Reproductive System, Female
Ovary - Atrophy
Narrative
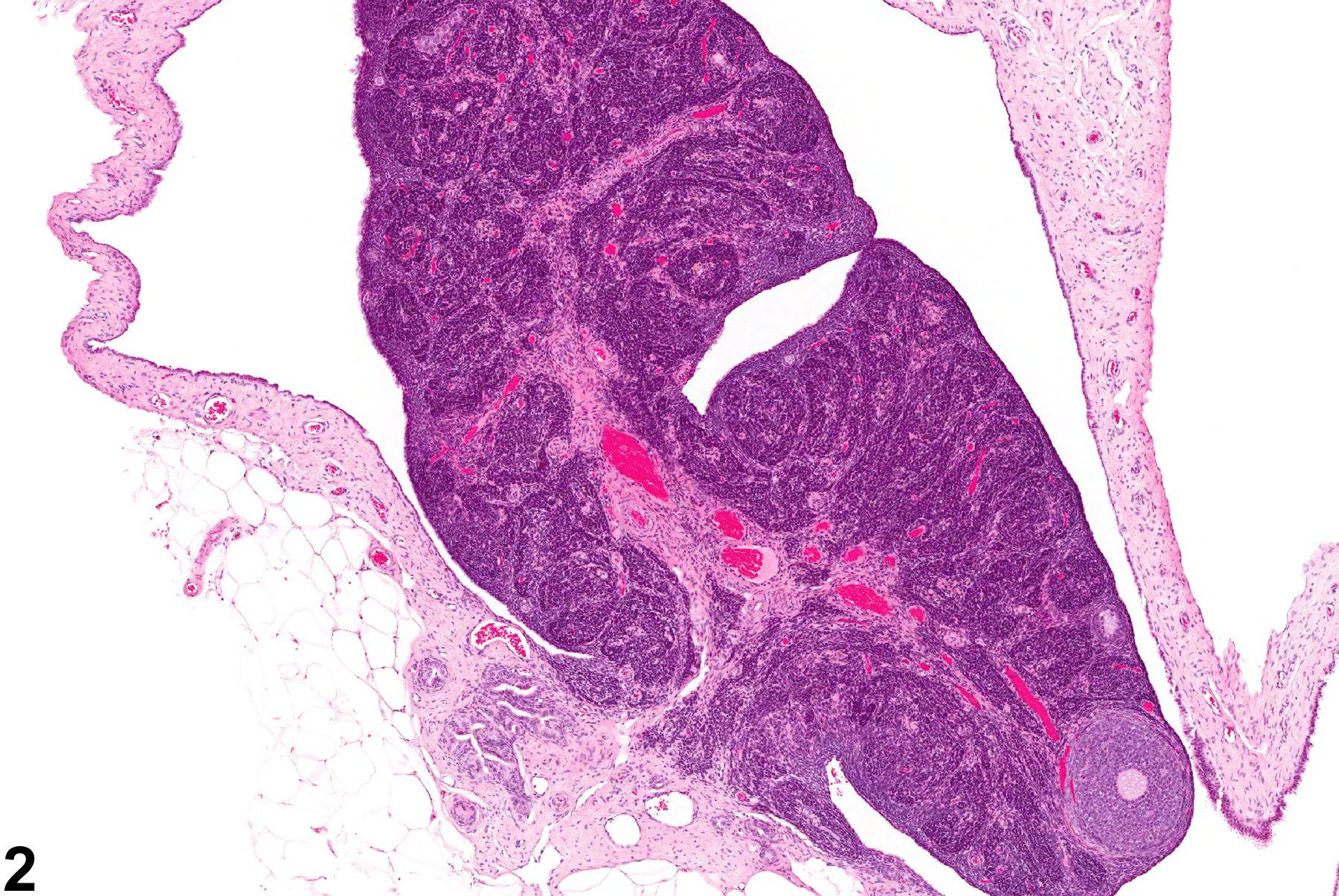
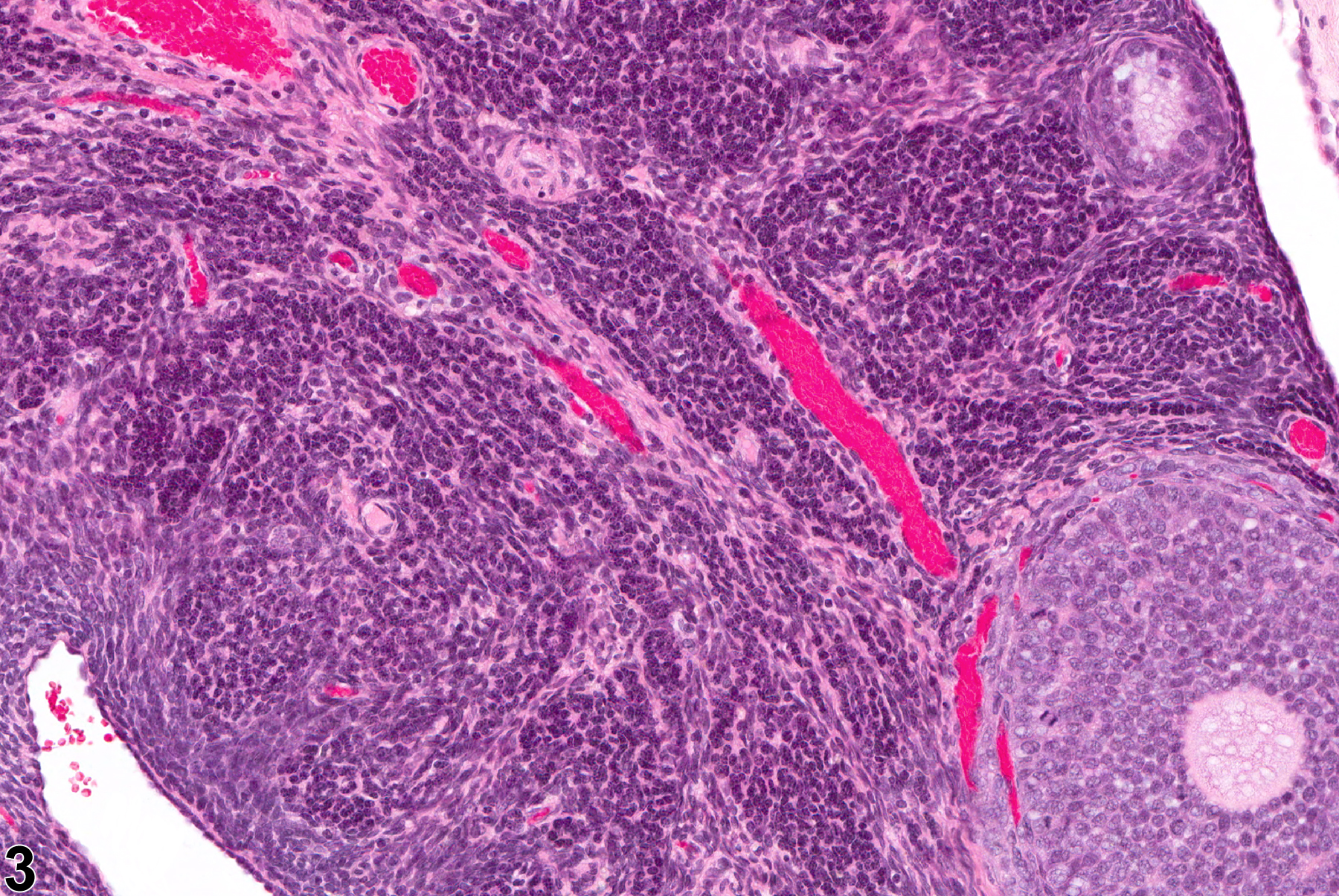
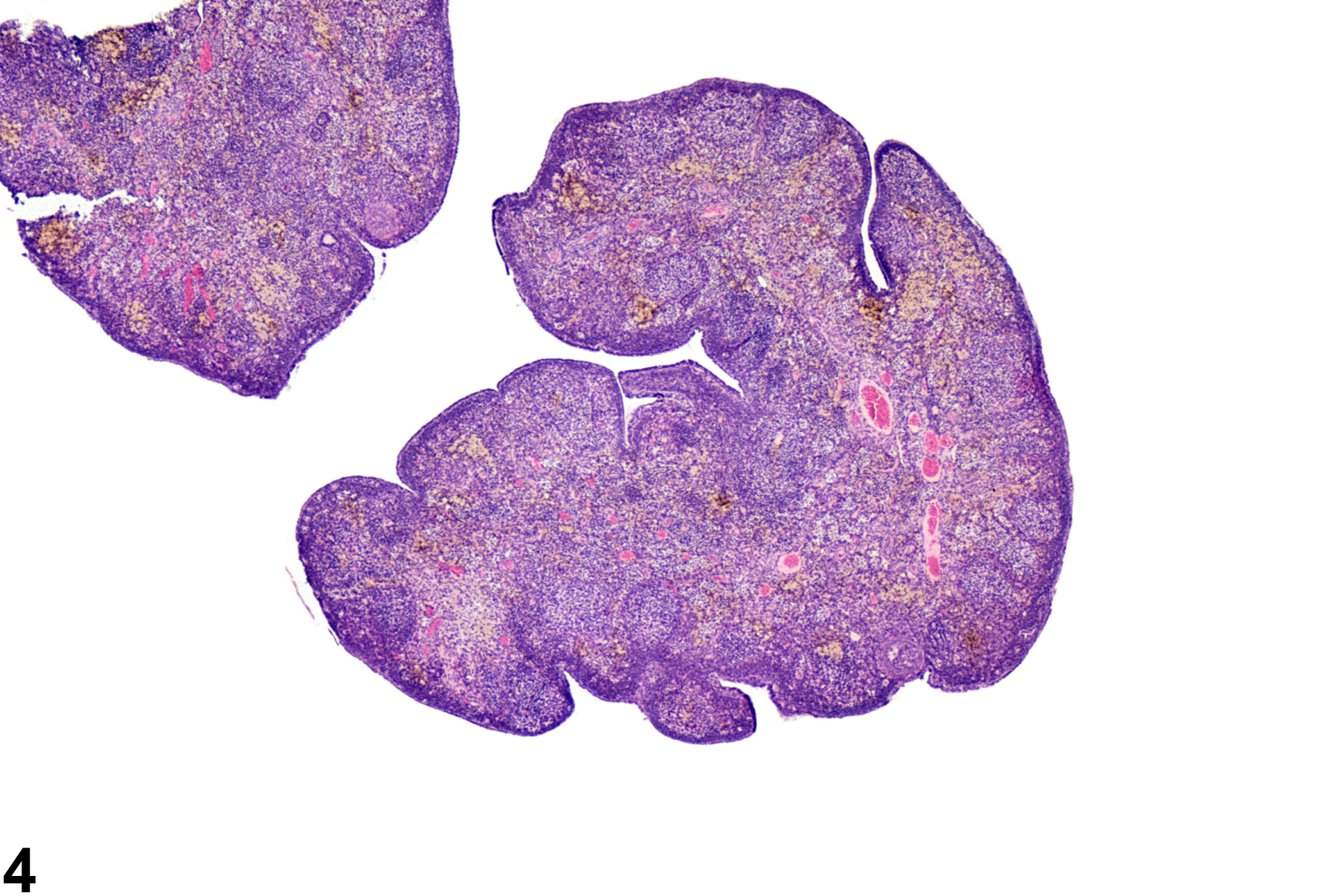
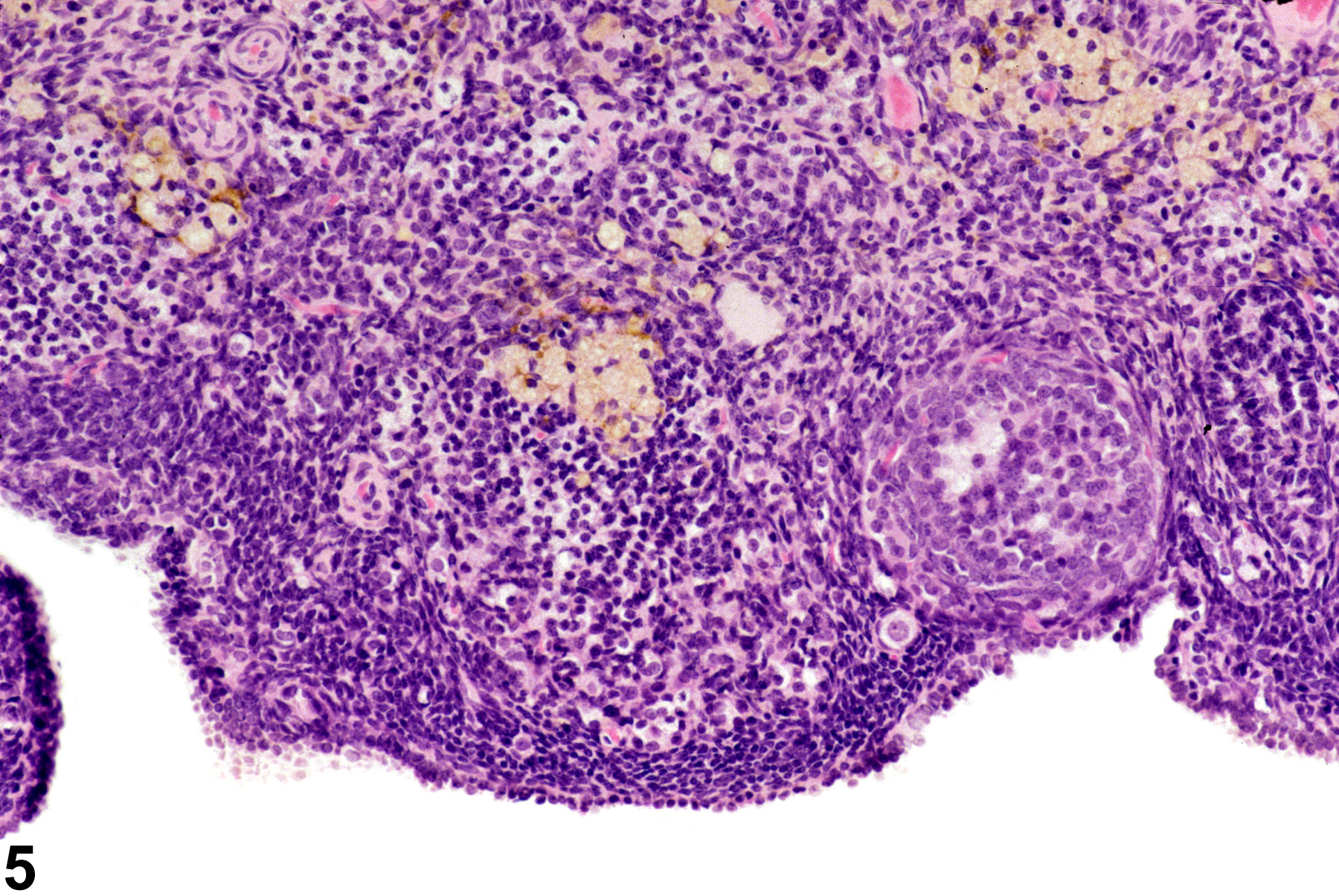
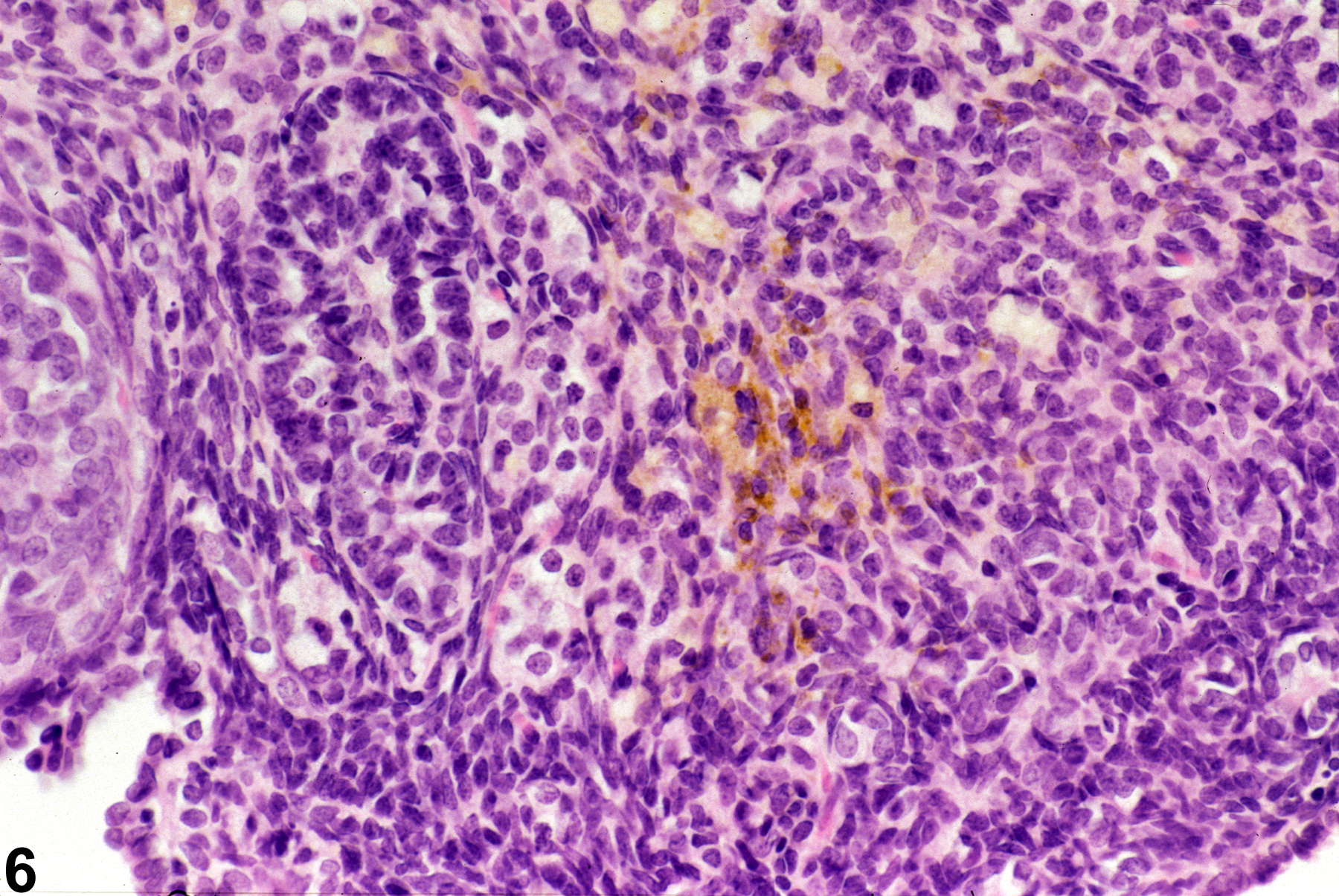
Ovarian atrophy occurs with age (senescence) or may result from administration of drugs or exposure to chemicals that block or suppress trophic control of the ovary. Ovarian atrophy is defined as decreased ovarian size and weight and decreased numbers of oocytes, developing follicles, and corpora lutea (Figure 1, Figure 2 and Figure 