Cardiovascular System
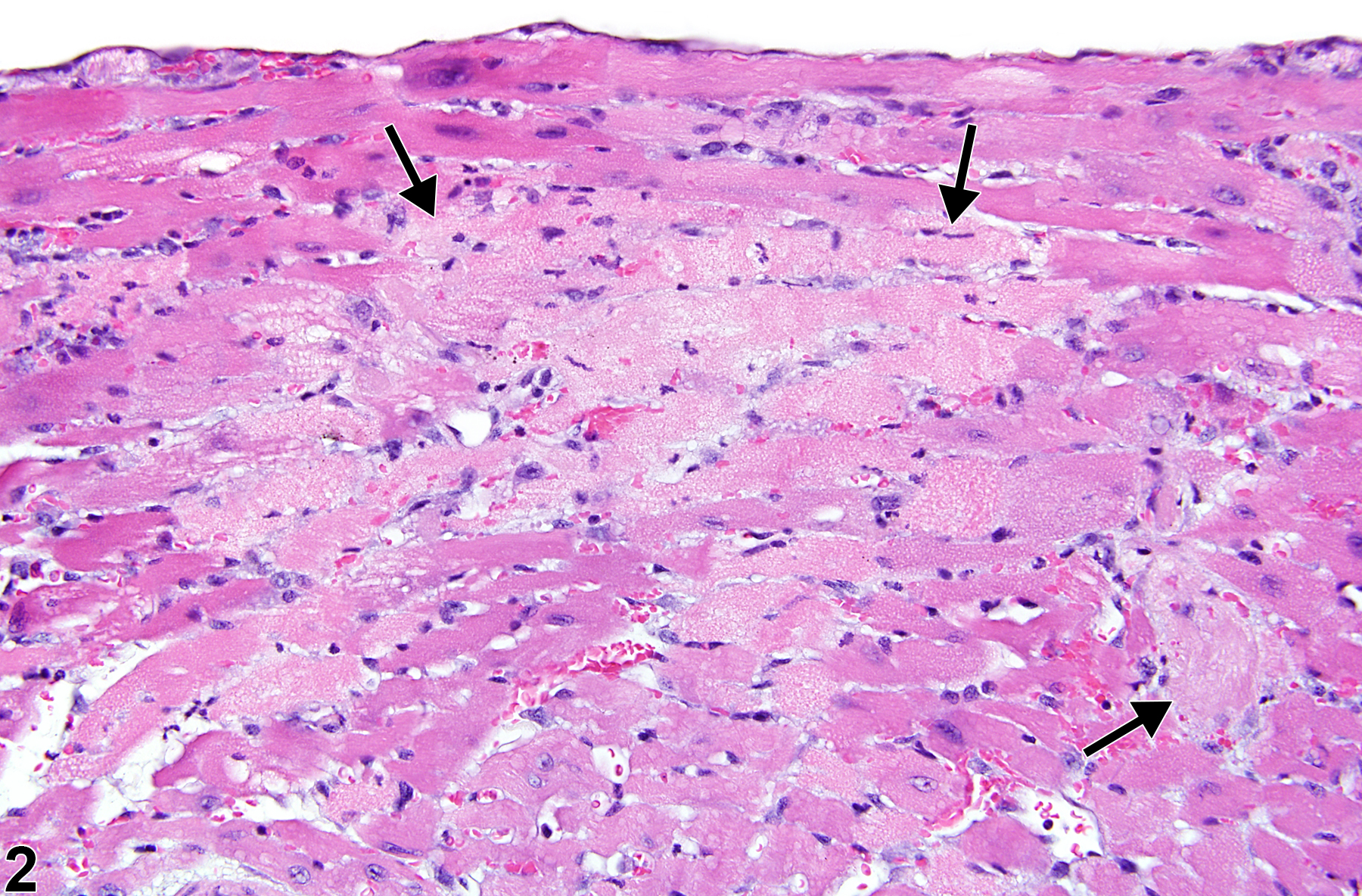
