Alimentary System
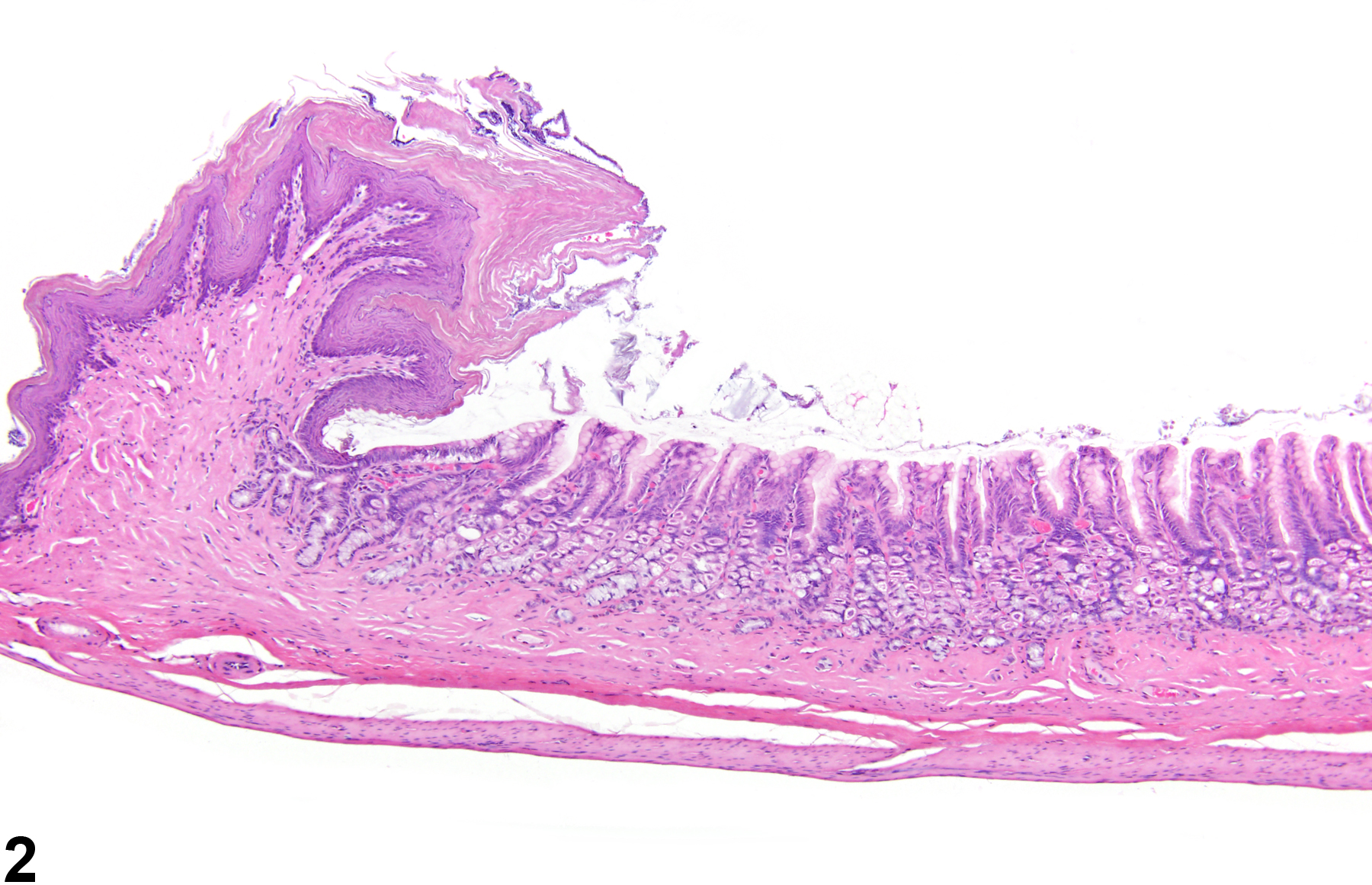
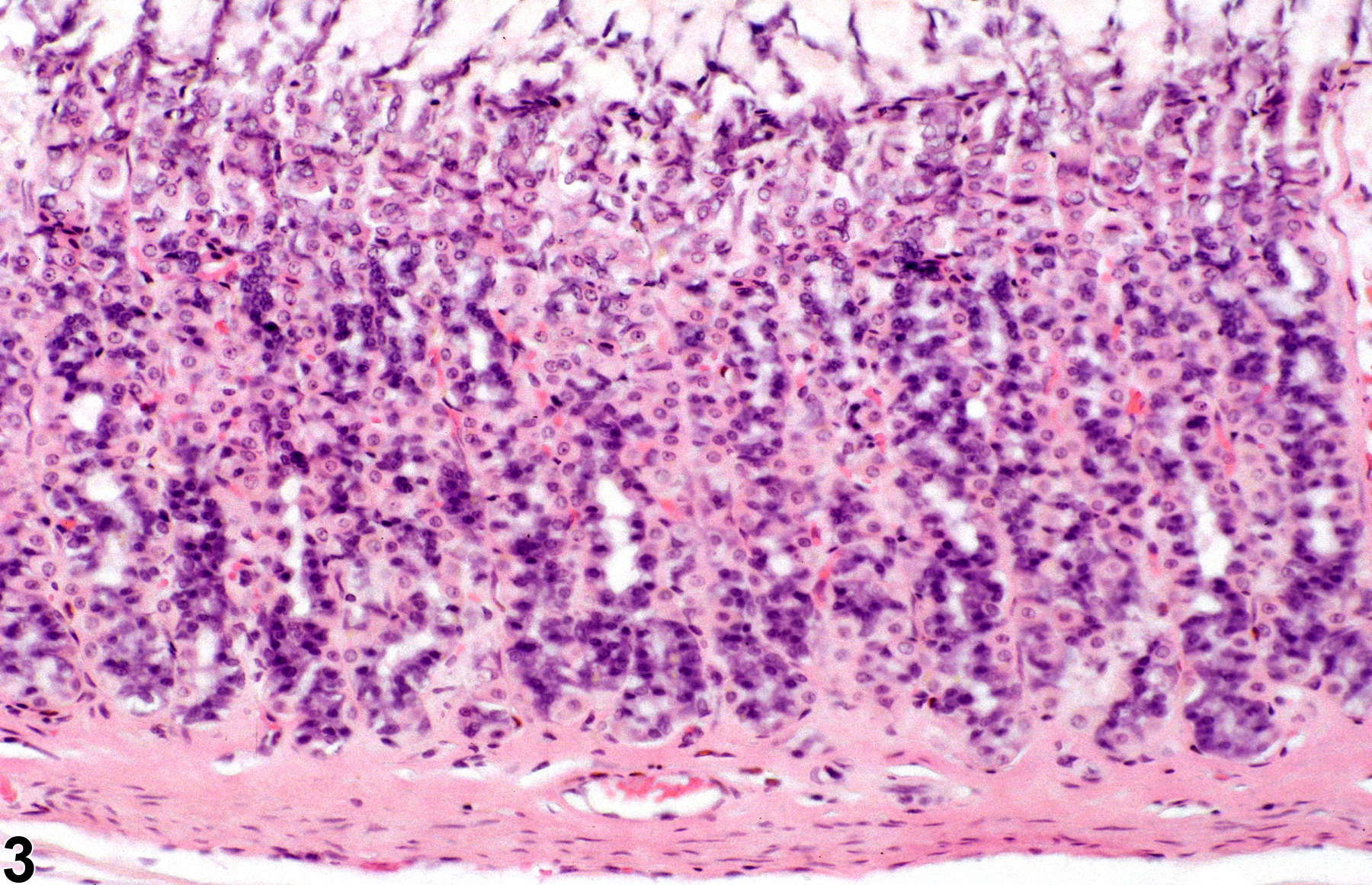
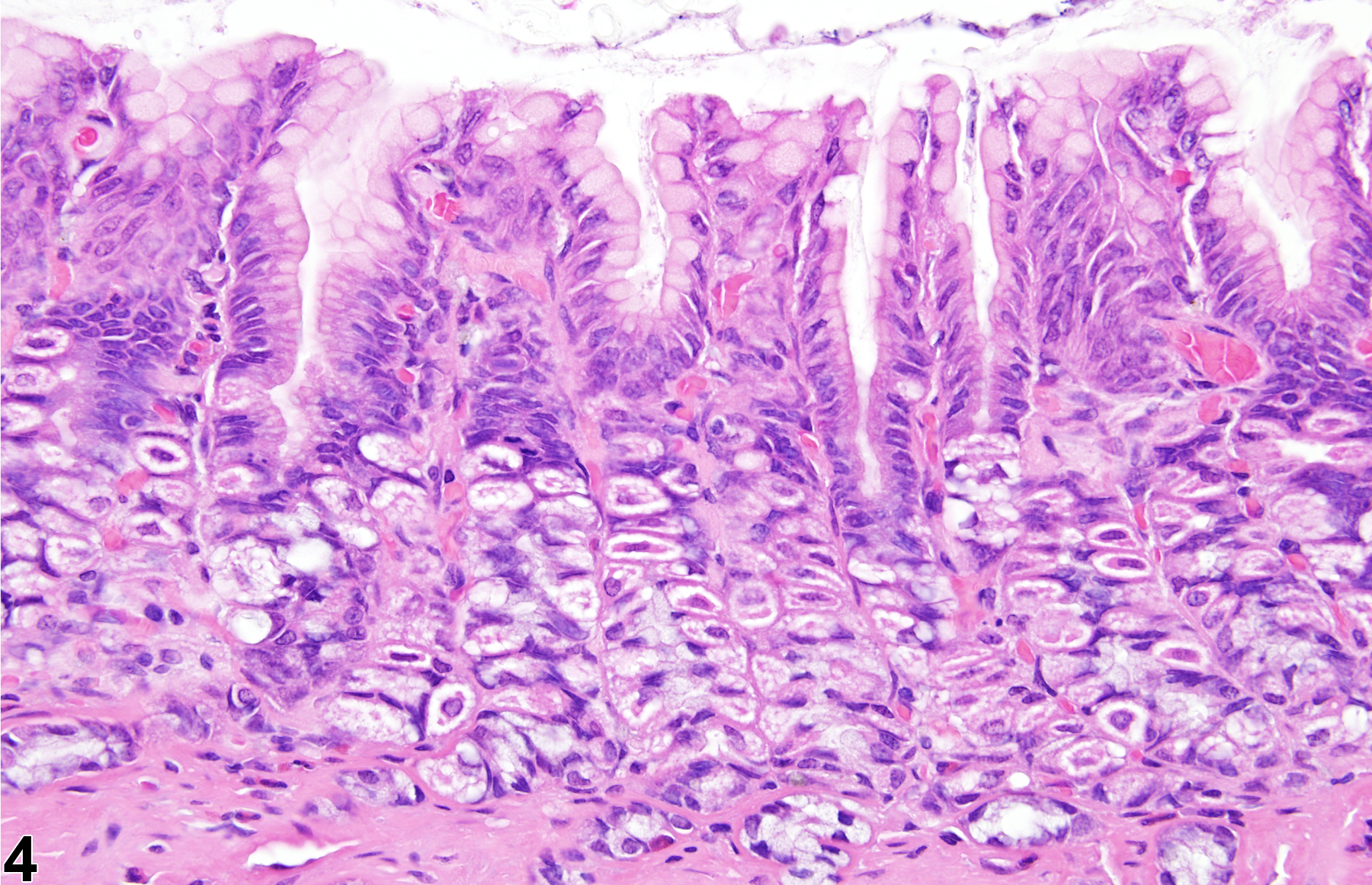
Stomach, Glandular Stomach - Atrophy
Narrative
Abdo KM, Cunningham ML, Snell ML, Herbert RA, Travlos GS, Eldridge SR, Bucher JR. 2001. 14-Week toxicity and cell proliferation of methyleugenol administered by gavage to F344 rats and B6C3F1 mice. Food Chem Toxicol 39:303-316.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11295478Bertram TA, Markovits JE, Juliana MM. 1996. Non-proliferative lesions of the alimentary canal in rats GI-1. In Guides for Toxicologic Pathology. STP/ARP/AFIP, Washington, DC, 1-16.
Full Text: https://www.toxpath.org/docs/SSNDC/GINonproliferativeRat.pdfBrown HR, Hardisty JF. 1990. Oral cavity, esophagus and stomach. In: Pathology of the Fischer Rat (Boorman GA, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 9-30.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Dethloff LA, Robertson DG, Tierney BM, Breider MA, Bestervelt LL. 1997. Gastric gland degeneration induced in monkeys by the CCK-B/gastrin receptor antagonist CI-988. Toxicol Pathol 25:441-448.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/9323831Eissele R, Patberg H, Koop H, Krack W, Lorenz W, McKnight AT, Arnold R. 1992. Effect of gastrin receptor blockade on endocrine cells in rats during achlorhydria. Gastroenterology 103:1596-1601.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/1426880Leininger JR, Jokinen MP, Dangler CA, Whiteley LO. 1999. Oral cavity, esophagus, and stomach. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, St Louis, MO, 29-48.
