. 2018; 13(2):e0191105. DOI: 10.1371/journal.pone.0191105 PubMed: 29462216
123. Sciome. GeniE. Research Triangle Park, NC: Sciome; 2021. https://www.sciome.com/genie/
124. National Toxicology Program (NTP). DrugMatrix/ToxFX. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2019. https://ntp.niehs.nih.gov/data/drugmatrix/
125. Illumina. BaseSpace Correlation Engine. 2021; https://www.illumina.com/products/by-type/informatics-products/basespace-correlation-engine.html
126. Zgheib E, Limonciel A, Jiang X, Wilmes A, Wink S, van de Water B, Kopp-Schneider A, Bois FY, Jennings P. Investigation of Nrf2, AhR and ATF4 activation in toxicogenomic databases. Front Genet. 2018; 9:429. DOI: 10.3389/fgene.2018.00429 PubMed: 30333853
127. Wang C, Gong B, Bushel PR, Thierry-Mieg J, Thierry-Mieg D, Xu J, Fang H, Hong H, Shen J, Su Z, et al. The concordance between RNA-seq and microarray data depends on chemical treatment and transcript abundance. Nat Biotechnol. 2014; 32(9):926-932. DOI: 10.1038/nbt.3001 PubMed: 25150839
128. Podtelezhnikov AA, Monroe JJ, Aslamkhan AG, Pearson K, Qin C, Tamburino AM, Loboda AP, Glaab WE, Sistare FD, Tanis KQ. Quantitative transcriptional biomarkers of xenobiotic receptor activation in rat liver for the early assessment of drug safety liabilities. Toxicol Sci. 2020; 175(1):98-112. DOI: 10.1093/toxsci/kfaa026 PubMed: 32119089
129. Oshida K, Vasani N, Jones C, Moore T, Hester S, Nesnow S, Auerbach S, Geter DR, Aleksunes LM, Thomas RS, et al. Identification of chemical modulators of the constitutive activated receptor (CAR) in a gene expression compendium. Nucl Recept Signal. 2015; 13(1). DOI: 10.1621/nrs.13002 PubMed: 25949234
130. Forbes KP, Kouranova E, Tinker D, Janowski K, Cortner D, McCoy A, Cui X. Creation and preliminary characterization of pregnane X receptor and constitutive androstane receptor knockout rats. Drug Metab Dispos. 2017; 45(10):1068-1076. DOI: 10.1124/dmd.117.075788 PubMed: 28716828
131. McMillian M, Nie A, Parker JB, Leone A, Kemmerer M, Bryant S, Herlich J, Yieh L, Bittner A, Liu X, et al. Drug-induced oxidative stress in rat liver from a toxicogenomics perspective. Toxicol Appl Pharmacol. 2005; 207(2 Suppl):171-178. DOI: 10.1016/j.taap.2005.02.031 PubMed: 15982685
132. Ganter B, Tugendreich S, Pearson CI, Ayanoglu E, Baumhueter S, Bostian KA, Brady L, Browne LJ, Calvin JT, Day GJ, et al. Development of a large-scale chemogenomics database to improve drug candidate selection and to understand mechanisms of chemical toxicity and action. J Biotechnol. 2005; 119(3):219-244. DOI: 10.1016/j.jbiotec.2005.03.022 PubMed: 16005536
133. Tonelli C, Chio IIC, Tuveson DA. Transcriptional regulation by Nrf2. Antioxid Redox Signal. 2018; 29(17):1727-1745. DOI: 10.1089/ars.2017.7342 PubMed: 28899199
134. Monroe JJ, Tanis KQ, Podtelezhnikov AA, Nguyen T, Machotka SV, Lynch D, Evers R, Palamanda J, Miller RR, Pippert T, et al. Application of a rat liver drug bioactivation transcriptional response assay early in drug development that informs chemically reactive metabolite formation and potential for drug-induced liver injury. Toxicol Sci. 2020; 177(1):281-299. DOI: 10.1093/toxsci/kfaa088 PubMed: 32559301
135. Liberzon A, Birger C, Thorvaldsdóttir H, Ghandi M, Mesirov JP, Tamayo P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 2015; 1(6):417-425. DOI: 10.1016/j.cels.2015.12.004 PubMed: 26771021
136. Gusenleitner D, Auerbach SS, Melia T, Gómez HF, Sherr DH, Monti S. Genomic models of short-term exposure accurately predict long-term chemical carcinogenicity and identify putative mechanisms of action. PLoS One. 2014; 9(7):e102579. DOI: 10.1371/journal.pone.0102579 PubMed: 25058030
137. Ellinger-Ziegelbauer H, Gmuender H, Bandenburg A, Ahr HJ. Prediction of a carcinogenic potential of rat hepatocarcinogens using toxicogenomics analysis of short-term in vivo studies. Mutat Res. 2008; 637(1-2):23-39. DOI: 10.1016/j.mrfmmm.2007.06.010 PubMed: 17689568
138. Ellinger-Ziegelbauer H, Stuart B, Wahle B, Bomann W, Ahr HJ. Characteristic expression profiles induced by genotoxic carcinogens in rat liver. Toxicol Sci. 2004; 77(1):19-34. DOI: 10.1093/toxsci/kfh016 PubMed: 14600272
139. Ellinger-Ziegelbauer H, Stuart B, Wahle B, Bomann W, Ahr HJ. Comparison of the expression profiles induced by genotoxic and nongenotoxic carcinogens in rat liver. Mutat Res. 2005; 575(1-2):61-84. DOI: 10.1016/j.mrfmmm.2005.02.004 PubMed: 15890375
140. Hill T, Rooney J, Abedini J, El-Masri H, Wood CE, Corton JC. Gene expression thresholds derived from short-term exposures identify rat liver tumorigens. Toxicol Sci. 2020; 177(1):41-59. DOI: 10.1093/toxsci/kfaa102 PubMed: 32603419
141. Auerbach SS, Shah RR, Mav D, Smith CS, Walker NJ, Vallant MK, Boorman GA, Irwin RD. Predicting the hepatocarcinogenic potential of alkenylbenzene flavoring agents using toxicogenomics and machine learning. Toxicol Appl Pharmacol. 2010; 243(3):300-314. DOI: 10.1016/j.taap.2009.11.021 PubMed: 20004213
142. Fielden MR, Adai A, Dunn RT 2nd, Olaharski A, Searfoss G, Sina J, Aubrecht J, Boitier E, Nioi P, Auerbach S, et al. Development and evaluation of a genomic signature for the prediction and mechanistic assessment of nongenotoxic hepatocarcinogens in the rat. Toxicol Sci. 2011; 124(1):54-74. DOI: 10.1093/toxsci/kfr202 PubMed: 21813463
143. Uehara T, Hirode M, Ono A, Kiyosawa N, Omura K, Shimizu T, Mizukawa Y, Miyagishima T, Nagao T, Urushidani T. A toxicogenomics approach for early assessment of potential non-genotoxic hepatocarcinogenicity of chemicals in rats. Toxicology. 2008; 250(1):15-26. DOI: 10.1016/j.tox.2008.05.013 PubMed: 18619722
144. Yamada F, Sumida K, Uehara T, Morikawa Y, Yamada H, Urushidani T, Ohno Y. Toxicogenomics discrimination of potential hepatocarcinogenicity of non-genotoxic compounds in rat liver. J Appl Toxicol. 2013; 33(11):1284-1293. DOI: 10.1002/jat.2790 PubMed: 22806939
145. Te JA. AbdulHameed MDM, Wallqvist A. Systems toxicology of chemically induced liver and kidney injuries: Histopathology-associated gene co-expression modules. J Appl Toxicol. 2016; 36(9):1137-1149. DOI: 10.1002/jat.3278 PubMed: 26725466
146. Schyman P, Xu Z, Desai V, Wallqvist A. TOXPANEL: A gene-set analysis tool to assess liver and kidney injuries. Front Pharmacol. 2021; 12:601511. DOI: 10.3389/fphar.2021.601511 PubMed: 33633572
147. Glaab WE, Holder D, He YD, Bailey WJ, Gerhold DL, Beare C, Erdos Z, Lane P, Michna L, Muniappa N, et al. Universal toxicity gene signatures for early identification of drug-induced tissue injuries in rats. Toxicol Sci. 2021; 181(2):148-159. DOI: 10.1093/toxsci/kfab038 PubMed: 33837425
148. Huang L, Heinloth AN, Zeng ZB, Paules RS, Bushel PR. Genes related to apoptosis predict necrosis of the liver as a phenotype observed in rats exposed to a compendium of hepatotoxicants. BMC Genomics. 2008; 9:288. DOI: 10.1186/1471-2164-9-288 PubMed: 18558008
149. AbdulHameed MDM. Ippolito DL, Stallings JD, Wallqvist A. Mining kidney toxicogenomic data by using gene co-expression modules. BMC Genomics. 2016; 17(1):790. DOI: 10.1186/s12864-016-3143-y PubMed: 27724849
150. Hoffmann D, Adler M, Vaidya VS, Rached E, Mulrane L, Gallagher WM, Callanan JJ, Gautier JC, Matheis K, Staedtler F, et al. Performance of novel kidney biomarkers in preclinical toxicity studies. Toxicol Sci. 2010; 116(1):8-22. DOI: 10.1093/toxsci/kfq029 PubMed: 20118187
151. Amin RP, Vickers AE, Sistare F, Thompson KL, Roman RJ, Lawton M, Kramer J, Hamadeh HK, Collins J, Grissom S, et al. Identification of putative gene based markers of renal toxicity. Environ Health Perspect. 2004; 112(4):465-479. DOI: 10.1289/ehp.6683 PubMed: 15033597
152. Yang J, Zhao YL, Wu ZQ, Si YL, Meng YG, Fu XB, Mu YM, Han WD. The single-macro domain protein LRP16 is an essential cofactor of androgen receptor. Endocr Relat Cancer. 2009; 16(1):139-153. DOI: 10.1677/ERC-08-0150 PubMed: 19022849
153. UniProt. UniProtKB - P22570 (ADRO_HUMAN). UniProt Consortium; 2022. https://www.uniprot.org/uniprot/P22570
154. UniProt. UniProtKB - P10275 (ANDR_HUMAN): Function. UniProt Consortium; 2022. https://www.uniprot.org/uniprot/P10275#function
155. UniProt. UniProtKB - Q8WWK9 (CKAP2_HUMAN). UniProt Consortium; 2022. https://www.uniprot.org/uniprot/Q8WWK9
156. Dunnick JK, Shockley KR, Pandiri AR, Kissling GE, Gerrish KE, Ton TV, Wilson RE, Brar SS, Brix AE, Waidyanatha S, et al. PBDE-47 and PBDE mixture (DE-71) toxicities and liver transcriptomic changes at PND 22 after in utero/postnatal exposure in the rat. Arch Toxicol. 2018; 92(11):3415-3433. DOI: 10.1007/s00204-018-2292-y PubMed: 30206662
157. Schoonjans K, Peinado-Onsurbe J, Lefebvre AM, Heyman RA, Briggs M, Deeb S, Staels B, Auwerx J. PPARalpha and PPARgamma activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. EMBO J. 1996; 15(19):5336-5348. DOI: 10.1002/j.1460-2075.1996.tb00918.x PubMed: 8895578
158. Dong H, Gill S, Curran IH, Williams A, Kuo B, Wade MG, Yauk CL. Toxicogenomic assessment of liver responses following subchronic exposure to furan in Fischer F344 rats. Arch Toxicol. 2016; 90(6):1351-1367. DOI: 10.1007/s00204-015-1561-2 PubMed: 26194646
159. Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012; 379(9822):1245-1255. DOI: 10.1016/S0140-6736(11)61347-0 PubMed: 22353262
160. Kim Y, Sills RC, Houle CD. Overview of the molecular biology of hepatocellular neoplasms and hepatoblastomas of the mouse liver. Toxicol Pathol. 2005; 33(1):175-180. DOI: 10.1080/01926230590522130 PubMed: 15805069
161. Dhanasekaran R, Bandoh S, Roberts LR. Molecular pathogenesis of hepatocellular carcinoma and impact of therapeutic advances [version 1; peer review: 4 approved]. F1000Res. 2016; 5(F1000 Faculty Rev):879. DOI: 10.12688/f1000research.6946.1
162. McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, et al. The Genome Analysis Toolkit: A MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010; 20(9):1297-1303. DOI: 10.1101/gr.107524.110 PubMed: 20644199
163. Kovi RC, Bhusari S, Mav D, Shah RR, Ton TV, Hoenerhoff MJ, Sills RC, Pandiri AR. Genome-wide promoter DNA methylation profiling of hepatocellular carcinomas arising either spontaneously or due to chronic exposure to Ginkgo biloba extract (GBE) in B6C3F1/N mice. Arch Toxicol. 2019; 93(8):2219-2235. DOI: 10.1007/s00204-019-02505-7 PubMed: 31278416
164. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of cumene (CAS No. 98-82-8) in F344/N rats and B6C3F1 mice (inhalation studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2009. NTP Technical Report No. 542. https://ntp.niehs.nih.gov/go/tr542abs PubMed: 19340095
165. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of 3,3',4,4'-tetrachloroazobenzene (TCAB) [CAS No. 14047-09-7] in Harlan Sprague-Dawley rats and B6C3F1 mice (gavage studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2010. NTP Technical Report No. 558. https://ntp.niehs.nih.gov/go/tr558abs PubMed: 21383777
166. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of ginkgo bilboa extract (CAS No. 90045-36-6) in F344/N rats and B6C3F1/N mice (gavage studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2013. NTP Technical Report No. 578. https://ntp.niehs.nih.gov/go/tr578abs PubMed: 23652021
167. National Toxicology Program (NTP). Toxicology studies of cobalt metal (CASRN 7440-48-4) in F344/N rats and B6C3F1/N mice and toxicology and carcinogenesis studies of cobalt metal in F344/NTac rats and B6C3F1/N mice (inhalation studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2014. NTP Technical Report No. 581. https://ntp.niehs.nih.gov/go/tr581abs PubMed: 33540953
168. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of vinylidene chloride (CASRN 75-35-4) in F344/N rats and B6C3F1/N mice (inhalation studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2015. NTP Technical Report No. 582. https://ntp.niehs.nih.gov/go/tr582abs PubMed: 33540956
169. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of oxazepam (CAS No. 604-75-1) in Swiss-Webster and B6C3F1 mice (feed studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 1993. NTP Technical Report No. 443. https://ntp.niehs.nih.gov/go/tr443abs PubMed: 12595920
170. National Toxicology Program (NTP). NTP toxicology and carcinogenesis studies of primidone (CAS No. 125-33-7) in F344/N rats and B6C3F1 mice (feed studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2000. NTP Technical Report No. 476. https://ntp.niehs.nih.gov/go/tr476abs PubMed: 12571687
171. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of anthraquinone (CAS No. 84-65-1) in F344/N rats and B6C3F1 mice (feed studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2005. NTP Technical Report No. 494. https://ntp.niehs.nih.gov/go/tr494abs PubMed: 16362060
172. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of bromochloroacetic acid (CAS No. 5589-96-8) in F344/N rats and B6C3F1 mice (drinking water studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health; 2009. NTP Technical Report No. 549. https://ntp.niehs.nih.gov/go/tr549abs PubMed: 19340096
173. National Toxicology Program (NTP). Toxicology studies of bromodichloroacetic acid (CASRN 71133-14-7) in F344/N rats and B6C3F1/N mice and toxicology and carcinogenesis studies of bromodichloroacetic acid in F344/NTac rats and B6C3F1/N mice (drinking water studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2015. NTP Technical Report No. 583. https://ntp.niehs.nih.gov/go/tr583abs PubMed: 33540954
174. National Toxicology Program (NTP). Toxicology studies of a pentabromodiphenyl ether mixture [DE-71 (technical grade)] (CASRN 32534-81-9) in F344/N rats and B6C3F1/N mice and toxicology and carcinogenesis studies of a pentabromodiphenyl ether mixture [DE-71 (technical grade)] in Wistar Han [Crl:WI(Han)] rats and B6C3F1/N mice (gavage studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2016. NTP Technical Report No. 589. https://ntp.niehs.nih.gov/go/tr589abs PubMed: 33557475
175. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of p-chloro-α,α,α-trifluorotoluene (CASRN 98-56-6) in Sprague Dawley (Hsd: Sprague Dawley SD) rats and B6C3F1/N mice (inhalation studies). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2018. NTP Technical Report No. 594. https://ntp.niehs.nih.gov/go/tr594abs PubMed: 33618492
176. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of di-n-butyl phthalate (CASRN 84-74-2) administered in feed to Sprague Dawley (Hsd:Sprague Dawley SD) rats and B6C3F1/N mice. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2021. NTP Technical Report No. 600. https://ntp.niehs.nih.gov/go/tr600abs PubMed: 35073285
177. National Toxicology Program (NTP). Toxicology and carcinogenesis studies of an isomeric mixture of tris(chloropropyl) phosphate administered in feed to Sprague Dawley (Hsd:Sprague Dawley SD) rats and B6C3F1/N mice. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program; 2023. NTP Technical Report No. 602. https://ntp.niehs.nih.gov/go/tr602abs PubMed: 37368980
178. Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009; 25(14):1754-1760. DOI: 10.1093/bioinformatics/btp324 PubMed: 19451168
179. Martin M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011; 17(1):10-12. DOI: 10.14806/ej.17.1.200
180. Mudge JM, Harrow J. Creating reference gene annotation for the mouse C57BL6/J genome assembly. Mamm Genome. 2015; 26(9-10):366-378. DOI: 10.1007/s00335-015-9583-x PubMed: 26187010
181. Frankish A, Diekhans M, Ferreira AM, Johnson R, Jungreis I, Loveland J, Mudge JM, Sisu C, Wright J, Armstrong J, et al. GENCODE reference annotation for the human and mouse genomes. Nucleic Acids Res. 2019; 47(D1):D766-D773. DOI: 10.1093/nar/gky955 PubMed: 30357393
182. GENCODE. Release M23 (GRCm38.p6). Hinxton, UK: EMBL-EBI; 2020. https://www.gencodegenes.org/mouse/release_M23.html
183. Patro R, Duggal G, Love MI, Irizarry RA, Kingsford C. Salmon provides fast and bias-aware quantification of transcript expression. Nat Methods. 2017; 14(4):417-419. DOI: 10.1038/nmeth.4197 PubMed: 28263959
184. Soneson C, Love MI, Robinson MD. Differential analyses for RNA-seq: Transcript-level estimates improve gene-level inferences [version 2; peer review: 2 approved]. F1000Res. 2015; 4:1521. DOI: 10.12688/f1000research.7563.2 PubMed: 26925227
185. Law CW, Chen Y, Shi W, Smyth GK. voom: Precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 2014; 15(2):R29. DOI: 10.1186/gb-2014-15-2-r29 PubMed: 24485249
186. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43(7):e47. DOI: 10.1093/nar/gkv007 PubMed: 25605792
187. Robinson MD, McCarthy DJ, Smyth GK. edgeR: A bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010; 26(1):139-140. DOI: 10.1093/bioinformatics/btp616 PubMed: 19910308
188. Andrews S. FastQC: A quality control tool for high throughput sequence data. 2010; https://www.bioinformatics.babraham.ac.uk/projects/fastqc
189. Smith T, Heger A, Sudbery I. UMI-tools: Modeling sequencing errors in Unique Molecular Identifiers to improve quantification accuracy. Genome Res. 2017; 27(3):491-499. DOI: 10.1101/gr.209601.116 PubMed: 28100584
190. Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, Batut P, Chaisson M, Gingeras TR. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics. 2013; 29(1):15-21. DOI: 10.1093/bioinformatics/bts635 PubMed: 23104886
191. Griffiths-Jones S, Grocock RJ, van Dongen S, Bateman A, Enright AJ. miRBase: MicroRNA sequences, targets and gene nomenclature. Nucleic Acids Res. 2006; 34 Suppl 1:D140-D144. DOI: 10.1093/nar/gkj112 PubMed: 16381832
192. Liao Y, Smyth GK, Shi W. featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014; 30(7):923-930. DOI: 10.1093/bioinformatics/btt656 PubMed: 24227677
193. Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R. The Sequence Alignment/Map format and SAMtools. Bioinformatics. 2009; 25(16):2078-2079. DOI: 10.1093/bioinformatics/btp352 PubMed: 19505943
194. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014; 15(12):550. DOI: 10.1186/s13059-014-0550-8 PubMed: 25516281
195. Blokzijl F, Janssen R, van Boxtel R, Cuppen E. MutationalPatterns: Comprehensive genome-wide analysis of mutational processes. Genome Med. 2018; 10(1):33. DOI: 10.1186/s13073-018-0539-0 PubMed: 29695279
196. Alexandrov LB, Stratton MR. Mutational signatures: The patterns of somatic mutations hidden in cancer genomes. Curr Opin Genet Dev. 2014; 24(100):52-60. DOI: 10.1016/j.gde.2013.11.014 PubMed: 24657537
197. Cingolani P, Platts A, Wang LL, Coon M, Nguyen T, Wang L, Land SJ, Lu X, Ruden DM. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin). 2012; 6(2):80-92. DOI: 10.4161/fly.19695 PubMed: 22728672
198. R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. European Environment Agency; 2020. https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006
199. Catalogue of Somatic Mutations in Cancer (COSMIC). Mutational signatures (v3.2 - March 2021): Single base substitution (SBS) signatures. 2021. https://cancer.sanger.ac.uk/signatures/sbs/
200. Hoenerhoff MJ, Pandiri AR, Snyder SA, Hong HH, Ton TV, Peddada S, Shockley K, Witt K, Chan P, Rider C, et al. Hepatocellular carcinomas in B6C3F1 mice treated with Ginkgo biloba extract for two years differ from spontaneous liver tumors in cancer gene mutations and genomic pathways. Toxicol Pathol. 2013; 41(6):826-841. DOI: 10.1177/0192623312467520 PubMed: 23262642
201. Aydinlik H, Nguyen TD, Moennikes O, Buchmann A, Schwarz M. Selective pressure during tumor promotion by phenobarbital leads to clonal outgrowth of beta-catenin-mutated mouse liver tumors. Oncogene. 2001; 20(53):7812-7816. DOI: 10.1038/sj.onc.1204982 PubMed: 11753661
202. Li J, Chanrion M, Sawey E, Wang T, Chow E, Tward A, Su Y, Xue W, Lucito R, Zender L, et al. Reciprocal interaction of Wnt and RXR-α pathways in hepatocyte development and hepatocellular carcinoma. PLoS One. 2015; 10(3):e0118480. DOI: 10.1371/journal.pone.0118480 PubMed: 25738607
203. Lin CY, Gustafsson JÅ. Targeting liver X receptors in cancer therapeutics. Nat Rev Cancer. 2015; 15(4):216-224. DOI: 10.1038/nrc3912 PubMed: 25786697
204. Rudraiah S, Zhang X, Wang L. Nuclear receptors as therapeutic targets in liver disease: Are we there yet? Annu Rev Pharmacol Toxicol. 2016; 56:605-626. DOI: 10.1146/annurev-pharmtox-010715-103209 PubMed: 26738480
Conclusions
Under the conditions of these 2-year feed studies, there was some evidence of carcinogenic activity of tris(chloropropyl) phosphate (TCPP) in male Hsd:Sprague Dawley SD rats based on the increased incidence of hepatocellular adenoma or carcinoma (combined). There was some evidence of carcinogenic activity of TCPP in female Hsd:Sprague Dawley SD rats based on the increased incidence of uterine adenoma or adenocarcinoma (combined). The marginal increase in the incidence of hepatocellular adenoma in female rats may have been related to exposure.
There was some evidence of carcinogenic activity of TCPP in male B6C3F1/N mice based on the increased incidence of hepatocellular carcinoma. There was clear evidence of carcinogenic activity of TCPP in female B6C3F1/N mice based on the increased incidences of hepatocellular adenoma, hepatocellular carcinoma, and hepatocellular adenoma or carcinoma (combined).
In the 2-year studies, exposure to TCPP resulted in increased incidences of nonneoplastic lesions in the liver of male and female rats and in female mice, and in the kidney of male mice.
Appendices
Appendix A. Chemical Characterization and Dose Formulation Studies
A.1.
Procurement and Characterization of Tris(chloropropyl) Phosphate
An isomeric mixture of tris(chloropropyl) phosphate (TCPP) was obtained from Albemarle (Orangeburg, SC) in two lots (101 and 134). Lot 101 was used in the 3-month rat and mouse studies. Lot 134 and a portion of lot 101 were blended to form lot M072911NP, which was used in the 2-year rat and mouse studies. Identity, purity, and stability analyses were conducted by the analytical chemistry laboratory at MRIGlobal (Kansas City, MO) for the study laboratory at Battelle (Columbus, OH). Reports on analyses performed in support of the TCPP studies are on file at the National Institute of Environmental Health Sciences.
The original drum of lot 101 was homogenized by blending the top, middle, and bottom portions of the drum for 10 to 15 minutes. After homogenization, all of lot 101 was transferred into 80 oz amber, glass, narrow-mouth containers, capped using a Teflon-lined lid, and sealed with white sealing tape under inert gas. All bulk containers were stored at room temperature. The purity of lot 101 was reevaluated once before, during, and after the 3-month studies and determined to be statistically similar to a frozen reference sample of the same lot.
Equal portions of lots 101 and 134 were mixed using a dual head peristaltic pump and Food and Drug Administration Norprene pump tubing combined with manual transfer. Lot 134 was filtered through the filter cartridge before mixing to remove particulate material present due to drum corrosion. The homogenized material (lot M072911NP) was delivered directly into two new, pre-cleaned, 55-gallon stainless-steel drums. Final homogenization was achieved by blending all portions of each 55-gallon drum (top, middle, bottom) with an air-driven stirrer at slow speed to prevent entrapment of air. Homogeneity between the two drums of lot M072911NP was confirmed and reported previously.35 The bulk test article was stored with an inert gas headspace at room temperature. The purity of lot M072911NP was reevaluated six times at time points before, during, and after the 2-year studies and was determined to be statistically similar to a frozen reference sample of the same lot at each reanalysis.
Lot 101 was identified as TCPP using proton (1H) nuclear magnetic resonance (NMR) and gas chromatography (GC) with mass spectrometry (MS) detection prior to the 3-month studies. Before the 2-year studies, all three lots (101, 134, and M072911NP) of the test chemical (clear, colorless, oily liquids) were identified as TCPP using 1H and carbon-13 (13C) NMR spectroscopy. In addition, lots 101 and M072911NP were identified as TCPP using infrared (IR) and ultraviolet-visible (UV/Vis) spectroscopy, GC/MS detection, and measurement of density. IR (Figure A-1), 1H NMR (Figure A-2), 13C NMR (Figure A-3), and UV/Vis (Figure A-4) spectra were consistent with the structure of TCPP. GC/MS identified one major peak and two isomer peaks for lot 101 before the 3-month studies (Table A-1, System B), and one major peak with three isomer peaks for lot M072911NP before the 2-year studies (Table A-1, System C). The major peak for both lots was identified as tris(1-chloro-2-propyl) phosphate (isomer 1, CASRN 13674-84-5) by comparison with a literature spectrum.114 The three other isomers in lot M072911NP were identified as bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (isomer 2, CASRN 76025-08-6), bis(2-chloropropyl) 2-chloroisopropyl phosphate (isomer 3, CASRN 76649-15-5), and tris(2-chloropropyl) phosphate (isomer 4, CASRN 6145-73-9) by comparing fragmentation patterns to structures of known isomers (Table 1, Table 2). The relative density of TCPP was determined to be 1.2936 ± 0.0004 (sd, standard deviation) at 21.0°C for lot 101 and 1.2959 ± 0.0001 (sd) at 21.5°C for lot M072911NP.
The moisture content as determined by Karl Fischer titration, the acid number, and the ester value were evaluated before the 3-month studies for lot 101 and before the 2-year studies for lot M072911NP. Acid number and ester value were determined for lots 101 and M072911NP using titration with standardized sodium hydroxide (approximately 0.001 N) and hydrochloric acid (approximately 0.5 N), respectively.
Analyses were conducted for lots 101 and 134 to ensure their acceptability before they were combined to form lot M072911NP, which was then used in the 2-year studies. These analyses included purity profiles of lots 101 and 134; elemental analysis of lot 101 for carbon, hydrogen, nitrogen, and chlorine conducted by ICON Development Services (Whitesboro, NY); and octonol:water partition coefficients (log P) for the two largest peaks in the profile of lot 101. After lots 101 and 134 were combined, the purity profile, elemental composition, and log P were determined for the combined lot, M072911NP.
For lot 101, Karl Fischer titration indicated a water content of 0.0997% ± 0.0019 (sd). Elemental analyses for carbon, hydrogen, nitrogen, and chlorine (33.02%, 5.64%, 0.14%, and 31.92%, respectively) were within 2% of theoretical values for TCPP. GC with flame ionization detection (FID) analysis (Table A-1, System E) detected four TCPP peaks with a combined relative area of 95.94%. Ten additional, unidentified impurities were present at concentrations of ≥0.05%. The largest peak in this analysis constituted 64.77% of the total peak area, and log P values for the two largest peaks in this profile were determined to be 2.69 and 2.74, respectively (Table A-1, System D). The average acid number for lot 101 was determined to be 0.011 ± 0.000 (sd) potassium hydroxide (KOH)/g, and the average ester value was calculated to be 104.7 mg KOH/g. The overall purity of lot 101 was determined to be approximately 95%.
