Alimentary System
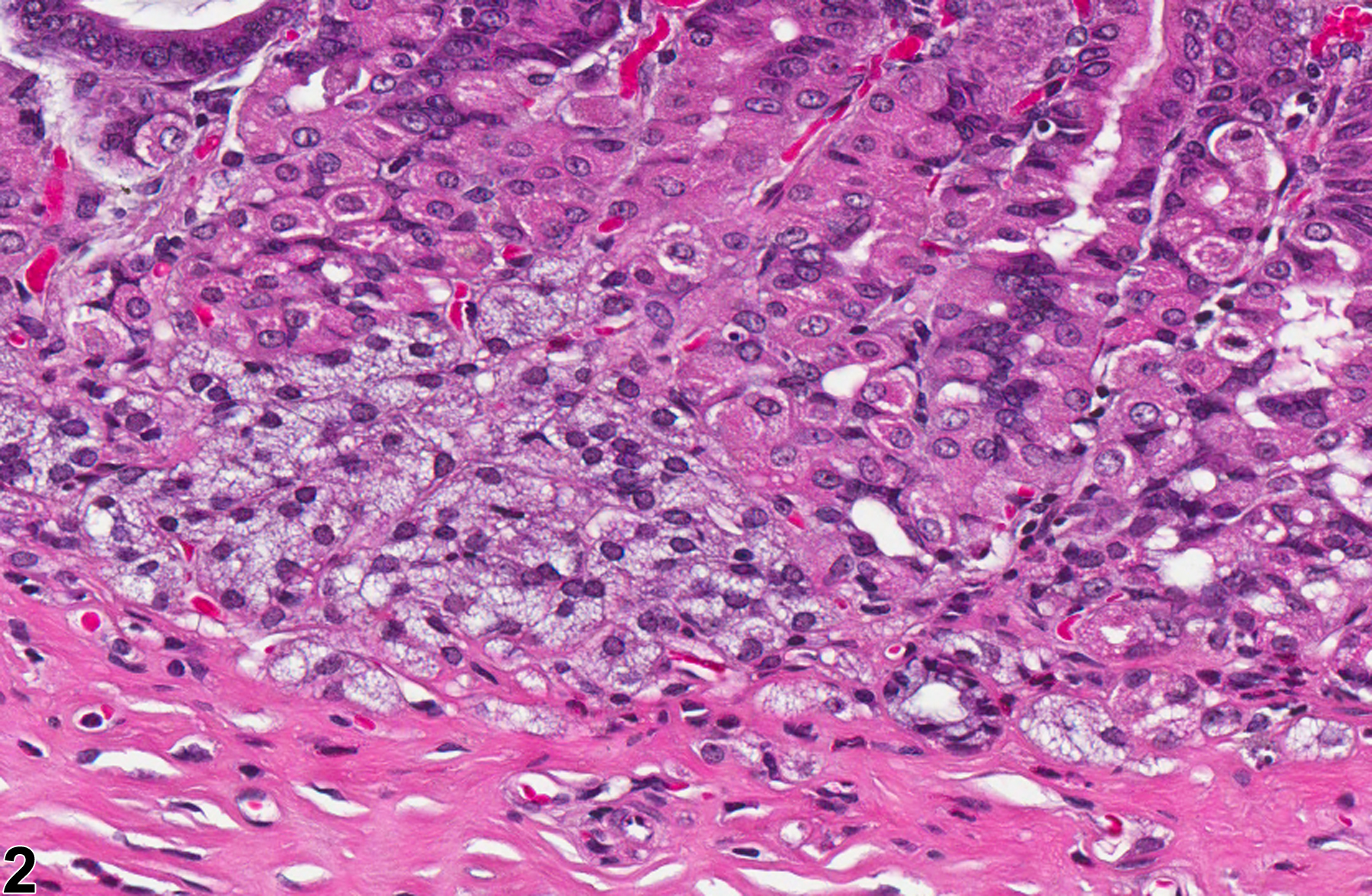
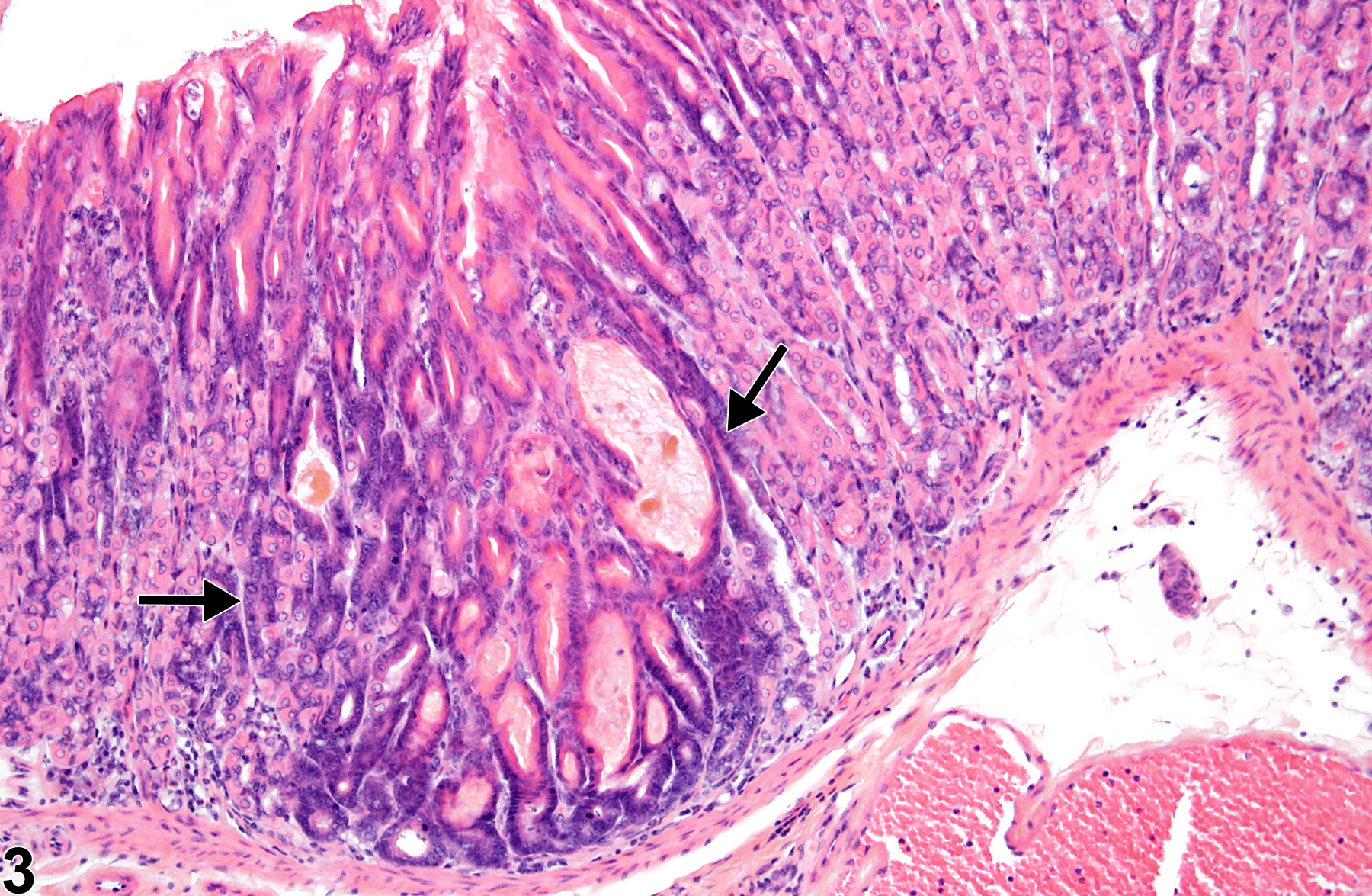
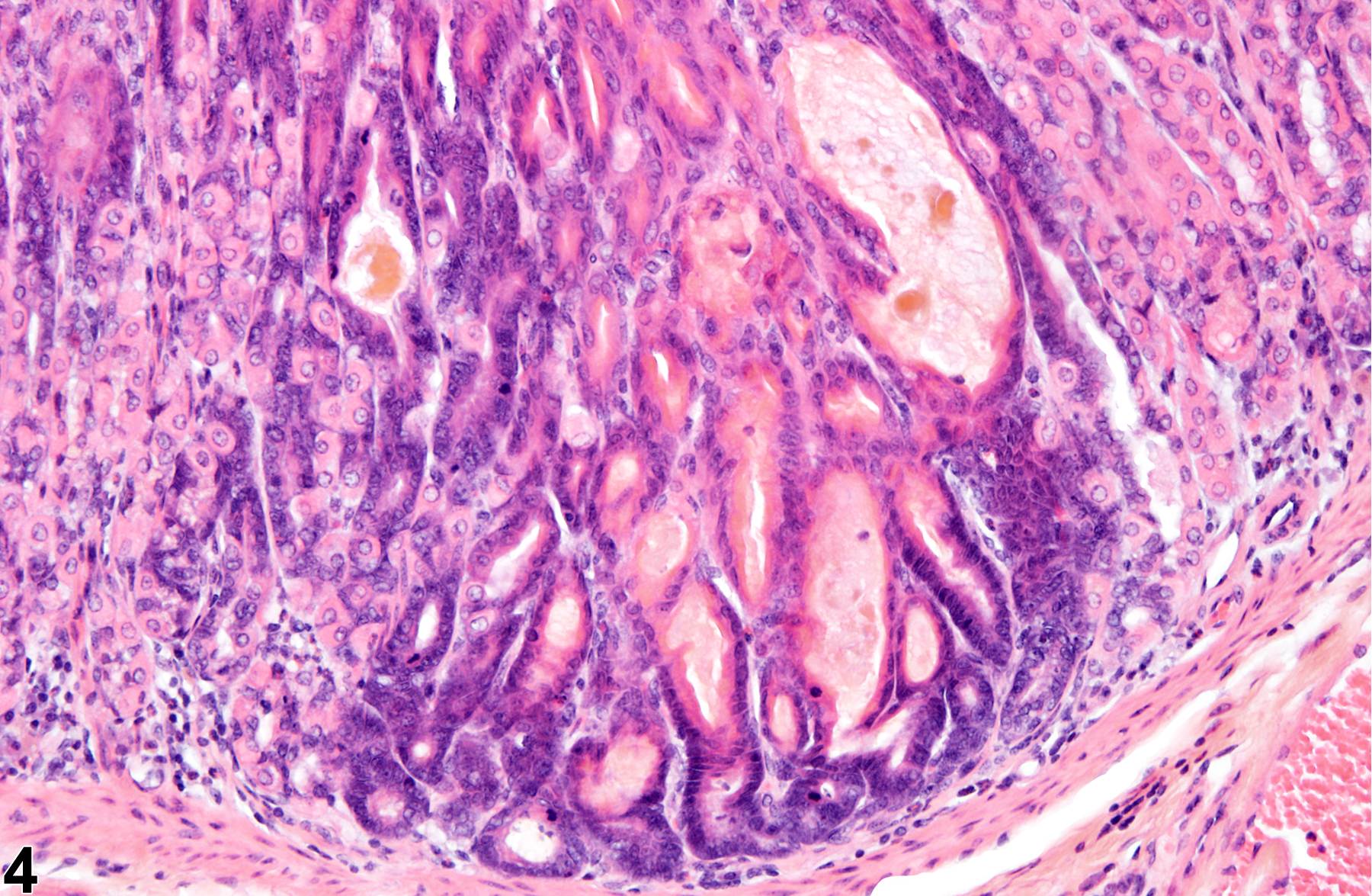
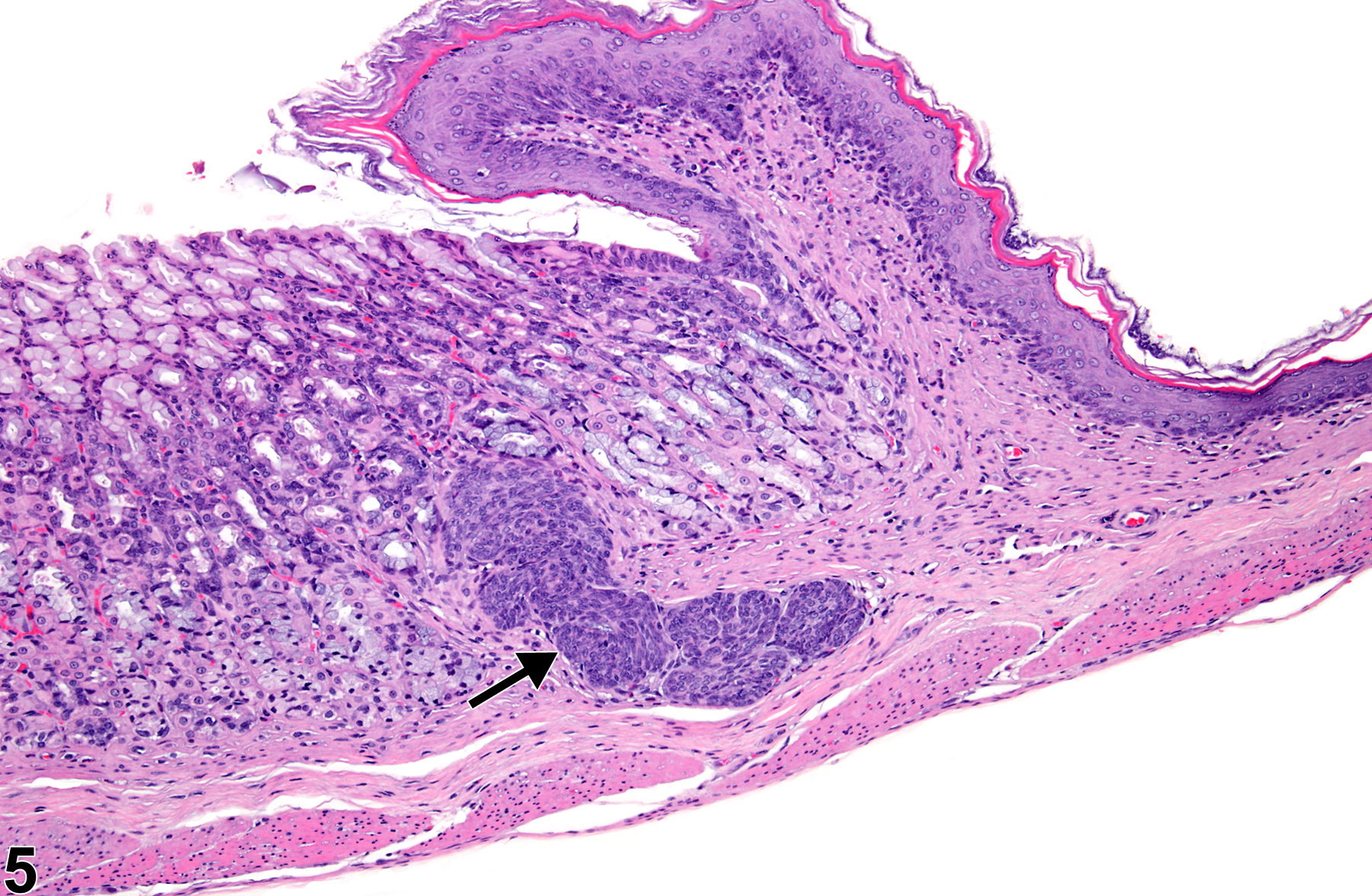
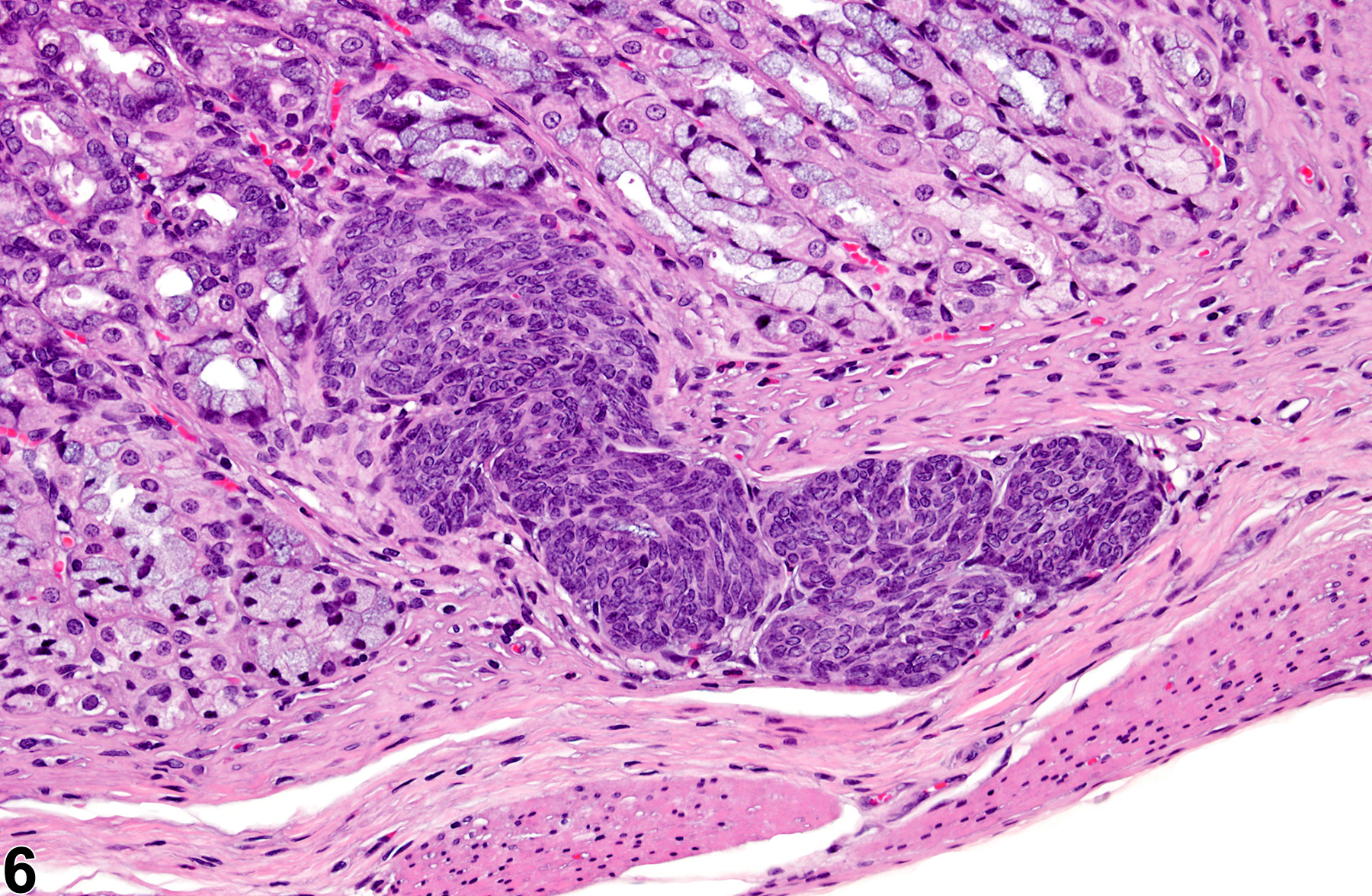
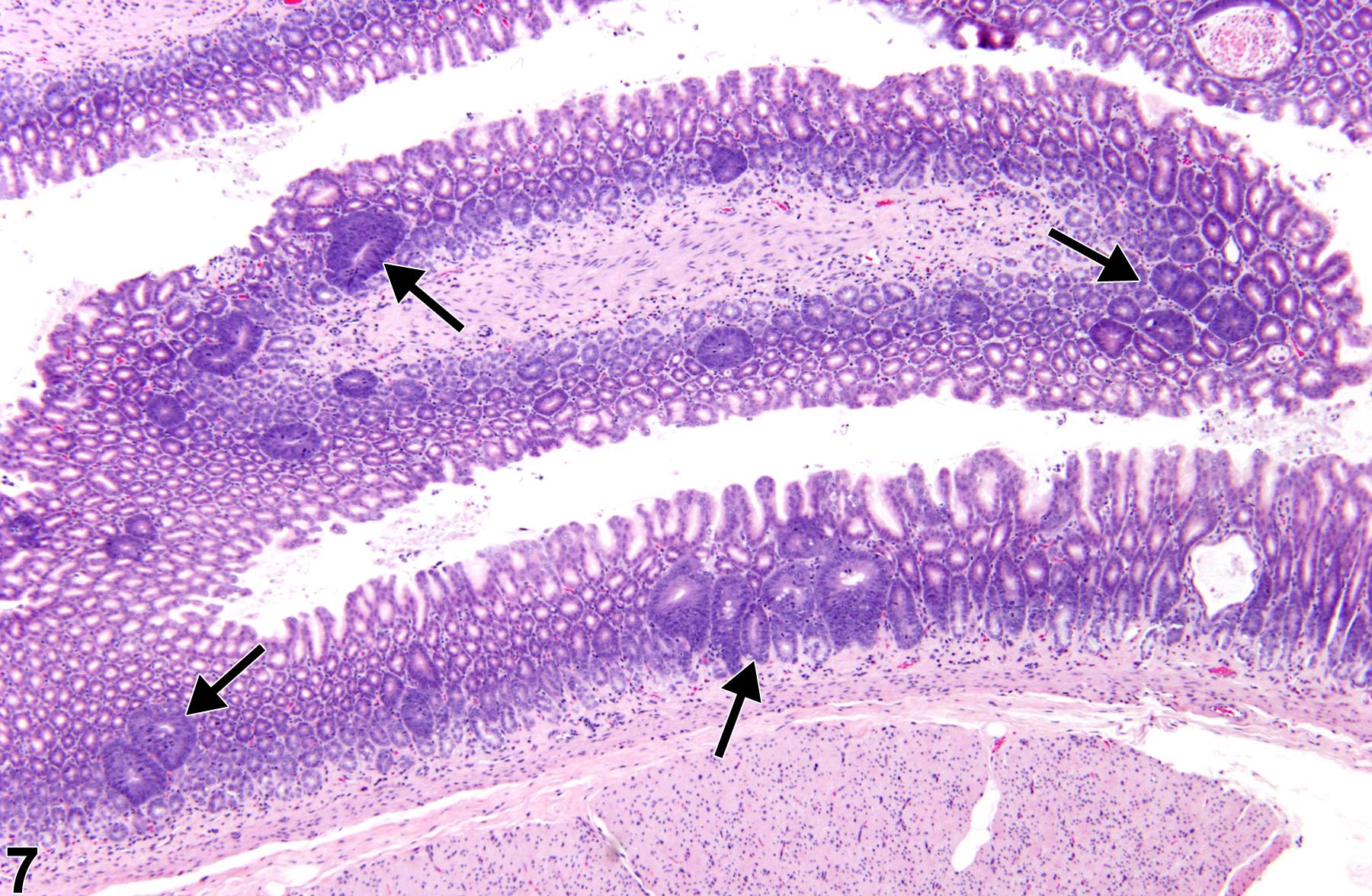