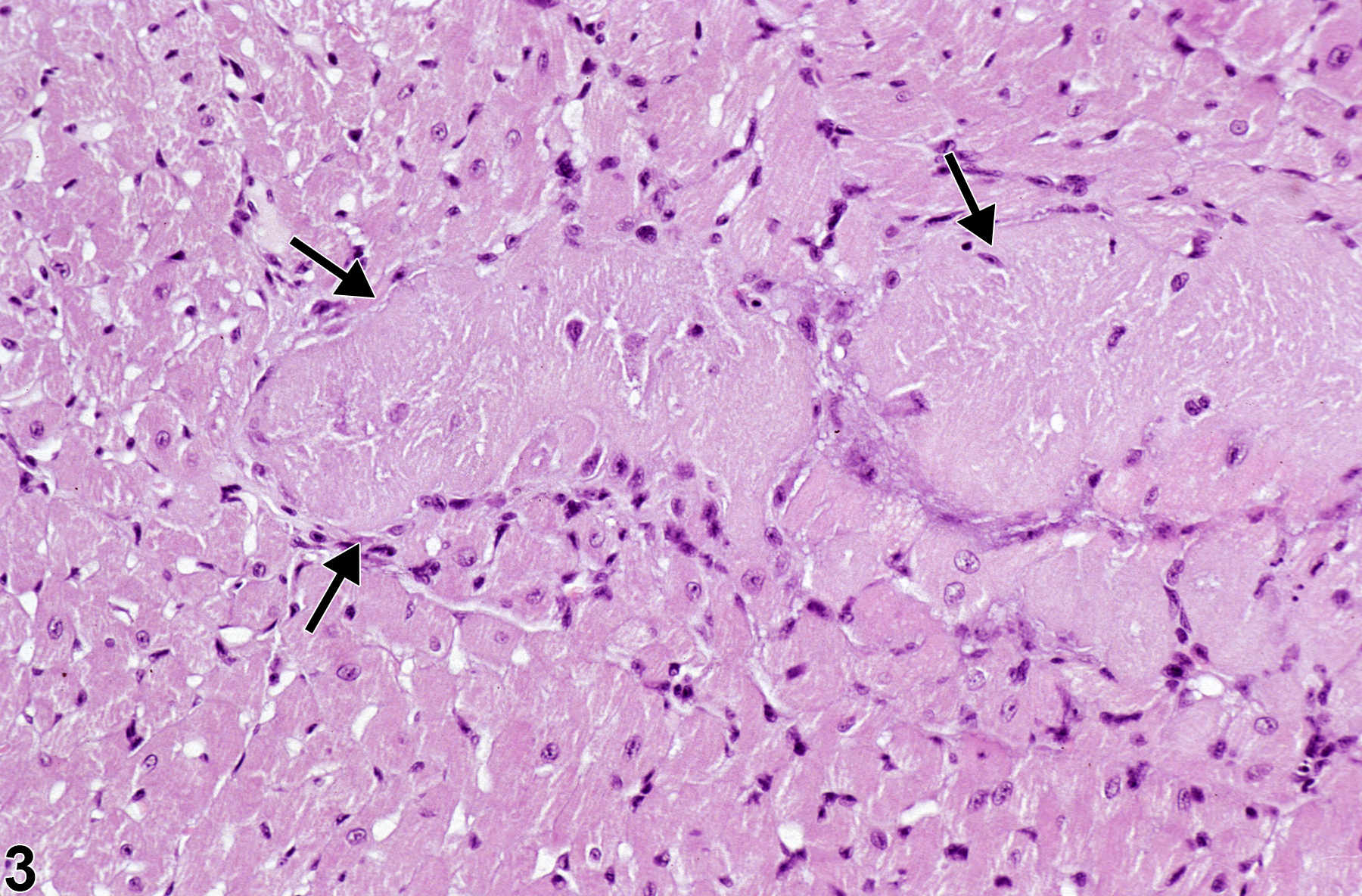
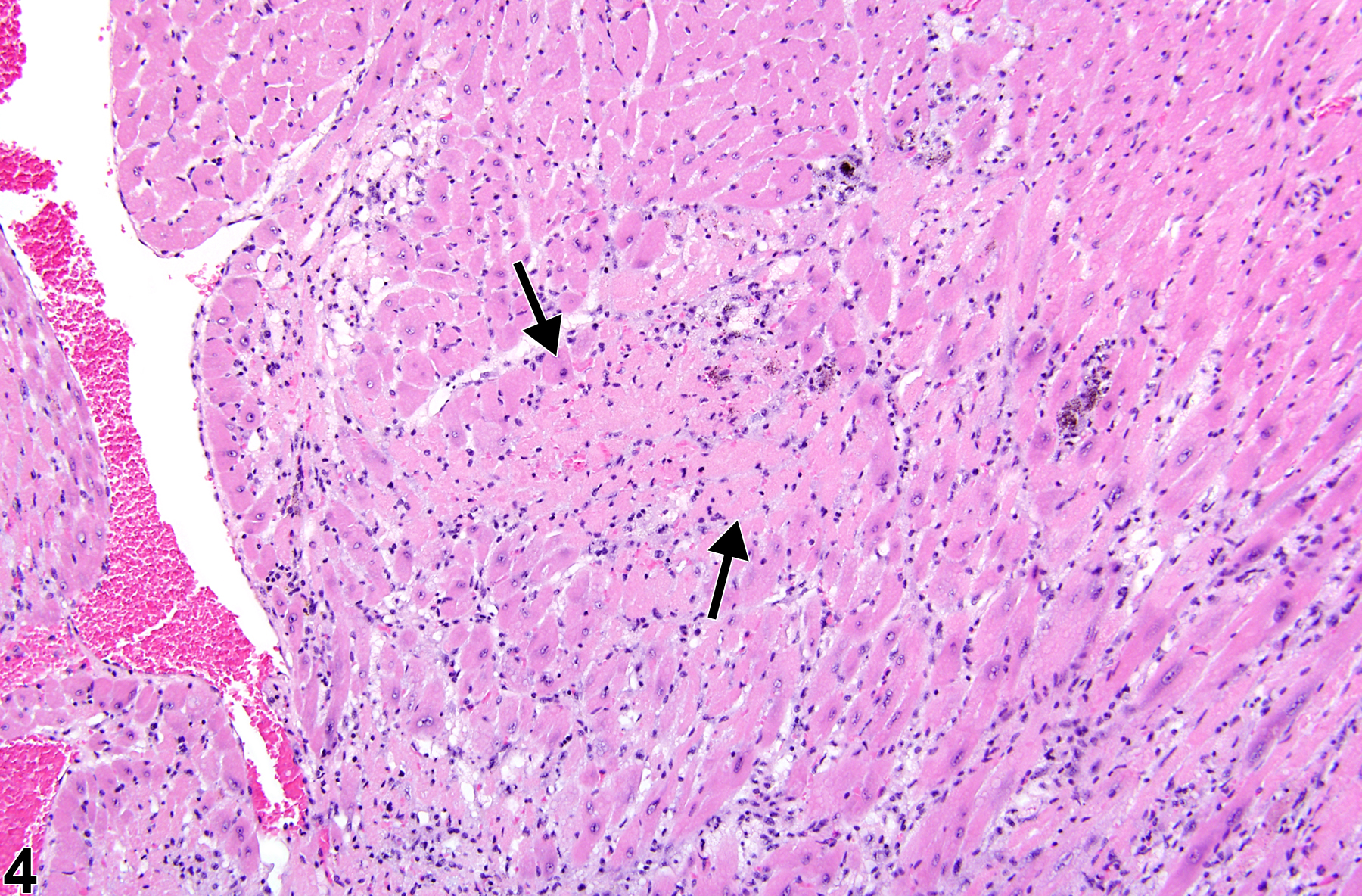