For lot 134, the GC/FID (Table A-1, System F) purity profile detected four TCPP peaks with a combined relative area of 98.79% and three reportable impurities ≥0.05% of the total peak area. The largest peak constituted 71.33% of the total peak area. 1H NMR and 13C NMR spectra of lot 134 were consistent with the anticipated structure of TCPP. Coupled with the purity analysis, these results indicated that lot 134 was suitable for blending with lot 101 to constitute lot M072911NP.
Lot M072911NP was determined to contain 0.039% water by Karl Fischer titration. Elemental analyses for carbon, hydrogen, nitrogen, and chlorine (33.01%, 5.58%, 0.10%, and 32.10%, respectively) were within 2% of the theoretical values for TCPP. GC/FID detected four TCPP peaks (Figure A-5) accounting for a combined relative area of 97.24% using System F and 97.71% using System G (Table A-1). Eight additional, unidentified impurities with peak areas ≥0.05% were present in lot M072911NP. Low-level impurity determination was attempted to identify four minor impurities using an alternate GC/MS system (Table A-1, System H). The impurities could not be identified using National Institute of Standards and Technology (NIST) library spectra; however, they each appeared to have some structural similarities to the TCPP isomers. The major peak in this analysis constituted 68.06%–68.35% of the total peak area, and log P values for the two largest peaks in this profile were determined to be 2.59 and 2.65, respectively (Table A-1, System D). The average acid number for lot M072911NP was determined to be 0.067 ± 0.006 (sd) mg KOH/g, and the average ester value was calculated to be 105.85 mg KOH/g. Additionally, the extinction coefficient (Єmax) at 280 nm of lot M072911NP was calculated using absorbance values obtained from UV-Vis spectroscopy with a 1% solution of TCPP in methanol. The average Єmax was calculated as 3.8 ± 1.3 (sd). The overall purity of lot M072911NP was determined to be ≥97%. A summary of these analyses is given in Table 2.
Accelerated stability studies of lot 101 were conducted by the analytical chemistry laboratory using GC/FID and TCPP-1 (tris[1-chloro-2-propyl] phosphate; TCIPP) as the marker (Table A-1, System F). A single vial was stored at refrigerated (approximately 5°C), room (approximately 25°C), and elevated (approximately 60°C) temperatures for 2 weeks, then analyzed for purity relative to a frozen sample (approximately −20°C). Stability of the bulk chemical was confirmed for at least 2 weeks when stored in glass vials sealed with Teflon-lined crimp caps at temperatures up to 60°C.
A.2.
Preparation and Analysis of Dose Formulations
The base diet was meal feed purchased from Zeigler Brothers, Inc. (Gardners, PA). The 3-month rat study used NIH-07 feed (one lot milled June 2009) in addition to NTP-2000 feed (four lots milled July–October 2009). The 3-month mouse study used NTP-2000 feed (four lots milled July–October 2009). The 2-year rat study used NIH-07 feed (two lots milled September and October 2011) in addition to NTP-2000 feed (24 lots milled October 2011–October 2013). The 2-year mouse study used only NTP-2000 feed (24 lots milled August 2011–September 2013). Other than the analyses performed to determine the suitability of each lot used for feeding the animals, no further feed analysis was performed.
Dose formulations for the 3-month studies were prepared monthly by thoroughly mixing TCPP with feed (Table A-2). For the rat study, dose formulations were prepared with lot 101 at concentrations of 0, 2,500, 5,000, 10,000, 20,000, and 40,000 ppm in NIH-07 feed (July 23 and August 12, 2009) and at concentrations of 0, 2,500, 5,000, 10,000, 20,000 ppm in NTP-2000 feed (August 25, September 22, October 21, and November 18, 2009). For the mouse study, dose formulations were prepared with lot 101 at concentrations of 0, 1,250, 2,500, 5,000, 10,000 and 20,000 ppm in NTP-2000 feed (August 25, September 22, October 21, and November 18, 2009). Dose formulations for the 2-year studies were prepared monthly by thoroughly mixing TCPP with feed (Table A-3). For the rat study, dose formulations were prepared with lot M072911NP at concentrations of 0, 2,500, 5,000, 10,000, and 20,000 ppm in NIH-07 feed (October 18, November 7, and November 18, 2011) and in NTP-2000 feed (27 formulations; November 2011–November 2013). For the 2-year mouse study, formulations were prepared with lot M072911NP at concentrations of 0, 1,250, 2,500, 5,000, and 10,000 ppm in NTP-2000 feed (27 formulations; October 2011–October 2013).
Formulation development studies were conducted by the analytical laboratory MRIGlobal (Kansas City, MO) with a lot (M063008NP; Albemarle) separate from those used for the 3-month and 2-year studies. Formulations were analyzed using GC/FID (Table A-1, System I) and TCPP-1 as the marker. Homogeneity of TCPP in NIH-07 and NTP-2000 feed at 3,040 ppm and 50,000 ppm was tested, and all formulations analyzed were determined to be homogenous and of appropriate concentrations, with relative standard deviations ranging from 0.8% to 1.5%. A 7-day simulated dosing study was conducted on a 3,000 ppm TCPP formulation in NIH-07 and NTP-2000 feed spiked with rodent urine and feces stored in a stainless-steel hopper, at approximately 25°C and 50% relative humidity, and with a 12-hour daily light cycle. The results indicated a loss of ≤5% of TCPP in the formulation after 7 days. Stability studies were conducted on a 3,000 ppm dose formulation of TCPP, using TCPP-1 as the marker, in NIH-07 and NTP-2000 feeds. Test article stability was measured after 42 days at room (approximately 25ºC), refrigerated (approximately 5ºC), and frozen (approximately −20ºC) temperatures. In both feeds, it was concluded that TCPP formulation could be stored up to 14 days under frozen temperatures with no significant loss and up to 42 days frozen with ≤4.4% loss of TCPP. For all studies, dose formulations prepared in NIH-07 and NTP-2000 feed were stored frozen (−15ºC to −30ºC) in sealed containers protected from light and were used within 42 days after preparation.
Before the 3-month studies, the study laboratory conducted homogeneity studies of the 2,500 and 40,000 ppm dose formulations in 25 kg NIH-07 batch sizes and 1,250 and 40,000 ppm dose formulations in 25 kg NTP-2000 batch sizes using GC/FID (Table A-1, System J). Additional homogeneity studies of lot M072911NP at the 2,500 and 20,000 ppm dose formulations in 65 kg NIH-07 batch sizes and the 1,250 and 20,000 ppm dose formulations in 84 kg NTP-2000 batch sizes were performed before the 2-year studies by the study laboratory using GC/FID (Table A-1, System J). All formulations were determined to be homogenous and of appropriate concentration.
For the 3-month studies, periodic analyses of the preadministration dose formulations of TCPP were conducted by the study laboratory every 1 to 2 months to determine purity, whereas postadministration (animal room) samples were analyzed every month (Table A-4, Table A-5) using GC/FID (Table A-1, System K). All preadministration dose formulations for rats and mice were within 10% of the target concentrations. For the 3-month rat study, the postadministration feeder samples from the 2,500, 5,000, and 10,000 ppm formulations prepared on September 22, 2009, were 17.3%, 12.5%, and 13.4% below the target concentrations, respectively. Additionally, the feeder sample from the 2,500 ppm formulation prepared on November 18, 2009, was 10.1% below the target concentration. For the 3-month mouse study, postadministration feeder samples from the 1,250, 2,500, and 5,000 ppm formulations and the bucket sample from the 1,250 ppm formulation prepared on September 22, 2009, were 14.9%, 13.1%, 10.9%, and 11.2% below the target concentrations, respectively. All other postadministration values were within 10% of the target concentrations for the 3-month studies in rats and mice.
For the 2-year studies, periodic analyses of the preadministration dose formulations of TCPP were conducted by the study laboratory every 1 to 3 months to determine purity, whereas postadministration (animal room) samples were analyzed every 1 to 7 months (Table A-6, Table A-7) using GC/FID (Table A-1, System K). All preadministration dose formulations for rats and mice were within 10% of the target concentrations. For the 2-year rat study, the postadministration barrel samples from the 2,500 and 5,000 ppm formulations prepared on January 2, 2013, were 16.0% and 10.3% below the target concentrations, respectively. All other postadministration values were within 10% of the target concentrations for the 2-year studies in rats and mice.
Table A-1. Gas Chromatography Systems Used in the Three-month and Two-year Feed Studies of Tris(chloropropyl) Phosphate
| Detection System | Column | Carrier Gas | Oven Temperature Program |
|---|---|---|---|
| System A | |||
| Mass spectrometry (EI) | Agilent HP-5 (30 m × 0.32 mm, 1.0 μm film thickness) | Helium at 2.0 mL/minute | 170°C for 0 minutes, then 0.5°C/minute to 198°C, no hold |
| System B | |||
| Mass spectrometry (EI) | Agilent DB-5MS (30 m × 0.32 mm, 1.0 μm film thickness) | Helium at 3.5 mL/minute | 170°C for 0 minutes, then 0.5°C/minute to 198°C, no hold |
| System C | |||
| Mass spectrometry (EI) | RTX-5 with Integra Guard (30 m × 0.25 mm, 0.10 μm film thickness) | Helium at 2.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 45 minutes |
| System D | |||
| Flame ionization (300ºC) | Agilent DB-5 (30 m × 0.53 mm ID, 1.5 µm film thickness) | Helium at 10.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 5 minutes |
| System E | |||
| Flame ionization (300°C) | J&W DB-5 (30 m × 0.53 mm ID, 1.5 µm film thickness) | Helium at 10.0 mL/minute | 170°C for 10 minutes, then 0.5°C/minute to 180°C, held for 10 minutes, then 10°C/minute to 200°C, no hold |
| System F | |||
| Flame ionization (300ºC) | J&W Scientific DB-5 (30 m × 0.53 mm ID, 1.5 µm film thickness) | Helium at 10.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 15 minutes |
| System G | |||
| Flame ionization (300ºC) | J&W Scientific DB-Wax (30 m × 0.53 mm ID, 1.0 μm film thickness) | Helium at 10.0 mL/minute | 200°C for 5 minutes, then 1°C/minute to 230°C, held for 15 minutes |
| System H | |||
| Mass spectrometry (EI) | RTX-5 with Integra Guard (30 m x 0.25 mm, 0.10 μm film thickness) | Helium at 2.0 mL/minute for 50 minutes, then 2.0 mL/minute to 10 mL/minute, held for 22 minutes | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 25 minutes, then 10°C/minute to 300°C, held for 10 minutes |
| System I | |||
| Flame ionization (300°C) | J&W Scientific DB-5 (30 m × 0.53 mm ID, 1.5 µm film thickness) | Helium at 10.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 5 minutes |
| System J | |||
| Flame ionization (300ºC) | Restek RTX-5 (30 m × 0.53 mm ID, 1.5 μm film thickness) | Helium at 10.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 7 minutes |
| System K | |||
| Flame ionization (300ºC) | Restek RTX-5 (30 m × 0.53 mm ID, 1.5 μm film thickness) | Helium at 10.0 mL/minute | 160°C for 5 minutes, then 1°C/minute to 180°C, held for 5 minutes, then 5°C/minute to 230°C, no hold |
Table A-2. Preparation and Storage of Dose Formulations Administered to Rats and Mice in the Three-month Feed Studies of Tris(chloropropyl) Phosphate
| Preparation |
|---|
| A concentrated premixed stock was made by first weighing a specified amount of untreated feed in a stainless-steel weighing container, weighing a specified amount of test article in a separate weighing container, and then transferring the weighed test article and a small portion of the weighed untreated premix feed into the mixing bowl of a Hobart mixer. The test article and the untreated feed were stirred thoroughly while small amounts of untreated feed were added continually until the mixture appeared to be slightly damp. The weighing container holding the test article was “rinsed” at least three times with more untreated premix feed, which was transferred into the mixing bowl. An amount of untreated premix feed was added to the mixing bowl that equaled the approximate amount of weighed test article. The mixture was stirred with a spatula, if needed, until the mixing paddle on the Hobart mixer was able to rotate. With the mixer at a setting of 2 or lower, untreated premix feed was added in equal portions until all of the premix feed had been incorporated with the test article. The mixture was stirred for roughly 5 minutes longer to ensure thorough mixing. To prepare the dose formulations, approximately half of the required untreated feed for each formulation concentration was added to a Patterson-Kelley 3-ft3 twin shell blender until it was distributed evenly. The premixed stock was then added to the blender and distributed evenly over the feed. The stock container was “rinsed” at least three times with additional untreated feed, and the “rinse” was added to the blender. The remaining untreated feed required to reach the desired formulation concentration was added to the blender evenly over the premix, the blender was sealed, and the formulation was mixed for ~15 minutes with the intensifier bar on. The dose formulations were prepared monthly in this manner. |
| Chemical Lot Number |
| 101 |
| Maximum Storage Time |
| 41 days (NIH-07); 42 days (NTP-2000) |
| Storage Conditions |
| Stored in sealed, plastic bag-lined buckets at −15°C to −30°C |
| Study Laboratory |
| Battelle (Columbus, OH) |
Table A-3. Preparation and Storage of Dose Formulations Administered to Rats and Mice in the Two-year Feed Studies of Tris(chloropropyl) Phosphate
| Preparation |
|---|
| A concentrated premixed stock was made by first weighing a specified amount of untreated feed in a stainless-steel weighing container, weighing a specified amount of test article in a separate weighing container, and then transferring the weighed test article and a small portion of the weighed untreated premix feed into the mixing bowl of a Hobart mixer. The test article and the untreated feed were stirred thoroughly while small amounts of untreated feed were added continually until the mixture appeared to be slightly damp. The weighing container holding the test article was “rinsed” at least three times with more untreated premix feed, which was transferred into the mixing bowl. An amount of untreated premix feed was added to the mixing bowl that equaled the approximate amount of weighed test article. The mixture was stirred with a spatula, if needed, until the mixing paddle on the Hobart mixer was able to rotate. With the mixer at a setting of 2 or lower, untreated premix feed was added in equal portions until all of the premix feed had been incorporated with the test article. The mixture was stirred for roughly 5 minutes longer to ensure thorough mixing. To prepare the dose formulations, approximately half of the required untreated feed for each formulation concentration was added to a Patterson-Kelley 3-ft3 twin shell blender until it was distributed evenly. The premixed stock was then added to the blender and distributed evenly over the feed. The stock container was “rinsed” at least three times with additional untreated feed, and the “rinse” was added to the blender. The remaining untreated feed required to reach the desired formulation concentration was added to the blender evenly over the premix, the blender was sealed, and the formulation was mixed for ~15 minutes with the intensifier bar on. The dose formulations were prepared monthly in this manner. |
| Chemical Lot Number |
| M072911NP |
| Maximum Storage Time |
| 42 days |
| Storage Conditions |
| Stored in plastic bags within sealed plastic barrels at −15°C to −30°C |
| Study Laboratory |
| Battelle (Columbus, OH) |
Table A-4. Results of Analyses of Dose Formulations Administered to Rats in the Perinatal and Three-month Feed Study of Tris(chloropropyl) Phosphate
| Date Prepared | Date Analyzed | Target Concentration (ppm) | Determined Concentration (ppm)a | Difference from Target (%) |
|---|---|---|---|---|
| July 23, 2009 | July 29, 2009 | 0 | BLOQ | NA |
| 2,500 | 2,520 ± 92.9 | 0.9 | ||
| 5,000 | 5,070 ± 125.0 | 1.5 | ||
| 10,000 | 10,300 ± 305.5 | 3.3 | ||
| 20,000 | 20,000 ± 0.0 | 0.0 | ||
| 40,000b | 40,200 ± 288.7 | 0.6 | ||
| August 25, 2009 | August 27, 2009 | 0 | BLOQ | NA |
| 2,500 | 2,580 ± 20.8 | 3.1 | ||
| 5,000 | 5,300 ± 60.8 | 5.4 | ||
| 10,000 | 10,100 ± 115.5 | 1.3 | ||
| 20,000 | 20,000 ± 152.8 | 0.2 | ||
| September 22, 2009 | September 25, 2009 | 0 | BLOQ | NA |
| 2,500 | 2,440 ± 26.5 | −2.4 | ||
| 5,000 | 5,030 ± NAc | 0.6 | ||
| 10,000 | 10,500 ± 839 | 5.3 | ||
| 20,000 | 20,000 ± 231 | −0.2 | ||
| November 18, 2009 | November 20, 2009 | 0 | BLOQ | NA |
| 2,500 | 2,510 ± 104.4 | 0.4 | ||
| 5,000 | 5,160 ± 234.4 | 3.3 | ||
| 10,000 | 9,760 ± 118.5 | −2.4 | ||
| 20,000 | 19,700 ± 793.7 | −1.5 | ||