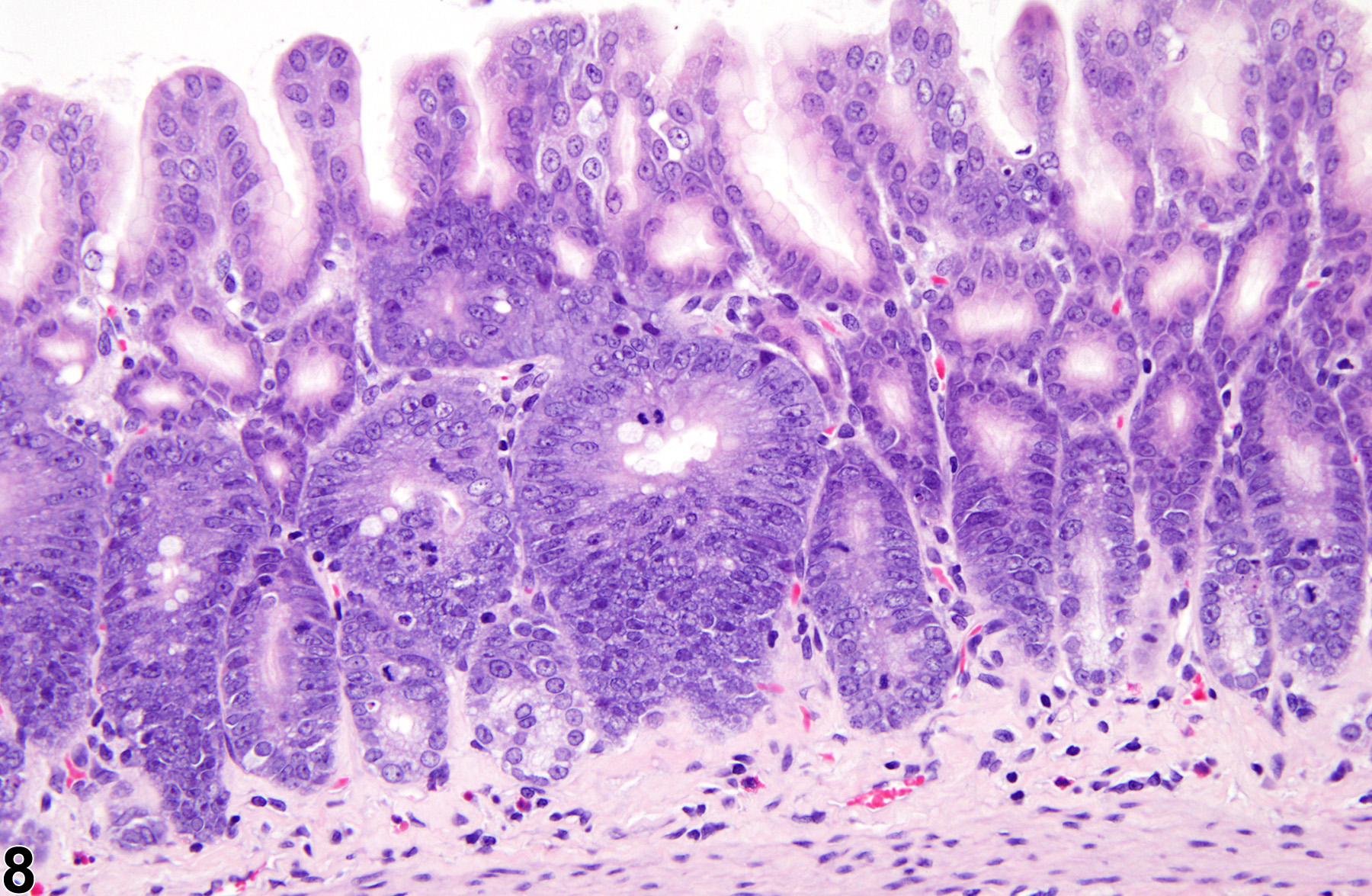
Stomach, Glandular Stomach, Epithelium - Hyperplasia
Narrative
Bertram TA, Markovits JE, Juliana MM. 1996. Non-proliferative lesions of the alimentary canal in rats GI-1. In Guides for Toxicologic Pathology. STP/ARP/AFIP, Washington, DC, 1-16.
