Hematopoietic System
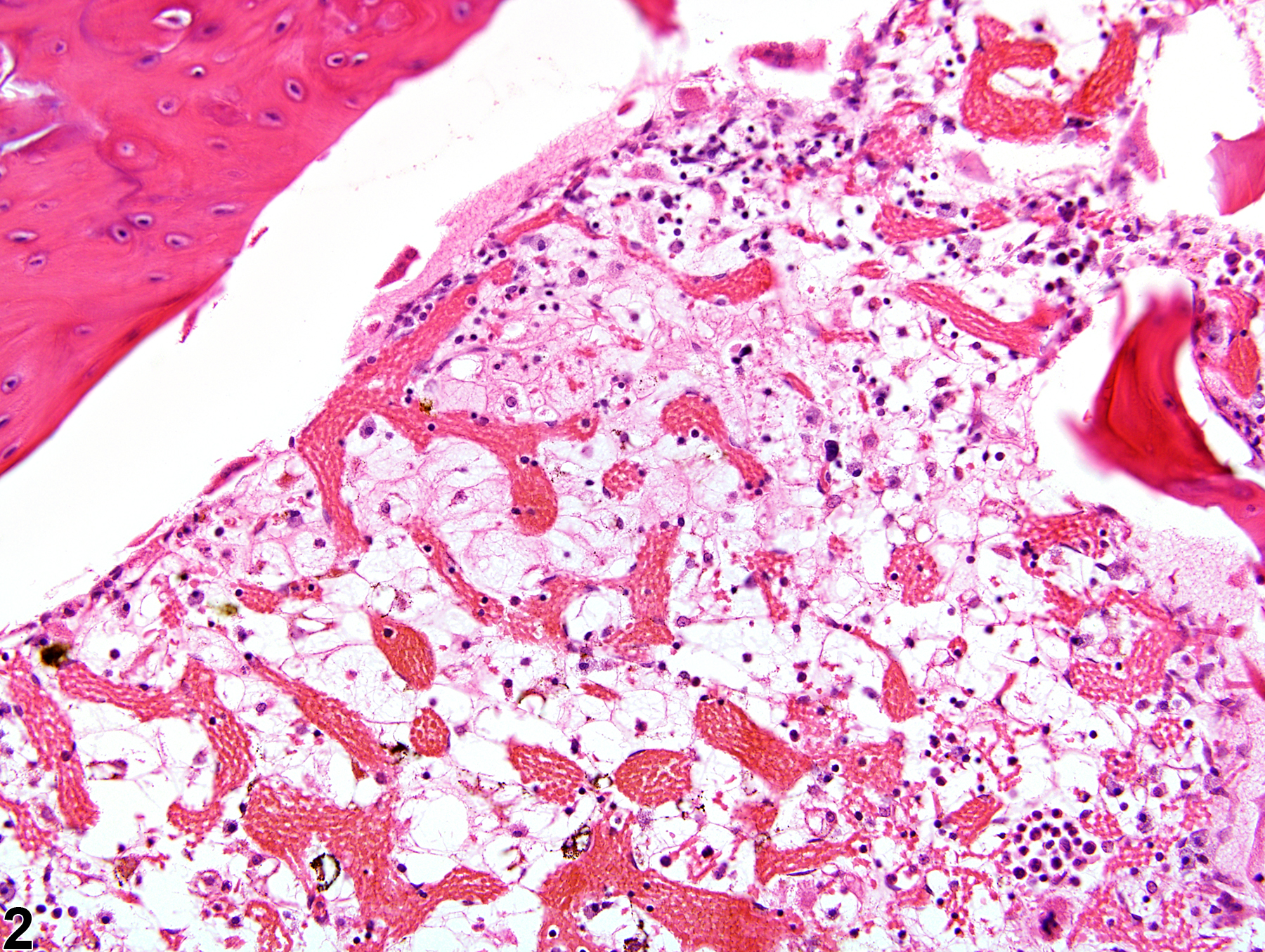
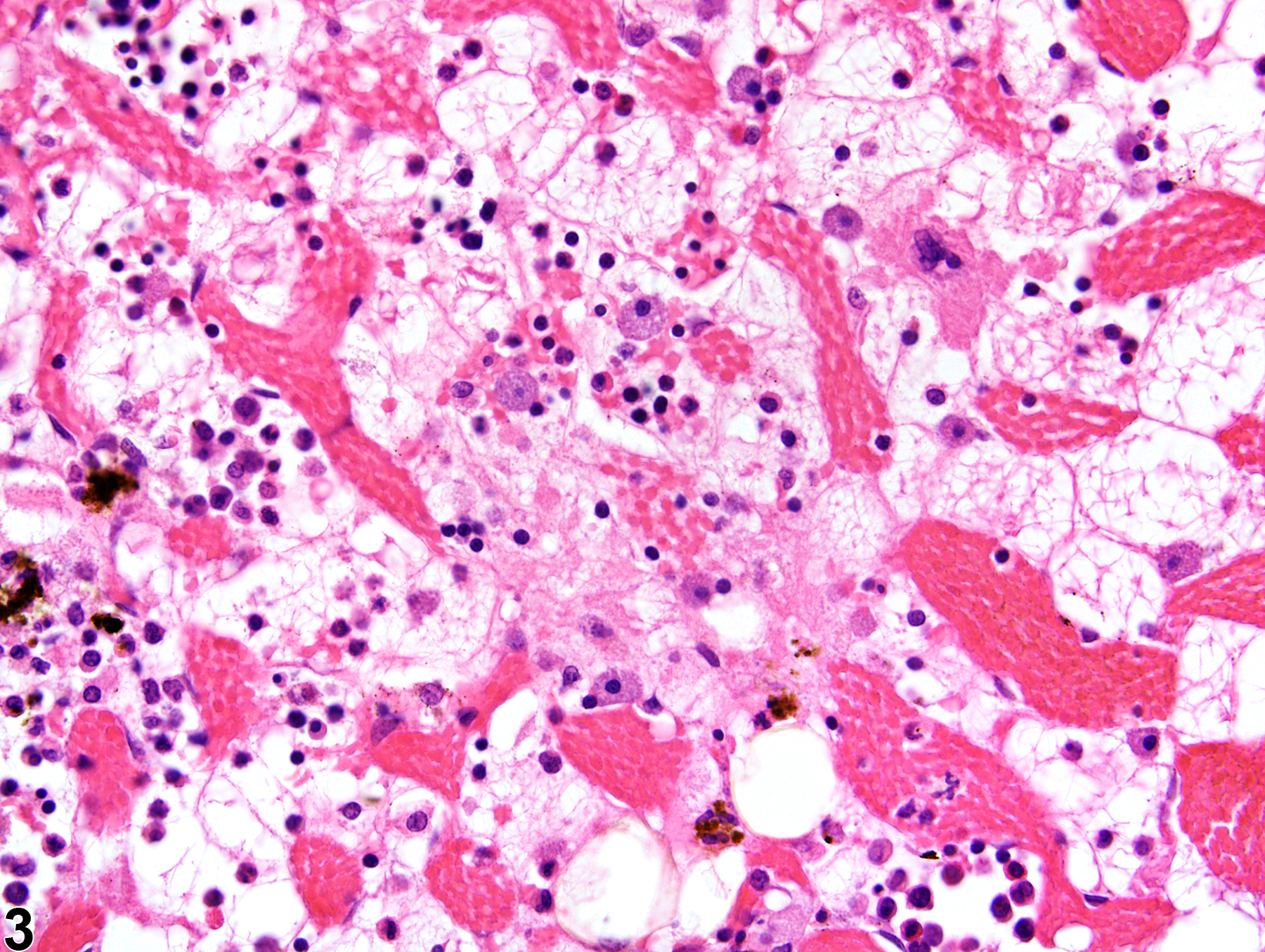
Bone Marrow - Gelatinous Transformation
Narrative
GMT has been described in debilitated rodents and in general is associated with cachexia (e.g., secondary to neoplasia, endocrinopathies) and advanced severe malnutrition (e.g., maldigestion). Under current husbandry standards, however, GMT is rarely encountered in NTP studies today. Although the pathogenesis of GMT remains unclear, its association with a wide range of diseases suggests it may result from basic bioregulatory processes that are activated in states of advanced illness. It has been suggested that the ground substance may be a replacement for the adipocytes that are used in catabolic states of disease, while the hematopoietic cell loss may be due to an unfavorable bone marrow microenvironment related to inadequate hematopoietic substrates and lack of stimulation by adipocytes and fibrocytes.
Bain BJ, Clark DM, Lampert IA, Wilkins BS. 2001. Bone Marrow Pathology, 3rd ed. Blackwell, Ames, IA, 90-140.
Abstract: http://onlinelibrary.wiley.com/book/10.1002/9780470757130Beeler-Marfisi J, Menoyo AG, Beck A, König J, Hewson J, Bienzle D. 2011. Gelatinous marrow transformation and hematopoietic atrophy in a miniature horse stallion. Vet Pathol 48:451-455.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/20587692Böhm J. 2000. Gelatinous transformation of the bone marrow - the spectrum of underlying diseases. Am J Surg Pathol 24:56-65.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/10632488Travlos GS. 2006. Histopathology of the bone marrow. Toxicol Pathol 34:566-598.