| Animal room samples | ||||
| July 23, 2009 | September 3, 2009 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,370 ± 17.3 | −5.2 | ||
| 5,000 | 4,840 ± 30.6 | −3.1 | ||
| 10,000 | 9,600 ± 64.3 | −3.9 | ||
| 20,000 | 19,600 ± 200.0 | −2.0 | ||
| 40,000b | NA | NA | ||
| July 23, 2009 | September 3, 2009 (bucket) | 0 | BLOQ | NA |
| 2,500 | 2,530 ± NAc | 1.2 | ||
| 5,000 | 4,970 ± 26.5 | −0.6 | ||
| 10,000 | 10,000 ± 202.1 | −0.3 | ||
| 20,000 | 19,600 ± 57.7 | −2.2 | ||
| 40,000 | 39,100 ± 300.0 | −2.3 | ||
| August 25, 2009 | October 9, 2009 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,340 ± 25.2 | −6.3 | ||
| 5,000 | 4,750 ± 5.8 | −4.9 | ||
| 10,000 | 9,340 ± 76.4 | −6.6 | ||
| 20,000 | 18,600 ± 152.8 | −6.8 | ||
| August 25, 2009 | October 9, 2009 (bucket) | 0 | BLOQ | NA |
| 2,500 | 2,450 ± 15.3 | −1.9 | ||
| 5,000 | 4,980 ± 15.3 | −0.3 | ||
| 10,000 | 9,760 ± 49.3 | −2.4 | ||
| 20,000 | 19,700 ± 288.7 | −1.3 | ||
| September 22, 2009 | November 6, 2009 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,070 ± 20.8 | −17.3 | ||
| 5,000 | 4,370 ± 66.6 | −12.5 | ||
| 10,000 | 8,660 ± 198.6 | −13.5 | ||
| 20,000 | 18,200 ± 260.6 | −9.0 | ||
| September 22, 2009 | November 6, 2009 (bucket) | 0 | BLOQ | NA |
| 2,500 | 2,280 ± 20.8 | −8.7 | ||
| 5,000 | 4,820 ± 10.0 | −3.6 | ||
| 10,000 | 9,370 ± 253.8 | −6.3 | ||
| 20,000 | 19,600 ± 351.2 | −2.2 | ||
| November 18, 2009 | December 18, 2009 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,250 ± 5.8 | −10.1 | ||
| 5,000 | 4,580 ± 20.0 | −8.4 | ||
| 10,000 | 9,260 ± 55.1 | −7.4 | ||
| 20,000 | 19,300 ± 360.6 | −3.5 | ||
| November 18, 2009 | December 18, 2009 (bucket) | 0 | BLOQ | NA |
| 2,500 | 2,390 ± 30.0 | −4.4 | ||
| 5,000 | 4,830 ± 10.0 | −3.4 | ||
| 10,000 | 9,850 ± 92.4 | −1.5 | ||
| 20,000 | 20,100 ± 1,006.6 | 0.3 | ||
Table A-5. Results of Analyses of Dose Formulations Administered to Mice in the Three-month Feed Study of Tris(chloropropyl) Phosphate
| Date Prepared | Date Analyzed | Target Concentration (ppm) | Determined Concentration (ppm)a | Difference from Target (%) |
|---|---|---|---|---|
| August 25, 2009 | August 27, 2009 | 0 | BLOQ | NA |
| 1,250 | 1,310 ± 15.3 | 4.5 | ||
| 2,500 | 2,580 ± 20.8 | 3.1 | ||
| 5,000 | 5,300 ± 60.8 | 5.4 | ||
| 10,000 | 10,100 ± 115.5 | 1.3 | ||
| 20,000 | 20,000 ± 152.8 | 0.2 | ||
| September 22, 2009 | September 25, 2009 | 0 | BLOQ | NA |
| 1,250 | 1,250 ± 32.1 | 0.3 | ||
| 2,500 | 2,440 ± 26.5 | −2.4 | ||
| 5,000 | 5,030 ± NAb | 0.6 | ||
| 10,000 | 10,500 ± 839 | 5.3 | ||
| 20,000 | 20,000 ± 231 | −0.2 | ||
| November 18, 2009 | November 20, 2009 | 0 | BLOQ | NA |
| 1,250 | 1,340 ± 25.2 | 6.9 | ||
| 2,500 | 2,510 ± 104.4 | 0.4 | ||
| 5,000 | 5,160 ± 234.4 | 3.3 | ||
| 10,000 | 9,760 ± 118.5 | −2.4 | ||
| 20,000 | 19,700 ± 793.7 | −1.5 | ||
| Animal room samples | ||||
| August 25, 2009 | October 9, 2009 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,260 ± 35.1 | 1.1 | ||
| 2,500 | 2,410 ± 10.0 | −3.6 | ||
| 5,000 | 5,010 ± 30.6 | 0.1 | ||
| 10,000 | 9,980 ± 125.8 | −0.2 | ||
| 20,000 | 19,200 ± 57.7 | −3.8 | ||
| August 25, 2009 | October 9, 2009 (bucket) | 0 | BLOQ | NA |
| 1,250 | 1,250 ± 15.3 | −0.3 | ||
| 2,500 | 2,490 ± 20.8 | −0.5 | ||
| 5,000 | 5,010 ± 11.5 | 0.1 | ||
| 10,000 | 9,940 ± 55.1 | −0.6 | ||
| 20,000 | 20,100 ± 173.2 | 0.5 | ||
| September 22, 2009 | November 6, 2009 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,060 ± 6 | −14.9 | ||
| 2,500 | 2,170 ± 123 | −13.1 | ||
| 5,000 | 4,460 ± 40 | −10.9 | ||
| 10,000 | 9,340 ± 242 | −6.6 | ||
| 20,000 | 18,900 ± 954 | −5.5 | ||
| September 22, 2009 | November 6, 2009 (bucket) | 0 | BLOQ | NA |
| 1,250 | 1,110 ± 10 | −11.2 | ||
| 2,500 | 2,300 ± 67 | −8.1 | ||
| 5,000 | 4,690 ± 74 | −6.3 | ||
| 10,000 | 9,530 ± 197 | −4.7 | ||
| 20,000 | 19,400 ± 115 | −3.2 | ||
| November 18, 2009 | December 18, 2009 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,160 ± 20.8 | −6.9 | ||
| 2,500 | 2,360 ± 86.2 | −5.7 | ||
| 5,000 | 4,880 ± 134.3 | −2.5 | ||
| 10,000 | 9,830 ± 378.6 | −1.7 | ||
| 20,000 | 19,200 ± 776.7 | −3.8 | ||
| November 18, 2009 | December 18, 2009 (bucket) | 0 | BLOQ | NA |
| 1,250 | 1,190 ± 20.8 | −4.5 | ||
| 2,500 | 2,400 ± 20.0 | −4.0 | ||
| 5,000 | 4,870 ± 23.1 | −2.7 | ||
| 10,000 | 9,970 ± 60.8 | −0.3 | ||
| 20,000 | 19,400 ± 351.2 | −3.2 | ||
Table A-6. Results of Analyses of Dose Formulations Administered to Rats in the Perinatal and Two-year Feed Study of Tris(chloropropyl) Phosphate
| Date Prepared | Date Analyzeda | Target Concentration (ppm)b | Determined Concentration (ppm)c | Difference from Target (%) |
|---|---|---|---|---|
| October 18, 2011 | October 18, 2011 | 0 | BLOQ | NA |
| 2,500 | 2,460 ± 10 | −1.6 | ||
| 5,000 | 5,000 ± 20 | 0.0 | ||
| 10,000 | 10,000 ± 100 | 0.1 | ||
| 20,000 | 20,100 ± 100 | 0.5 | ||
| November 10, 2011 | November 11, 2011 | 0 | BLOQ | NA |
| 2,500 | 2,480 ± 10 | −0.7 | ||
| 2,500 | 2,490 ± 40 | −0.5 | ||
| 5,000 | 4,950 ± 70 | −1.1 | ||
| 5,000 | 5,120 ± 120 | 2.3 | ||
| 10,000 | 9,990 ± 90 | −0.1 | ||
| 10,000 | 10,200 ± 300 | 1.8 | ||
| 20,000 | 20,100 ± 200 | 0.7 | ||
| 20,000 | 20,200 ± 300 | 1.2 | ||
| January 31, 2012 | February 2, 2012 | 0 | BLOQ | NA |
| 2,500 | 2,490 ± 10 | −0.4 | ||
| 2,500 | 2,500 ± 10 | 0.1 | ||
| 5,000 | 5,050 ± 80 | 0.9 | ||
| 5,000 | 4,980 ± 20 | −0.5 | ||
| 10,000 | 9,860 ± 40 | −1.4 | ||
| 10,000 | 9,890 ± 10 | −1.1 | ||
| 20,000 | 19,800 ± 200 | −1.0 | ||
| 20,000 | 19,700 ± 300 | −1.3 | ||
| March 28, 2012 | March 29, 2012 | 0 | BLOQ | NA |
| 2,500 | 2,480 ± 0 | −0.8 | ||
| 2,500 | 2,500 ± 60 | 0.0 | ||
| 5,000 | 4,920 ± 20 | −1.6 | ||
| 5,000 | 4,950 ± 70 | −0.9 | ||
| 10,000 | 9,970 ± 20 | −0.3 | ||
| 10,000 | 9,900 ± 40 | −1.0 | ||
| 20,000 | 19,900 ± 100 | −0.5 | ||
| 20,000 | 19,800 ± 0 | −1.0 | ||
| June 19, 2012 | June 20, 2012 | 0 | BLOQ | NA |
| 2,500 | 2,370 ± 30 | −5.2 | ||
| 2,500 | 2,380 ± 30 | −4.7 | ||
| 5,000 | 4,850 ± 20 | −3.1 | ||
| 5,000 | 4,820 ± 40 | −3.6 | ||
| 10,000 | 9,640 ± 30 | −3.6 | ||
| 10,000 | 9,680 ± 60 | −3.2 | ||
| 20,000 | 19,400 ± 200 | −2.8 | ||
| 20,000 | 19,200 ± 200 | −4.2 | ||
| August 14, 2012 | August 14, 2012 | 0 | BLOQ | NA |
| 2,500 | 2,380 ± 100 | −4.7 | ||
| 2,500 | 2,490 ± 20 | −0.3 | ||
| 5,000 | 5,050 ± 20 | 1.1 | ||
| 5,000 | 5,020 ± 30 | 0.3 | ||
| 10,000 | 10,200 ± 100 | 2.3 | ||
| 10,000 | 10,200 ± 100 | 1.7 | ||
| 20,000 | 20,800 ± 200 | 3.8 | ||
| 20,000 | 20,200 ± 200 | 1.0 | ||
| November 8, 2012 | November 8, 2012 | 0 | BLOQ | NA |
| 2,500 | 2,510 ± 10 | 0.3 | ||
| 2,500 | 2,490 ± 10 | −0.4 | ||
| 5,000 | 4,960 ± 10 | −0.8 | ||
| 5,000 | 4,950 ± 20 | −1.1 | ||
| 10,000 | 10,000 ± 200 | 0.2 | ||
| 10,000 | 9,930 ± 150 | −0.7 | ||
| 20,000 | 19,700 ± 100 | −1.7 | ||
| 20,000 | 19,600 ± 100 | −1.8 | ||
| January 2, 2013 | January 4, 2013 | 0 | BLOQ | NA |
| 2,500 | 2,500 ± 10 | 0.1 | ||
| 2,500 | 2,510 ± 10 | 0.5 | ||
| 5,000 | 5,000 ± 10 | −0.1 | ||
| 5,000 | 5,020 ± 0 | 0.4 | ||
| 10,000 | 10,100 ± 100 | 0.5 | ||
| 10,000 | 10,100 ± 100 | 0.7 | ||
| 20,000 | 20,400 ± 600 | 1.8 | ||
| 20,000 | 20,000 ± 200 | −0.2 | ||
| March 26, 2013 | March 27, 2013 | 0 | BLOQ | NA |
| 2,500 | 2,550 ± 10 | 1.9 | ||
| 2,500 | 2,560 ± 10 | 2.5 | ||
| 5,000 | 4,990 ± 40 | −0.2 | ||
| 5,000 | 5,020 ± 20 | 0.4 | ||
| 10,000 | 10,200 ± 200 | 2.0 | ||
| 10,000 | 9,990 ± 90 | −0.1 | ||
| 20,000 | 19,900 ± 100 | −0.5 | ||
| 20,000 | 19,800 ± 100 | −0.8 | ||
| May 22, 2013 | May 23, 2013 | 0 | BLOQ | NA |
| 2,500 | 2,560 ± 20 | 2.3 | ||
| 2,500 | 2,580 ± 110 | 3.1 | ||
| 5,000 | 5,080 ± 20 | 1.5 | ||
| 5,000 | 4,980 ± 10 | −0.3 | ||
| 10,000 | 10,100 ± 100 | 0.8 | ||
| 10,000 | 10,100 ± 100 | 0.7 | ||
| 20,000 | 20,000 ± 100 | −0.2 | ||
| 20,000 | 20,700 ± 500 | 3.3 | ||
| August 15, 2013 | August 15, 2013 | 0 | BLOQ | NA |
| 2,500 | 2,530 ± 0 | 1.2 | ||
| 2,500 | 2,530 ± 30 | 1.1 | ||
| 5,000 | 4,970 ± 30 | −0.6 | ||
| 5,000 | 4,960 ± 30 | −0.7 | ||
| 10,000 | 9,970 ± 30 | −0.3 | ||
| 10,000 | 9,980 ± 30 | −0.2 | ||
| 20,000 | 19,800 ± 300 | −1.0 | ||
| 20,000 | 19,700 ± 400 | −1.7 | ||
| October 16, 2013 | October 15, 2013 | 0 | BLOQ | NA |
| 2,500 | 2,560 ± 20 | 2.5 | ||
| 2,500 | 2,550 ± 20 | 2.1 | ||
| 5,000 | 5,000 ± 80 | 0.1 | ||
| 5,000 | 5,000 ± 10 | 0.0 | ||
| 10,000 | 10,000 ± 100 | 0.2 | ||
| 10,000 | 9,950 ± 50 | −0.5 | ||
| 20,000 | 19,900 ± 100 | −0.5 | ||
| 20,000 | 19,900 ± 200 | −0.3 | ||
| Animal room samples | ||||
| October 18, 2011 | November 21, 2011 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,480 ± 30 | −0.9 | ||
| 5,000 | 4,930 ± 50 | −1.5 | ||
| 10,000 | 9,950 ± 130 | −0.5 | ||
| 20,000 | 20,000 ± 300 | 0.0 | ||
| October 18, 2011 | November 21, 2011 (barrel) | 0 | BLOQ | NA |
| 2,500 | 2,490 ± 20 | −0.5 | ||
| 5,000 | 4,990 ± 30 | −0.2 | ||
| 10,000 | 10,000 ± 100 | 0.2 | ||
| 20,000 | 20,100 ± 100 | 0.5 | ||
| November 10, 2011 | December 20, 2011 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,350 ± 20 | −6.1 | ||
| 5,000 | 4,740 ± 10 | −5.3 | ||
| 10,000 | 9,320 ± 170 | −6.8 | ||
| 20,000 | 18,200 ± 100 | −8.8 | ||
| November 10, 2011 | December 20, 2011 (barrel) | 0 | BLOQ | NA |
| 2,500 | 2,470 ± 30 | −1.1 | ||
| 5,000 | 5,000 ± 90 | −0.1 | ||
| 10,000 | 10,600 ± 600 | 6.0 | ||
| 20,000 | 20,300 ± 800 | 1.5 | ||
| June 19, 2012 | July 31, 2012 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,390 ± 20 | −4.5 | ||
| 5,000 | 5,000 ± 10 | 0.0 | ||
| 10,000 | 10,000 ± 100 | 0.0 | ||
| 20,000 | 20,600 ± 300 | 3.0 | ||
| June 19, 2012 | July 31, 2012 (barrel) | 0 | BLOQ | NA |
| 2,500 | 2,560 ± 20 | 2.5 | ||
| 5,000 | 4,700 ± 40 | −5.9 | ||
| 10,000 | 9,310 ± 100 | −6.9 | ||
| 20,000 | 19,000 ± 200 | −4.8 | ||
| January 2, 2013 | February 14, 2013 (feeder) | |||
| 2,500 | 2,100 ± 50 | −16.0 | ||
| 5,000 | 4,490 ± 170 | −10.3 | ||
| 10,000 | 9,400 ± 40 | −6.0 | ||
| 20,000 | 19,400 ± 100 | −2.8 | ||
| January 2, 2013 | February 14, 2013 (barrel) | 0 | BLOQ | NA |
| 2,500 | 2,450 ± 20 | −2.0 | ||
| 5,000 | 4,950 ± 20 | −1.0 | ||
| 10,000 | 9,960 ± 50 | −0.4 | ||
| 20,000 | 19,900 ± 100 | −0.7 | ||
| August 15, 2013 | September 24, 2013 (feeder) | 0 | BLOQ | NA |
| 2,500 | 2,400 ± 20 | −4.0 | ||
| 5,000 | 4,700 ± 40 | −6.1 | ||
| 10,000 | 9,800 ± 100 | −2.0 | ||
| 20,000 | 19,200 ± 300 | −3.8 | ||
| August 15, 2013 | September 24, 2013 (barrel) | 0 | BLOQ | NA |
| 2,500 | 2,520 ± 10 | 0.7 | ||
| 5,000 | 4,970 ± 20 | −0.7 | ||
| 10,000 | 9,780 ± 70 | −2.2 | ||
| 20,000 | 18,900 ± 100 | −5.3 | ||
Table A-7. Results of Analyses of Dose Formulations Administered to Mice in the Two-year Feed Study of Tris(chloropropyl) Phosphate
| Date Prepared | Date Analyzeda | Target Concentration (ppm)b | Determined Concentration (ppm)c | Difference from Target (%) |
|---|---|---|---|---|
| October 11, 2011 | October 11, 2011 | 0 | BLOQ | NA |
| 1,250 | 1,270 ± 10 | 1.3 | ||
| 2,500 | 2,500 ± 10 | 0.0 | ||
| 5,000 | 4,960 ± 40 | −0.9 | ||
| 10,000 | 9,960 ± 120 | −0.4 | ||
| November 10, 2011 | November 11, 2011 | 0 | BLOQ | NA |
| 1,250 | 1,250 ± 20 | 0.3 | ||
| 2,500 | 2,480 ± 10 | −0.7 | ||
| 2,500 | 2,490 ± 40 | −0.5 | ||
| 5,000 | 4,950 ± 70 | −1.1 | ||
| 5,000 | 5,120 ± 120 | 2.3 | ||
| 10,000 | 9,990 ± 90 | −0.1 | ||
| 10,000 | 10,200 ± 300 | 1.8 | ||
| January 31, 2012 | February 2, 2012 | 0 | BLOQ | NA |
| 1,250 | 1,260 ± 10 | 0.5 | ||
| 2,500 | 2,490 ± 10 | −0.4 | ||
| 2,500 | 2,500 ± 10 | 0.1 | ||
| 5,000 | 5,050 ± 80 | 0.9 | ||
| 5,000 | 4,980 ± 20 | −0.5 | ||
| 10,000 | 9,860 ± 40 | −1.4 | ||
| 10,000 | 9,890 ± 10 | −1.1 | ||
| March 28, 2012 | March 29, 2012 | 0 | BLOQ | NA |
| 1,250 | 1,210 ± 10 | −2.9 | ||
| 2,500 | 2,480 ± 0 | −0.8 | ||
| 2,500 | 2,500 ± 60 | 0.0 | ||
| 5,000 | 4,920 ± 20 | −1.6 | ||
| 5,000 | 4,950 ± 70 | −0.9 | ||
| 10,000 | 9,970 ± 20 | −0.3 | ||
| 10,000 | 9,900 ± 40 | −1.0 | ||
| June 19, 2012 | June 20, 2012 | 0 | BLOQ | NA |
| 1,250 | 1,170 ± 10 | −6.7 | ||
| 2,500 | 2,370 ± 30 | −5.2 | ||
| 2,500 | 2,380 ± 30 | −4.7 | ||
| 5,000 | 4,850 ± 20 | −3.1 | ||
| 5,000 | 4,820 ± 40 | −3.6 | ||
| 10,000 | 9,640 ± 30 | −3.6 | ||
| 10,000 | 9,680 ± 60 | −3.2 | ||
| August 14, 2012 | August 14, 2012 | 0 | BLOQ | NA |
| 1,250 | 1,210 ± 10 | −3.2 | ||
| 2,500 | 2,380 ± 100 | −4.7 | ||
| 2,500 | 2,490 ± 20 | −0.3 | ||
| 5,000 | 5,050 ± 20 | 1.1 | ||
| 5,000 | 5,020 ± 30 | 0.3 | ||
| 10,000 | 10,200 ± 100 | 2.3 | ||
| 10,000 | 10,200 ± 100 | 1.7 | ||
| November 8, 2012 | November 8, 2012 | 0 | BLOQ | NA |
| 1,250 | 1,260 ± 20 | 0.5 | ||
| 2,500 | 2,510 ± 10 | 0.3 | ||
| 2,500 | 2,490 ± 10 | −0.4 | ||
| 5,000 | 4,960 ± 10 | −0.8 | ||
| 5,000 | 4,950 ± 20 | −1.1 | ||
| 10,000 | 10,000 ± 200 | 0.2 | ||
| 10,000 | 9,930 ± 150 | −0.7 | ||
| January 2, 2013 | January 4, 2013 | 0 | BLOQ | NA |
| 1,250 | 1,260 ± 10 | 0.5 | ||
| 2,500 | 2,500 ± 10 | 0.1 | ||
| 2,500 | 2,510 ± 10 | 0.5 | ||
| 5,000 | 5,000 ± 10 | −0.1 | ||
| 5,000 | 5,020 ± 0 | 0.4 | ||
| 10,000 | 10,100 ± 100 | 0.5 | ||
| 10,000 | 10,100 ± 100 | 0.7 | ||
| March 26, 2013 | March 27, 2013 | 0 | BLOQ | NA |
| 1,250 | 1,320 ± 10 | 5.6 | ||
| 2,500 | 2,550 ± 10 | 1.9 | ||
| 2,500 | 2,560 ± 10 | 2.5 | ||
| 5,000 | 4,990 ± 40 | −0.2 | ||
| 5,000 | 5,020 ± 20 | 0.4 | ||
| 10,000 | 10,200 ± 200 | 2.0 | ||
| 10,000 | 9,990 ± 90 | −0.1 | ||
| May 22, 2013 | May 23, 2013 | 0 | BLOQ | NA |
| 1,250 | 1,330 ± 10 | 6.1 | ||
| 2,500 | 2,560 ± 20 | 2.3 | ||
| 2,500 | 2,580 ± 110 | 3.1 | ||
| 5,000 | 5,080 ± 20 | 1.5 | ||
| 5,000 | 4,980 ± 10 | −0.3 | ||
| 10,000 | 10,100 ± 100 | 0.8 | ||
| 10,000 | 10,100 ± 100 | 0.7 | ||
| August 15, 2013 | August 15, 2013 | 0 | BLOQ | NA |
| 1,250 | 1,280 ± 10 | 2.7 | ||
| 2,500 | 2,530 ± 0 | 1.2 | ||
| 2,500 | 2,530 ± 30 | 1.1 | ||
| 5,000 | 4,970 ± 30 | −0.6 | ||
| 5,000 | 4,960 ± 30 | −0.7 | ||
| 10,000 | 9,970 ± 30 | −0.3 | ||
| 10,000 | 9,980 ± 30 | −0.2 | ||
| October 16, 2013 | October 15, 2013 | 0 | BLOQ | NA |
| 1,250 | 1,310 ± 0 | 4.8 | ||
| 2,500 | 2,560 ± 20 | 2.5 | ||
| 2,500 | 2,550 ± 20 | 2.1 | ||
| 5,000 | 5,000 ± 80 | 0.1 | ||
| 5,000 | 5,000 ± 10 | 0.0 | ||
| 10,000 | 10,000 ± 100 | 0.2 | ||
| 10,000 | 9,950 ± 50 | −0.5 | ||
| Animal room samples | ||||
| October 11, 2011 | November 22, 2011 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,150 ± 10 | −8.3 | ||
| 2,500 | 2,320 ± 10 | −7.1 | ||
| 5,000 | 4,570 ± 110 | −8.5 | ||
| 10,000 | 9,180 ± 20 | −8.2 | ||
| October 11, 2011 | November 22, 2011 (barrel) | 0 | BLOQ | NA |
| 1,250 | 1,190 ± 20 | −4.5 | ||
| 2,500 | 2,430 ± 20 | −2.9 | ||
| 5,000 | 4,970 ± 100 | −0.6 | ||
| 10,000 | 9,780 ± 30 | −2.2 | ||
| November 10, 2011 | December 20, 2011 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,210 ± 10 | −3.5 | ||
| 2,500 | 2,410 ± 20 | −3.7 | ||
| 5,000 | 4,810 ± 10 | −3.8 | ||
| 10,000 | 9,010 ± 60 | −9.9 | ||
| November 10, 2011 | December 20, 2011 (barrel) | 0 | BLOQ | NA |
| 1,250 | 1,260 ± 10 | 0.5 | ||
| 2,500 | 2,470 ± 30 | −1.1 | ||
| 5,000 | 5,000 ± 90 | −0.1 | ||
| 10,000 | 10,600 ± 600 | 6.0 | ||
| June 19, 2012 | July 31, 2012 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,290 ± 10 | 2.9 | ||
| 2,500 | 2,470 ± 10 | −1.2 | ||
| 5,000 | 4,890 ± 50 | −2.2 | ||
| 10,000 | 9,430 ± 80 | −5.7 | ||
| June 19, 2012 | July 31, 2012 (barrel) | 0 | BLOQ | NA |
| 1,250 | 1,290 ± 10 | 2.9 | ||
| 2,500 | 2,560 ± 20 | 2.5 | ||
| 5,000 | 4,700 ± 40 | −5.9 | ||
| 10,000 | 9,310 ± 100 | −6.9 | ||
| January 2, 2013 | February 14, 2013 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,170 ± 20 | −6.4 | ||
| 2,500 | 2,450 ± 10 | −2.0 | ||
| 5,000 | 4,930 ± 10 | −1.3 | ||
| 10,000 | 9,380 ± 70 | −6.2 | ||
| January 2, 2013 | February 14, 2013 (barrel) | 0 | BLOQ | NA |
| 1,250 | 1,210 ± 10 | −3.5 | ||
| 2,500 | 2,450 ± 20 | −2.0 | ||
| 5,000 | 4,950 ± 20 | −1.0 | ||
| 10,000 | 9,960 ± 50 | −0.4 | ||
| August 15, 2013 | September 24, 2013 (feeder) | 0 | BLOQ | NA |
| 1,250 | 1,250 ± 10 | −0.3 | ||
| 2,500 | 2,420 ± 10 | −3.1 | ||
| 5,000 | 4,790 ± 60 | −4.1 | ||
| 10,000 | 9,510 ± 70 | −4.9 | ||
| August 15, 2013 | September 24, 2013 (barrel) | 0 | BLOQ | NA |
| 1,250 | 1,220 ± 30 | −2.1 | ||
| 2,500 | 2,520 ± 10 | 0.7 | ||
| 5,000 | 4,970 ± 20 | −0.7 | ||
| 10,000 | 9,780 ± 70 | −2.2 | ||
Figure A-1. Infrared Absorption Spectrum of Tris(chloropropyl) Phosphate (Lot M072911NP)
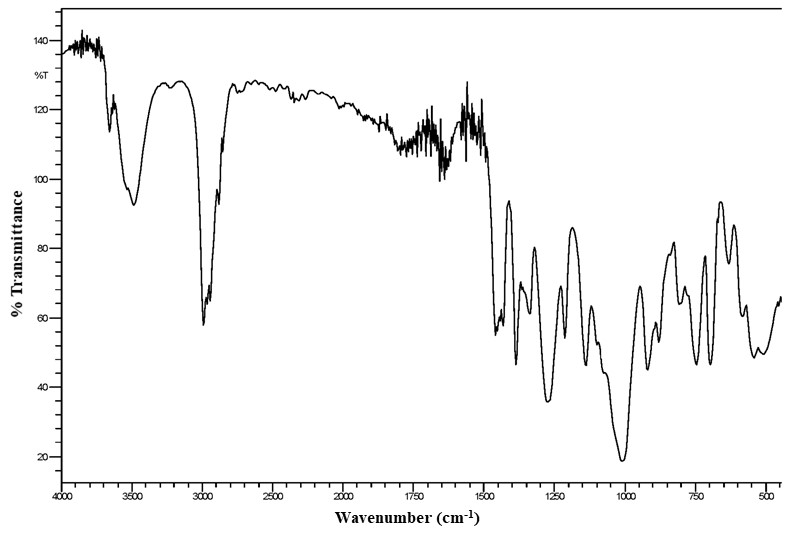
Figure A-2. 1H Nuclear Magnetic Resonance Spectrum of Tris(chloropropyl) Phosphate (Lot M072911NP)
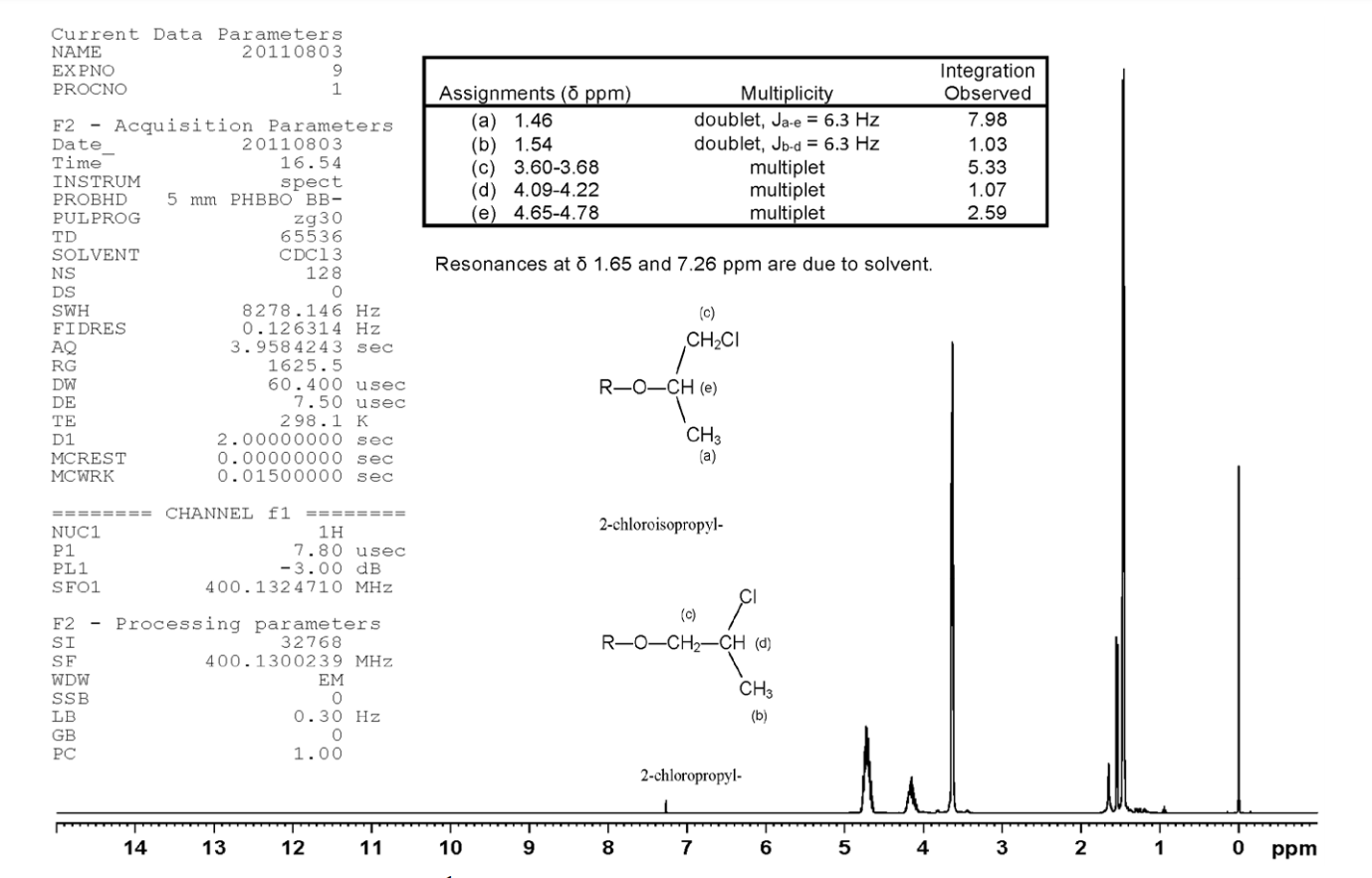
Figure A-3. 13C Nuclear Magnetic Resonance Spectrum of Tris(chloropropyl) Phosphate (Lot M072911NP)
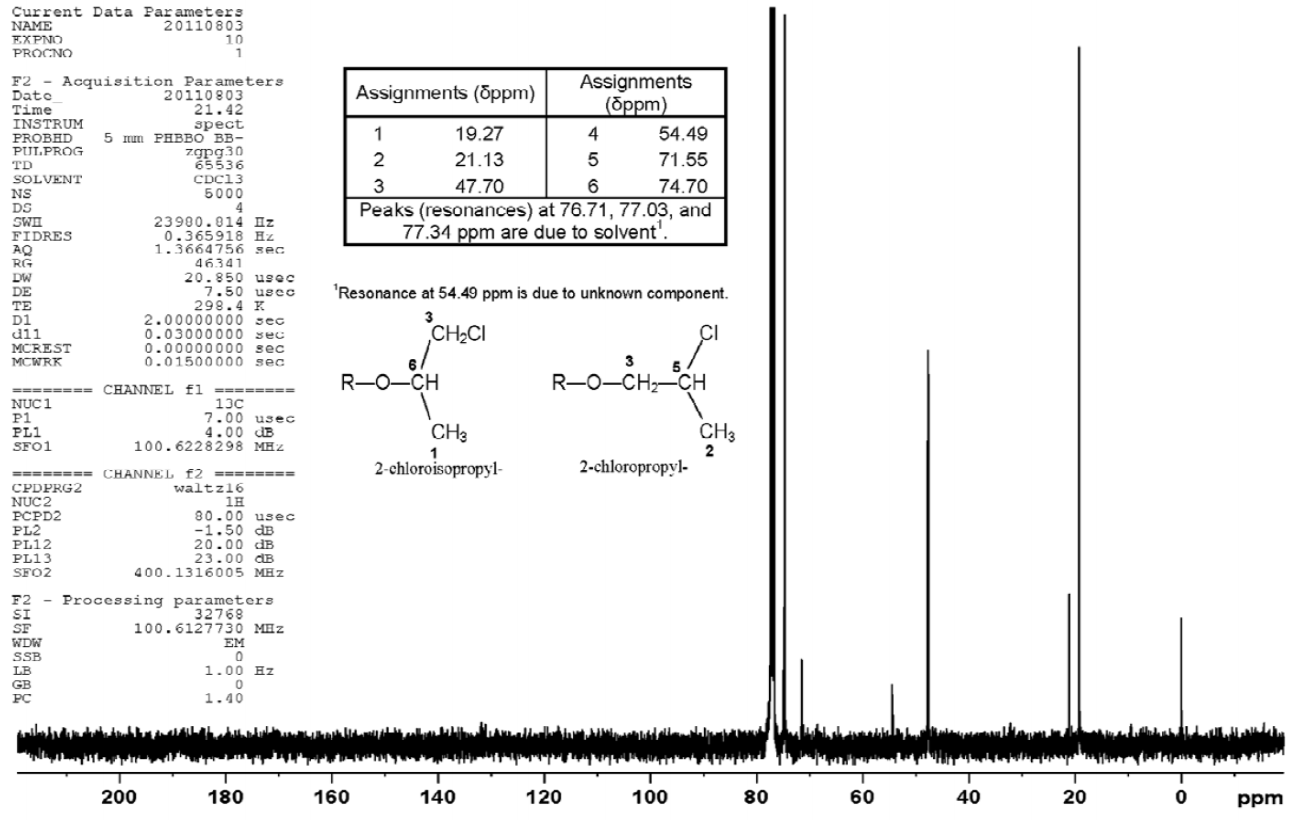
Figure A-4. Ultraviolet/Visible Spectrum of Tris(chloropropyl) Phosphate (Lot M072911NP)
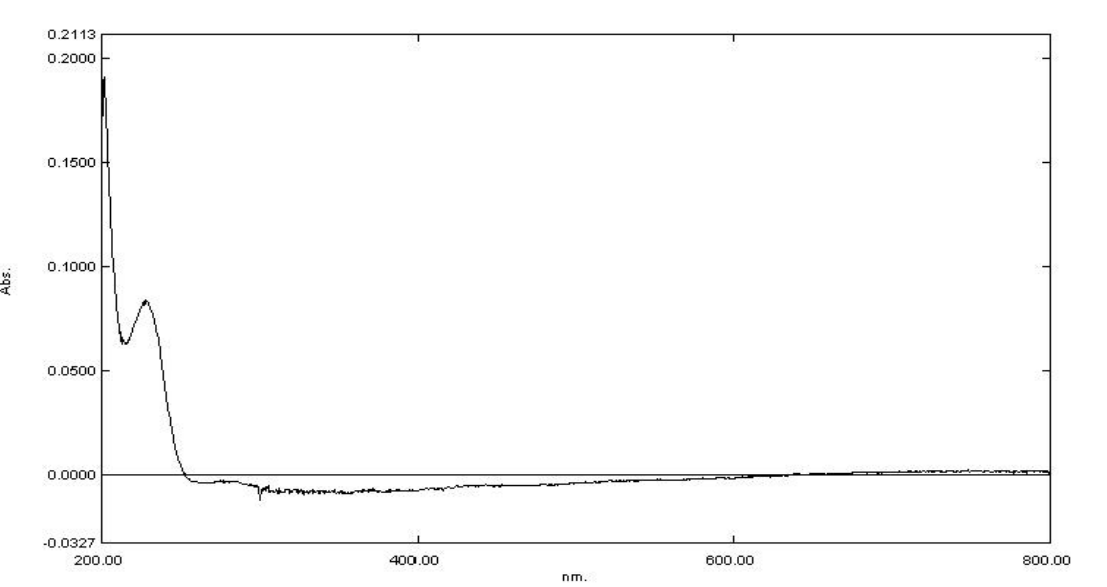
Figure A-5. Gas Chromatogram of Tris(chloropropyl) Phosphate (Lot M072911NP)
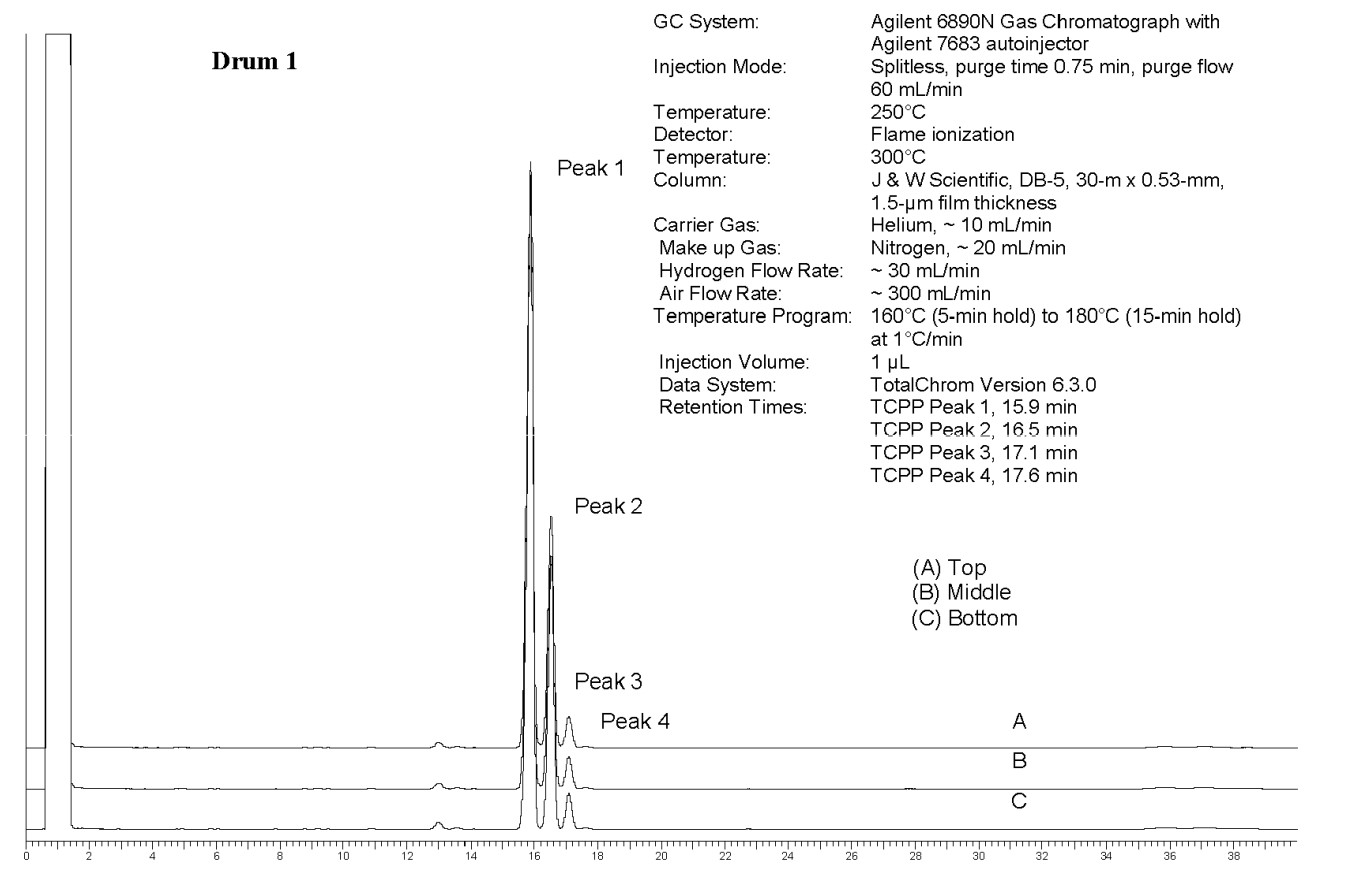
Appendix B. Ingredients, Nutrient Composition, and Contaminant Levels in NIH-07 and NTP-2000 Rat and Mouse Ration
B.1.