Full Text: https://www.toxpath.org/docs/SSNDC/GINonproliferativeRat.pdfBrown HR, Hardisty JF. 1990. Oral cavity, esophagus and stomach. In: Pathology of the Fischer Rat (Boorman GA, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 9-30.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Greaves P. 2007. Digestive system. In: Histopathology of Preclinical Toxicity Studies, 3rd ed. Academic Press, London, 334-456.
Hirose M, Hakoi K, Takahashi S, Hoshiya T, Akagi K, Lin C, Saito K, Kaneko H, Shirai T. 1999. Sequential morphological and biological changes in the glandular stomach induced by oral administration of catechol to male F344 rats. Toxicol Pathol 27:448-455.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/10485826Hirose M, Wada S, Yamaguchi S, Masuda A, Okazaki S, Ito N. 1992. Reversibility of catechol-induced rat glandular stomach lesions. Cancer Res 52:787-790.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/1737338Johnson JD, Ryan MJ, Toft JD II, Graves SW, Hejtmancik MR, Cunningham ML, Herbert R, Abdo KM. 2000. Two-year toxicity and carcinogenicity study of methyleugenol in F344/N rats and B6C3F1 mice. J Agric Food Chem 48:3620-3632.
Abstract: http://pubs.acs.org/doi/abs/10.1021/jf000364aLeininger JR, Jokinen MP, Dangler CA, Whiteley LO. 1999. Oral cavity, esophagus, and stomach. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, St Louis, MO, 29-48.
