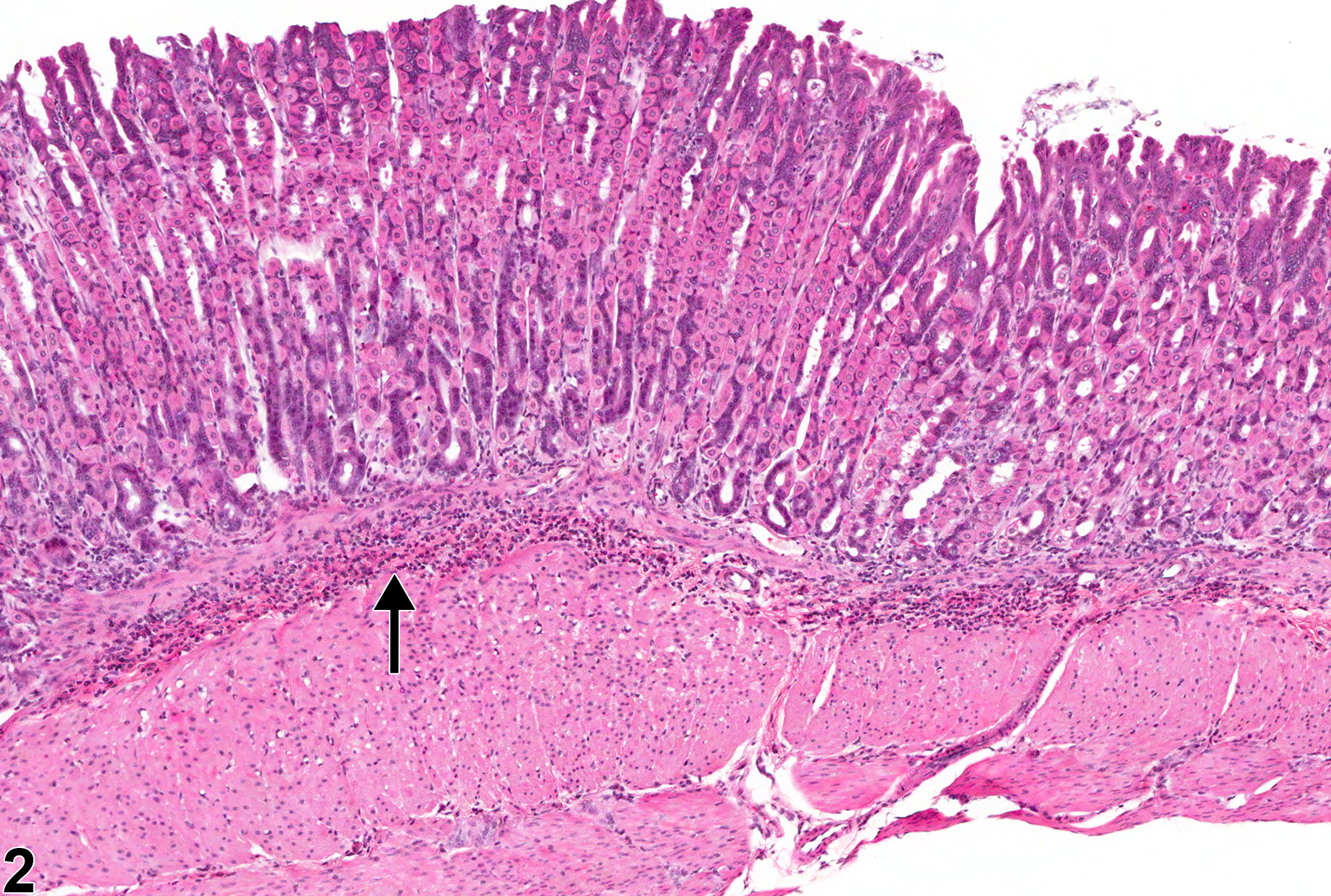
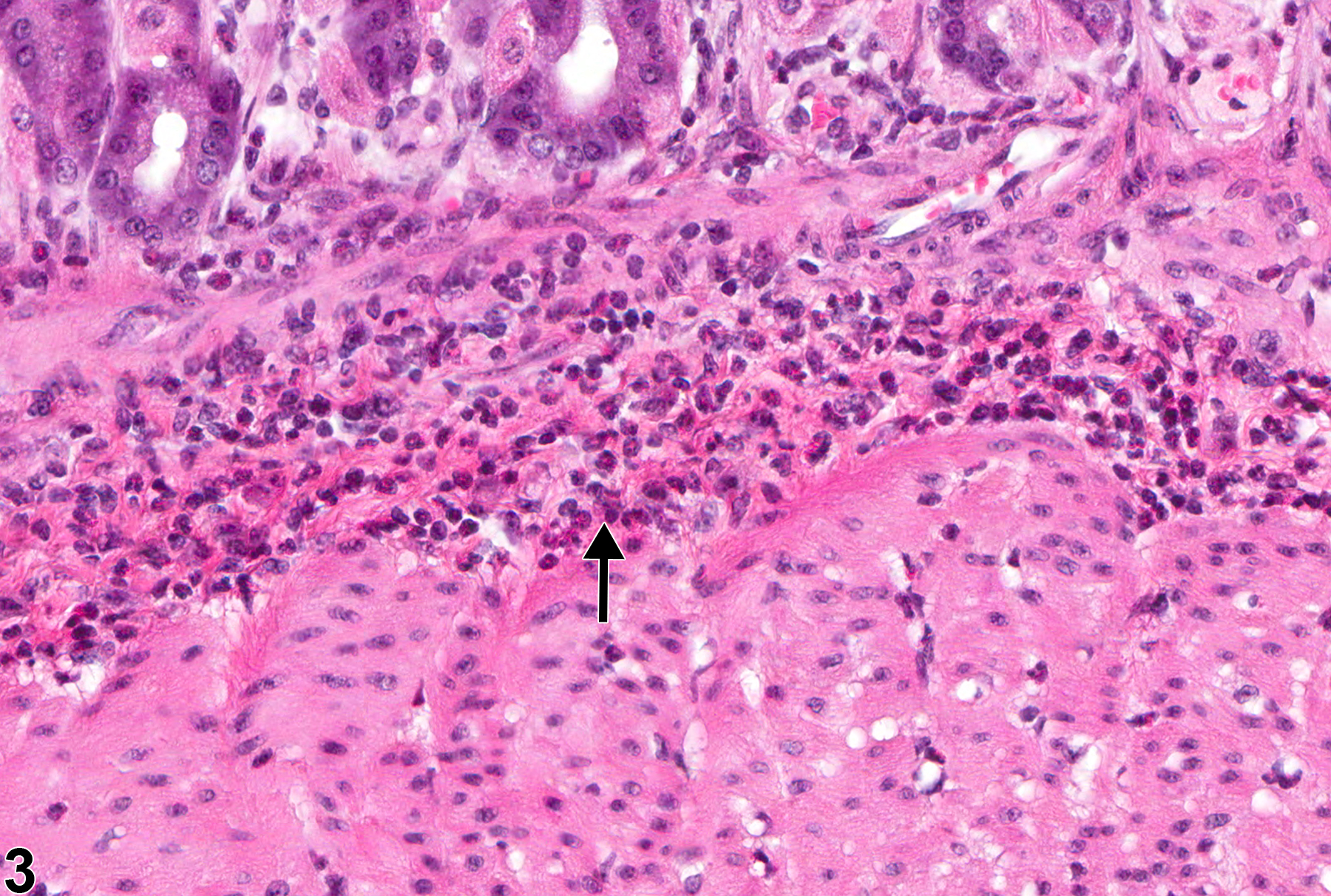
Alimentary System
Stomach, Glandular Stomach - Infiltration, Cellular
Narrative
Focal collections of mast cells can occasionally be found in the submucosa of the stomach. Mast cells have been shown to contribute importantly to acute allergic reactions, late-phase reactions, and chronic allergic inflammation. Focal accumulations of mast cells in the submucosa of the gastrointestinal tract are usually considered an incidental lesion in NTP studies. Such accumulations may be confused with mast cell tumor. Focal accumulations of mast cells tend to be smaller and less well circumscribed than mast cell tumors and infiltrate between structures rather than compressing or effacing adjacent structures.
Ackermann MR. 2007. Acute inflammation. In: Pathologic Basis of Veterinary Disease (McGavin MD, Zachary JF, eds). Mosby, St Louis, MO, 101-152.
Algood HM, Gallo-Romero J, Wilson KT, Peek RM Jr, Cover TL. 2007. Host response to Helicobacter pylori infection before initiation of the adaptive immune response. FEMS Immunol Med Microbiol 51:577-586.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/17919297Ferrero RL, Ave P, Radcliff FJ, Labigne A, Huerre MR. 2000. Outbred mice with long-term Helicobacter felis infection develop both gastric lymphoid tissue and glandular hyperplastic lesions. J Pathol 191:333-340.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/10878557Leininger JR, Jokinen MP, Dangler CA, Whiteley LO. 1999. Oral cavity, esophagus, and stomach. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, St Louis, MO, 29-48.
