Cardiovascular System
Blood Vessel - Polyarteritis Nodosa
Narrative
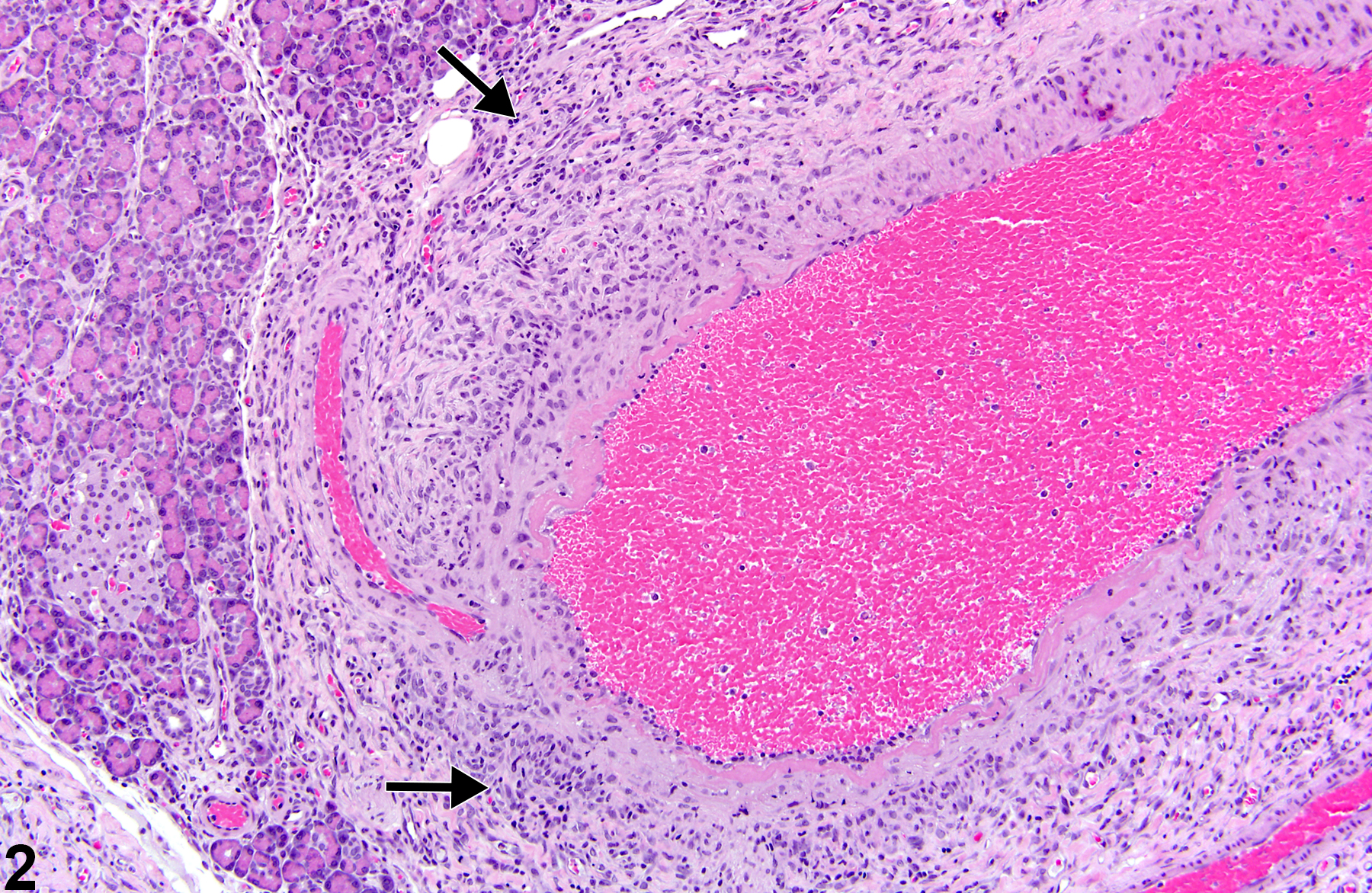
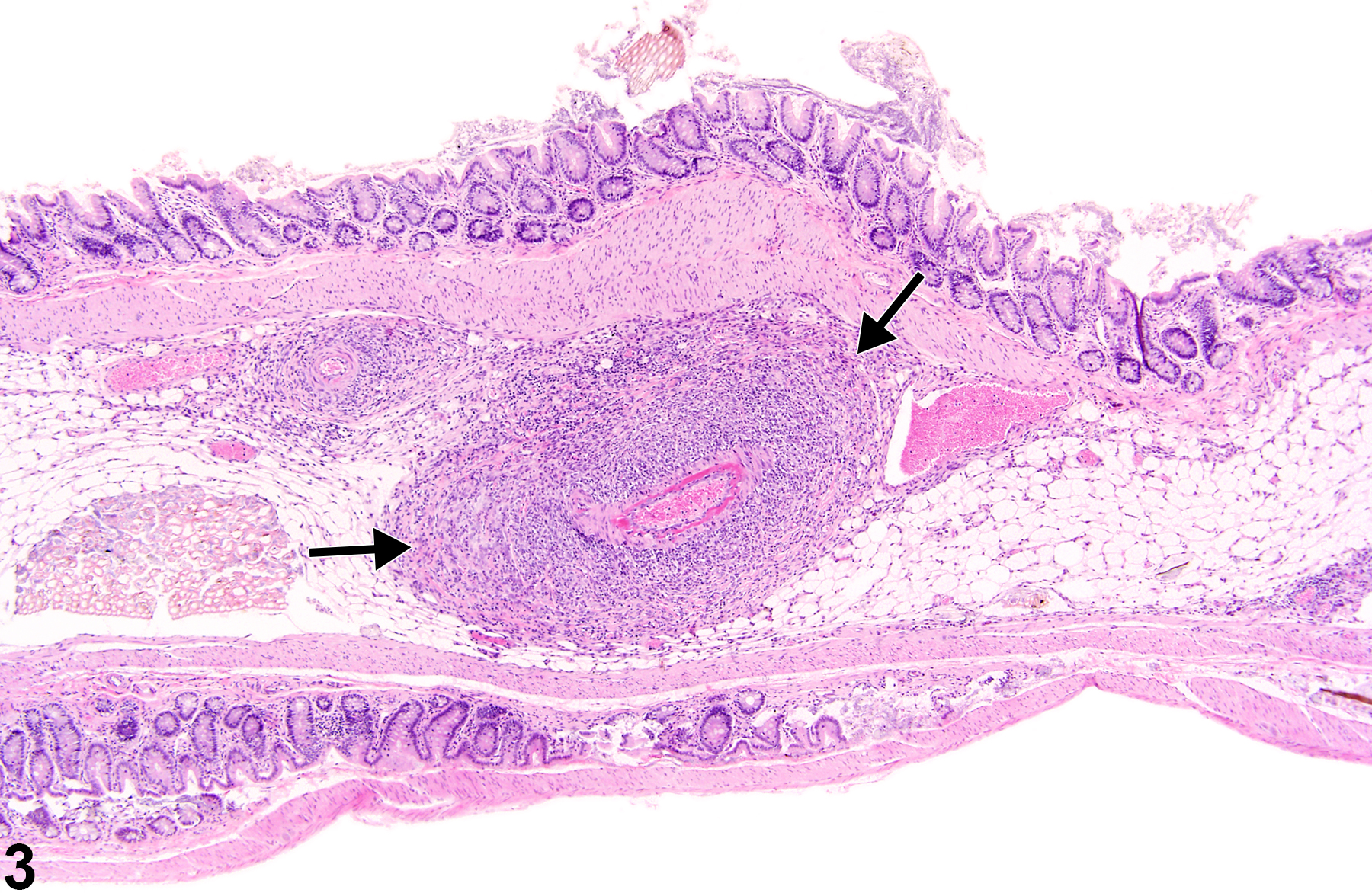
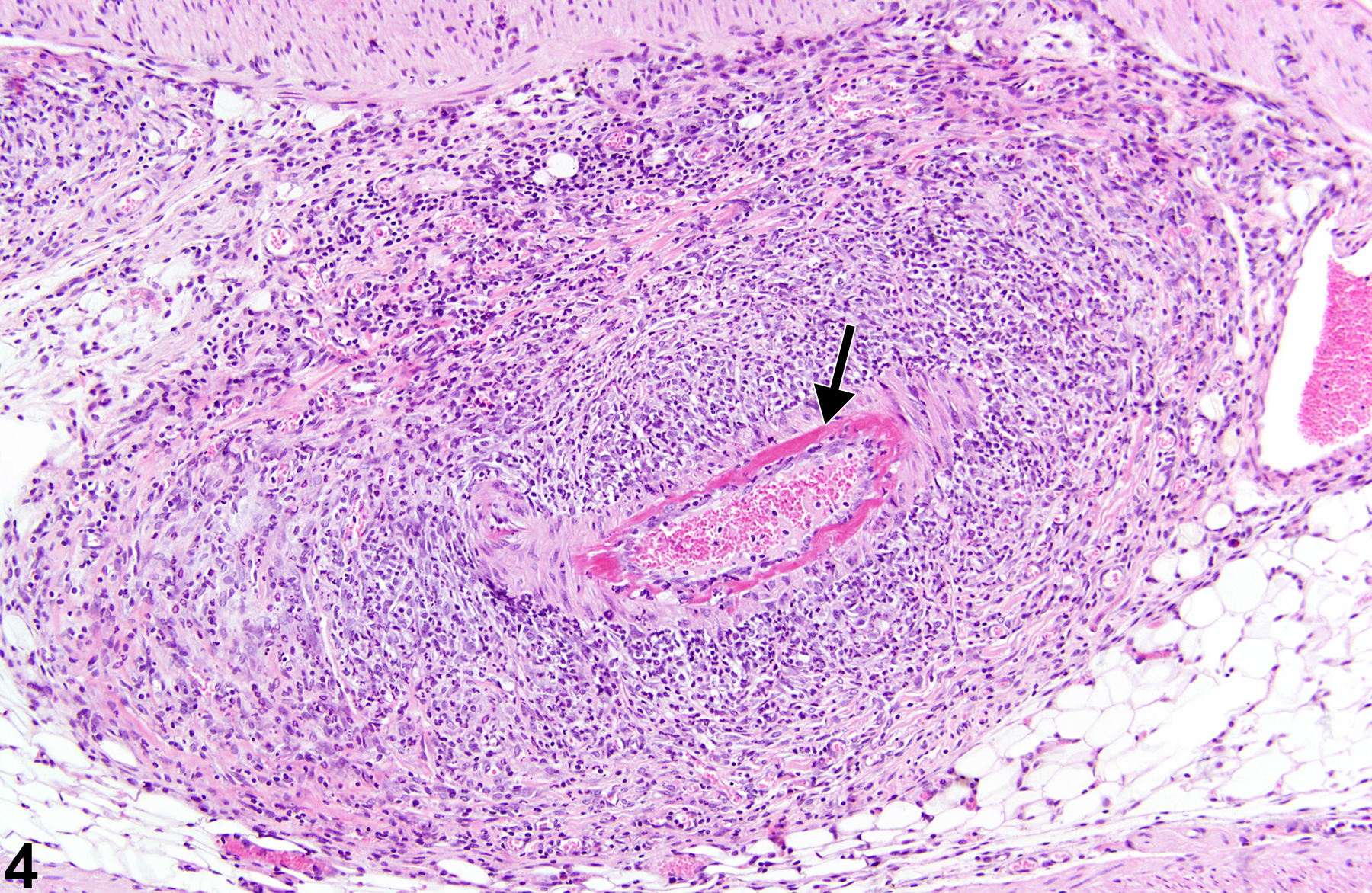
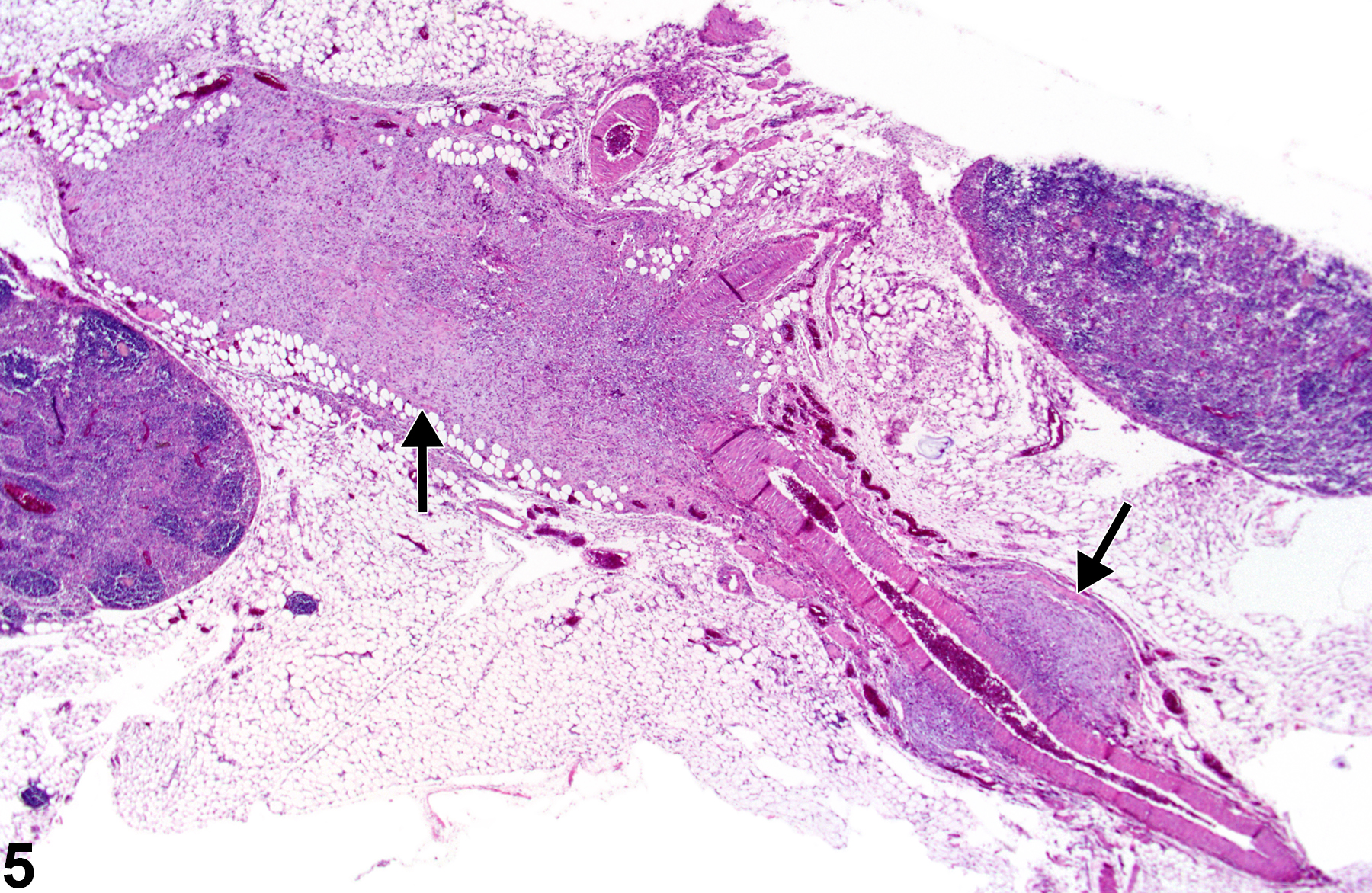
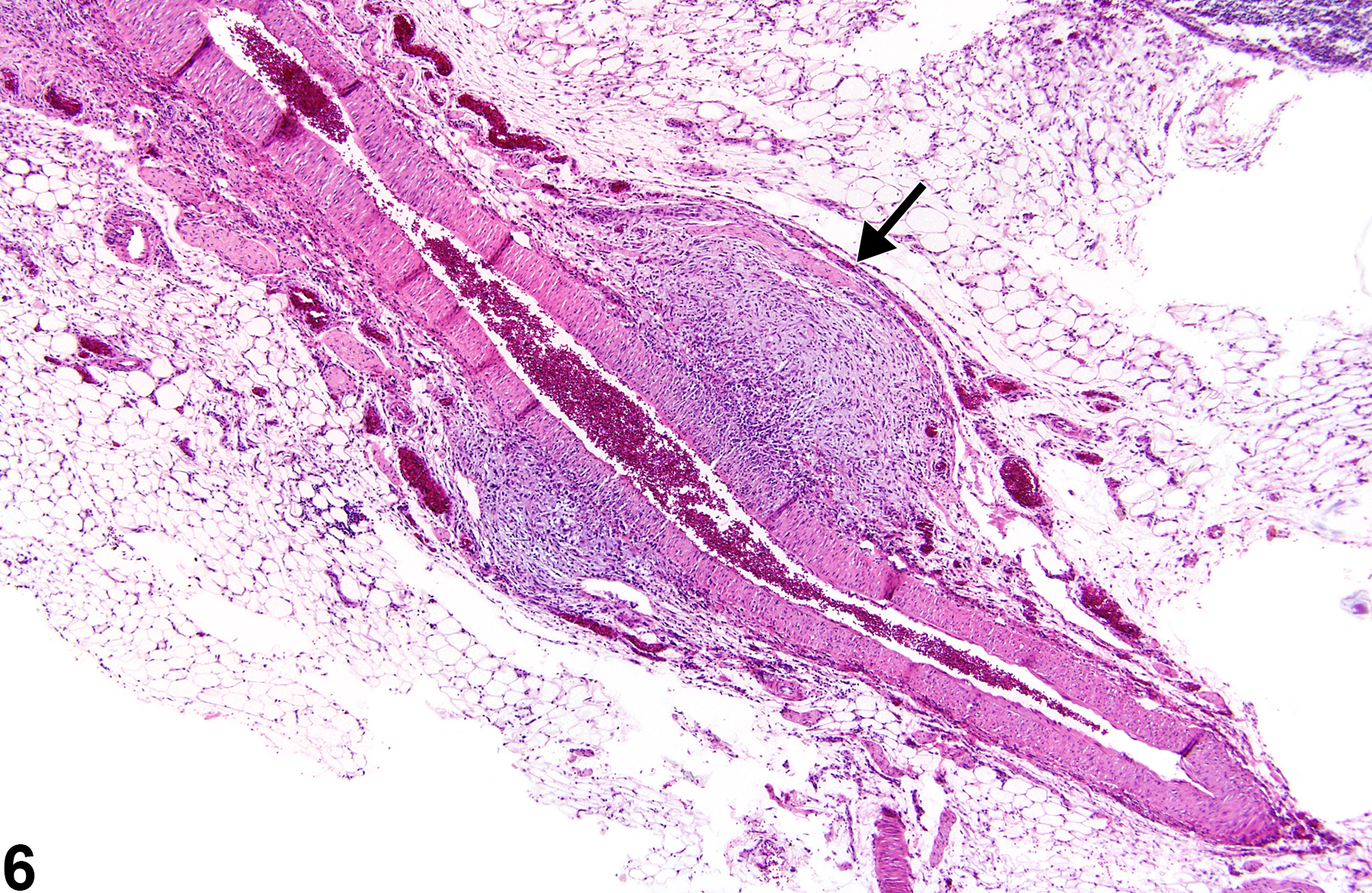
Polyarteritis nodosa (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, and Figure 9) has many synonyms, including polyarteritis, arteritis, periarteritis, spontaneous necrotizing arteriopathy, panarteritis, polyangitis, chronic arteritis, and necrotizing vasculitis. The