Morson BC, Sobin LH, Grundmann E, Johansen A, Nagayo T, Serck-Hanssen A. 1980. Precancerous conditions and epithelial dysplasia in the stomach. J Clin Pathol 33:711-721.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1146204/Nagayo T. 1981. Dysplasia of the gastric mucosa and its relation to the precancerous state. Jpn J Cancer Res 72:813-823.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/7341333Streett CS, Cimprich RE, Robertson JL. 1984. Pathologic findings in the stomach of rats treated with the H2-receptor antagonist tiotidine. Scand J Gastroenterol Suppl 101:190-117.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/6152748Tsiftsis D, Jass JR, Filipe MI, Wastell C. 1980. Altered patterns of mucin secretion in the precancerous lesions induced in the glandular part of the rat stomach by the carcinogen N-methyl-N-nitrosoguanidine. Invest Cell Pathol 3:399-408.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/7462020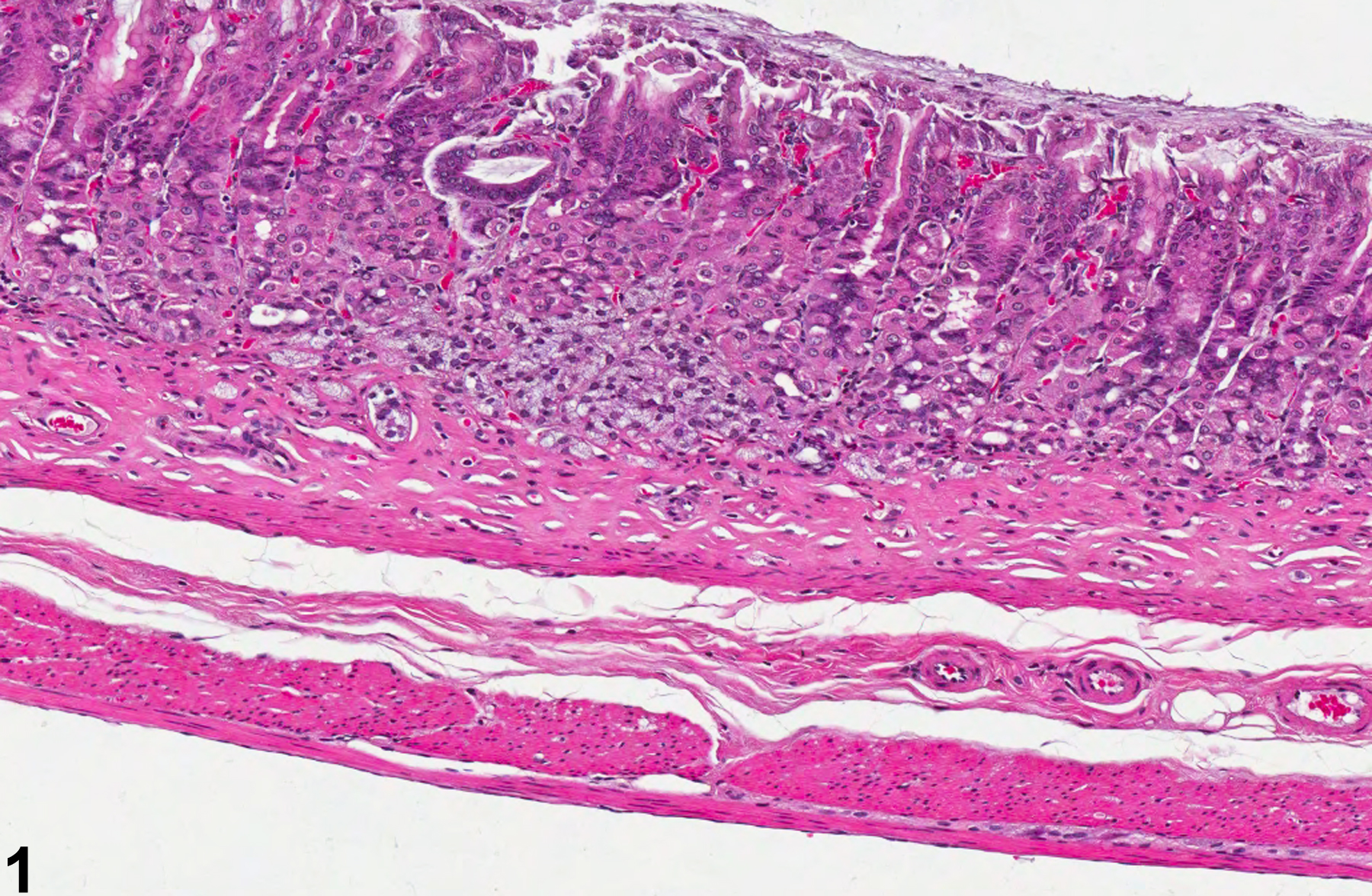
Stomach, Glandular stomach, Epithelium - Hyperplasia in female F344/N rat from a chronic study. There is a focal area of hyperplasia in the mucosa.