3). Atrophic ovaries are small, with a conspicuous absence of follicles and corpora lutea and the presence of increased stroma, with frequent prominent pigment in the interstitium (Figure 4, Figure 5 and Figure 6). Atrophy occurs more frequently in B6C3F1 mice than in F344 rats in NTP carcinogenicity studies. Additional findings associated with ovarian atrophy may be diagnosed separately if deemed relevant and may provide information as to pathogenesis of atrophy in toxicity or carcinogenicity studies. These findings may include interstitial cell hyperplasia, necrosis of follicular epithelium, increased numbers of cystic follicles, accumulation of pigment or lipid, cholesterol clefts, hyaline degeneration of corpora lutea, fibrosis, and mineralization.
Ovary - Atrophy should be recorded and graded only when treatment related. It is important to distinguish treatment-related atrophy from senescence, and atrophic lesions associated with senescence should not be diagnosed. When atrophy is treatment related, it is important to describe the changes present in the ovary in the pathology narrative. The pathologist should also check for secondary atrophy of uterus and vagina and associated changes in mammary tissue. Ovary - Atrophy is a suitable diagnosis for a short-term study. If treatment-related ovarian atrophy is diagnosed, notify NTP that more detailed studies (e.g., counts of primordial, primary, and antral follicles and corpora lutea) may be warranted.
Alison RH, Morgan KT, Montgomery CA. 1990. Ovary. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 429-442.
Davis BJ, Dixon D, Herbert RA. 1999. Ovary, oviduct, uterus, cervix and vagina. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 409-444.
Faccini JM, Abbott DP, Paulus GJJ. 1990. Female genital tract. In: Mouse Histopathology: A Glossary for Use in Toxicity and Carcinogenicity Studies (Greaves P, Faccini JM, eds). Elsevier, Amsterdam, 147-168.
Greaves P. 2012. Female genital tract. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 4th ed. Elsevier, Amsterdam, 667-724.
Maekawa A, Maita K, Harleman JH. 1996. Changes in the ovary. In: Pathobiology of the Aging Mouse (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 451-467.
Maekawa A, Yoshida A. 1996. Susceptibility of the female genital system to toxic substances. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 481-493.