NIH-07 Feed
Table B-1. Ingredients of NIH-07 Rat Ration
| Ingredients | Percent by Weight |
|---|---|
| Ground hard winter wheat | 23.00 |
| Ground #2 yellow shelled corn | 24.25 |
| Wheat middlings | 10.0 |
| Oat hulls | 0.0 |
| Alfalfa meal (dehydrated, 17% protein) | 4.0 |
| Purified cellulose | 0.0 |
| Soybean meal (47% protein) | 12.0 |
| Fish meal (62% protein) | 10.0 |
| Corn oil (without preservatives) | 0.0 |
| Soy oil (without preservatives) | 2.5 |
| Dried brewer’s yeast | 2.0 |
| Calcium carbonate (USP) | 0.5 |
| Vitamin premixa | 0.25 |
| Mineral premixb | 0.15 |
| Calcium phosphate, dibasic (USP) | 1.25 |
| Sodium chloride | 0.5 |
| Choline chloride (70% choline) | 0.10 |
| Dried skim milk | 5.00 |
| Dried molasses | 1.50 |
| Corn gluten meal (60% protein) | 3.00 |
| Methionine | 0.0 |
Table B-2. Vitamins and Minerals in NIH-07 Rat Ration
| Amounta | Source | ||
|---|---|---|---|
| Vitamins | |||
| Vitamin A | 6,062 IU | Stabilized vitamin A palmitate or acetate | |
| Vitamin D | 5,070 IU | D-activated animal sterol | |
| Vitamin K | 3.1 mg | Menadione sodium bisulfite complex | |
| Vitamin E | 22 IU | α-Tocopheryl acetate | |
| Niacin | 33 mg | – | |
| Folic acid | 2.4 mg | – | |
| d-Pantothenic acid | 19.8 mg | d-Calcium pantothenate | |
| Riboflavin | 3.8 mg | – | |
| Thiamine | 11 mg | Thiamine mononitrate | |
| B12 | 50 μg | – | |
| Pyridoxine | 6.5 mg | Pyridoxine hydrochloride | |
| Biotin | 0.15 mg | d-Biotin | |
| Minerals | |||
| Iron | 132 mg | Iron sulfate | |
| Zinc | 18 mg | Zinc oxide | |
| Manganese | 66 mg | Manganese oxide | |
| Copper | 4.4 mg | Copper sulfate | |
| Iodine | 2.0 mg | Calcium iodate | |
| Cobalt | 0.44 mg | Cobalt carbonate | |
Table B-3. Nutrient Composition of NIH-07 Rat Ration
| Nutrient | Mean ± Standard Deviation | Range | Number of Samples |
|---|---|---|---|
| Protein (% by weight) | 24.28 ± 1.03 | 23.2–25.6 | 4 |
| Crude fat (% by weight) | 5.2 ± 0.183 | 5.0–5.4 | 4 |
| Crude fiber (% by weight) | 3.155 ± 0.160 | 3.0–3.36 | 4 |
| Ash (% by weight) | 6.29 ± 0.361 | 5.92–6.67 | 4 |
| Amino acids (% of total diet) | |||
| Arginine | 1.278 ± 0.343 | 0.258–1.49 | 11 |
| Cystine | 0.307 ± 0.059 | 0.153–0.372 | 11 |
| Glycine | 1.065 ± 0.289 | 0.217–1.31 | 11 |
| Histidine | 0.482 ± 0.121 | 0.125–0.553 | 11 |
| Isoleucine | 0.914 ± 0.233 | 0.214–1.03 | 11 |
| Leucine | 1.873 ± 0.485 | 0.423–2.13 | 11 |
| Lysine | 1.140 ± 0.345 | 0.111–1.32 | 11 |
| Methionine | 0.453 ± 0.117 | 0.102–0.515 | 11 |
| Phenylalanine | 1.023 ± 0.245 | 0.286–1.12 | 11 |
| Threonine | 0.850 ± 0.228 | 0.168–0.961 | 11 |
| Tryptophan | 0.259 ± 0.064 | 0.076–0.326 | 11 |
| Tyrosine | 0.801 ± 0.200 | 0.209–0.894 | 11 |
| Valine | 1.055 ± 0.264 | 0.262–1.17 | 11 |
| Essential fatty acids (% of total diet) | |||
| Linoleic | 2.436 ± 0.489 | 0.199–3.77 | 11 |
| Linolenic | 0.367 ± 0.397 | 0.214–1.56 | 11 |
| Vitamins | |||
| Vitamin A (IU/kg) | 5,178 ± 117 | 3,940–3,400 | 4 |
| Vitamin D (IU/kg) | 1,000a | – | – |
| α-Tocopherol (ppm) | 6,097 ± 20,067 | 31.36–66,600 | 11 |
| Thiamine (ppm)b | 14.975 ± 2.346 | 11.7–17.1 | 4 |
| Riboflavin (ppm) | 13.54 ± 4.438 | 4.2–19.8 | 11 |
| Niacin (ppm) | 95.02 ± 16.30 | 51.9–112.0 | 11 |
| Pantothenic acid (ppm) | 40.69 ± 12.76 | 3.8–51.1 | 11 |
| Pyridoxine (ppm)b | 11.74 ± 4.81 | 0.42–19.7 | 11 |
| Folic acid (ppm) | 2.38 ± 0.571 | 1.37–3.09 | 11 |
| Biotin (ppm) | 0.300 ± 0.187 | 0.0–0.638 | 11 |
| B12 (ppb) | 45.27 ± 15.14 | 4.0–61.6 | 11 |
| Choline (as chloride) (ppm) | 1,719.0 ± 386.0 | 700.0–2,200.0 | 11 |
| Minerals | |||
| Calcium (%) | 1.096 ± 0.159 | 0.949–1.29 | 4 |
| Phosphorus (%) | 0.894 ± 0.073 | 0.819–0.970 | 4 |
| Potassium (%) | 0.762 ± 0.226 | 0.088–0.88 | 11 |
| Chloride (%) | 0.656 ± 0.102 | 0.411–0.8 | 11 |
| Sodium (%) | 0.409 ± 0.112 | 0.318–0.721 | 11 |
| Magnesium (%) | 0.171 ± 0.053 | 0.0162–0.218 | 11 |
| Iron (ppm) | 353.3 ± 117.5 | 35.7–469.0 | 11 |
| Manganese (ppm) | 82.88 ± 27.28 | 3.53–104.0 | 11 |
| Zinc (ppm) | 58.75 ± 20.3 | 4.74–89.2 | 11 |
| Copper (ppm) | 12.91 ± 4.73 | 0.683–21.1 | 11 |
| Iodine (ppm) | 1.647 ± 1.088 | 0.0–3.45 | 11 |
| Chromium (ppm) | 3.95 ± 0.035 | 3.89–4.0 | 11 |
| Cobalt (ppm) | 0.470 ± 0.296 | 0.01–0.963 | 11 |
Table B-4. Contaminant Levels in NIH-07 Rat Ration
| Mean ± Standard Deviation | Range | Number of Samples | |
|---|---|---|---|
| Contaminants | |||
| Arsenic (ppm) | 0.370 ± 0.121 | 0.267–0.545 | 4 |
| Cadmium (ppm) | 0.079 ± 0.012 | 0.065–0.092 | 4 |
| Lead (ppm) | 0.089 ± 0.021 | 0.065–0.116 | 4 |
| Mercury (ppm) | 0.0185 ± 0.005 | 0.014–0.026 | 4 |
| Selenium (ppm) | 0.463 ± 0.041 | 0.431–0.521 | 4 |
| Aflatoxins (ppb)a | <5.0 | – | 4 |
| Nitrate nitrogen (ppm)b | 22.0 ± 7.040 | 15.2–30.8 | 4 |
| Nitrite nitrogen (ppm)a,b | <0.61 | – | 4 |
| BHA (ppm)a,c | <1.0 | – | 4 |
| BHT (ppm)a,c | <1.0 | – | 4 |
| Aerobic plate count (CFU/g) | <10.0 | – | 4 |
| Coliform (MPN/g) | <3.0 | – | 4 |
| Escherichia coli (MPN/g)a | <10.0 | – | 4 |
| Salmonella sp. (MPN/g) | Negative | – | 4 |
| Total nitrosamines (ppb)d | 11.48 ± 3.19 | 7.8–15.1 | 4 |
| N-Nitrosodimethylamine (ppb)d | 6.075 ± 3.92 | 2.5–9.9 | 4 |
| N-Nitrosopyrrolidine (ppb)d | 5.4 ± 1.359 | 3.9–7.2 | 4 |
| Pesticides (ppm) | |||
| α-BHCa | <0.01 | – | 4 |
| β-BHCa | <0.02 | – | 4 |
| γ-BHCa | <0.01 | – | 4 |
| δ-BHCa | <0.01 | – | 4 |
| Heptachlora | <0.01 | – | 4 |
| Aldrina | <0.01 | – | 4 |
| Heptachlor epoxidea | <0.01 | – | 4 |
| DDEa | <0.01 | – | 4 |
| DDDa | <0.01 | – | 4 |
| DDTa | <0.01 | – | 4 |
| HCBa | <0.01 | – | 4 |
| Mirexa | <0.01 | – | 4 |
| Methoxychlora | <0.05 | – | 4 |
| Dieldrina | <0.01 | – | 4 |
| Endrina | <0.01 | – | 4 |
| Telodrina | <0.01 | – | 4 |
| Chlordanea | <0.05 | – | 4 |
| Toxaphenea | <0.10 | – | 4 |
| Estimated PCBsa | <0.20 | – | 4 |
| Ronnela | <0.01 | – | 4 |
| Ethiona | <0.02 | – | 4 |
| Trithiona | <0.05 | – | 4 |
| Diazinona | <0.10 | – | 4 |
| Methyl chlorpyrifos | 0.065 ± 0.054 | 0.022–0.143 | 4 |
| Methyl parathiona | <0.02 | – | 4 |
| Ethyl parathiona | <0.02 | – | 4 |
| Malathion | 0.027 ± 0.011 | 0.02–0.044 | 4 |
| Endosulfan Ia | <0.01 | – | 4 |
| Endosulfan IIa | <0.01 | – | 4 |
| Endosulfane sulfatea | <0.03 | – | 4 |
B.2.
NTP-2000 Feed
Table B-5. Ingredients of NTP-2000 Rat and Mouse Ration
| Ingredients | Percent by Weight |
|---|---|
| Ground hard winter wheat | 23.00 |
| Ground #2 yellow shelled corn | 22.44 |
| Wheat middlings | 15.0 |
| Oat hulls | 8.5 |
| Alfalfa meal (dehydrated, 17% protein) | 7.5 |
| Purified cellulose | 5.5 |
| Soy protein concentrate | 4.0 |
| Fish meal (60% protein) | 4.0 |
| Corn oil (without preservatives) | 3.0 |
| Soy oil (without preservatives) | 3.0 |
| Dried brewer’s yeast | 1.0 |
| Calcium carbonate (USP) | 0.9 |
| Vitamin premixa | 0.5 |
| Mineral premixb | 0.5 |
| Calcium phosphate, dibasic (USP) | 0.4 |
| Sodium chloride | 0.3 |
| Choline chloride (70% choline) | 0.26 |
| Methionine | 0.2 |
Table B-6. Vitamins and Minerals in NTP-2000 Rat and Mouse Ration
| Amounta | Source | |
|---|---|---|
| Vitamins | ||
| Vitamin A | 4,000 IU | Stabilized vitamin A palmitate or acetate |
| Vitamin D | 1,000 IU | D-activated animal sterol |
| Vitamin K | 1.0 mg | Menadione sodium bisulfite complex |
| α-Tocopheryl acetate | 100 IU | – |
| Niacin | 23 mg | – |
| Folic acid | 1.1 mg | – |
| d-Pantothenic acid | 10 mg | d-Calcium pantothenate |
| Riboflavin | 3.3 mg | – |
| Thiamine | 4 mg | Thiamine mononitrate |
| B12 | 52 μg | – |
| Pyridoxine | 6.3 mg | Pyridoxine hydrochloride |
| Biotin | 0.2 mg | d-Biotin |
| Minerals | ||
| Magnesium | 514 mg | Magnesium oxide |
| Iron | 35 mg | Iron sulfate |
| Zinc | 12 mg | Zinc oxide |
| Manganese | 10 mg | Manganese oxide |
| Copper | 2.0 mg | Copper sulfate |
| Iodine | 0.2 mg | Calcium iodate |
| Chromium | 0.2 mg | Chromium acetate |
Table B-7. Nutrient Composition of NTP-2000 Rat and Mouse Ration
| Nutrient | Mean ± Standard Deviation | Range | Number of Samples |
|---|---|---|---|
| Protein (% by weight) | 14.67 ± 0.530 | 13.9–16.8 | 30 |
| Crude fat (% by weight) | 8.48 ± 0.422 | 7.5–9.7 | 30 |
| Crude fiber (% by weight) | 9.437 ± 0.703 | 7.49–11.3 | 30 |
| Ash (% by weight) | 5.242 ± 1.70 | 4.6–14.2 | 30 |
| Amino acids (% of total diet) | |||
| Arginine | 0.806 ± 0.074 | 0.67–0.97 | 30 |
| Cystine | 0.220 ± 0.021 | 0.15–0.25 | 30 |
| Glycine | 0.702 ± 0.037 | 0.62–0.8 | 30 |
| Histidine | 0.341 ± 0.07 | 0.277–0.68 | 30 |
| Isoleucine | 0.548 ± 0.039 | 0.43–0.66 | 30 |
| Leucine | 1.096 ± 0.062 | 0.96–1.24 | 30 |
| Lysine | 0.70 ± 0.103 | 0.31–0.86 | 30 |
| Methionine | 0.409 ± 0.041 | 0.26–0.49 | 30 |
| Phenylalanine | 0.623 ± 0.046 | 0.471–0.72 | 30 |
| Threonine | 0.513 ± 0.041 | 0.43–0.61 | 30 |
| Tryptophan | 0.156 ± 0.026 | 0.11–0.2 | 30 |
| Tyrosine | 0.423 ± 0.065 | 0.28–0.54 | 30 |
| Valine | 0.666 ± 0.039 | 0.55–0.73 | 30 |
| Essential fatty acids (% of total diet) | |||
| Linoleic | 3.939 ± 0.233 | 3.49–4.55 | 30 |
| Linolenic | 0.306 ± 0.030 | 0.21–0.368 | 30 |
| Vitamins | |||
| Vitamin A (IU/kg) | 3,624 ± 71.5 | 2,030–5,170 | 30 |
| Vitamin D (IU/kg) | 1,000a | – | – |
| α-Tocopherol (ppm) | 2,376 ± 12,602 | 13.6–69,100 | 30 |
| Thiamine (ppm)b | 7.42 ± 0.819 | 5.8–10.1 | 30 |
| Riboflavin (ppm) | 8.17 ± 2.792 | 4.2–17.5 | 30 |
| Niacin (ppm) | 79.19 ± 8.50 | 66.4–98.2 | 30 |
| Pantothenic acid (ppm) | 26.33 ± 10.87 | 17.4–81.0 | 30 |
| Pyridoxine (ppm)b | 9.72 ± 2.02 | 6.44–14.3 | 30 |
| Folic acid (ppm) | 1.60 ± 0.440 | 1.15–3.27 | 30 |
| Biotin (ppm) | 0.330 ± 0.097 | 0.2–0.704 | 30 |
| B12 (ppb) | 50.1 ± 34.34 | 18.3–174.0 | 30 |
| Choline (as chloride) (ppm) | 2,572.0 ± 634 | 1,160–3,790 | 30 |
| Minerals | |||
| Calcium (%) | 0.918 ± 0.0357 | 0.858–1.02 | 30 |
| Phosphorus (%) | 0.540 ± 0.094 | 0.0531–0.602 | 30 |
| Potassium (%) | 0.668 ± 0.029 | 0.626–0.733 | 30 |
| Chloride (%) | 0.391 ± 0.044 | 0.3–0.517 | 30 |
| Sodium (%) | 0.194 ± 0.027 | 0.153–0.283 | 30 |
| Magnesium (%) | 0.217 ± 0.053 | 0.185–0.49 | 30 |
| Iron (ppm) | 190.4 ± 36.11 | 135.0–311.0 | 30 |
| Manganese (ppm) | 50.02 ± 9.27 | 21.0–73.1 | 30 |
| Zinc (ppm) | 56.81 ± 25.25 | 42.5–184.0 | 30 |
| Copper (ppm) | 7.61 ± 2.457 | 3.21–16.3 | 30 |
| Iodine (ppm) | 0.514 ± 0.217 | 0.0–0.972 | 30 |
| Chromium (ppm) | 1.119 ± 1.157 | 0.33–3.97 | 30 |
| Cobalt (ppm) | 0.219 ± 0.150 | 0.0857–0.864 | 30 |
Table B-8. Contaminant Levels in NTP-2000 Rat and Mouse Ration
| Mean ± Standard Deviation | Range | Number of Samples | |
|---|---|---|---|
| Contaminants | |||
| Arsenic (ppm) | 0.200 ± 0.039 | 0.147–0.285 | 30 |
| Cadmium (ppm) | 0.051 ± 0.007 | 0.038–0.067 | 30 |
| Lead (ppm) | 0.132 ± 0.010 | 0.06–0.474 | 30 |
| Mercury (ppm) | 0.012 ± 0.003 | 0.01–0.021 | 30 |
| Selenium (ppm) | 0.181 ± 0.053 | 0.029–0.333 | 30 |
| Aflatoxins (ppb)a | <5.0 | – | 30 |
| Nitrate nitrogen (ppm)b | 16.57 ± 6.45 | 10.0–29.6 | 30 |
| Nitrite nitrogen (ppm)a,b | <0.61 | – | 30 |
| BHA (ppm)a,c | <1.00 | – | 30 |
| BHT (ppm)c | 1.007 ± 0.040 | 1.0–1.22 | 30 |
| Aerobic plate count (CFU/g) | <10.0 | – | 30 |
| Coliform (MPN/g) | <3.0 | – | 30 |
| Escherichia coli (MPN/g)a | <10.0 | – | 30 |
| Salmonella sp. (MPN/g) | Negative | – | 30 |
| Total nitrosamines (ppb)d | 10.28 ± 7.32 | 1.5–33.8 | 30 |
| N-Nitrosodimethylamine (ppb)d | 2.07 ± 3.28 | 0.0–17.6 | 30 |
| N-Nitrosopyrrolidine (ppb)d | 8.22 ± 5.19 | 1.4–20.0 | 30 |
| Pesticides (ppm) | |||
| α-BHCa | <0.01 | – | 30 |
| β-BHCa | <0.02 | – | 30 |
| γ-BHCa | <0.01 | – | 30 |
| δ-BHCa | <0.01 | – | 30 |
| Heptachlora | <0.01 | – | 30 |
| Aldrina | <0.01 | – | 30 |
| Heptachlor epoxidea | <0.01 | – | 30 |
| DDEa | <0.01 | – | 30 |
| DDDa | <0.01 | – | 30 |
| DDTa | <0.01 | – | 30 |
| HCBa | <0.01 | – | 30 |
| Mirexa | <0.01 | – | 30 |
| Methoxychlora | <0.05 | – | 30 |
| Dieldrina | <0.01 | – | 30 |
| Endrina | <0.01 | – | 30 |
| Telodrina | <0.01 | – | 30 |
| Chlordanea | <0.05 | – | 30 |
| Toxaphenea | <0.10 | – | 30 |
| Estimated PCBsa | <0.20 | – | 30 |
| Ronnela | <0.01 | – | 30 |
| Ethiona | <0.02 | – | 30 |
| Trithiona | <0.05 | – | 30 |
| Diazinona | <0.10 | – | 30 |
| Methyl chlorpyrifos | 0.109 ± 0.125 | 0.02–0.611 | 30 |
| Methyl parathiona | <0.02 | – | 30 |
| Ethyl parathiona | <0.02 | – | 30 |
| Malathion | 0.091 ± 0.093 | 0.02–0.385 | 30 |
| Endosulfan Ia | <0.01 | – | 30 |
| Endosulfan IIa | <0.01 | – | 30 |
| Endosulfane sulfatea | <0.03 | – | 30 |
Appendix C. Sentinel Animal Program
C.1.
Methods
Rodents used in the National Toxicology Program are produced in optimally clean facilities to eliminate potential pathogens that might affect study results. The Sentinel Animal Program is part of the periodic monitoring of animal health that occurs during the toxicological evaluation of test compounds. Under this program, the disease state of the rodents is monitored via sera or feces from extra (sentinel) or exposed animals in the study rooms. The sentinel animals and the study animals are subject to identical environmental conditions. Furthermore, the sentinel animals come from the same production source and weanling groups as the animals used for the studies of test compounds.
For these toxicology and carcinogenesis studies, blood samples were collected from each sentinel animal and allowed to clot, and the serum was separated. Additionally, fecal samples were collected and tested for endoparasites and Helicobacter species. All samples were processed appropriately with serology and Helicobacter testing performed by IDEXX BioResearch (formerly Rodent Animal Diagnostic Laboratory [RADIL], University of Missouri), Columbia, MO, for determination of the presence of pathogens. Evaluation for endo- and ectoparasites was performed in-house by the testing laboratory.
The laboratory methods and agents for which testing was performed are tabulated below; the times at which samples were collected during the studies are also listed (Table C-1; Table C-2).
C.2.
Results
Rats: Positive for endoparasites, pinworms (Syphacia spp.). All other test results were negative.
Mice: All test results were negative.
Table C-1. Methods and Results for Sentinel Animal Testing in Male and Female Rats
| Collection Time Points | Three-month Study | Two-year Study | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quarantinea | Perinatalb | Study Termination | Quarantinea | Quarantinec | Perinatalb | 1 Monthd | 6 Months | 12 Months | 16 Months | 18 Months | Study Termination | |
| Number examined (males/females) | 0/10 | 0/10 | 5/5 | 0/10 | 0/1 | 0/10 | 5/5 | 5/5 | 5/5 | 1/0 | 5/5 | 5/5 |
| Method/test | ||||||||||||
| Multiplex fluorescent immunoassay (MFI) | ||||||||||||
| Kilham rat virus (KRV) | – | – | – | – | – | – | – | – | – | – | – | – |
| Mycoplasma pulmonis | – | – | – | – | – | – | – | – | – | – | – | – |
| Parvo NS-1 | – | – | – | NT | NT | NT | NT | NT | NT | NT | NT | NT |
| Pneumonia virus of mice (PVM) | – | – | – | – | – | – | – | – | – | – | – | – |
| Rat coronavirus/ sialodacryoadenitis virus (RCV/SDA) | – | – | – | – | – | – | – | – | – | – | – | – |
| Rat minute virus (RMV) | – | – | – | – | – | – | – | – | – | – | – | – |
| Rat parvo virus (RPV) | – | – | – | – | – | – | – | – | – | – | – | – |
| Rat theilovirus (RTV) | – | – | – | – | – | – | – | – | – | – | – | – |
| Sendai | – | – | – | – | – | – | – | – | – | – | – | – |
| Theiler's murine encephalomyelitis virus (TMEV) | – | – | – | – | – | – | – | – | – | – | – | – |
| Toolan's H-1 | – | – | – | – | – | – | – | – | – | – | – | – |
| Immunofluorescence assay (IFA) | ||||||||||||
| Rat parvo virus (RPV) | NT | NT | NT | NT | NT | NT | NT | – | NT | NT | NT | NT |
| In-house evaluation | ||||||||||||
| Endoparasite evaluation (evaluation of cecal content) | NT | NT | NT | – | – | NT | – | NT | – | – | + | NT |
| Ectoparasite evaluation (evaluation of perianal surface) | NT | NT | NT | – | – | NT | – | NT | – | – | + | NT |
Table C-2. Methods and Results for Sentinel Animal Testing in Male and Female Mice
| Collection Time Points | Three-month Study | Two-year Study | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 Montha | Study Termination | Quarantine | 1 Month | 6 Months | 12 Months | 18 Months | Study Termination | |
| Number examined (males/females) | 5/5 | 5/5 | 5/5 | 5/5 | 5/5 | 5/5 | 5/5 | 5/5 |
| Method/test | ||||||||
| Multiplex fluorescent immunoassay (MFI) | ||||||||
| Ectromelia virus | – | – | – | – | – | – | – | – |
| Epizootic diarrhea of infant mice (EDIM) | – | – | – | – | – | – | – | – |
| Lymphocytic choriomeningitis virus (LCMV) | – | – | – | – | – | – | – | – |
| Mycoplasma pulmonis | – | – | – | – | – | – | – | – |
| Mouse hepatitis virus (MHV) | – | – | – | – | – | – | – | – |
| Mouse norovirus (MNV) | – | – | – | – | – | – | – | – |
| Parvo NS-1 | – | – | – | – | – | – | – | – |
| Mouse parvovirus (MPV) | – | – | – | – | – | – | – | – |
| Minute virus of mice (MVM) | – | – | – | – | – | – | – | – |
| Pneumonia virus of mice (PVM) | – | – | – | – | – | – | – | – |
| Reovirus (REO3) | – | – | – | – | – | – | – | – |
| Sendai | – | – | – | – | – | – | – | – |
| Theiler's murine encephalomyelitis virus (TMEV) GDVII | – | – | – | – | – | – | – | – |
| Polymerase chain reaction (PCR) | ||||||||
| Helicobacter species | NT | NT | NT | NT | NT | NT | – | NT |
| In-house evaluation | ||||||||
| Endoparasite evaluation (evaluation of cecal content) | NT | NT | – | – | NT | – | – | NT |
| Ectoparasite evaluation (evaluation of perianal surface) | NT | NT | – | – | NT | – | – | NT |
Appendix D. Genetic Toxicology
D.1.
Evaluation Protocol
The National Toxicology Program (NTP) considers biological as well as statistical factors to determine an overall assay result. For an individual assay, the statistical procedures for data analysis are described in the following protocols. There have been instances, however, in which multiple samples of a chemical were tested in the same assay, and different results were obtained among these samples and/or among laboratories. In such cases, all the data are critically evaluated with attention given to possible protocol variations in determining the weight of evidence for an overall conclusion of chemical activity in an assay. For in vitro assays conducted with and without exogenous metabolic activation, results obtained in the absence of activation are analyzed separately from results obtained in the presence of activation. The summary table in the abstract of this Technical Report presents NTP’s scientific judgment regarding the overall evidence for activity of the chemical in an assay.
D.2.
Bacterial Mutagenicity
D.2.1.
Bacterial Mutagenicity Test Protocol
Testing procedures were modified from those originally reported by Zeiger et al.90 Two independent studies were conducted. In the first study, coded samples of the isomeric mixture of tris(chloropropyl) phosphate (TCPP) were incubated with the Salmonella typhimurium (TA97, TA98, TA100, TA1535, TA1537) tester strains either in buffer or S9 mix (metabolic activation enzymes and cofactors from Aroclor 1254-induced male Sprague Dawley rat or Syrian hamster liver) for 20 minutes at 37°C. In the second study, blinded samples of TCPP (the same chemical lot that was used in the 2-year bioassays) were incubated with S. typhimurium strains TA98 and TA100 or Escherichia coli WP2 uvrA (pKM101) either in buffer or S9 mix (metabolic activation enzymes and cofactors from phenobarbital/benzoflavone-induced male Sprague Dawley rat liver) for 20 minutes at 37°C. In both studies, top agar supplemented with L-histidine (or tryptophan for the E. coli strain) and d-biotin was added, and the contents of the tubes were mixed and poured onto the surfaces of minimal glucose agar plates. Histidine- or tryptophan-independent mutant colonies arising on these plates were counted after incubation for 2 days at 37°C.
Each trial consisted of triplicate plates of concurrent positive and negative controls and of at least five doses of TCPP. Dose-limiting toxicity was observed in all S. typhimurium strains in the first study. In the second study, dose-limiting toxicity was observed in both S. typhimurium strains but not in the E. coli strain. All trials were repeated.
In this assay, a positive response is defined as a reproducible, dose-related increase in histidine-independent (revertant) colonies in any one strain/activation combination. An equivocal response is defined as an increase in revertants that is not dose-related, is not reproducible, or is not of sufficient magnitude to support a determination of mutagenicity. A negative response is obtained when no increase in revertant colonies is observed after chemical treatment. No minimum percentage or fold increase is required for a chemical to be judged positive or weakly positive, although positive calls are typically reserved for increases in mutant colonies that are at least twofold over background.
D.2.2.
Results
TCPP did not induce mutations, with or without the addition of exogenous metabolic activation enzymes supplied by induced rat or hamster liver S9 in various concentrations, in any of several strains of bacteria (S. typhimurium and E. coli), in two bacterial mutagenicity studies (Table D-1, Table D-2). In Study 1 (study number 815918), the highest dose tested was limited by toxicity to 1,000 µg/plate; in Study 2 (study number G20263C), although toxicity was again observed in some trials at 1,000 µg/plate, higher doses (ranging up to 6,000 µg/plate) could be tested, particularly in the E. coli strain.