Abstract: http://tpx.sagepub.com/content/34/5/566.abstractWeiss DJ. 2010. Chronic inflammation and secondary myelofibrosis. In: Schalm’s Veterinary Hematology, 5th ed (Weiss DJ, Wardrop KJ, eds). Wiley-Blackwell, Ames, IA, 112-117.
Abstract: http://vet.sagepub.com/content/40/2/223.1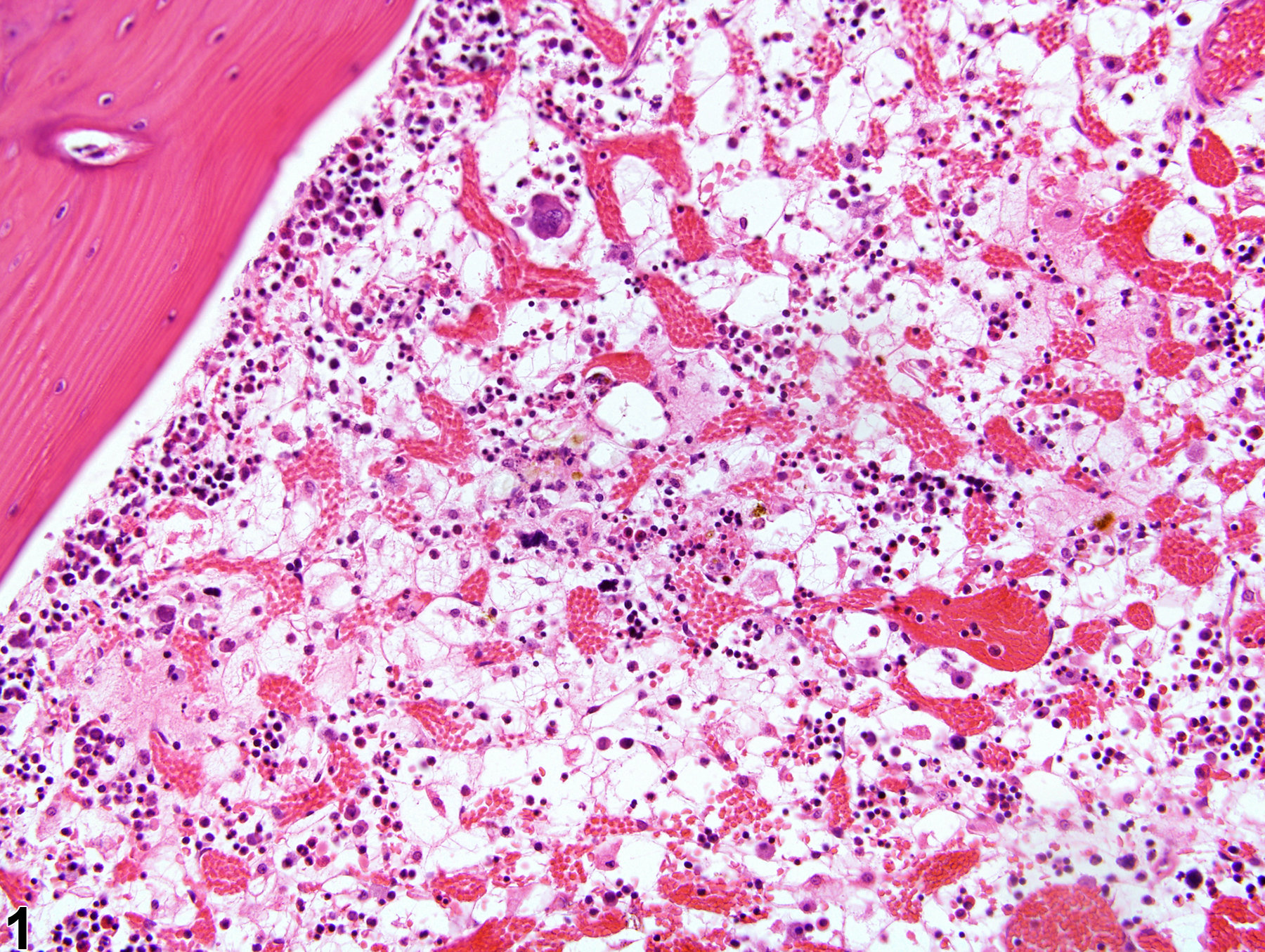
Gelatinous transformation of bone marrow in a female F344/N rat from a subchronic study. The marrow is characterized by severe hypocellularity, atrophied fat cells, and the presence of eosinophilic granular ground substance.