incidence of polyarteritis nodosa varies among different strains of rats and is seen most commonly in Sprague Dawley, August, and Mendel-Sherman rats and less frequently in Wistar and Long-Evans rats. It is even less common in F344 rats. In all rat strains it is more common in older males than in females. The blood vessels most commonly affected are vessels of the mesentery, pancreas, and testes, but lesions may also occur in the hepatic, coronary, uterine, cerebral, adrenal, and renal arteries. The pathogenesis is unknown but is suspected to be immunologic. Dietary content and dietary restriction have been shown to affect the incidence of polyarteritis nodosa in rats and mice. Lesions similar to polyarteritis nodosa have been seen in repeatedly bred rats and in rats with induced hypertension. In mice, lesions are similar to those occurring in the rat but are more frequently distributed in renal blood vessels. Specifically, in CD-1 mice polyarteritis nodosa has been noted in the thymus, ovary, uterus, kidney, and heart. Polyarteritis nodosa is also observed in the aged hamster, with renal, testicular, and coronary arteries affected.
Blood vessel necrosis in laboratory animals is most often seen as one component of the spectrum of lesions of polyarteritis nodosa and rarely presents as an individual lesion without other associated changes. Fibrinoid necrosis as part of spontaneous polyarteritis nodosa often involves the pancreatic and mesenteric vessels of rats. Interestingly, administration of cardiovascular drugs such as vasodilators and vasoconstrictor substances causes medial necrosis of the same vascular beds.