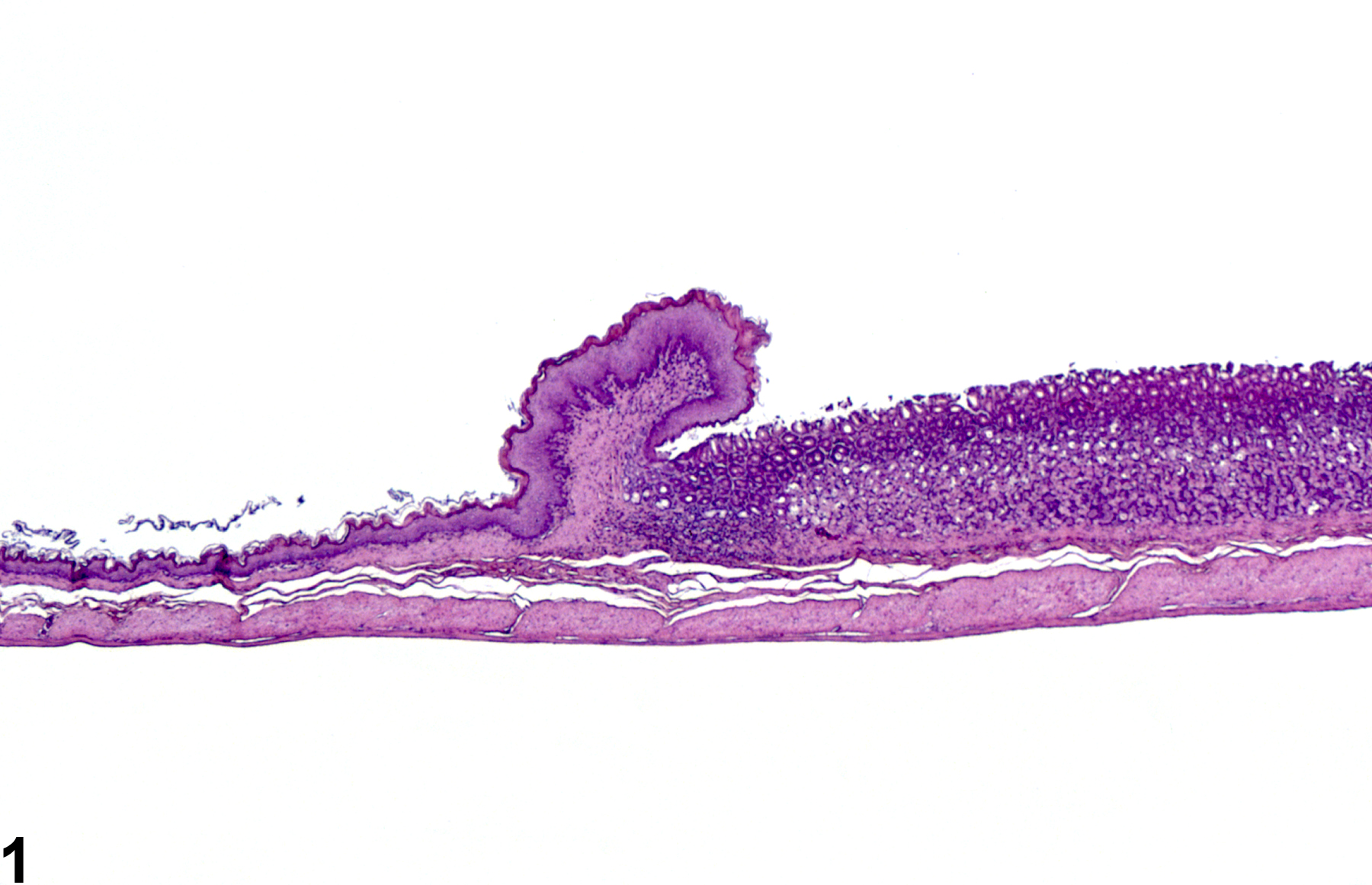
Normal glandular stomach in a male F344/N rat from a subchronic study.