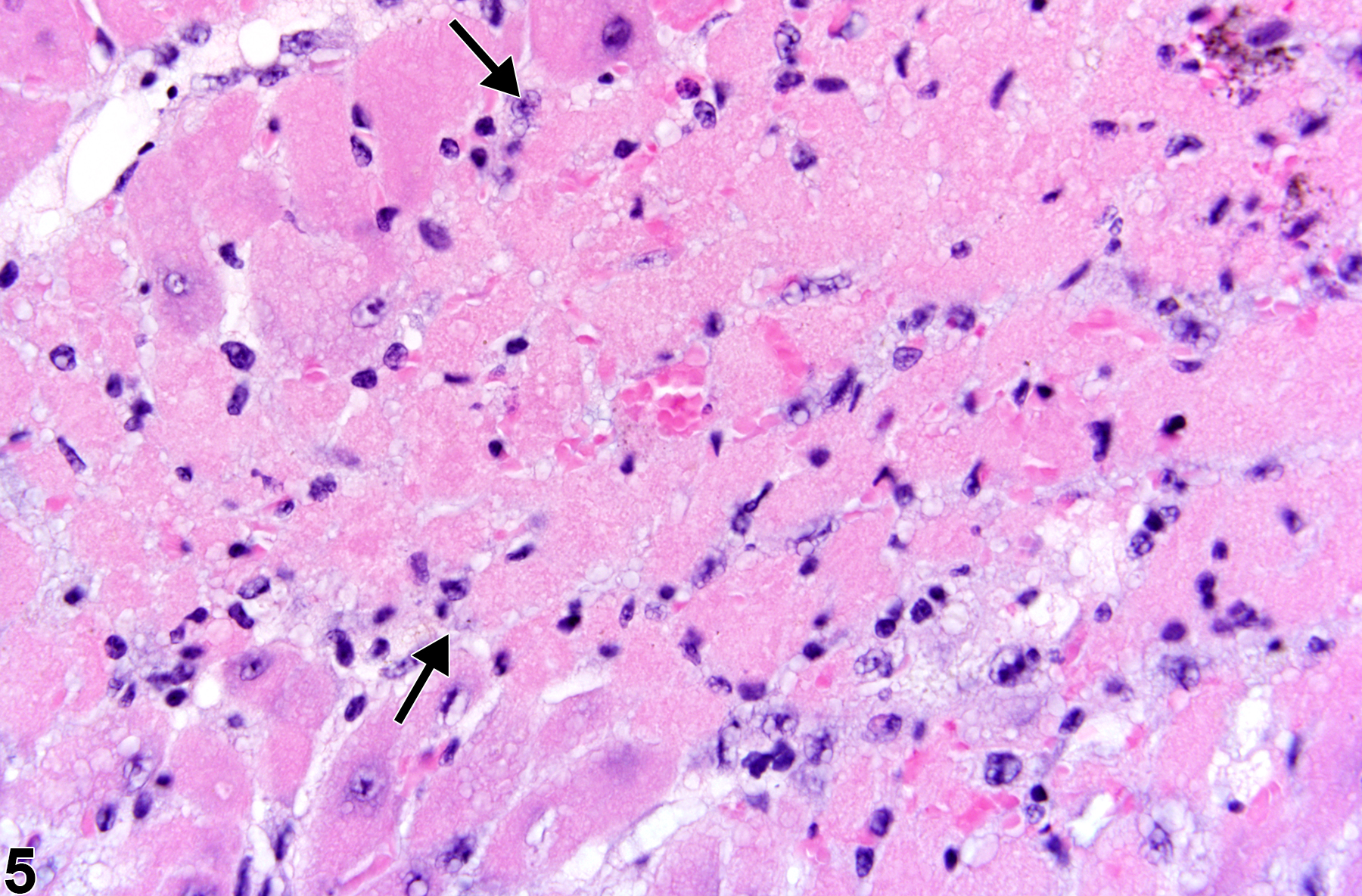