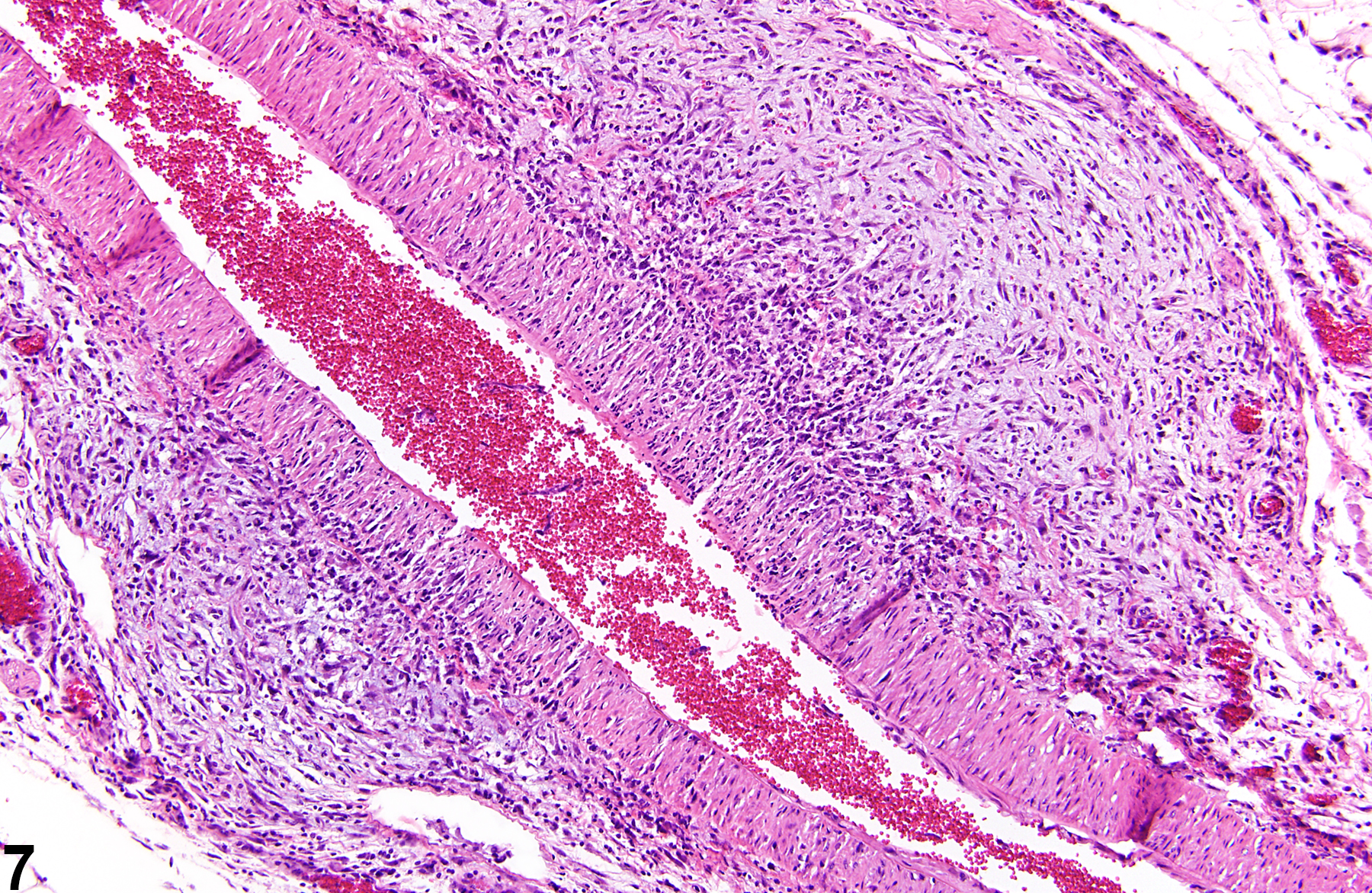
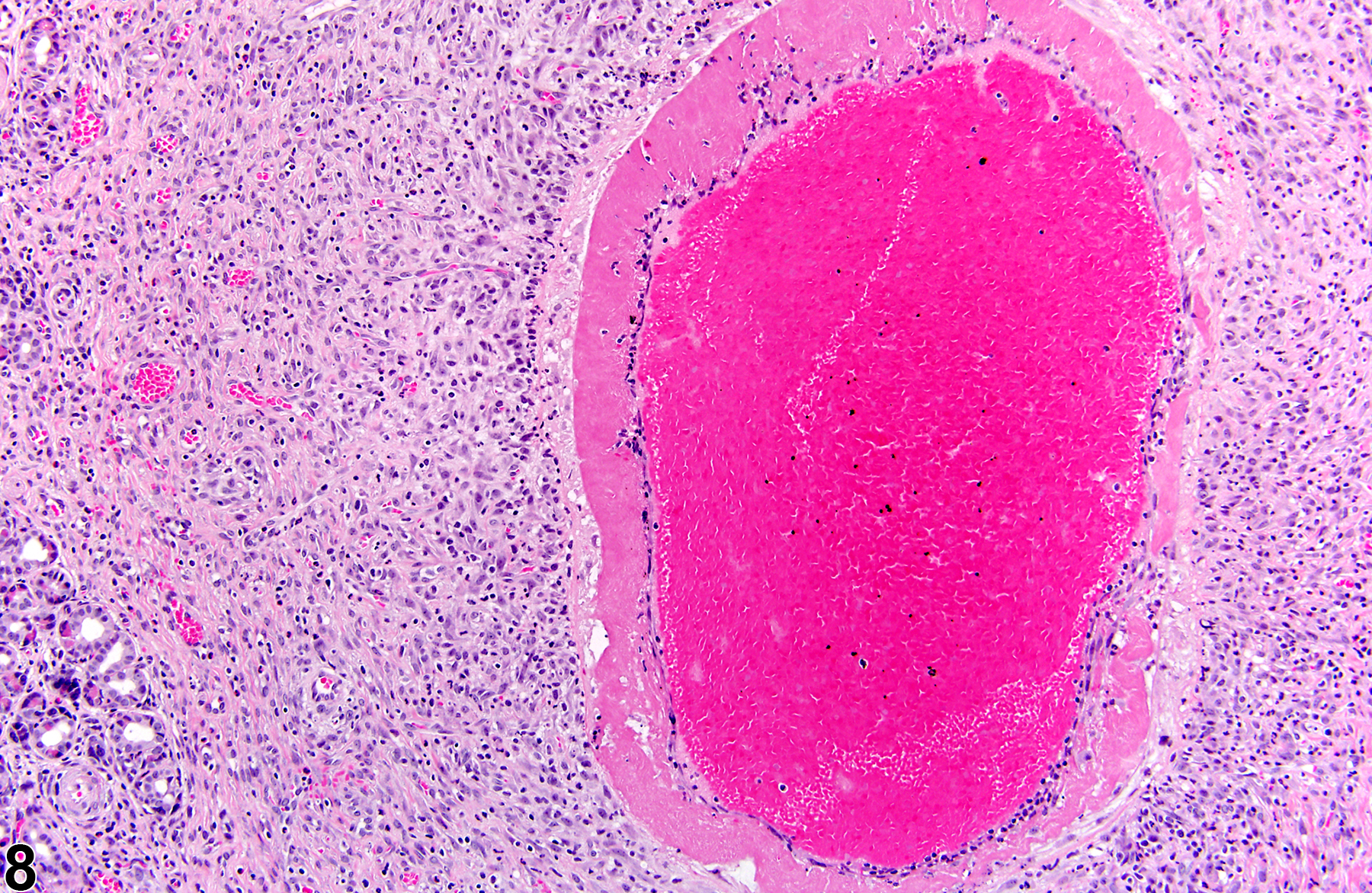
In the acute phase, lesions are typified by fibrinoid necrosis of the arterial walls, with a marked neutrophilic infiltrate. As the lesions progress, medial and adventitial fibrosis becomes more prominent, numbers of lymphocytes and plasma cells increase, and intimal proliferation may occur. The arterial lumen may narrow due to thrombus formation. Complete occlusion, aneurysm formation, and recanalization of thrombi may occur in later stages. Acute and chronic vascular changes often coexist in the same animal and can be found in the same artery.
Though it is generally considered a spontaneous disease, polyarteritis nodosa may be exacerbated by administration of xenobiotics. Additionally, the lesions of polyarteritis nodosa may be induced by administration of some xenobiotics, which may prove difficult to distinguish from the spontaneous disease. Comparison to the concurrent controls may help in differentiating spontaneous lesions from induced lesions. The anatomic vascular sites affected may differ with the type of compound administered.
Polyarteritis nodosa may present segmentally, and multiple stages of inflammation (acute, chronic, and healed lesions) may be encountered within the same animal. The five types of inflammation most commonly used to represent the cell types and temporal association with lesions are acute, suppurative, chronic, chronic-active, and granulomatous. In acute inflammation, the predominant cell type is the neutrophil, though fewer macrophages and lymphocytes may also be present. Neutrophils are also the predominant cell type in suppurative inflammation, but they are aggregated, and many are degenerate. Suppurative inflammation often contains cellular debris, proteinaceous fluid, and fibrin. Lymphocytes predominate in chronic inflammation, and macrophages, plasma cells, and eosinophils may also be present. Attempts at healing, typified by fibrosis, may also be present in chronic polyarteritis. Lymphocytes are also the predominant cell type in chronic-active inflammation, though a significant acute inflammatory component must also be present, notably fibrin and neutrophils. Granulomatous inflammation is a form of chronic inflammation in which the predominant cell type is the activated macrophage. Activated macrophages may take on an epithelial appearance (epithelioid macrophage) or present as multinucleated giant cells. Granulomatous inflammation is usually in response to foreign material. An example of this type of reaction was noted in pigs undergoing transcoronary intervention: fragments of embolized surgical gauze produced granulomatous lesions in blood vessels. Vascular mineralization may be a prominent feature in both chronic inflammatory processes.