Table D-1. Mutagenicity of Tris(chloropropyl) Phosphate in Bacterial Tester Strains (Study 1)a
| Strain | Concentration (μg/plate) | Without S9 | Without S9 | With 5% Rat S9 | With 10% Rat S9 | With 10% Rat S9 | With 30% Rat S9 | With 30% Rat S9 | With 10% Hamster S9 | With 10% Hamster S9 | With 30% Hamster S9 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TA97 | 0 | 79 ± 4.1 | 97 ± 0.9 | – | 110 ± 3.7 | – | 170 ± 15.5 | – | 106 ± 6.8 | – | 91 ± 11.6 |
| 3.3 | – | 89 ± 4.9 | – | – | – | – | – | – | – | – | |
| 10 | 68 ± 6.2 | 76 ± 9.5 | – | 121 ± 3.2 | – | 167 ± 7.8 | – | 116 ± 3.5 | – | 92 ± 2.1 | |
| 33 | 71 ± 1.9 | 91 ± 5.8 | – | 118 ± 11.1 | – | 170 ± 5.0 | – | 92 ± 2.9 | – | 94 ± 3.8 | |
| 100 | 76 ± 4.5 | 90 ± 4.3 | – | 114 ± 2.8 | – | 172 ± 6.1 | – | 117 ± 7.2 | – | 85 ± 1.5 | |
| 333 | 70 ± 8.4 | 91 ± 10.1 | – | 102 ± 3.6 | – | 159 ± 12.8 | – | 116 ± 2.0 | – | 99 ± 13.6 | |
| 666 | – | – | – | 86 ± 3.7b | – | – | – | 92 ± 4.1b | – | – | |
| 667 | Toxic | – | – | – | – | – | – | – | – | – | |
| 1,000 | – | – | – | – | – | 62 ± 7.0b | – | – | – | Toxic | |
| Trial summary | Negative | Negative | – | Negative | – | Negative | – | Negative | – | Negative | |
| Positive controlc | 434 ± 3.2 | 246 ± 28.7 | – | 1,137 ± 7.8 | – | 556 ± 32.5 | – | 804 ± 22.1 | – | 474 ± 24.0 | |
| TA98 | 0 | 16 ± 0.9 | 14 ± 1.5 | – | 30 ± 6.1 | – | 29 ± 3.8 | – | 29 ± 2.3 | – | 30 ± 3.5 |
| 3.3 | – | 16 ± 1.5 | – | – | – | – | – | – | – | – | |
| 10 | 17 ± 1.5 | 13 ± 3.8 | – | 33 ± 3.7 | – | 36 ± 1.8 | – | 28 ± 2.1 | – | 37 ± 3.8 | |
| 33 | 19 ± 0.9 | 15 ± 1.2 | – | 21 ± 3.5 | – | 29 ± 2.5 | – | 23 ± 3.5 | – | 36 ± 1.5 | |
| 100 | 17 ± 3.7 | 13 ± 2.3 | – | 22 ± 1.5 | – | 28 ± 0.9 | – | 23 ± 3.6 | – | 29 ± 3.9 | |
| 333 | 18 ± 1.5 | 18 ± 0.6 | – | 23 ± 0.3 | – | 23 ± 1.2 | – | 26 ± 2.5 | – | 37 ± 3.9 | |
| 666 | – | – | – | 26 ± 3.1b | – | – | – | 28 ± 0.9 | – | – | |
| 667 | 18 ± 1.0 | – | – | – | – | – | – | – | – | – | |
| 1,000 | – | – | – | – | – | 27 ± 1.2b | – | – | – | 24 ± 4.9b | |
| Trial summary | Negative | Negative | – | Negative | – | Negative | – | Negative | – | Negative | |
| Positive control | 154 ± 4.4 | 527 ± 38.2 | – | 284 ± 52.8 | – | 84 ± 2.0 | – | 237 ± 3.2 | – | 88 ± 5.7 | |
| TA100 | 0 | 152 ± 6.9 | 162 ± 4.7 | – | 135 ± 7.0 | – | 128 ± 2.9 | – | 127 ± 3.2 | 94 ± 3.5 | 142 ± 6.1 |
| 3.3 | – | 118 ± 7.6 | – | – | – | – | – | – | – | – | |
| 10 | 157 ± 2.4 | 126 ± 29.0 | – | 138 ± 7.2 | – | 131 ± 6.4 | – | 126 ± 9.4 | 91 ± 3.3 | 157 ± 20.5 | |
| 33 | 144 ± 6.4 | 156 ± 2.5 | – | 139 ± 7.2 | – | 133 ± 5.0 | – | 157 ± 2.9 | 83 ± 4.1 | 130 ± 2.3 | |
| 100 | 145 ± 4.9 | 127 ± 2.8 | – | 128 ± 8.6 | – | 135 ± 6.1 | – | 176 ± 8.7 | 105 ± 2.1 | 113 ± 12.0 | |
| 333 | 145 ± 2.6 | 126 ± 1.0 | – | 119 ± 6.6 | – | 148 ± 0.7 | – | 186 ± 9.2 | 131 ± 7.1 | 162 ± 8.6 | |
| 666 | – | – | – | 84 ± 4.9b | – | – | – | 132 ± 4.5b | 100 ± 4.0 | – | |
| 667 | 128 ± 2.6b | – | – | – | – | – | – | – | – | – | |
| 1,000 | – | – | – | – | – | 111 ± 23.3b | – | – | – | 112 ± 9.5b | |
| Trial summary | Negative | Negative | – | Negative | – | Negative | – | Equivocal | Equivocal | Negative | |
| Positive control | 468 ± 13.8 | 351 ± 91.9 | – | 801 ± 92.3 | – | 398 ± 5.0 | – | 719 ± 41.6 | 541 ± 7.2 | 494 ± 5.3 | |
| TA1535 | 0 | 35 ± 0.9 | 24 ± 1.8 | 12 ± 0.3 | 12 ± 1.5 | 13 ± 3.0 | 15 ± 3.6 | 11 ± 3.2 | 13 ± 0.9 | 10 ± 1.2 | 9 ± 2.3 |
| 3.3 | – | 20 ± 4.0 | – | – | – | – | – | – | – | – | |
| 10 | 36 ± 1.7 | 24 ± 3.0 | – | – | 12 ± 2.3 | 16 ± 0.3 | – | 10 ± 3.8 | 12 ± 2.7 | 10 ± 2.6 | |
| 33 | 38 ± 3.4 | 25 ± 1.8 | 11 ± 2.3 | 14 ± 1.9 | 8 ± 0.7 | 15 ± 1.7 | 13 ± 2.2 | 12 ± 0.6 | 8 ± 1.9 | 17 ± 2.6 | |
| 100 | 34 ± 5.3 | 26 ± 1.5 | 11 ± 0.3 | 10 ± 2.0 | 10 ± 1.2 | 17 ± 3.1 | 9 ± 1.0 | 10 ± 1.8 | 11 ± 1.9 | 12 ± 2.3 | |
| 333 | 37 ± 6.0 | 23 ± 1.7 | 7 ± 0.6 | 7 ± 1.0 | 11 ± 2.3 | 28 ± 3.6 | 12 ± 1.0 | 20 ± 1.5 | 18 ± 4.8 | 10 ± 1.9 | |
| 666 | – | – | – | – | 10 ± 1.5b | – | – | 23 ± 0.3b | 16 ± 4.3b | – | |
| 667 | 11 ± 10.5b | – | 11 ± 1.9b | 8 ± 1.3 | – | – | 9 ± 1.0 | – | – | – | |
| 1,000 | – | – | 6 ± 1.5b | 5 ± 0.3b | – | 24 ± 3.2b | 7 ± 2.0b | – | – | 9 ± 3.2b | |
| Trial summary | Negative | Negative | Negative | Negative | Negative | Equivocal | Negative | Equivocal | Negative | Negative | |
| Positive control | 210 ± 0.6 | 60 ± 5.1 | 129 ± 7.1 | 155 ± 2.5 | 113 ± 4.2 | 134 ± 8.0 | 143 ± 6.7 | 281 ± 4.9 | 50 ± 2.8 | 48 ± 5.5 | |
| TA1537 | 0 | 4 ± 0.3 | – | – | – | – | 6 ± 0.0 | – | – | – | 8 ± 1.5 |
| 10 | 7 ± 0.6 | – | – | – | – | 9 ± 3.2 | – | – | – | 6 ± 1.5 | |
| 33 | 6 ± 1.7 | – | – | – | – | 7 ± 1.2 | – | – | – | 5 ± 2.6 | |
| 100 | 6 ± 1.9 | – | – | – | – | 8 ± 1.9 | – | – | – | 8 ± 2.3 | |
| 333 | 5 ± 1.9 | – | – | – | – | 7 ± 1.5 | – | – | – | 5 ± 0.9 | |
| 667 | 6 ± 0.9b | – | – | – | – | – | – | – | – | – | |
| 1,000 | – | – | – | – | – | 5 ± 1.2b | – | – | – | 5 ± 0.3b | |
| Trial summary | Negative | – | – | – | – | Negative | – | – | – | Negative | |
| Positive control | 258 ± 7.9 | – | – | – | – | 76 ± 4.7 | – | – | – | 55 ± 1.8 | |
Table D-2. Mutagenicity of Tris(chloropropyl) Phosphate in Bacterial Tester Strains (Study 2)a
| Strain | Concentration (μg/plate) | Without S9 | Without S9 | With 10% Rat S9 | With 10% Rat S9 |
|---|---|---|---|---|---|
| TA98 | 0 | 18 ± 2.1 | 15 ± 3.1 | 23 ± 2.3 | 18 ± 2.4 |
| 40 | – | 10 ± 2.1 | – | – | |
| 80 | – | 11 ± 2.3 | – | 24 ± 4.6 | |
| 100 | 15 ± 3.1 | – | 16 ± 0.9 | – | |
| 200 | – | 15 ± 3.2 | – | 19 ± 2.3 | |
| 250 | 14 ± 2.6 | – | 15 ± 3.8 | – | |
| 500 | 20 ± 1.5 | 18 ± 4.0 | 24 ± 0.9 | 13 ± 2.0 | |
| 750 | – | 19 ± 2.0 | – | – | |
| 1,000 | 18 ± 3.5 | 13 ± 1.7 | 18 ± 0.6 | 15 ± 3.5 | |
| 2,000 | Toxic | Toxic | – | – | |
| 3,000 | Toxic | – | 15 ± 4.0 | 5 ± 0.9 | |
| 6,000 | – | – | 16 ± 8.2 | 6 ± 3.7 | |
| Trial summary | Negative | Negative | Negative | Negative | |
| Positive controlb | 697 ± 9.5 | 548 ± 7.0 | 1,659 ± 73.5 | 1,089 ± 22.2 | |
| TA100 | 0 | 88 ± 1.5 | 75 ± 4.6 | 81 ± 4.2 | 85 ± 3.2 |
| 40 | – | 73 ± 4.1 | – | – | |
| 80 | – | 67 ± 5.8 | – | – | |
| 100 | 83 ± 3.8 | – | 89 ± 3.5 | – | |
| 200 | – | 85 ± 1.3 | – | 83 ± 4.3 | |
| 250 | 80 ± 11.9 | – | 94 ± 4.6 | – | |
| 300 | – | – | – | 78 ± 2.6 | |
| 500 | 81 ± 6.2 | 79 ± 2.6 | 83 ± 5.0 | – | |
| 750 | – | 58 ± 4.2 | – | – | |
| 1,000 | Toxic | Toxic | 67 ± 7.6 | 80 ± 5.0 | |
| 2,000 | Toxic | Toxic | – | Toxic | |
| 3,000 | Toxic | – | Toxic | Toxic | |
| 6,000 | – | – | Toxic | Toxic | |
| Trial summary | Negative | Negative | Negative | Negative | |
| Positive control | 654 ± 20.9 | 541 ± 10.7 | 791 ± 24.9 | 422 ± 16.1 | |
| Escherichia coli WP2 uvrA (pKM101) | 0 | 114 ± 1.9 | 99 ± 8.2 | 149 ± 4.6 | 139 ± 5.6 |
| 100 | 123 ± 10.7 | 108 ± 6.7 | 155 ± 5.2 | 145 ± 12.7 | |
| 250 | 110 ± 3.3 | 100 ± 1.9 | 150 ± 4.3 | 157 ± 3.2 | |
| 500 | 104 ± 12.7 | 95 ± 5.9 | 150 ± 6.2 | 133 ± 5.8 | |
| 1,000 | 100 ± 3.7 | 96 ± 4.9 | 168 ± 1.9 | 119 ± 9.3 | |
| 3,000 | 86 ± 7.5 | 74 ± 8.2 | 111 ± 5.8 | 94 ± 4.8 | |
| 6,000 | 73 ± 7.6 | 88 ± 2.9 | 111 ± 2.5 | 92 ± 3.5 | |
| Trial summary | Negative | Negative | Negative | Negative | |
| Positive control | 2,291 ± 125.2 | 2,313 ± 73.3 | 978 ± 16.1 | 895 ± 39.4 | |
D.3.
Micronucleus Assay
D.3.1.
Peripheral Blood Micronucleus Test Protocol
At termination of the 3-month toxicity studies of TCPP, blood samples (approximately 200 μL) were collected from male and female rats and mice, placed in ethylenediaminetetraacetic acid (EDTA)-coated tubes, and shipped overnight to the testing laboratory. Upon arrival, blood samples were fixed in ultracold methanol using a MicroFlowPLUS Kit (Litron Laboratories, Rochester, NY) according to the manufacturer’s instructions. Fixed samples were stored in a −80°C freezer until analysis. Thawed blood samples were analyzed for frequency of micronucleated immature erythrocytes (i.e., reticulocytes or polychromatic erythrocytes [PCEs]) and mature erythrocytes (i.e., normochromatic erythrocytes [NCEs]) using a flow cytometer115; both the mature and the immature erythrocyte populations can be analyzed separately by employing special cell surface markers to differentiate the two cell types. Because the very young reticulocyte subpopulation (CD71+ cells) can be targeted using this technique, rat blood samples can be analyzed for damage that occurred in the bone marrow within the past 24–48 hours, before the rat spleen appreciably alters the percentage of PCEs in circulation.116 In mice, both the immature and mature erythrocyte populations can be evaluated for micronucleus frequency because the mouse spleen does not sequester and eliminate damaged erythrocytes. Damaged erythrocytes achieve steady state in the peripheral blood of mice after 4 weeks of continuous exposure. Approximately 20,000 PCEs and 1 × 106 NCEs were analyzed per animal for frequency of micronucleated cells, and the percentage of immature erythrocytes (% PCE) was calculated as a measure of bone marrow toxicity resulting from chemical exposure.
Prior experience with the large number of cells scored using flow cytometric scoring techniques117 suggests it is reasonable to assume that the proportion of micronucleated reticulocytes is approximately normally distributed. The statistical tests selected for trend and for pairwise comparisons with the control group depend on whether the variances among the groups are equal. The Levene test at α = 0.05 is used to test for equal variances. In the case of equal variances, linear regression is used to test for a linear trend with exposure concentration and the Williams test is used to test for pairwise differences between each exposed group and the control group. In the case of unequal variances, the Jonckheere test is used to test for linear trend and the Dunn test is used for pairwise comparisons of each exposed group with the control group. To correct for multiple pairwise comparisons, the p value for each comparison with the control group is multiplied by the number of comparisons made. In the event that this product is >1.00, it is replaced with 1.00. Trend tests and pairwise comparisons with the control group are considered statistically significant at p ≤ 0.025.
In the micronucleus test, it is preferable to base a positive result on the presence of both a positive trend as well as on at least one significantly elevated exposed group compared with the corresponding control group. In addition, historical control data are used to evaluate the biological significance of any observed response. Both statistical significance and biological significance are considered when arriving at a call. The presence of either a positive trend or a single significant exposed group generally results in an equivocal call. The absence of both a trend and any significant differences between exposed groups and the control group results in a negative call. Ultimately, the scientific staff determines the final call after considering the results of statistical analyses, reproducibility of any effects observed (in acute studies), and the magnitudes of those effects.
D.3.2.
Results
Micronuclei are biomarkers of chromosomal changes, either in chromosome number or structure (breaks). In the in vivo peripheral blood micronucleus test, TCPP exposure for 3 months via dosed feed did not result in an increase in micronucleated PCEs in male or female Sprague Dawley rats (Table D-3). However, the percentage of PCEs among total erythrocytes was increased in an exposure concentration-related manner in both sexes, suggesting a stimulation of erythropoiesis in the rats exposed to TCPP. In female B6C3F1/N mice, exposure to TCPP for 3 months via dosed feed did not result in an increase in micronucleated PCEs or NCEs (Table D-4). In male mice, the values for micronucleated red blood cells were within the laboratory historical control 95% confidence interval and the absolute increase in micronucleated NCEs and PCEs was quite small, amounting to an increase over control values of approximately 17% or 23%, respectively. Due to the questionable biological significance of the observed increase in micronucleated cells, the results in male mice were judged to be equivocal (Table D-4). In both male and female mice, the percentage of PCEs among total erythrocytes was increased in an exposure concentration-related manner, similar to what was observed in the rat samples, suggesting that exposure to TCPP also produced a stimulation of erythropoiesis in mice.
Table D-3. Frequency of Micronuclei in Peripheral Blood Erythrocytes of Male and Female Rats in the Three-month Feed Study of Tris(chloropropyl) Phosphatea
| Number of Rats with Erythrocytes Scored | Micronucleated PCEs/1,000 PCEsb | P Valuec | Micronucleated NCEs/1,000 NCEsb | P Valuec | PCEs (%)b | P Valuec | |
|---|---|---|---|---|---|---|---|
| Male | |||||||
| Exposure concentration (ppm) | |||||||
| 0 | 5 | 0.388 ± 0.077 | 0.031 ± 0.003 | 0.869 ± 0.047 | |||
| 2,500 | 5 | 0.500 ± 0.096 | 0.3646 | 0.030 ± 0.008 | 1.0000 | 0.859 ± 0.078 | 1.0000 |
| 5,000 | 5 | 0.370 ± 0.075 | 0.4331 | 0.029 ± 0.004 | 1.0000 | 1.048 ± 0.056 | 0.0701 |
| 10,000 | 5 | 0.420 ± 0.085 | 0.4612 | 0.031 ± 0.006 | 1.0000 | 1.324 ± 0.057 | <0.001 |
| Trendd | p = 0.5255 | p = 0.5268 | p < 0.001 | ||||
| Female | |||||||
| Exposure concentration (ppm) | |||||||
| 0 | 5 | 0.570 ± 0.051 | 0.073 ± 0.018 | 0.899 ± 0.069 | |||
| 2,500 | 5 | 0.676 ± 0.134 | 0.7823 | 0.085 ± 0.031 | 0.6955 | 1.040 ± 0.119 | 0.4322 |
| 5,000 | 5 | 0.380 ± 0.064 | 0.8558 | 0.050 ± 0.013 | 0.7793 | 1.129 ± 0.181 | 0.4080 |
| 10,000 | 5 | 0.460 ± 0.060 | 0.8851 | 0.057 ± 0.009 | 0.8131 | 1.030 ± 0.090 | 0.4362 |
| 20,000 | 5 | 0.310 ± 0.068 | 0.8970 | 0.033 ± 0.008 | 0.8297 | 1.522 ± 0.138 | 0.0049 |
| Trend | p = 0.9930 | p = 0.9711 | p = 0.0035 | ||||
Table D-4. Frequency of Micronuclei in Peripheral Blood Erythrocytes of Male and Female Mice in the Three-month Feed Study of Tris(chloropropyl)a
| Number of Rats with Erythrocytes Scored | Micronucleated PCEs/1,000 PCEsb | P Valuec | Micronucleated NCEs/1,000 NCEsb | P Valuec | PCEs (%)b | P Valuec | |
|---|---|---|---|---|---|---|---|
| Male | |||||||
| Exposure concentration (ppm) | |||||||
| 0 | 5 | 2.630 ± 0.170 | 1.462 ± 0.028 | 1.360 ± 0.084 | |||
| 1,250 | 5 | 2.920 ± 0.200 | 0.3523 | 1.452 ± 0.012 | 1.0000 | 1.252 ± 0.029 | 1.0000 |
| 2,500 | 5 | 2.770 ± 0.137 | 0.4180 | 1.490 ± 0.011 | 1.0000 | 1.348 ± 0.036 | 1.0000 |
| 5,000 | 5 | 2.550 ± 0.328 | 0.4465 | 1.502 ± 0.030 | 0.8303 | 1.234 ± 0.044 | 1.0000 |
| 10,000 | 5 | 2.940 ± 0.183 | 0.2091 | 1.549 ± 0.022 | 0.1015 | 1.368 ± 0.050 | 0.9831 |
| 20,000 | 5 | 3.236 ± 0.217 | 0.0363 | 1.710 ± 0.028 | 0.0016 | 1.730 ± 0.052 | <0.001 |
| Trendd | p = 0.0225 | p < 0.001 | p < 0.001 | ||||
| Female | |||||||
| Exposure concentration (ppm) | |||||||
| 0 | 5 | 2.860 ± 0.171 | 1.133 ± 0.020 | 1.120 ± 0.019 | |||
| 1,250 | 5 | 1.950 ± 0.110 | 0.9998 | 1.101 ± 0.041 | 0.9117 | 1.285 ± 0.087 | 0.2611 |
| 2,500 | 5 | 1.980 ± 0.107 | 1.0000 | 1.077 ± 0.009 | 0.9560 | 1.225 ± 0.084 | 0.3118 |
| 5,000 | 5 | 1.680 ± 0.133 | 1.0000 | 1.107 ± 0.024 | 0.9677 | 1.631 ± 0.106 | 0.0078 |
| 10,000 | 5 | 1.750 ± 0.177 | 1.0000 | 1.030 ± 0.041 | 0.9730 | 1.346 ± 0.107 | 0.0072 |
| 20,000 | 5 | 1.710 ± 0.189 | 1.0000 | 1.018 ± 0.018 | 0.9769 | 1.716 ± 0.089 | <0.001 |
| Trend | p = 0.9946 | p = 0.9984 | p < 0.001 | ||||
Appendix E. Bioactivity Screening of Tris(chloropropyl) Phosphate Using Tox21 In Vitro Assay Data
E.1.
Introduction
The National Toxicology Program (NTP) has integrated diverse data sources derived from experimental systems with varying biological complexity to understand the mechanisms of toxicity and carcinogenicity induced by the isomeric mixture of tris(chloropropyl) phosphate (TCPP). These diverse data sources include quantitative high-throughput in vitro screening (qHTS) data, short-term toxicogenomic data, and next-generation sequencing (NGS) data of the mouse liver tumors. This appendix summarizes the activity of TCPP using a qHTS approach.
Countless chemical substances exist in the world, but only a small fraction of these have been adequately assessed for their potential toxicity to humans. The Toxicology in the 21st Century program, or Tox21, is a unique collaboration among several federal agencies established to develop new methods to rapidly test the potential for thousands of substances to adversely affect human health. One method to rapidly generate compound-induced, human-relevant toxicity data is to use the qHTS approach on a large number of substances employing in vitro, human cell-based assays. In Tox21 qHTS, to achieve better confidence in the compound potency data, each substance is tested using 15 concentrations (generally from 5 nM to 92 µM) in three different batches; viability is run in a single batch. Since 2011, approximately 10,000 substances have been screened in more than 70 assays covering mostly human stress response and nuclear receptor pathways. The data provide rich resources from which to query activities of compounds of interest, such as TCPP.
E.2.
Materials and Methods
The data were analyzed as described in a publication by Hsieh et al.118 The concentration-response data per substance at each batch (each batch was conducted on different days) was analyzed separately using the Curvep algorithm in the Rcurvep package (v.1.2.0, https://cran.r-project.org/web/packages/Rcurvep/index.html) and activity metrics including the point of departure (POD), maximum response (Emax), and weighted area under the curve (wAUC) were calculated. The POD is the compound potency at which the elicited response exceeds the noise threshold. The Emax is in the percentage scale, relative to the response elicited by the respective assay positive control (PC). In Tox21 assays, the baseline value of Emax is set to 0%. A positive or negative Emax value indicates an increasing or decreasing effect, respectively, and a higher absolute Emax value indicates a stronger effect. The wAUC is a summarized activity value that includes both potency and efficacy information and is weighted by both POD and the testing concentration range, and thus allows for proper across-chemical comparison.119,120 The activities from batches were summarized using the median and known artifacts were flagged using the integrative analysis on existing multiple data sources (e.g., readout data in the same screen, counter-screens, and/or external data). Additionally, the substance quality control (QC) information (compound identity, purity, and concentration) was included to remove data with poor QC. The final output from the analysis using the current data sets includes a total of 209 endpoints, each of which has the activity calls at the compound level with summary statistics and warning flags. The activity values at the compound level were summarized (mean) when testing substances had the same activity call (e.g., all active) and acceptable QC. For POD, values were calculated as the mean of log10(M) and then converted back to µM. The activity values resulting from the 209 endpoints were considered directly associated with the annotated target by excluding the current known assay artifacts.
The results were deposited into the NTP database and can be retrieved using the Tox21 Data Application Programming Interface (API) (https://rstudio.niehs.nih.gov/tox21_qhts_api/). The current Tox21 data sets cover 86 protocols and include a total of 209 endpoints. Out of these endpoints for which data were collected, 89 are for detecting nonspecific effects (e.g., cytotoxicity) and 37 are for identifying autofluorescent chemical structures in various conditions. The remaining 83 endpoints are for detecting specific effects such as covering activation/inhibition of nuclear receptor pathways, activation of stress response pathways, and inhibition of cytochrome P450 (CYP).
E.3.
Results
E.3.1.
Activity of Tris(chloropropyl) Phosphate in Tox21 Assays
In the Tox21 library, two independent substances (Tox21_202982 and Tox21_303533) were identified as TCPP and were tested in the Tox21 assays. Both substances had acceptable QC (https://tripod.nih.gov/tox/samples) and their data were first used to generate activity results at the substance level then at the compound level. The activity results at the compound level are discussed throughout the report.
TCPP showed activities in 10 endpoints from the Tox21 qHTS assessment. Seven of 10 endpoints were related to xenobiotic homeostasis, particularly activation of the pregnane X receptor (PXR) signaling pathway and the constitutive androstane receptor (CAR) pathway, and the inhibition of CYP 1A2, 2C19, 2C9, 3A4, and 2D6. TCPP induced cytotoxicity in two separate assays and was associated with inhibition of the progesterone receptor (PR) signaling pathway involved in sex hormone homeostasis. The POD and Emax of the activities in these 10 endpoints are plotted in Figure E-1. The underlying activity data in Figure E-1 are provided in Table E-1, including the experimental protocol information deposited in PubChem (https://pubchem.ncbi.nlm.nih.gov/). For the 10 endpoints that TCPP showed activity, the concentration-response curves associated with each endpoint, in addition to the activity results at the testing substance level, are provided in Figure E-1 to Figure E-12. The activation of the PXR pathway was also supported by other orthogonal Toxicity Forecasting (ToxCast) assays including Attagene and Novascreen2 (available at https://comptox.epa.gov/dashboard).
Table E-1. Tris(chloropropyl) Phosphate Activity Data in Tox21 Quantitative High-throughput Screening Assays
| Endpoint | Target | Target Group | Lowest Tested Concentration (µM) | Highest Tested Concentration (µM) | POD (µM) | Emax (%) | # of Activity Calls of Substances | # of Activity Calls without Artifact Flags | PubChem AID |
|---|---|---|---|---|---|---|---|---|---|
| tox21-car-agonist-p1_car-agonist_1a | car-agonist | xenobiotic homeostasis | 0.001121 | 92.34215 | 44.55 | 24.58 | 2 | 2 | 1224892 |
| tox21-p450-1a2-p1_1a2-inhibitor_1b | 1a2-inhibitor | xenobiotic homeostasis | 0.001398 | 115.1596 | 2.63 | −84.72 | 2 | 2 | 1671199 |
| tox21-p450-2c9-p1_2c9-inhibitor_1c | 2c9-inhibitor | xenobiotic homeostasis | 0.001398 | 115.1596 | 37.88 | −56.40 | 2 | 2 | 1671198 |
| tox21-p450-2c19-p1_2c19-inhibitor_1d | 2c19-inhibitor | xenobiotic homeostasis | 0.001398 | 115.1596 | 14.36 | −122.41 | 2 | 2 | 1671197 |
| tox21-p450-2d6-p1_2d6-inhibitor_1e | 2d6-inhibitor | xenobiotic homeostasis | 0.001398 | 115.1596 | 65.80 | −25.21 | 2 | 2 | 1671196 |
| tox21-p450-3a4-p1_3a4-inhibitor_1f | 3a4-inhibitor | xenobiotic homeostasis | 0.001474 | 115.1596 | 92.81 | −32.72 | 2 | 2 | 1671201 |
| tox21-pr-bla-antagonist-p1_pr-antagonist_1g | pr-antagonist | sex hormone homeostasis | 0.001121 | 87.55884 | 36.94 | −46.43 | 2 | 1 | 1347031 |
| tox21-pxr-p1_pxr-agonist_1h | pxr-agonist | xenobiotic homeostasis | 0.001121 | 92.34215 | 20.07 | 38.66 | 2 | 2 | 1347033 |
| tox21-rt-viability-hek293-p1_viability@glo_40h_1i | viability@glo_40h | cytotoxicity | 9.32E-04 | 76.7715 | 65.83 | −18.32 | 2 | 2 | 1224874 |
| tox21-rt-viability-hek293-p1_viability@glo_1j | viability@glo | cytotoxicity | 9.32E-04 | 76.7715 | 31.75 | −23.99 | 2 | 2 | NA |
Figure E-1. Tris(chloropropyl) Phosphate Activity in Tox21 Quantitative High-throughput Screening Assays
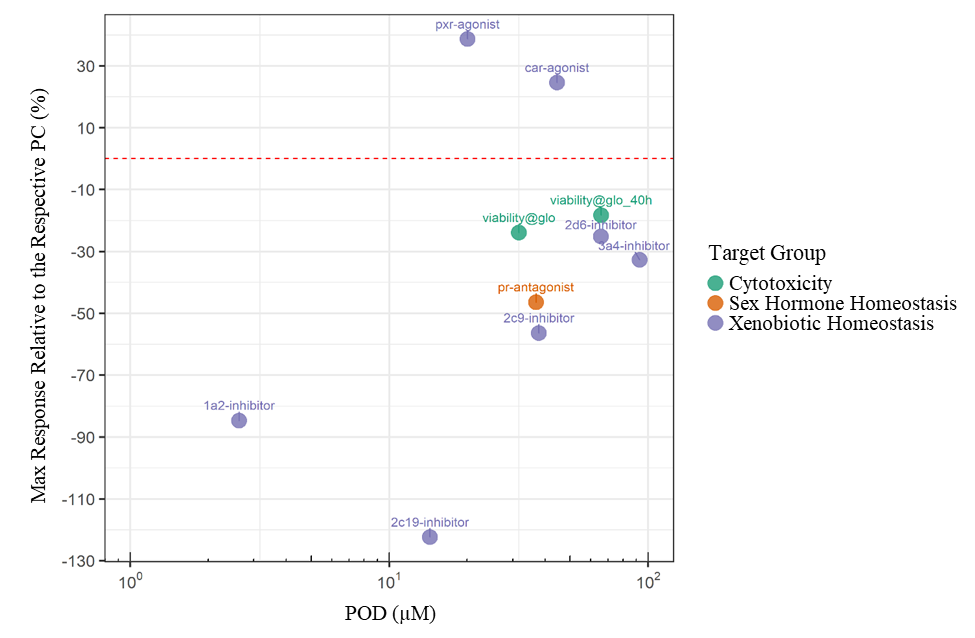
The red dashed line represents 0%; data points above this line represent activation while points below represent inhibition. 2d6 = cytochrome P450 (CYP) 2D6; 3a4 = CYP 3A4; 2c9 = CYP 2C9; 1a2 = CYP 1A2; 2c19 = CYP 2C19; CAR = constitutive androstane receptor; glo = RealTime-Glo MT Cell Viability Assay over time; glo_40h = RealTime-Glo MT Cell Viability Assay at 40 hours of exposure; PC = positive control; PR = progesterone receptor; PXR = pregnane X receptor; POD = point of departure.