Heart, Myocardium - Necrosis
Narrative
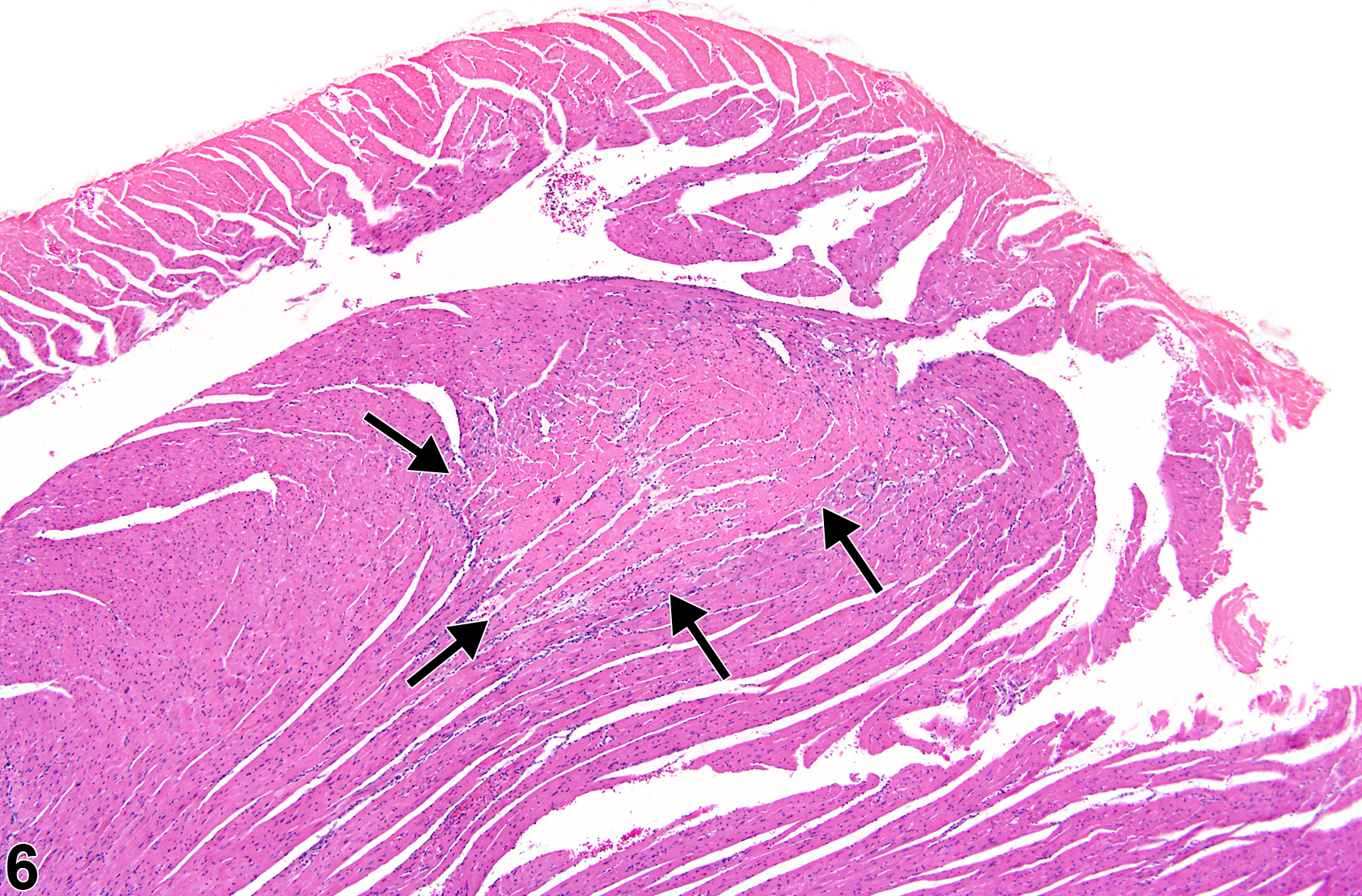