Park JH, Hong JJ, Park JH. 2003. Experimental infection of mice with tightly coiled spiral bacteria ("Candidatus Helicobacter suis") originating from the pig stomach. J Comp Pathol 129:54-160.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/12921721Wedemeyer J, Galli SJ. 2000. Mast cells and basophils in acquired immunity. Br Med Bull 56:936-955.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11359630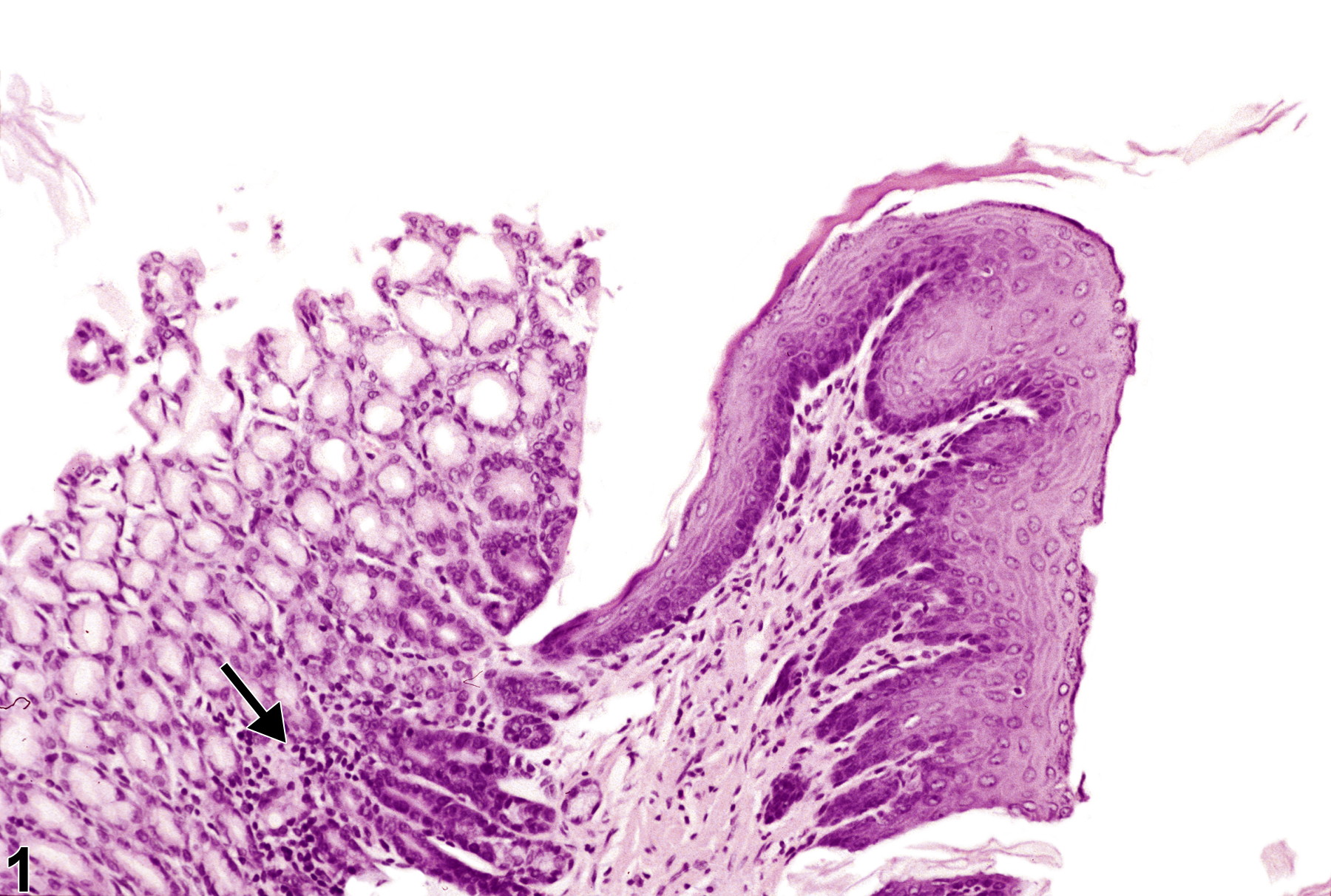
Stomach, Glandular stomach - Infiltration, Cellular, Lymphocyte in a male F344/N rat from a chronic study. Lymphocytic cellular infiltrate is present in the deep mucosa of glandular stomach (arrow).