Figure E-2. Concentration-response Curves of Tris(chloropropyl) Phosphate Agonism of the Constitutive Androstane Receptor in HepG2 Cells
Results from 15 tested concentrations in three Tox21 batches for tox21-car-agonist-p1_car-agonist_1 are shown above for A) active agonism of the constitutive androstane receptor (CAR) by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 54.66 µm, and a maximum response (Emax) of 27%; and B) active agonism of CAR by TCPP (Tox21_303533) with no artifact flags, a POD of 36.29 µm, and an Emax of 22.17%. The compound level POD and Emax are 44.55 µm and 24.58%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-3. Concentration-response Curves of Tris(chloropropyl) Phosphate Inhibition of Cytochrome P450 1a2

Results from 15 tested concentrations in three Tox21 batches for tox21-p450-1a2-p1_1a2-inhibitor_1 are shown above for A) active inhibition of cytochrome P450 (CYP) 1a2 by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 1.89 µm, and a maximum response (Emax) of −84.78%; and B) active inhibition of CYP1a2 by TCPP (Tox21_303533) with no artifact flags, a POD of 3.66 µm, and an Emax of −84.65%. The compound level POD and Emax are 2.63 µm and −84.72%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-4. Concentration-response Curves of Tris(chloropropyl) Phosphate Inhibition of Cytochrome P450 2c19
Results from 15 tested concentrations in three Tox21 batches for tox21-p450-2c19-p1_2c19-inhibitor_1 are shown above for A) active inhibition of cytochrome P450 (CYP) 2c19 by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 15.53 µm, and a maximum response (Emax) of −130.64%; and B) active inhibition of CYP2c19 by TCPP (Tox21_303533) with no artifact flags, a POD of 13.27 µm, and an Emax of −114.17%. The compound level POD and Emax are 14.36 µm and −122.41%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-5. Concentration-response Curves of Tris(chloropropyl) Phosphate Inhibition of Cytochrome P450 2c9
Results from 15 tested concentrations in three Tox21 batches for tox21-p450-2c9-p1_2c9-inhibitor_1 are shown above for A) active inhibition of cytochrome P450 (CYP) 2c9 by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 36.12 µm, and a maximum response (Emax) of −58.06%; and B) active inhibition of CYP2c9 by TCPP (Tox21_303533) with no artifact flags, a POD of 39.72 µm, and an Emax of −54.74%. The compound level POD and Emax are 37.88 µm and −56.4%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-6. Concentration-response Curves of Tris(chloropropyl) Phosphate Inhibition of Cytochrome P450 2d6
Results from 15 tested concentrations in three Tox21 batches for tox21-p450-2d6-p1_2d6-inhibitor_1 are shown above for A) active inhibition of cytochrome P450 (CYP) 2d6 by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 104.45 µm, and a maximum response (Emax) of −22.76%; and B) active inhibition of CYP2d6 by TCPP (Tox21_303533) with no artifact flags, a POD of 41.45 µm, and an Emax of −27.65%. The compound level POD and Emax are 65.80 µm and −25.21%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-7. Concentration-response Curves of Tris(chloropropyl) Phosphate Inhibition of Cytochrome P450 3a4
Results from 15 tested concentrations in three Tox21 batches for tox21-p450-3a4-p1_3a4-inhibitor_1 are shown above for A) active inhibition of cytochrome P450 (CYP) 3a4 by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 93.28 µm, and a maximum response (Emax) of −33.86%; and B) active inhibition of CYP3a4 by TCPP (Tox21_303533) with no artifact flags, a POD of 92.32 µm, and an Emax of −31.59%. The compound level POD and Emax are 92.81 µm and −32.72%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-8. Concentration-response Curves of Tris(chloropropyl) Phosphate Agonism of the Progesterone Receptor in HEK293 Cells
Results from 15 tested concentrations in three Tox21 batches for tox21-pr-bla-antagonist-p1_pr-antagonist_1 are shown above for A) inconclusive antagonism of the progesterone receptor (PR) by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with an artifact flag of potency_overlap_with_counterscreen, a point of departure (POD) of 39.14 µm, and a maximum response (Emax) of −60.02%; and B) active antagonism of PR by TCPP (Tox21_303533) with no artifact flags, a POD of 36.94 µm, and an Emax of −46.43%. The compound level POD and Emax are 36.94 µm and −46.94%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch. Ch1 = channel 1, background readout; Ch2 = channel 2, reporter gene readout; Ratio = the ratio of Ch2:Ch1.
Figure E-9. Concentration-response Curves of Tris(chloropropyl) Phosphate Agonism of the Pregnane X Receptor in HepG2 Cells
Results from 15 tested concentrations in three Tox21 batches for tox21-pxr-p1_pxr-agonist_1 are shown above for A) active agonism of the pregnane X receptor (PXR) by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 31.02 µm, and a maximum response (Emax) of 37.08%; and B) active agonism of PXR by TCPP (Tox21_303533) with no artifact flags, a POD of 12.99 µm, and an Emax of 40.24%. The compound level POD and Emax are 20.07 µm and 38.66%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
Figure E-10. Concentration-response Curves of Tris(chloropropyl) Phosphate on Viability in HEK293 Cells at 0, 8, 16, 24, 32, and 40 Hours
Results from 15 tested concentrations in one Tox21 batch for tox21-rt-viability-hek293-p1_viability@glo_1 are shown above for A) decreased viability by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 14.06 µm, and a maximum response (Emax) of −32.68%; and B) decreased viability by TCPP (Tox21_303533) with no artifact flags, a POD of 71.66 µm, and an Emax of −15.30%. The compound level POD and Emax are 31.75 µm and −23.99%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch. Glo_0h_n = cell viability readout at 0 hours; Glo_16h_n = cell viability readout at 16 hours; Glo_24h_n = cell viability readout at 24 hours; Glo_32h_n = cell viability readout at 32 hours; Glo_40h_n = cell viability readout at 40 hours; Glo_8h_n = cell viability readout at 8 hours.
Figure E-11. Concentration-response Curves of Tris(chloropropyl) Phosphate on Viability in HEK293 Cells at 40 Hours
Results from 15 tested concentrations in one Tox21 batch for tox21-rt-viability-hek293-p1_viability@glo_40h_1 are shown above for A) decreased viability by tris(chloropropyl) phosphate (TCPP; Tox21_202982) with no artifact flags, a point of departure (POD) of 60.46 µm, and a maximum response (Emax) of −21.33%; and B) decreased viability by TCPP (Tox21_303533) with no artifact flags, a POD of 71.66 µm, and an Emax of −15.3%. The compound level POD and Emax are 65.83 µm and −18.32%, respectively, with an overall activity call of active. A dose-response line of best fit is shown for each batch.
E.3.2.
Activity of Tris(chloropropyl) Phosphate Compared with Reference Chemicals
Among the activities that TCPP displayed in Tox21 qHTS assays were clear effects on PXR and CAR activation (Figure E-1, Figure E-2, Figure E-9). The degree of the effect was compared with reference chemicals that were also tested in the Tox21 assays. The reference chemicals for activation of PXR and CAR were selected from Judson et al.121 The reference chemicals selected included those with the highest number of available supporting reports and were also active in the related Tox21 assays. For PXR activation, rifampicin was selected (support # = 13); for CAR activation, bisphenol A (support # = 3) and methoxychlor (support # = 3) were selected.
The effect of PXR or CAR activation by the reference chemicals was compared with that of TCPP in relation to all the other active chemicals in the related Tox21 screens (Figure E-12) using the wAUC activity metric. In Figure E-12A, the effect of rifampicin is within the top 5% of active chemicals to activate PXR, and TCPP is within the top 50%. In Figure E-12B, the effects of methoxychlor and bisphenol A are within the top 0.2% and 4%, respectively, of active chemicals to activate CAR, and TCPP is within the top 66%.
Figure E-12. Comparison of Tris(chloropropyl) Phosphate Potency to Assay Reference Chemicals in Tox21
A) Pregnane X receptor (PXR) and B) constitutive androstane receptor (CAR) activation in Tox21 assays (gray dots) plotted based on weighted area under the curve (wAUC) value. Each individual gray dot represents all active chemicals in the related Tox21 screens, organized by wAUC values. Tris(chloropropyl) phosphate (TCPP) activity versus rifampicin activity (PXR reference chemical) is highlighted in panel A (red dots). TCPP activity versus bisphenol A and methoxychlor activity (CAR reference chemicals) is highlighted in panel B (red dots).
E.4.
Summary
In summary, the in vitro data suggest that TCPP activates PXR and CAR, although to a lesser degree than do the reference chemicals rifampicin for PXR and bisphenol A and methoxychlor for CAR.
Appendix F. Five-day Rat Toxicogenomic Study of Tris(chloropropyl) Phosphate
F.1.
Introduction
The National Toxicology Program (NTP) has integrated diverse data sources derived from experimental systems with varying biological complexity to understand the mechanisms of toxicity and carcinogenicity induced by the isomeric mixture of tris(chloropropyl) phosphate (TCPP). These diverse data sources include quantitative high-throughput in vitro screening (qHTS) data, short-term toxicogenomic data, and next-generation sequencing (NGS) data of the mouse liver tumors. This appendix summarizes the toxicogenomic results following a 5-day exposure to TCPP.
A 5-day repeat-dose in vivo genomic dose-response study in male rats was performed to characterize TCPP’s biological potency as a means of estimating its toxicological potency in guideline toxicological assessments. In this study, TCPP was shown to increase absolute and relative liver weights at doses ≥1,000 mg TCPP/kg body weight/day (mg/kg/day) with a benchmark dose (BMD) of approximately 589 mg/kg/day.106 The most sensitive gene set (gene ontology biological process) BMD of 1 standard deviation (BMD1SD) value identified was approximately 26 mg/kg/day in liver. In addition, the relative liver weight change BMD1SD was 253 mg/kg/day. In this appendix, the transcriptomics data generated in the 5-day study were employed to characterize specific mechanisms of toxicity with the goal of providing mechanistic context to the bioassay findings.
F.2.
Material and Methods
A detailed description of the in vivo study, transcriptomic data generation, and quality control can be found in Gwinn et al.106 In sum, doses of 0, 18.75, 37.5, 75, 150, 300, 600, 1,000, or 2,000 mg/kg/day were administered by gavage for 5 consecutive days to groups of four male Sprague Dawley (Hsd:Sprague Dawley SD) rats and the liver and kidney were collected 24 hours after the last dose. RNA was extracted from the liver and kidney and subjected to high-throughput transcriptomics using the rat TempO-Seq S1500+ platform.122
Normalized and log2-transformed S1500+ data from the Gwinn et al.106 study were subject to whole-genome extrapolation using the GeniE tool.123 GeniE-extrapolated data were used for dose-response analysis as described below. Dose-response analyses of extrapolated gene expression values were performed using BMDExpress 2.3. Data were prefiltered (one data set per chemical) using a Williams trend test (p < 0.05, 1 × 104 dose-level permutations) in combination with a fold-change cutoff of >ǀ2ǀ to remove detector oligos (DOs; two DOs/transcript) that did not demonstrate a response to chemical exposure following BMD analysis. DOs that passed the prefilter were fit to multiple continuous models (linear, exponential 2–5, polynomial of degree 2, and power). An assumption of constant variance across dose groups was made due to the log transformation of the data. To be considered further, model fits had to demonstrate convergent BMD, BMDL (BMD lower 95% confidence limit), and BMDU (BMD upper 95% confidence limit) values. For the models with convergent values, the one with the lowest Akaike information criterion was chosen as the best fit model for each DO. A benchmark response (BMR) of 1 standard deviation (1SD) from the modeled response for the control group for each DO was used to identify a BMD, BMDL, and BMDU. Prior to analysis, DOs were removed that mapped to more than one gene, had a global goodness of fit p value of <0.0001, had a BMD greater than the highest dose level, or had a BMD/BMDL ratio of >20. DOs were then mapped to genes using an individual gene analysis to yield 1 BMD per gene. If more than 1 DO met the above criteria and were mapped to a gene, the average BMD and BMDL of the DO values for that gene were reported as the gene BMD.
Sets of biomarkers were identified by expert review of the literature, signatures were derived from the NTP DrugMatrix Database,124 and analysis of the existing data sets was conducted using the Illumina Correlation Engine.125 To ensure that all genes in a set reflect an active molecular process (as opposed to loss of activity), they were selected on the basis of their upregulation during a process (i.e., activation of peroxisome proliferator-activated receptor alpha [PPARα]). A list of the genes in each biomarker set are listed in Table F-1. Each biomarker set was evaluated for response in its target tissue and compared with reference chemical responses when appropriate. Importantly, the sets of biomarkers were not intended to diagnose a toxicological effect or mode of action explicitly but instead were meant to provide a weight of evidence, specifically the greater the number of genes responding in a biomarker set (i.e., upregulated with an associated BMD value), the greater confidence that a specific biological or toxicological process was occurring following exposure to the test article. To provide context, results from reference chemicals (DE-71, fenofibrate, and furan) were included in the biomarker set analysis when it was deemed that TCPP elicited a moderate-to-strong response of a biomarker gene set.
Table F-1. Gene Biomarker Sets Evaluated in the Five-day Toxicogenomic Study of Tris(chloropropyl) Phosphate
| Biomarker Set | Tissue Specificity | Genes in Biomarker Set | |
|---|---|---|---|
| AhR activation126-128 | Liver | Cyp1a2, Cyp1b1, Cyp1a1, Aldh3a1, Fmo1, Cyp2a1, Nqo1, Ahrr | |
| CAR/PXR activation127-130 | Liver | Cyp2b1, Cyp2b2, Ces2c, Abcc3, Aldh1a7, Zdhhc2, Gstp1, Akr7a3, Cyp3a23/3a1, Aldh1a1, Cyp3a2 | |
| PPARα activation127,128,131,132 | Liver | Acot1, Fabp3, Acot2, Cpt1b, Hdc, Vnn1, Vldr, Cidea, Pex11a, Lpl, Ehhadh, Chrna1, Cyp4a1, Acaa1a | |
| Nrf2 activation126,128,131,133,134 | Liver | Hmox1, Nqo1, Gsta2, Gsta5, Gstt3, Gstm1, Txnrd1, Gstp1, Ephx1, Prdx1, Gclc, Gclm, Srxn1, Gpx2 | |
| Estrogen receptor activation127,128 | Liver | Rgs3, Ablim3, Orm1, Rbp7, Ctr9, Lifr, Gdf15, Cited4 | |
| Inflammation132,135 | Any | Lcn2, Cxcl9, A2m, S100a9, Wfdc21, Pla1a, Il1b, Eb13, Marcks, Cd44, Ccl3, Tnfaip3, Serpinb1a, Timp1 | |
| Cell proliferation132,135 | Any | Mcm6, Mki67, Ccna2, Cdk1, Ccnb1, Top2a, Pclaf, Aurka, Rrm2, Bub1, Dscc1, Spc25 | |
| Genotoxic hepatocarcinogenicity128,136-139 | Liver | Ccng1, Mdm2, Ecn1, Adam8, Aen, Tyms, Phlda3, Bax, Abcb1b, cdkn1a, Nhej, Atp6v1d, Mgmt, Btg2, Pln | |
| Nongenotoxic hepatocarcinogenicity136,137,139-144 | Liver | Acot1, Abcc3, Knstm, Aldh1a1, Abcb1b, Adh7, Gdf15, Akr7a3, Ces2c, Zdhhc2 | |
| General liver pathology132,145,146 | Liver | Pla2g12, Aabcc3, Cyp1a1, Gucy2c, Gadd45b, Ces2c, Wfdc21, Ablim3, Slc4a4, Cidea, Enc1, Zdhhc2, Dact2, Ppl | |
| Overt liver toxicity (e.g., cell death, necrosis)132,147,148 | Liver | Abcb1b, Abcc3, Btg3, Hmox1, Rbm3, Snx10, Gnai1, Gpx2, Gpnmb, Lcn2, Ccl2, Pvr, Anxa2, Hspb1, Tnfrs12a, Enc1 | |
| Acute kidney injury145,146,149-151 | Kidney | Havcr1, Clu, Timp1, Cp, Anxa2, Gpnmb, Lcn2, Pqlc3, Fgb, Postn | |
F.3.
Results
Five days of oral exposure to TCPP caused an upregulated 122 genes in the liver compared to 727 genes by DE-71 (doses ≤500 mg/kg/day for 5 days), 616 genes by fenofibrate (doses ≤1,000 mg/kg/day for 5 days), and 631 genes by furan (doses ≤16 mg/kg/day for 5 days) in adult male rats (Appendix H). Of the upregulated genes in the liver following exposure to TCPP, the Ar (androgen receptor) had the lowest BMD1SD at 10 mg/kg/day (Figure F-1). Ar “steroid hormone receptors are ligand-activated transcription factors that regulate eukaryotic gene expression and affect cellular proliferation and differentiation in target tissues.”152-154 TCPP also upregulated 20 genes in rat kidney following 5 days of repeated exposure. Relative to chemicals that cause kidney toxicity and injury,106 the response to TCPP in the kidney was weak. In the kidney, the most sensitive upregulated gene was Ckap2 (cytoskeleton associated protein 2), which had a BMD1SD of 97 mg/kg/day (Figure F-2). Ckap2 “possesses microtubule stabilizing properties,” and “is involved in regulating aneuploidy, cell cycling, and cell death in a p53/TP53-dependent manner.”155 Notably, there were 26 upregulated genes with a BMD1SD below 97 mg/kg/day in rat liver indicating that the liver is likely a more sensitive target for TCPP than the kidney when administered by the oral route.
Figure F-1. Dose-response Plots for Ar in Rat Liver and Ckap2 in Rat Kidney Following Exposure to Tris(chloropropyl) Phosphate
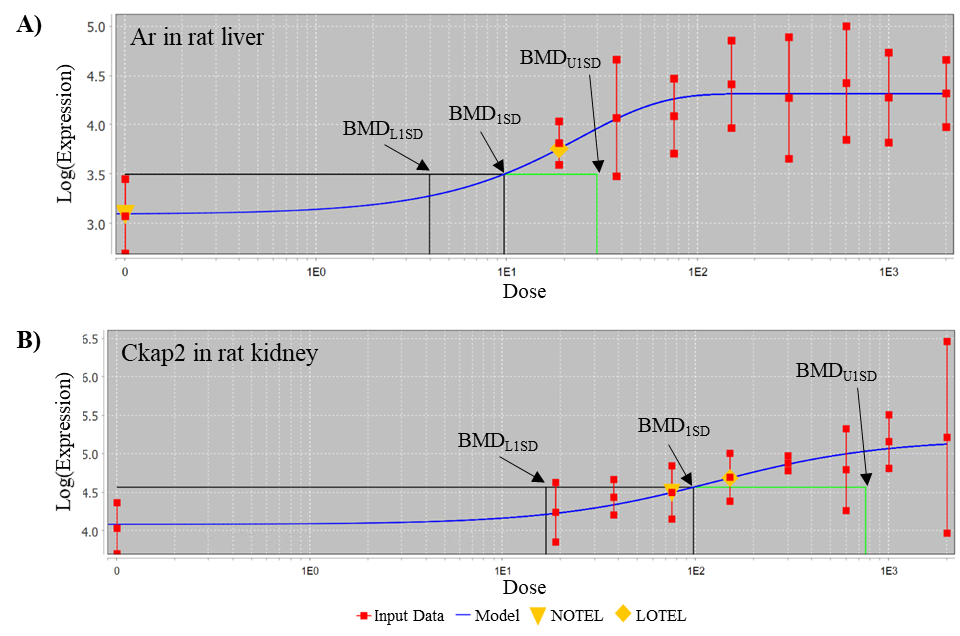
Doses are plotted along the horizontal axis and log2 counts per million expression levels are plotted on the vertical axis. A LOTEL is the lowest dose that demonstrates a significant change relative to the control group (t-test, p < 0.05). A) The BMD1SD, BMDL1SD, and BMDU1SD for Ar in rat liver are 10, 4, and 30 mg/kg/day, respectively. B) The BMD1SD, BMDL1SD, and BMDU1SD for Ckap2 in rat kidney are 97, 17, and 756 mg/kg/day, respectively. BMD1SD = benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean; BMDL1SD = benchmark dose lower confidence limit corresponding to a benchmark response set to 1 standard deviation from the mean; BMDU1SD = benchmark dose upper confidence limit corresponding to a benchmark response set to 1 standard deviation from the mean; LOTEL = lowest-observed-transcriptional-effect level; NOTEL = no-observed-transcriptional-effect level.
TCPP upregulated 7 out of 11 CAR and PXR-related biomarker genes in the liver with a low BMD1SD of 250 mg/kg/day. The genes in the biomarker set that were responsive to TCPP included Cyp3a23/3a1, Aldh1a1, Cyp2b1, Ces2c, Cyp2b2, Aldh1a7, and Zdhhc2 (Figure F-2). By comparison, the known CAR and PXR activator, DE-71,156 caused upregulation of 10 of the 11 CAR and PXR-related biomarker genes in the liver with a low BMD1SD of 0.3 mg/kg/day. In addition, TCPP also upregulated 8 out of 14 PPARα-related biomarker genes with a low BMD1SD of 105 mg/kg/day. The genes in the biomarker set that were responsive to TCPP included Ehhadh, Hdc, Vnn1, Cyp4a1, Acot2, Acaa1a, Cidea, and Acot1 (Figure F-3). By comparison, the known PPARα activator, fenofibrate,157 caused the upregulation of 12 out of 14 PPARα-related biomarker genes in the liver with the lowest, Ehaddh, having a BMD1SD of 0.6 mg/kg/day. There was moderate activity of the nuclear erythroid 2-related factor 2 (Nrf2) biomarker set with 4 out of 14 genes responding to TCPP administration with the lowest sensitive genes, Gsta2 and Gsta5, having a BMD1SD of 576 mg/kg/day. The genes in the biomarker set that were responsive to TCPP included Gsta5, Gsta2, Gstt3, and Gclm. By comparison, the known Nrf2 activator, furan,158 caused the upregulation of 6 out of 14 Nrf2 biomarker genes in the liver with the lowest having a BMD1SD of 3.1 mg/kg/day (Figure F-4). These data also demonstrated no activity in key biomarker genes in the liver related to aryl hydrocarbon receptor (AhR), estrogen receptor (ER), genotoxicity, inflammation, and cell proliferation signaling pathways (data not shown).
Figure F-2. Identification of Constitutive Androstane Receptor and Pregnane X Receptor-related Biomarker Genes within the Benchmark Dose Median Accumulation Plot of 123 Upregulated Genes in Rat Liver Following Tris(chloropropyl) Phosphate Exposure for Five Days
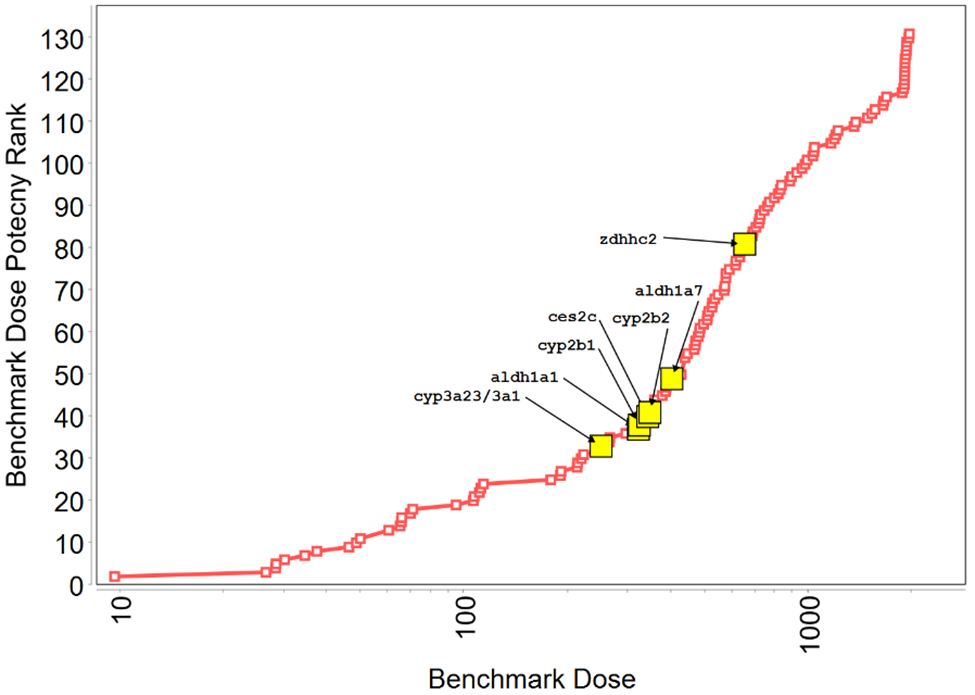
Seven out of 11 constitutive androstane receptor (CAR) and pregnane X receptor (PXR)-related biomarker genes were upregulated in response to tris(chloropropyl) phosphate (labeled in the plot and shown in yellow). Of the seven upregulated biomarker genes, Cyp3a23/3a1 had the lowest benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean (BMD1SD) of 250 mg/kg/day. By comparison, the prototype CAR and PXR activator, DE-71, showed upregulation of 10 out of 11 biomarker genes in the liver in a 5-day study. Red squares indicate each individual upregulated gene.
Figure F-3. Identification of Peroxisome Proliferator-activated Receptor Alpha-related Biomarker Genes within the Benchmark Dose Median Accumulation Plot of 123 Upregulated Genes in Rat Liver Following Tris(chloropropyl) Phosphate Exposure for Five Days
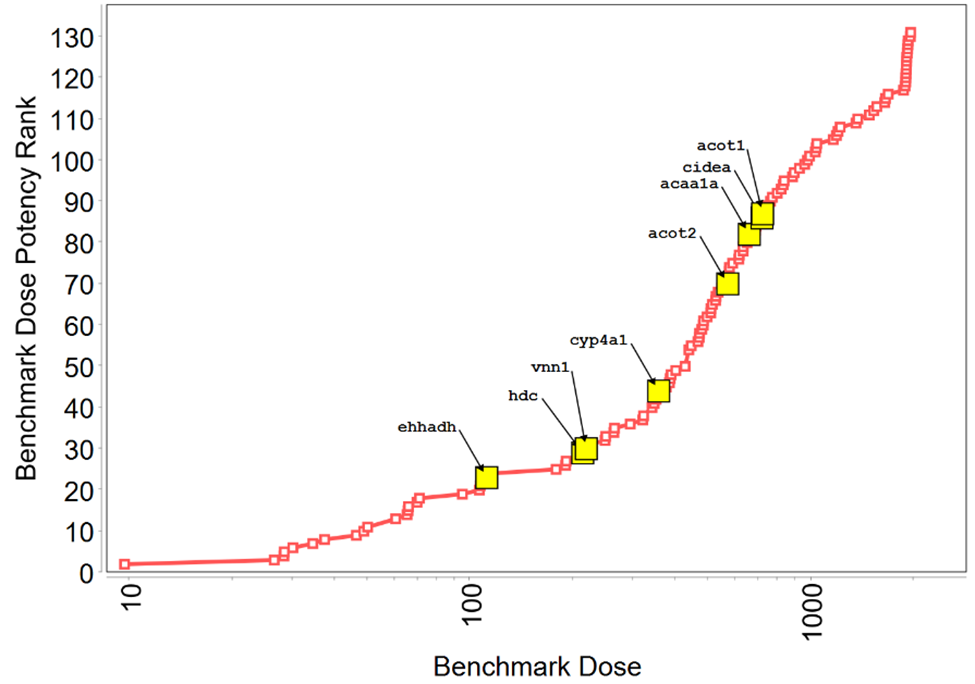
Eight out of 14 peroxisome proliferator-activated receptor alpha (PPARα)-related biomarker genes were upregulated in response to tris(chloropropyl) phosphate (labeled in the plot and shown in yellow). Of the eight upregulated biomarker genes, Ehhadh had the lowest benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean (BMD1SD) of 105 mg/kg/day. By comparison, the prototype PPARα activator, fenofibrate, showed upregulation of 12 out of 14 biomarker genes in the liver in a 5-day study. Red squares indicate each individual upregulated gene.