Whenever present, polyarteritis nodosa should be diagnosed and graded based on the extent of lesion. It should be diagnosed in the organs in which it appears (e.g., Mesentery - Polyarteritis nodosa). The individual components of polyarteritis nodosa (e.g., inflammation, vascular necrosis, fibrosis) should not be diagnosed separately unless they are considered lesions of a differing pathogenesis in animals with polyarteritis nodosa. The pathologist should attempt to differentiate spontaneous vascular lesions from those induced by compound administration, taking into account the strain, sex, and anatomic location of the lesions. If a more detailed analysis needs to be performed, the NTP pathologist should be consulted.
Clemo FAS, Evering WE, Snyder PW, Albassam MA. 2003. Differentiating spontaneous from drug-induced vascular injury in the dog. Toxicol Pathol 31:25-31.
Full Text: http://tpx.sagepub.com/content/31/1_suppl/25.full.pdfCohen JK, Cai LQ, Zhu YS, La Perle KM. 2007. Pancreaticoduodenal arterial rupture and hemoabdomen in ACI/SegHsd rats with polyarteritis nodosa. Comp Med 57(4):370-376.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/17803051Johansson S. 1981. Cardiovascular lesions in Sprague-Dawley rats induced by long-term treatment with caffeine. Acta Pathol Microbiol Scand A 89(3):185-191.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/6118996Louden C, Morgan DG. 2001. Pathology and pathophysiology of drug-induced arterial injury in laboratory animals and its implication on the evaluation of novel chemical entities for human clinical trials. Pharmacol Toxicol 89:158-170.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11881965Maita K, Hirano M, Harada T, Mitsumori K, Yoshida A, Takahashi K, Nakashima N, Kitazawa T, Enomoto A, Inui K, Shirasu Y. 1988. Mortality, major cause of moribundity, and spontaneous tumors in CD-1 mice. Toxicol Pathol 16:340-349.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/3194656Maxie MG, Robinson WF. 2007. Cardiovascular system. In: Jubb, Kennedy and Palmer's Pathology of Domestic Animals, Vol 3, 5th ed (Maxie MG, ed). Saunders Elsevier, Edinburgh, 69-72.