Montgomery CA, Alison RH. 1987. Non-neoplastic lesions of the ovary in Fischer 344 rats and B6C3F1 mice. Environ Health Perspect 73:53-75.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1474552/National Toxicology Program. 1988. NTP TR-339. Toxicology and Carcinogenesis Studies of 2-Amino-4-nitrophenol (CAS No. 99-57-0) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/10784Peluso JJ, Gordon LR. 1992. Nonneoplastic and neoplastic changes in the ovary. In: Pathobiology of the Aging Rat (Mohr U, Dungworth DL, Capen CC, eds). ILSI Press, Washington, DC, 351-364.
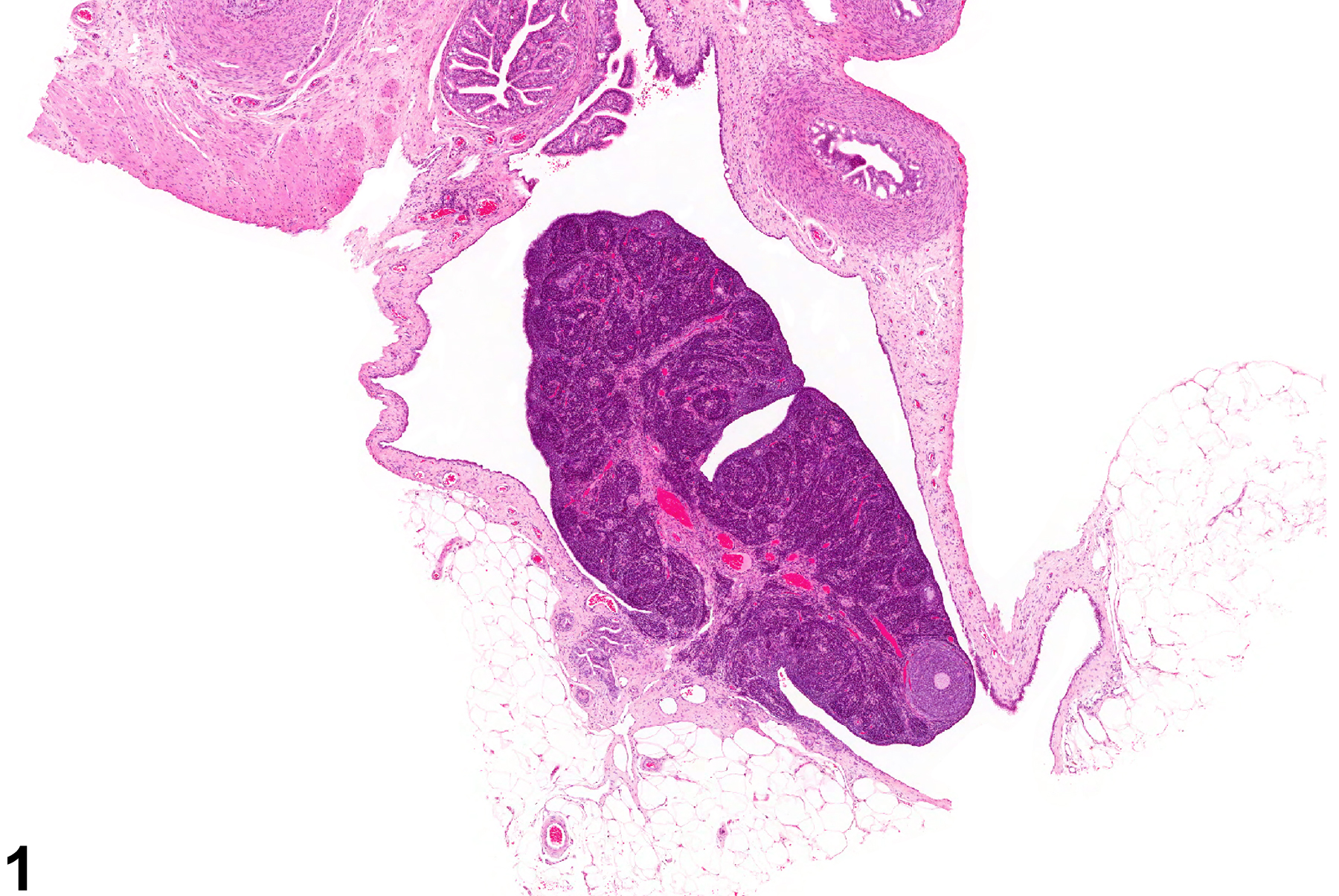
Ovary - Atrophy in a female F344/N rat from a chronic study. The ovary is small and lacking follicles and corpora lutea.