Figure F-4. Identification of Nuclear Erythroid 2-related Factor 2-related Biomarker Genes within the Benchmark Dose Median Accumulation Plot of 123 Upregulated Genes in Rat Liver Following Tris(chloropropyl) Phosphate Exposure for Five Days
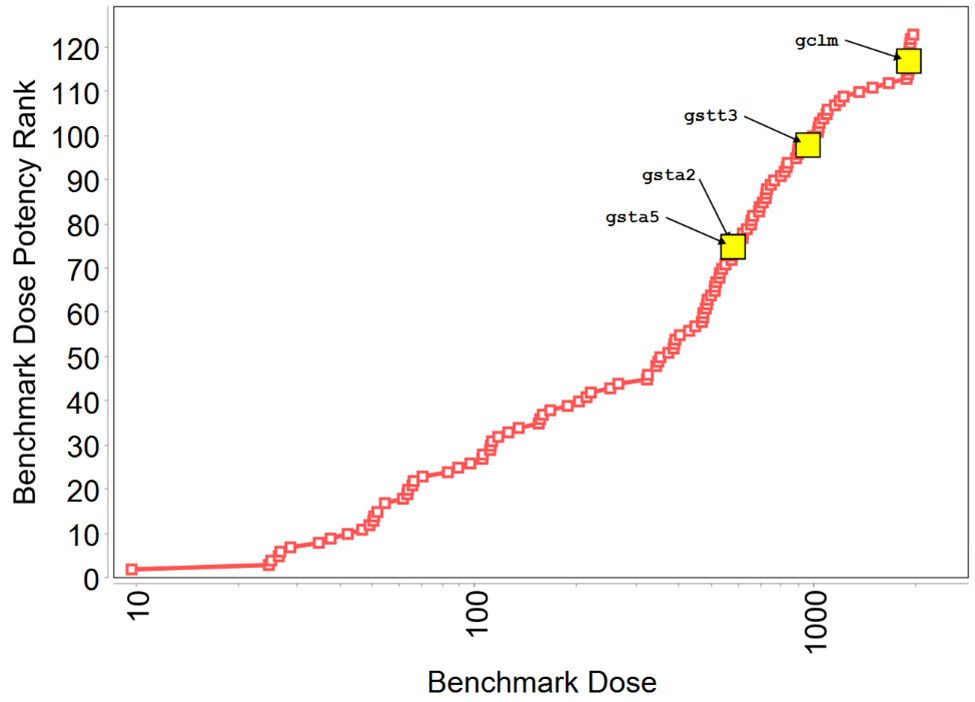
Four out of 14 nuclear erythroid 2-related factor 2 (Nrf2) related biomarker genes were upregulated in response to tris(chloropropyl) phosphate (labeled in the plot and shown in yellow). Of the four upregulated biomarker genes, Gsta2 and Gsta5 had the lowest benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean (BMD1SD) of 575 mg/kg/day. By comparison, the prototype Nrf2 activator, furan, showed upregulation of 6 out of 14 biomarker genes in the liver in a 5-day study. Red squares indicate each individual upregulated gene.
The TCPP liver gene expression data were also assessed for several biomarker sets associated with adverse effects. Four out of 14 biomarkers genes (Dact2, Ces2c, Zdhhc2, Cidea) indicative of general liver pathology were responsive to TCPP exposure in the liver with the lowest, Dact2, having a BMD1SD of 133 mg/kg/day (Figure F-5). By comparison, the prototypes DE-71, fenofibrate, and furan (all known to cause pathology in rat liver) caused upregulation of 10, 10, and 6 out of 14 general liver pathology biomarker genes in rat liver in the 5-day study, respectively. This finding suggests some potential for TCPP to cause liver pathology consistent with what is documented in the main body of the report. Notably, only one of the biomarker genes indicative of overt liver toxicity was responsive, denoting that pathological manifestations produced by TCPP are unlikely to induce overt hepatocellular injury at doses as high as 2,000 mg/kg/day (data not shown). Five out of 10 biomarkers genes (Aldh1a1, Ces2c, Zdhhc2, Acot1, Adh7) indicative of nongenotoxic hepatocarcinogenicity were responsive to TCPP exposure in the liver with the lowest, Aldh1a1, having a BMD1SD of 321 mg/kg/day (Figure F-6). By comparison, the prototypes DE-71, fenofibrate, and furan (all known to cause pathology in rat liver) caused upregulation of 7, 5, and 9 out of 10 nongenotoxic hepatocarcinogenicity biomarker genes in rat liver in the 5-day study, respectively. This response of the nongenotoxic hepatocarcinogenicity biomarker genes to TCPP was considered a moderate response and is consistent with the moderate, albeit dose-related, increases in hepatocellular carcinoma in the cancer bioassay. Finally, gene expression data from the kidney showed no upregulation of acute kidney injury biomarker genes.
Figure F-5. Identification of General Liver Pathology-related Biomarker Genes within the Benchmark Dose Median Accumulation Plot of 123 Upregulated Genes in Rat Liver following Tris(chloropropyl) Phosphate Exposure for Five Days
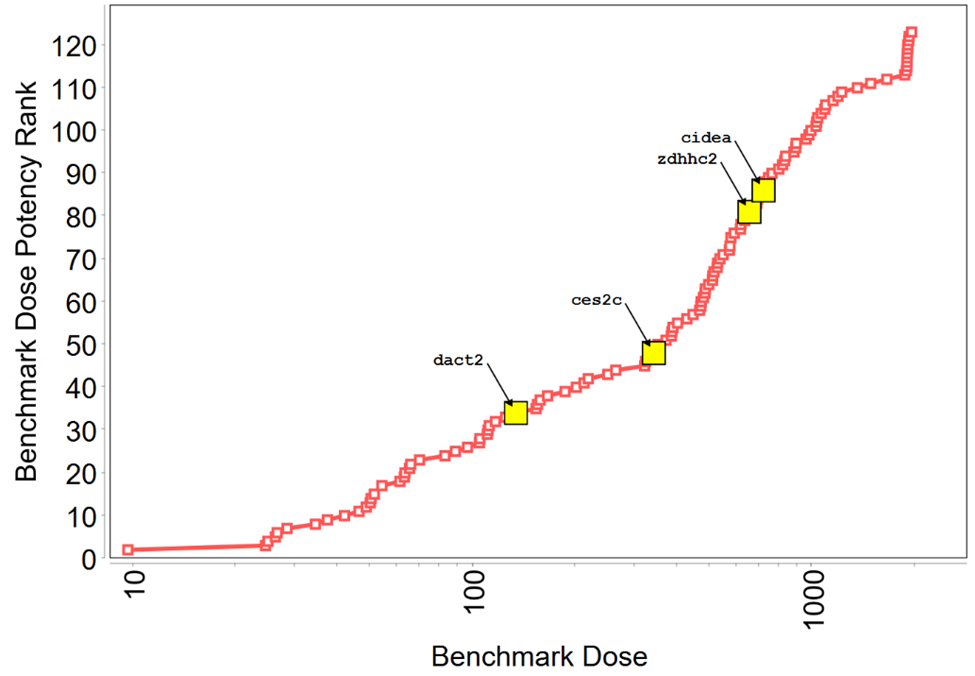
Four out of 14 general liver pathology-related biomarker genes were upregulated in response to tris(chloropropyl) phosphate (labeled in the plot and shown in yellow). Of the four upregulated biomarker genes, Dact2 had the lowest benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean (BMD1SD) of 134 mg/kg/day. By comparison, the prototypes DE-71, fenofibrate, and furan (all known to cause pathology in rat liver) caused upregulation of 10, 10, and 6 out of 14 biomarker genes in the liver in a 5-day study, respectively. Red squares indicate each individual upregulated gene.
Figure F-6. Identification of Nongenotoxic Hepatocarcinogenicity-related Biomarker Genes within the Benchmark Dose Median Accumulation Plot of 123 Upregulated Genes in Rat Liver following Tris(chloropropyl) Phosphate Exposure for Five Days
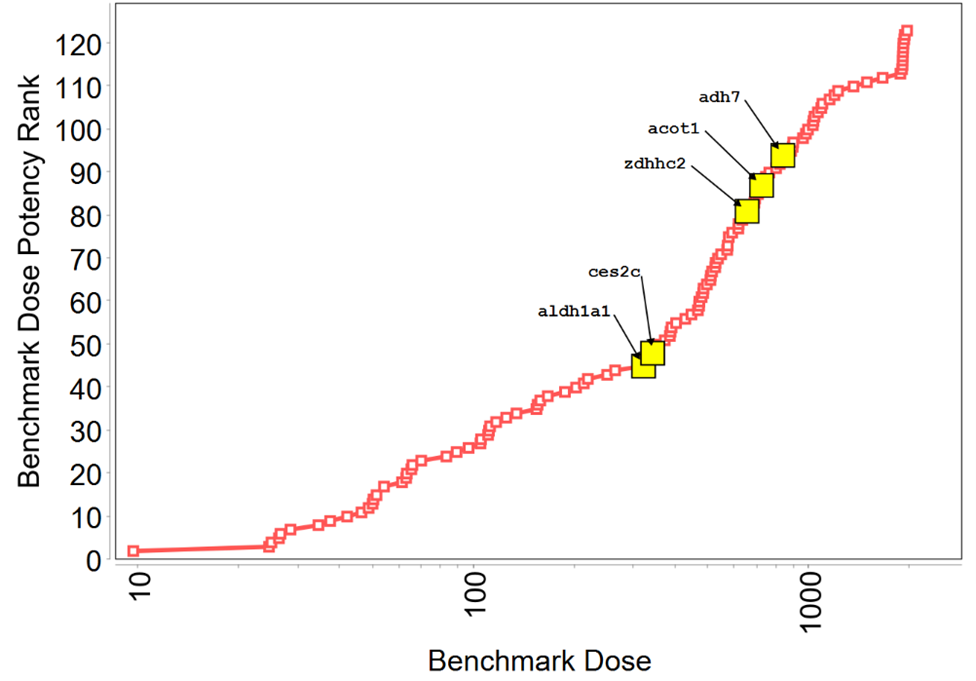
Five out of 10 nongenotoxic hepatocarcinogenicity-related biomarker genes were upregulated in response to tris(chloropropyl) phosphate (labeled in the plot and shown in yellow). Of the five upregulated biomarker genes, Aldh1a1 had the lowest benchmark dose corresponding to a benchmark response set to 1 standard deviation from the mean (BMD1SD) of 321 mg/kg/day. By comparison, the prototypes DE-71, fenofibrate, and furan (all known to cause pathology in rat liver) caused upregulation of 7, 5, and 9 out of 10 biomarker genes in the liver in a 5-day study, respectively. Red squares indicate each individual upregulated gene.
F.4.
Summary
In summary, the mechanistic and toxicological assessment of the transcriptomic data from the 5-day study suggest the liver is more responsive (i.e., overall number of genes responding) and potentially more sensitive to the oral effects of TCPP than is the kidney. The androgen receptor (Ar) in the liver had the lowest benchmark dose of one standard deviation (BMD1SD) of 10 mg/kg/day (95% confidence interval: 3.9 to 29.6 mg/kg/day) and the most sensitive endpoint in the kidney was cytoskeleton associated protein 2 (Ckap2) with a BMD1SD of 97 mg/kg/day (95% confidence interval: 16.7 to 756 mg/kg/day), suggesting the liver may be more sensitive than the kidney to TCPP exposure via the oral route. TCPP is a CAR, PXR, and PPARα activator in the rat liver, although to a lesser degree than the reference chemicals, DE-71 and fenofibrate. Other observations included possible Nrf2 activation in rat liver at higher doses.
Appendix G. Multiomics Evaluation of B6C3F1/N Mouse Hepatocellular Carcinomas Arising Spontaneously or Following Chronic Exposure to Tris(chloropropyl) Phosphate
G.1.
Introduction
The National Toxicology Program (NTP) has integrated diverse data sources derived from experimental systems with varying biological complexity to understand the mechanisms of toxicity and carcinogenicity induced by the isomeric mixture of tris(chloropropyl) phosphate (TCPP). These diverse data sources include quantitative high-throughput in vitro screening (qHTS) data, short-term toxicogenomic data, and next-generation sequencing (NGS) data of the mouse liver tumors. This appendix summarizes the NGS data on mouse hepatocellular carcinomas (HCCs) arising spontaneously or following chronic exposure to TCPP.
HCC is the third leading cause of cancer-related death in humans across the world.159 It is also the most common primary liver tumor in B6C3F1/N mice and may arise either spontaneously or due to chronic chemical exposure, and male mice have twice the incidence rate compared with female mice.160 Hepatocellular carcinogenesis is a multistep process and chemical exposure can modulate the pathogenesis. Genetic and epigenetic alterations that progressively accumulate over a lifetime can contribute to various stages in the carcinogenic process.161 Assessment of molecular alterations in rodent HCCs from vehicle control and chemical-exposed experimental animals can provide some insight into the mechanisms of chemical-induced carcinogenesis.160
Current developments in sequencing technologies have dramatically changed the field of genomics research. NGS encompasses different techniques, such as whole-genome/exome sequencing, RNA sequencing (RNA-seq), and miRNA sequencing (miRNA-seq). Integrated analytic approaches applied to one or more data platforms (whole-exome sequencing, DNA copy number analyses, DNA methylation, RNA, miRNA, and proteomic expression) can provide a better understanding of the carcinogenic process.162,163
G.2.
Materials and Methods
G.2.1.
Multiomics Analyses on Mouse Hepatocellular Carcinomas Arising Spontaneously or Following Chronic Exposure to Tris(chloropropyl) Phosphate
G.2.1.1.
Animals and Tissue Sampling
HCCs arising either spontaneously or following chemical exposure in B6C3F1/N mice were obtained from the current TCPP 2-year bioassay. HCCs from male mice exposed for 2 years via feed dosed with 1,250 ppm (n = 5) or 5,000 ppm (n = 5) of TCPP (Table G-1) were selected for this study. Samples of nontumor livers and spontaneous HCCs from age-matched mice were obtained from the current study or other NTP studies run within the last 10 years. At necropsy, liver tumors larger than 0.5 mm were flash frozen in liquid nitrogen and stored at −80°C until DNA/RNA isolation. The morphology of liver tumor samples selected for molecular analyses was confirmed independently by two board-certified pathologists. Male B6C3F1/N mouse samples of HCC from TCPP-exposed mice (n = 10), spontaneous HCCs (n = 21), and age-matched nontumor livers from vehicle control groups (n = 10) were included in the molecular analyses.
Table G-1. Summary of Hepatocellular Carcinomas and Nontumor Liver Samples from B6C3F1/N Male Mice in the Two-year Feed Study of Tris(chloropropyl) Phosphate and Nontumor Liver Samples from Male Mice in Two-year Feed Studies
| TCPP Exposure Concentration (ppm) | n | HCC Type |
|---|---|---|
| 0 | 10a | Nontumor tissue |
| 0 | 21b | Spontaneous |
| 1,250 | 5c | TCPP exposed |
| 5,000 | 5c | TCPP exposed |
G.2.1.2.
DNA Extraction, Whole-exome Sequencing, and Variant Calling
Genomic DNA was extracted and purified using a Puregene Gentra Kit (Qiagen, Valencia, CA). Exome-enriched genomic libraries were prepared from all 41 collected samples (Table G-1) using the SureSelectXT Mouse All Exon kit (Agilent, CA) following the manufacturer’s recommendations with an input of 1 µg DNA. Sequencing was performed on the Illumina HiSeq 2500 at LabCorp (Seattle, WA). Paired-end whole-exome sequencing data were assessed with FastQC (v.0.11.5) for quality control, and sequencing adapters were trimmed with Cutadapt (v.1.12). The short reads were aligned with the Burrows-Wheeler Alignment (BWA) tool with maximal exact matches178 against reference mouse genome (mm10). Alignment bam files have gone through base calling quality recalibration with the GATK toolkit.162 Variant calling was accomplished by Mutect2 tumor-only mode and against an in-house panel of normal single nucleotide variants (SNV; genomic sequence in vehicle control mice used as a filter) informed by the age-matched nontumor liver samples. Known germline mutations obtained from an in-house whole-genome sequencing project (data not shown) and a mm10 dbSNP database were also excluded. A mutation was only called if it was within the captured regions of 100 base pairs (bp) upstream and downstream of the target exon regions. Only Mutect2 PASS SNVs were reported.
G.2.1.3.
RNA Isolation, RNA Sequencing, miRNA Sequencing, and Gene Expression Analysis
Total RNA was extracted using miRNeasy Mini Kit (Qiagen, Valencia, CA). RNA integrity was measured with Tapestation (Agilent Technologies, Santa Clara, CA) and all samples had an RNA integrity number (RIN) >7. RNA-seq libraries were prepared for 22 samples (TCPP-exposed HCCs [n = 5], spontaneous HCCs [n = 11], and age-matched nontumor livers from vehicle control mice [n = 6]) (Table G-1) by following the TruSeq Stranded Total RNA Library Prep Gold Kit (Illumina, San Diego, CA) manufacturer’s protocol with an input of 250 ng total RNA. The libraries were sequenced on the Illumina NovaSeq system at the National Institute of Environmental Health Sciences (NIEHS) Epigenomics and DNA Sequencing core facility (Durham, NC).
Raw reads (stranded, 151 bp, paired-end) were first filtered with a custom Perl script to remove reads of average quality score of ≤20, and then processed using the Cutadapt (v.1.12)179 program to remove adapter sequences. Mouse nucleotide sequences of all transcripts were retrieved from GENCODE release M23.180-182 The mapping-based mode of the Salmon (v1.0.0)183 tool was deployed for fast transcript quantification from RNA-seq data to the reference mouse genome (mm10). The quantified transcripts were imported using the R package tximport184 and also summarized to gene level with countsFromAbundance = “lengthScaledTPM.” Differential transcript and/or gene expression between TCPP-exposed or spontaneous HCC samples and the control samples was analyzed using Limma-Voom procedure (R package limma, v3.42.2).185-187 Genes were considered differentially expressed at an absolute fold-change of two using a false-discovery rate (FDR) adjusted p value (or q value) of 0.05 and AvgExpr more than 0 threshold.
miRNA-seq libraries were prepared for all 41 samples (Table G-1) by following the NEXTFLEX Small RNA-Seq Kit v3 (PerkinElmer Applied Genomics, Waltham, MA) manufacturer’s protocol with an input of 1 µg total RNA. The library preparation was sequenced on the Illumina NextSeq system at the NIEHS Epigenomics and DNA Sequencing core facility (Durham, NC).
FastQC software was used to assess overall sequencing quality.188 Unique molecular identifiers (UMI) were extracted using the UMI-tools software.189 Reads were aligned to the mm10 genome with the STAR aligner v.2.6.0.190 Mouse miRNAs and coordinates were obtained from the miRBase repository.191 The featureCounts utility from the Subread package was used to assign reads to miRNAs with -M and -R flags set to detect multimapped (e.g., same alignment position and UMI) and unassigned reads, respectively.192 The samtools view function was subsequently used to discard unassigned reads and duplicates were removed with UMI-tools dedup.193 Final UMI counts per miRNA were quantified with featureCounts.192 The DESeq2 R package v.1.26 was used to detect differentially expressed miRNAs. Genes were considered differentially expressed at an absolute fold-change of two using a 0.01 FDR threshold.194
G.2.1.4.
Mutational Signature Analysis and Cancer Driver Gene Analysis
The MutationalPatterns v.3.2.0 R package195 was used to identify mutational processes196 by fitting the mutational signatures published in the Catalogue of Somatic Mutations in Cancer (COSMIC) catalog2 to the mutational profiles of the somatic SNVs in each tumor (https://cancer.sanger.ac.uk/signatures/). The latter were obtained by classifying SNVs into 96 categories according to six different possible base-pair substitutions: C > A, C > G, C > T, T > A, T > C, and T > G (interpreting the pyrimidine base in the Watson–Crick pair as the reference base and the bases immediately 5′ and 3′ to the mutated base in the reference genome).196 Fitting of mutational signatures to somatic variants was initially performed using all 60 published COSMIC signatures. SnpEff was used to predict and annotate the effects of variants on genes.197 Substitutions were selected that altered coding sequence: missense, synonymous, splice site mutations, start lost/gained, or stop lost/gained substitutions. Raw sequence read data were uploaded to the National Center for Biotechnology Information Sequence Read Archive (accession: PRJNA779957).
G.2.1.5.
Functional Enrichment Analysis of Genes
To identify the biological function of the differentially expressed genes and related toxicity difference between TCPP-exposed HCCs and spontaneous HCCs, Toxicity Lists (Tox Lists) analysis was performed using Ingenuity pathway analysis (IPA) (QIAGEN Ingenuity Systems, Redwood City, CA). Tox Lists are sets of molecules that are known to be involved in particular types of toxicological pathways.
G.2.2.
Statistical Analysis
Fisher’s exact test was conducted to test for significant differences in the number of mutations between TCPP-exposed HCCs and spontaneous HCCs. Plots were created using R198 with packages ggplot2, pheatmap, and VennDiagram.
G.3.
Results
G.3.1.
Comparison of the Mutation Landscape between Hepatocellular Carcinomas Arising Spontaneously or Following Tris(chloropropyl) Phosphate Exposure
In male mice, the mutation burden of HCCs arising spontaneously was compared with exposure to low dose (1,250 ppm) and high dose (5,000 ppm) of TCPP. On average, the sequence depth for the whole-exome sequencing was approximately 150-fold. There were no differences in mutation burden between HCCs arising spontaneously and following chronic TCPP exposure (Figure G-1). In addition, there were no differences in mutation burden between the highest (5,000 ppm) and the lowest (1,250 ppm) TCPP exposures.
Figure G-1. Mutation Burden of Hepatocellular Carcinomas in B6C3F1/N Male Mice Arising Spontaneously or Following Chronic (Two-year) Exposure to Tris(chloropropyl) Phosphate
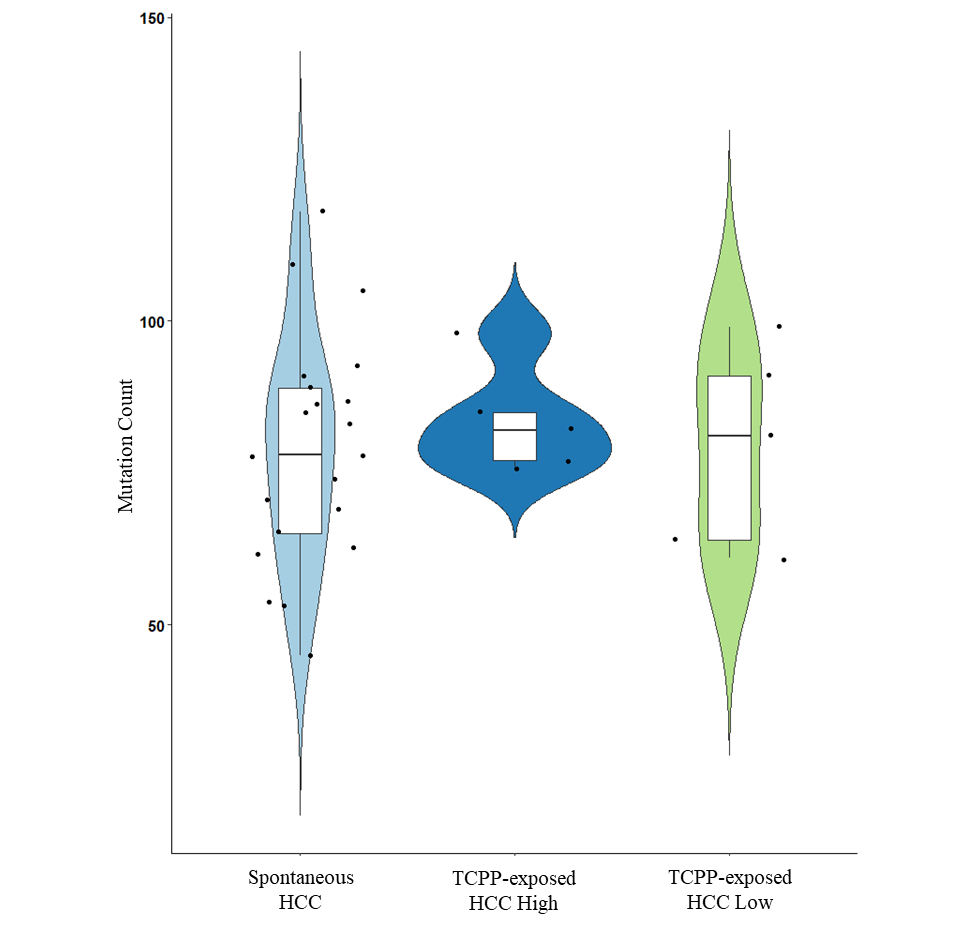
HCC = hepatocellular carcinoma; TCPP-exposed HCC = HCC following tris(chloropropyl) phosphate exposure; High = 5,000 mg TCPP/kg body weight/day (mg/kg/day); Low = 1,250 mg/kg/day.
To determine whether TCPP might preferentially induce mutations in specific nucleotides, mutational spectra were generated according to the trinucleotide context as described in the methods section of this appendix. The nonnegative matrix factorization plots (Figure G-2) show that the mutation spectra for HCCs arising spontaneously or following chronic high or low TCPP exposure were similar to each other, mainly showing C > T or G > A transitions. A cosine correlation similarity was used as a measure of closeness between human COSMIC signature and the mutational spectra of HCCs arising spontaneously or due to chronic TCPP exposure. Three signatures were identified with a cosine similarity higher than 0.70 with a COSMIC signature; the best COSMIC signature matches were SBS5 in TCPP-exposed HCCs and spontaneous HCCs, and SBS40 and SBS3 in TCPP-exposed HCCs (Table G-2). In human cancers, SBS40 and SBS5 are considered clock-like signatures and are correlated with the patient’s age.199 SBS3 has been suggested to indicate defective homologous recombination-based DNA damage repair.199
In addition to the mutation signatures, mutations in cancer driver genes were documented that identify biological pathways that promote cancer. Hras, Ube2c, Sfr1, and Muc4 were found to be highly mutated in both TCPP-exposed HCCs and spontaneous HCCs. The most frequently mutated genes are indicated in Figure G-3. Interestingly, Ctnnb1 was highly mutated only in spontaneous HCCs but not in TCPP-exposed HCCs.
Figure G-2. Mutational Spectra of Hepatocellular Carcinomas in B6C3F1/N Male Mice Arising Spontaneously or Following Chronic (Two-year) Exposure to Tris(chloropropyl) Phosphate
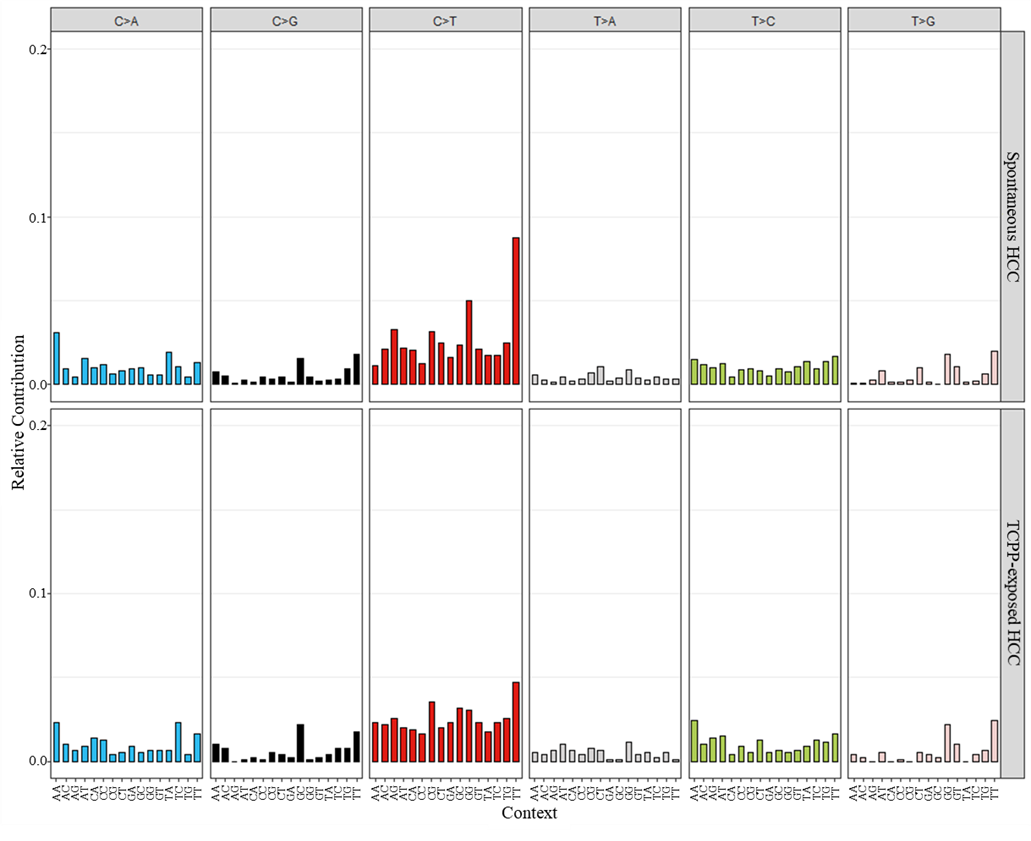
C = cytosine; A = adenine; G = guanine; T = thymine; HCC = hepatocellular carcinoma; TCPP-exposed HCC = HCC following tris(chloropropyl) phosphate exposure.