Mecklenburg L, Heuser A, Juengling T, Kohler M, Foell R, Ockert D, Tuch K, Bode G. 2006. Mesenteritis precedes vasculitis in the rat mesentery after subacute administration of a phosphodiesterase type 4 inhibitor. Toxicol Lett 163(1):54-64.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16246509Mitsomori K. 1990. Blood and lymphatic vessels. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 475-483.
Morton D, Safron JA, Glosson J, Rice DW, Wilson DM, White RD. 1997. Histologic lesions associated with intravenous infusions of large volumes of isotonic saline solution in rats for 30 days. Toxicol Pathol 25:390-394.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/9280122Moyer CF, Kodavanti UP, Haseman JK, Costa DL, Nyska A. 2002. Systemic vascular disease in male B6C3F1 mice exposed to particulate matter by inhalation: Studies conducted by the National Toxicology Program. Toxicol Pathol 30:427-434.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/12187935Nyska A, Herbert RA, Chan PC, Haseman JK, Hailey JR. 1998. Theophylline induced periarteritis in F344/N rats. Arch Toxicol 72(11):731-737.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/9879811Nyska A, Jokinen MP, Brix AE, Sells DM, Wyde ME, Orzech D, Haseman JK, Flake G, Walker NJ. 2004. Exocrine pancreatic pathology in female Harlan Sprague-Dawley rats after chronic treatment with 2,3,7,8-tetrachlorodibenzo-p-dioxin and dioxin-like compounds. Environ Health Perspect 112:903-910.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1242020/Peters BS, Kuttler B, Beineke A, Lorenz G, Thiele A, Nicolai O, Rettig R, Mullins JJ, Peters J. 2010. The renin-angiotensin system as a primary cause of polyarteritis nodosa in rats. J Cell Mol Med 14(6A):1318-1327.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3828848/Ramot Y, Amir G, Willenz EP, Nyska A. 2008. Foreign-body granulomas within intramyocardial arteries in a transcoronary safety assessment in pigs. Toxicol Pathol 36:385-387.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/18467686Ramot Y, Manno RA, Okazaki Y, Krakovsky M, Lamensdorf I, Meiron M, Toren A, Zehavi-Goldstein E, Vezzali E, Nyska A. 2009. Spontaneous aortitis in the BALB/c mouse. Toxicol Pathol 37:667-671.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/19474416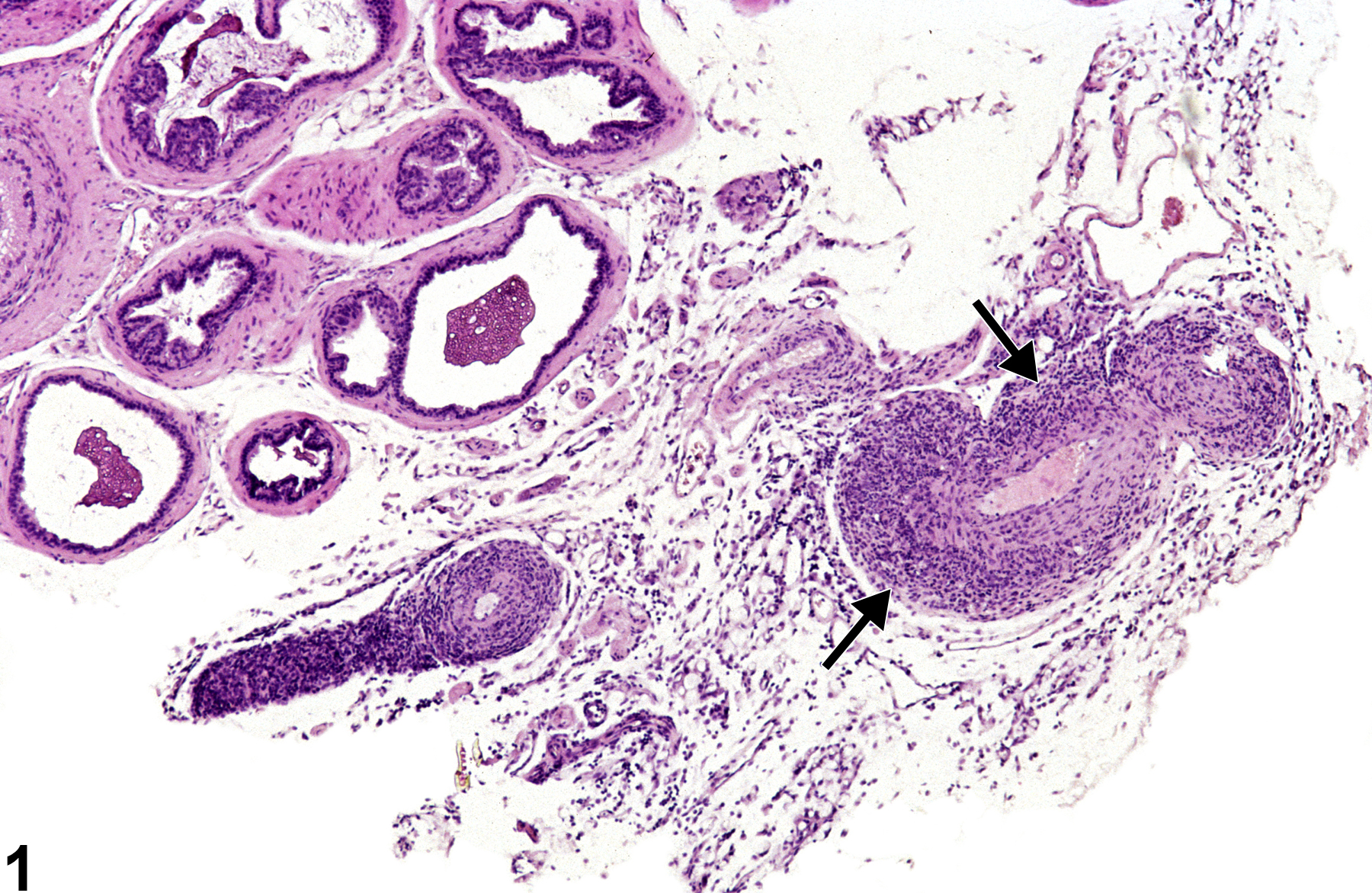
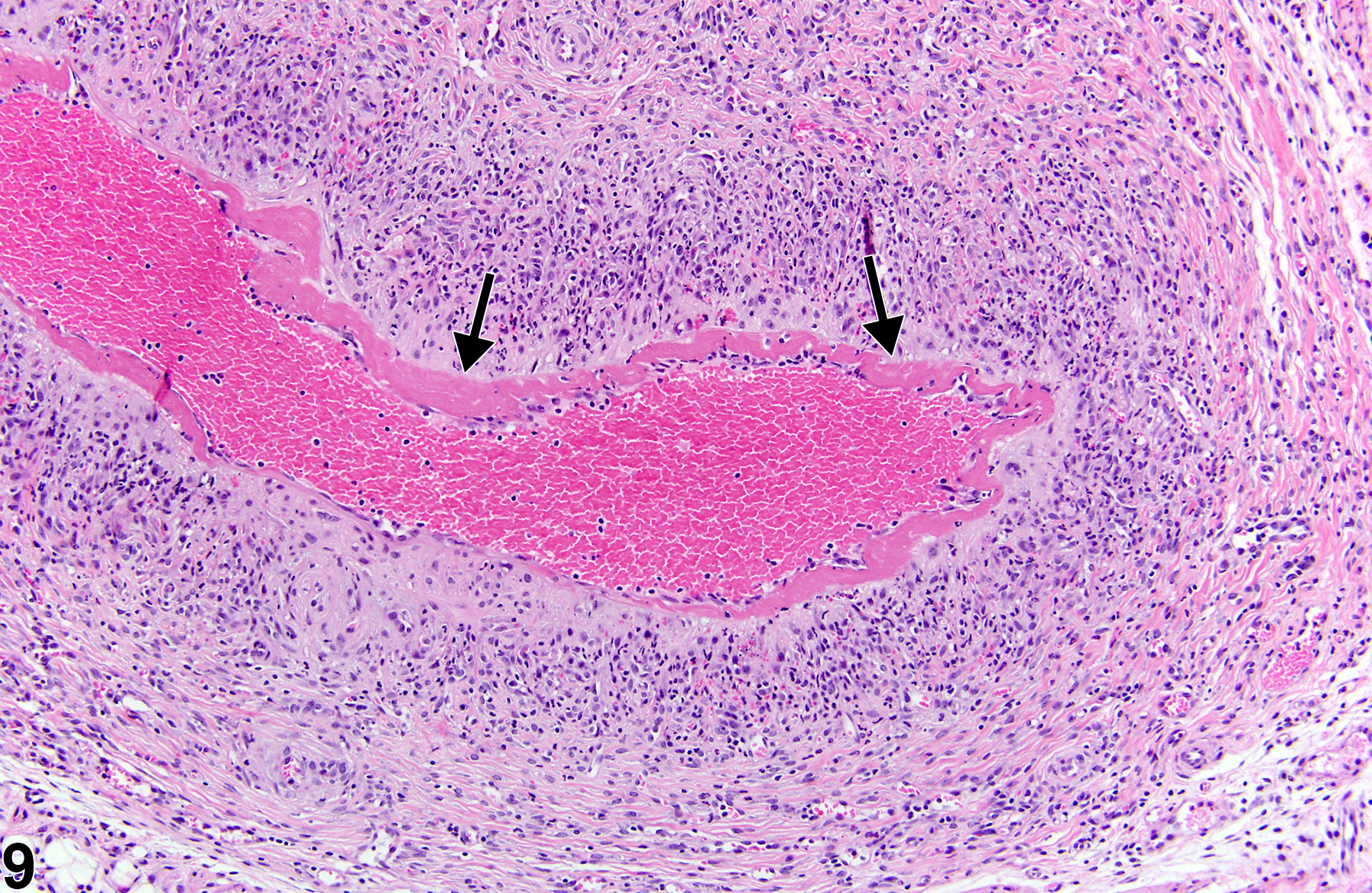
Prostate - Polyarteritis nodosa in a male B6C3F1/N mouse from a subchronic study. Abundant inflammatory cells surround a prostatic artery (arrows).