Table G-2. Comparison of Mutational Spectra of Spontaneous and Tris(chloropropyl) Phosphate-exposed Hepatocellular Carcinomas in B6C3F1/N Male Mice from the Two-year Study to Human Signatures
| COSMIC Signature Best Match | Cosine Similarity | |
|---|---|---|
| Spontaneous HCC | TCPP-exposed HCC | |
| SBS5 | 0.713609169 | 0.82649 |
| SBS40 | 0.689305018 | 0.766232 |
| SBS3 | 0.595527185 | 0.705439 |
| SBS89 | 0.550977772 | 0.662781 |
| SBS25 | 0.548747391 | 0.625432 |
| SBS6 | 0.58110974 | 0.619277 |
| SBS32 | 0.587950468 | 0.61254 |
| SBS42 | 0.467218617 | 0.601773 |
| SBS30 | 0.492207486 | 0.577837 |
| SBS44 | 0.463672159 | 0.569612 |
| SBS11 | 0.531883449 | 0.551167 |
| SBS84 | 0.406918893 | 0.508751 |
| SBS15 | 0.523454849 | 0.504139 |
| SBS23 | 0.42163454 | 0.500457 |
| SBS87 | 0.45417571 | 0.498804 |
Figure G-3. Most Frequently Mutated Genes in Hepatocellular Carcinomas in B6C3F1/N Male Mice
This list shows results for hepatocellular carcinomas (A) arising spontaneously or (B) following chronic (2-year) exposure to tris(chloropropyl) phosphate.
G.3.2.
Gene Expression Profiling and Functional Enrichment Analysis
Genome-wide gene expression profiling of 21 samples was performed to identify genes that were differentially expressed across different groups. Principal component analysis (PCA) of mouse mRNA expression profiles demonstrated spontaneous HCC and TCPP-exposed HCCs clustered separately from age-matched vehicle control nontumor liver (Figure G-4A). As seen in the unsupervised hierarchical clustering analysis of the transcriptomic data, the gene expression patterns were similar in HCCs arising spontaneously or following chronic TCPP exposure (Figure G-4B). The PCA indicates the tumor heterogeneity in HCCs arising spontaneously or following chronic TCPP exposure. HCCs arising spontaneously or following chronic TCPP exposure had 789 or 173 uniquely altered genes, respectively, and 1,295 differentially altered genes in common (Figure G-4C).
Figure G-4. Differential Gene Expression Profiles of Hepatocellular Carcinomas in B6C3F1/N Male Mice Arising Spontaneously or Following Chronic (Two-year) Exposure to Tris(chloropropyl) Phosphate
A) A principal components analysis (PCA) (dark blue = vehicle control nontumor liver; orange = spontaneous hepatocellular carcinomas (HCCs); green = HCCs from TCPP-exposed mice. B) A hierarchical cluster analysis showing the comparable gene expression changes in HCCs arising spontaneously or following TCPP exposure. C) Venn diagram showing the number of common or differentially expressed genes that are unique to HCCs arising spontaneously or due to TCPP exposure. HCC = hepatocellular carcinoma; TCPP-exposed HCC = HCC following tris(chloropropyl) phosphate exposure.
To link the biological function of the differentially expressed genes to TCPP-exposed HCCs, Tox Lists analysis in IPA was performed. Pathways related to the development of HCCs included liver hyperplasia and/or hyperproliferation, aryl hydrocarbon receptor (AhR) signaling, lipopolysaccharide and interleukin-1 (LPS/IL-1)-mediated inhibition of retinoid x receptor (RXR) function, xenobiotic metabolism signaling, and hepatic fibrosis; these pathways were common to both HCCs arising spontaneously or following chronic TCPP exposure. Top unique Toxicity List (pathways) altered in HCCs arising spontaneously or following exposure to TCPP were related to fatty acid metabolism and liver proliferation, respectively (Figure G-5).
Figure G-5. Top 10 Tox Lists Derived from Ingenuity Pathway Analysis of Hepatocellular Carcinomas in B6C3F1/N Male Mice Arising Spontaneously or Following Chronic (Two-year) Exposure to Tris(chloropropyl) Phosphate
LPS/IL-1 = lipopolysaccharide/interleukin-1; RXR = retinoid X receptor; LXR = liver X receptor; NRF2 = nuclear factor-erythroid factor 2-related factor 2; FXR = farnesoid X receptor; RAR = retinoic acid receptor; HCC = hepatocellular carcinoma; TCPP-exposed HCC = HCC following tris(chloropropyl) phosphate exposure.
G.3.3.
miRNA Expression Profiles
Differentially expressed miRNA profiles of HCCs arising spontaneously or following chronic TCPP exposure relative to age-matched vehicle control nontumor liver tissue were examined. PCA of mouse global miRNA expression profiles demonstrated spontaneous HCC and TCPP-exposed HCCs clustered separate from age-matched vehicle control nontumor liver (Figure G-6A). The unsupervised hierarchical clustering analysis of miRNA expression showed similar patterns for both HCCs arising spontaneously or following chronic TCPP exposure (Figure G-6B). HCCs arising spontaneously or following chronic TCPP exposure had 49 and 17 miRNA uniquely altered, respectively, and had 128 differentially altered miRNA in common (Figure G-6C).
Figure G-6. Differential miRNA Expression Profiles of Hepatocellular Carcinomas in B6C3F1/N Male Mice Arising Spontaneously or Following Chronic (Two-year) Exposure to Tris(chloropropyl) Phosphate
A) A principal components analysis (PCA) (blue = vehicle control nontumor liver tissue; orange = spontaneous hepatocellular carcinomas (HCCs); green = TCPP-exposed HCCs). B) A hierarchical cluster analysis showing the comparable miRNA alterations in HCCs arising spontaneously or following TCPP exposure. C) Venn diagram showing the number of common or differentially expressed miRNA that are unique to spontaneous HCCs and/or TCPP-exposed HCCs. TCPP-exposed HCC = HCC following tris(chloropropyl) phosphate exposure.
G.4.
Summary
Conclusions from NTP animal studies and bacterial assays have determined that TCPP is largely nongenotoxic (e.g., negative Ames assay, micronucleus assay in male and female rats, and micronucleus assay in female mice; equivocal micronucleus test in male mice) (Appendix H); thus, TCPP likely does not directly cause mutations and initiate carcinogenesis. The evaluation of NGS data on B6C3F1/N mouse HCCs suggested that the mutation burden and mutation spectrum of TCPP-exposed HCCs were similar to spontaneous HCCs. Additional mutation signatures (SBS3 and SBS40) in TCPP-exposed HCCs may reflect additional genetic events related to tumor promotion, such as deficits in DNA damage repair, but further studies are needed to confirm this hypothesis. The cancer driver genes identified in this study were common to both HCCs arising spontaneously or following chronic TCPP exposure. The homologues of Hras, Muc4, and Ctnnb1 are also cancer driver genes in human cancers. The lack of enrichment for Ctnnb1 mutations in TCPP-exposed HCCs compared with spontaneous HCCs suggest that TCPP may not be a strong CAR or PXR activator compared with ginkgo biloba extract200 or phenobarbital201 given that there were higher frequencies of Ctnnb1 mutations in mouse HCCs from these exposures. The gene expression pattern and miRNA expression pattern of TCPP-exposed HCCs were also similar to spontaneous HCCs and the Toxicity List (pathways) of TCPP-exposed HCCs and spontaneous HCCs analyzed in IPA were comparable. The differentially expressed genes and miRNAs that are unique to TCPP-exposed HCCs may be considered as potential biomarkers to distinguish TCPP-exposed HCCs from spontaneous HCCs; however, further validation by reverse transcription polymerase chain reaction (RT-PCR) is pending. The differential gene expression data suggest that TCPP may act as a tumor promoter by weakly interacting with multiple nuclear receptors such as CAR, PXR, PPARα, AhR, and RXR in rodent hepatocellular carcinogenesis.202-204 Further validation and mechanistic studies are needed, however, to confirm their definitive roles in TCPP-induced hepatocellular carcinogenesis.
Appendix H. Supplemental Data
Tables with supplemental data can be found here: https://doi.org/10.22427/NTP-DATA-TR-602.
About This Report
Foreword
The National Toxicology Program (NTP), established in 1978, is an interagency program within the Public Health Service of the U.S. Department of Health and Human Services. Its activities are executed through a partnership of the National Institute for Occupational Safety and Health (part of the Centers for Disease Control and Prevention), the Food and Drug Administration (primarily at the National Center for Toxicological Research), and the National Institute of Environmental Health Sciences (part of the National Institutes of Health), where the program is administratively located. NTP offers a unique venue for the testing, research, and analysis of agents of concern to identify toxic and biological effects, provide information that strengthens the science base, and inform decisions by health regulatory and research agencies to safeguard public health. NTP also works to develop and apply new and improved methods and approaches that advance toxicology and better assess health effects from environmental exposures.
The Technical Report series began in 1976 with carcinogenesis studies conducted by the National Cancer Institute. In 1981, this bioassay program was transferred to NTP. The studies described in the NTP Technical Report series are designed and conducted to characterize and evaluate the toxicological potential, including carcinogenic activity, of selected substances in laboratory animals (usually two species, rats and mice). Substances (e.g., chemicals, physical agents, and mixtures) selected for NTP toxicity and carcinogenicity studies are chosen primarily on the basis of human exposure, level of commercial production, and chemical structure. The interpretive conclusions presented in NTP Technical Reports are derived solely from the results of these NTP studies, and extrapolation of the results to other species, including characterization of hazards and risks to humans, requires analyses beyond the intent of these reports. Selection for study per se is not an indicator of a substance’s carcinogenic potential.
NTP conducts its studies in compliance with its laboratory health and safety guidelines and the Food and Drug Administration Good Laboratory Practice Regulations and meets or exceeds all applicable federal, state, and local health and safety regulations. Animal care and use are in accordance with the Public Health Service Policy on Humane Care and Use of Laboratory Animals. Studies are subjected to retrospective quality assurance audits before they are presented for public review. Draft reports undergo external peer review before they are finalized and published.
NTP Technical Reports are available free of charge on the NTP website and cataloged in PubMed, a free resource developed and maintained by the National Library of Medicine (part of the National Institutes of Health). Data for these studies are included in NTP’s Chemical Effects in Biological Systems database.
For questions about the reports and studies, please email NTP or call 984-287-3211.
Collaborators
Division of Translational Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina, USA
Designed studies, evaluated and interpreted results, and reported findings
K.R. Ryan, Ph.D., Study Scientist
A.R. Pandiri, B.V.Sc., Ph.D., Study Pathologist
S.S. Auerbach, Ph.D.
C.R. Blystone, Ph.D.
M.F. Cesta, D.V.M., Ph.D.
B.J. Collins, M.S.P.H.
M.C. Cora, D.V.M.
H.C. Cunny, Ph.D.
D.R. Germolec, Ph.D.
M.J. Hooth, Ph.D.
J-H. Hsieh, Ph.D.
A.P. King-Herbert, D.V.M.
D.E. Malarkey, D.V.M., Ph.D. (Retired)
B.S. McIntyre, Ph.D.
S.S. Mehta, Dr.P.H.
G.K. Roberts, Ph.D.
K.A. Shipkowski, Ph.D.
K.R. Shockley, Ph.D.
S.L. Smith-Roe, Ph.D.
M.D. Stout, Ph.D.
G.S. Travlos, D.V.M.
S. Waidyanatha, Ph.D.
N.J. Walker, Ph.D.
K.L. Witt, M.S. (Retired)
M. Xu, Ph.D.
Provided oversight for data management
J.M. Fostel, Ph.D.
Battelle, Columbus, Ohio, USA
Conducted studies and evaluated pathology findings
M.R. Hejtmancik, Ph.D., Principal Investigator
B.R. Sparrow, Ph.D., Principal Investigator
M.A. Buccellato, D.V.M., Ph.D.
D.M. Fallacara, Ph.D.
L.M. Fomby, D.V.M., Ph.D.
M.J. Ryan, D.V.M., Ph.D.
A.J. Skowronek, D.V.M., Ph.D.
D.Y. Vasconcelos, D.V.M., Ph.D.
Conducted prestart chemistry activities and dose formulations
W.M. Black, B.S.
B.L. Burback, Ph.D.
K.A. Carrico, B.A.
D.A. Contos, M.S.
S.W. Graves, B.S.
J.S. Richey, M.S.
MRIGlobal, Kansas City, Missouri, USA
Conducted preliminary chemistry activities, dose formulations, and biological sample chemistry analyses
K.L. Aillon, Ph.D., Principal Investigator
J.W. Algaier, Ph.D., Principal Investigator
J. Cookinham, B.S.
A. Kazerooni, M.S.
S.P. Kerns, M.S.
M. Kroenke, B.S.
A.J. Mainey, B.S.
B. O’Brien, M.S.
D. Slade, Ph.D.
W.R. Vandaveer, Ph.D.
Experimental Pathology Laboratories, Inc., Research Triangle Park, North Carolina, USA
Provided pathology review
K.Y. Cimon, D.V.M., M.S.
E.M. Quist, D.V.M., Ph.D.
Coordinated Pathology Working Group on perinatal and 2-year rats (October 8, 2019) and Pathology Peer Review on uterine lesions in perinatal and 2-year rats (November 7, 2019)
E.M. Quist, D.V.M., Ph.D.
Coordinated Pathology Working Group on 2-year mice (October 10, 2019)
K.Y. Cimon, D.V.M., M.S.
Integrated Laboratory Systems, LLC, Research Triangle Park, North Carolina, USA
Coordinated Pathology Peer Reviews on perinatal and 3-month rats and 3-month mice (May 1, 2012) and on thymus lesions in perinatal and 3-month rats (January 26, 2015)
D.K. Giri, D.V.M., Ph.D.
RTI International, Research Triangle Park, North Carolina, USA
Provided sperm count and vaginal cytology evaluation (SCVCE) analysis
R.W. Tyl, Ph.D., Principal Investigator
C.S. Sloan, M.S.
ICF, Fairfax, Virginia, USA
Conducted technical writing and data integration and ensured report quality
K.S. Duke, Ph.D.
Contributors
Division of Translational Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina, USA
Provided oversight of external peer review
M.S. Wolfe, Ph.D.
M.L. Brownlow, Ph.D.
Kelly Government Solutions, Research Triangle Park, North Carolina, USA
Supported external peer review
E.A. Maull, Ph.D.
Pathology Working Group, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina, USA
Participated in Pathology Working Group on perinatal and 2-year rats (October 8, 2019)
A.E. Brix, D.V.M., Ph.D., Experimental Pathology Laboratories, Inc.
M.F. Cesta, D.V.M., Ph.D., National Institute of Environmental Health Sciences
D. Dixon, D.V.M., Ph.D., National Institute of Environmental Health Sciences
S.A. Elmore, D.V.M., M.S., National Institute of Environmental Health Sciences
M.R. Elwell, D.V.M., Ph.D., Apex ToxPath, LLC
D.E. Malarkey, D.V.M., Ph.D., National Institute of Environmental Health Sciences
A.R. Pandiri, B.V.Sc., Ph.D., National Institute of Environmental Health Sciences
Participated in Pathology Working Group on 2-year mice (October 10, 2019)
A.E. Brix, D.V.M., Ph.D., Experimental Pathology Laboratories, Inc.
M.F. Cesta, D.V.M., Ph.D., National Institute of Environmental Health Sciences
D. Dixon, D.V.M., Ph.D., National Institute of Environmental Health Sciences
S.A. Elmore, D.V.M., M.S., National Institute of Environmental Health Sciences
M.R. Elwell, D.V.M., Ph.D., Apex ToxPath, LLC
D.E. Malarkey, D.V.M., Ph.D., National Institute of Environmental Health Sciences
A.R. Pandiri, B.V.Sc., Ph.D., National Institute of Environmental Health Sciences
Pathology Peer Review, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina, USA
Participated in Pathology Peer Review on uterine lesions in perinatal and 2-year rats (November 7, 2019)
A.E. Brix, D.V.M., Ph.D., Experimental Pathology Laboratories, Inc.
M.F. Cesta, D.V.M., Ph.D., National Institute of Environmental Health Sciences
D. Dixon, D.V.M., Ph.D., National Institute of Environmental Health Sciences
S.A. Elmore, D.V.M., M.S., National Institute of Environmental Health Sciences
A.R. Pandiri, B.V.Sc., Ph.D., National Institute of Environmental Health Sciences
Participated in Pathology Peer Review on perinatal and 3-month rats and 3-month mice (May 1, 2012)
M.F. Cesta, D.V.M., Ph.D., National Institute of Environmental Health Sciences
K.Y. Cimon, D.V.M., M.S., Experimental Pathology Laboratories, Inc.
M.C. Cora, D.V.M., National Institute of Environmental Health Sciences
S.A. Elmore, D.V.M., M.S., National Institute of Environmental Health Sciences
G.P. Flake, M.D., National Institute of Environmental Health Sciences
A.R. Pandiri, B.V.Sc., Ph.D., National Institute of Environmental Health Sciences
Participated in Pathology Peer Review on thymus lesions in perinatal and 3-month rats (January 26, 2015)
M.F. Cesta, D.V.M., Ph.D., National Institute of Environmental Health Sciences
G.P. Flake, M.D., National Institute of Environmental Health Sciences
R.A. Herbert, D.V.M., Ph.D., National Institute of Environmental Health Sciences
D.E. Malarkey, D.V.M., Ph.D., National Institute of Environmental Health Sciences
A.R. Pandiri, B.V.Sc., Ph.D., National Institute of Environmental Health Sciences
Experimental Pathology Laboratories, Inc., Research Triangle Park, North Carolina, USA
Supervised pathology review
G.A. Willson, B.V.M.S., Principal Investigator
Integrated Laboratory Systems, LLC, Research Triangle Park, North Carolina, USA
Conducted micronucleus and bacterial mutagenicity assays
L. Recio, Ph.D., Principal Investigator
C.A. Hobbs, Ph.D.
C.D. Swartz, D.V.M., Ph.D.
BioReliance Corporation, Rockville, Maryland, USA
Conducted bacterial mutagenicity assays
S. Wagner, M.S., Principal Investigator
CSS Corporation, Research Triangle Park, North Carolina, USA
Prepared quality assessment audits
S. Brecher, Ph.D., Principal Investigator
S. Iyer, B.S.
V.S. Tharakan, D.V.M.
Social & Scientific Systems, a DLH Company, Research Triangle Park, North Carolina, USA
Provided statistical analyses
S.J. McBride, Ph.D., Principal Investigator
L.J. Betz, M.S.
S.F. Harris, M.S.
J.D. Krause, Ph.D.
ASRC Federal, Research Triangle Park, North Carolina, USA
Prepared data for report
J. Berke, B.S.
P. Brown, B.S.
C. Martini, B.S.
C. Myers, M.S.
N. Sayers, B.S.
ICF, Fairfax, Virginia, USA
Provided contract oversight
D. Burch, M.E.M., Principal Investigator
J.A. Wignall, M.S.P.H.
Prepared and edited report
T. Hamilton, M.S.
K.L. McKinley, M.E.M.
M.E. McVey, Ph.D.
K.T. O’Donovan, B.A.
A.E. Peppriell, Ph.D.
J.I. Powers, M.A.P.
L.M. Prince, Ph.D.
J.R. Rochester, Ph.D.
J.W. Tracy, M.H.S.
S.J. Snow, Ph.D.
Supported external peer review
A.P. Shirzadi, M.P.H.
L.M. Green, M.P.H.
Explanation of Levels of Evidence of Carcinogenic Activity
The National Toxicology Program (NTP) describes the results of individual experiments on a chemical agent and notes the strength of the evidence for conclusions regarding each study. Negative results, in which the study animals do not have a greater incidence of neoplasia than control animals, do not necessarily mean that a chemical is not a carcinogen, in as much as the experiments are conducted under a limited set of conditions. Positive results demonstrate that a chemical is carcinogenic for laboratory animals under the conditions of the study and indicate that exposure to the chemical has the potential for hazard to humans. Other organizations, such as the International Agency for Research on Cancer, assign a strength of evidence for conclusions based on an examination of all available evidence, including animal studies such as those conducted by NTP, epidemiologic studies, and estimates of exposure. Thus, the actual determination of risk to humans from chemicals found to be carcinogenic in laboratory animals requires a wider analysis that extends beyond the purview of these studies.
Five categories of evidence of carcinogenic activity are used in the Technical Report series to summarize the strength of evidence observed in each experiment: two categories for positive results (clear evidence and some evidence); one category for uncertain findings (equivocal evidence); one category for no observable effects (no evidence); and one category for experiments that cannot be evaluated because of major flaws (inadequate study). These categories of interpretative conclusions were first adopted in June 1983 and then revised in March 1986 for use in the Technical Report series to incorporate more specifically the concept of actual weight of evidence of carcinogenic activity. For each separate experiment (male rats, female rats, male mice, female mice), one of the following five categories is selected to describe the findings. These categories refer to the strength of the experimental evidence and not to potency or mechanism.
-
Clear evidence of carcinogenic activity is demonstrated by studies that are interpreted as showing a dose-related (i) increase of malignant neoplasms, (ii) increase of a combination of malignant and benign neoplasms, or (iii) marked increase of benign neoplasms if there is an indication from this or other studies of the ability of such tumors to progress to malignancy.
-
Some evidence of carcinogenic activity is demonstrated by studies that are interpreted as showing a chemical-related increased incidence of neoplasms (malignant, benign, or combined) in which the strength of the response is less than that required for clear evidence.
-
Equivocal evidence of carcinogenic activity is demonstrated by studies that are interpreted as showing a marginal increase of neoplasms that may be chemical related.
-
No evidence of carcinogenic activity is demonstrated by studies that are interpreted as showing no chemical-related increases in malignant or benign neoplasms.
-
Inadequate study of carcinogenic activity is demonstrated by studies that, because of major qualitative or quantitative limitations, cannot be interpreted as valid for showing either the presence or absence of carcinogenic activity.
For studies showing multiple chemical-related neoplastic effects that if considered individually would be assigned to different levels of evidence categories, the following convention has been adopted to convey completely the study results. In a study with clear evidence of carcinogenic activity at some tissue sites, other responses that alone might be deemed some evidence are indicated as “were also related” to chemical exposure. In studies with clear or some evidence of carcinogenic activity, other responses that alone might be termed equivocal evidence are indicated as “may have been” related to chemical exposure.
When a conclusion statement for a particular experiment is selected, consideration must be given to key factors that would extend the actual boundary of an individual category of evidence. Such consideration should allow for incorporation of scientific experience and current understanding of long-term carcinogenesis studies in laboratory animals, especially for those evaluations that may be on the borderline between two adjacent levels. These considerations should include:
-
adequacy of the experimental design and conduct;
-
occurrence of common versus uncommon neoplasia;
-
progression (or lack thereof) from benign to malignant neoplasia as well as from preneoplastic to neoplastic lesions;
-
some benign neoplasms have the capacity to regress but others (of the same morphologic type) progress. At present, it is impossible to identify the difference. Therefore, where progression is known to be a possibility, the most prudent course is to assume that benign neoplasms of those types have the potential to become malignant;
-
combining benign and malignant tumor incidence known or thought to represent stages of progression in the same organ or tissue;
-
latency in tumor induction;
-
multiplicity in site-specific neoplasia;
-
metastases;
-
supporting information from proliferative lesions (hyperplasia) in the same site of neoplasia or other experiments (same lesion in another sex or species);
-
presence or absence of dose relationships;
-
statistical significance of the observed tumor increase;
-
concurrent control tumor incidence as well as the historical control rate and variability for a specific neoplasm;
-
survival-adjusted analyses and false positive or false negative concerns;
-
structure-activity correlations; and
-
in some cases, genetic toxicology.
Peer Review
The National Toxicology Program (NTP) conducted a peer review of the draft NTP Technical Report on the Toxicology and Carcinogenesis Studies of an Isomeric Mixture of Tris(chloropropyl) Phosphate Administered in Feed to Sprague Dawley (Hsd:Sprague Dawley SD) Rats and B6C3F1/N Mice by letter in November 2022 by the experts listed below. Reviewer selection and document review followed established NTP practices. The reviewers were charged to:
-
Peer review the draft NTP Technical Report on the Toxicology and Carcinogenesis Studies of an Isomeric Mixture of Tris(chloropropyl) Phosphate Administered in Feed to Sprague Dawley (Hsd:Sprague Dawley SD) Rats and B6C3F1/N Mice.
-
Comment on NTP’s interpretations of the data.
NTP carefully considered reviewer comments in finalizing this report.
Peer Reviewers
Lois Lehman-McKeeman, Ph.D.
Vice President
Bristol-Myers Squibb Company, New Jersey, USA
Richard Peterson II, D.V.M., Ph.D., DACVP
Research Fellow and Associate Director, Pathology
AbbVie, Illinois, USA
Russell Cattley, V.M.D., Ph.D., DAVCP, FIATP
Emeritus Professor and Fellow
Auburn University and Alaplains Pathology LCC, Alabama, USA
Publication Details
Publisher: National Toxicology Program
Publishing Location: Research Triangle Park, NC
ISSN: 2378-8925
DOI: https://doi.org/10.22427/NTP-TR-602
Report Series: NTP Technical Report Series
Report Series Number: 602
Official citation: National Toxicology Program (NTP). 2023. NTP technical report on the toxicology and carcinogenesis studies of an isomeric mixture of tris(chloropropyl) phosphate administered in feed to Sprague Dawley (Hsd:Sprague Dawley SD) rats and B6C3F1/N mice. Research Triangle Park, NC: National Toxicology Program. Technical Report 602.
Acknowledgments
This work was supported by the Intramural Research Program (ES103316, ES103318, and ES103319) at the National Institute of Environmental Health Sciences, National Institutes of Health and performed for the National Toxicology Program, Public Health Service, U.S. Department of Health and Human Services under contracts HHSN273201800006C, HHSN271201800012I, HHSN273201600011C, HHSN273201600020C, GS00Q14OADU417 (Order No. HHSN273201600015U), HHSN273201500006C, HHSN273201500014C, HHSN273201400015C, HHSN273201400020C, HHSN273201300009C, HHSN316201200054W, HHSN273201100001C, HHSN291200555548C, N01-ES-35514, N01-ES-45517, N01-ES-55541, N01-ES-55552, and N01-ES-65557.
Overview
Tris(chloropropyl) phosphate (TCPP) is a flame retardant commonly used in consumer products. Commercial TCPP is an isomeric mixture, and variations in manufacturing methods result in commercial formulations that contain different ratios of four isomers. TCPP trade names include but are not limited to Amgard TMCP, Antiblaze 80, Antiblaze TMCP, AP 33, Fyrol PCF, and Hostaflam OP 820. The most abundant isomer in the TCPP lots used in the current studies was tris(1-chloro-2-propyl) phosphate (TCIPP; CASRN 13674-84-5; 65%–68%). Other isomers were bis(2-chloro-1-methylethyl) 2-chloropropyl phosphate (CASRN 76025-08-6; 25%–27%), bis(2-chloropropyl) 2-chloroisopropyl phosphate (CASRN 76649-15-5; 4%), and tris(2-chloropropyl) phosphate (CASRN 6145-73-9; 0.2%). The isomeric mixture of TCPP and commercial products are commonly referred to by CASRN 13674-84-5, which is the CASRN of the major isomer, TCIPP.
TCPP was nominated to the National Toxicology Program (NTP) by the U.S. Consumer Product Safety Commission (CPSC) for toxicological testing. This nomination is part of a larger effort to populate data on several flame retardants of interest (https://ntp.niehs.nih.gov/go/nm-n20608). The NTP research program on TCPP is evaluating toxicity on various cellular or molecular targets in vitro (e.g., high-throughput screening) and in vivo. TCPP effects on prenatal development in rats have been reported in NTP Developmental and Reproductive Toxicity Technical Report (DART)-01 and immunotoxicity assessments in TCPP-exposed rats are in progress. Further information on the NTP research program on TCPP is available at the Program’s website.1 The purpose of this report is to summarize and discuss the effects of TCPP subchronic and chronic exposure in rats and mice. Complementary research of these subchronic and chronic studies includes analysis of Tox21 (Toxicology in the 21st Century program) in vitro screening data (summary results of these data are provided in Appendix E), 5-day rat toxicogenomic study (summary results of these data are provided in Appendix F), and multiomics evaluation of hepatocellular carcinomas in mice (summary results of these data are provided in Appendix G).