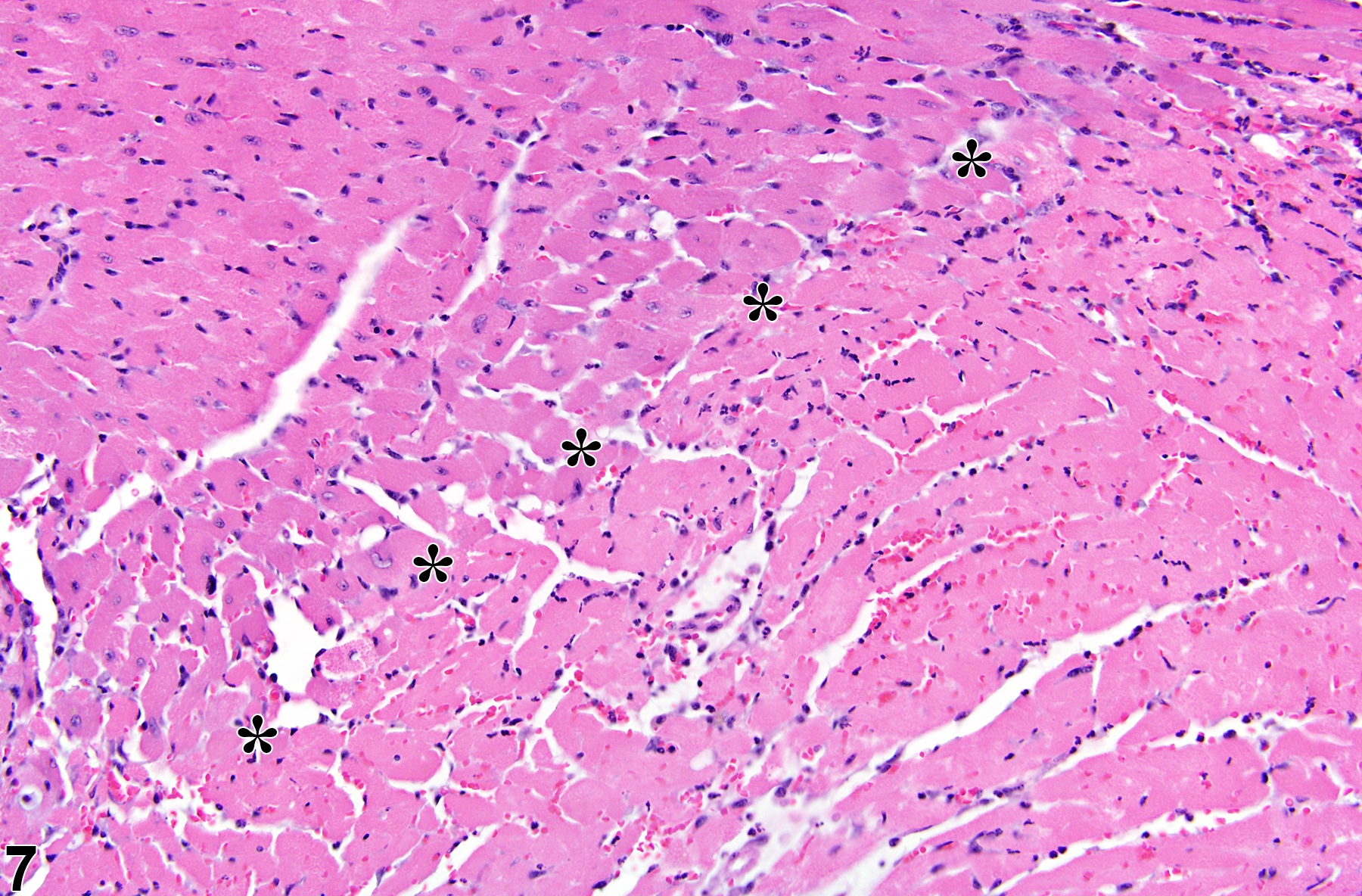
Necrosis can be one of the morphologic manifestations seen in cardiotoxicity (i.e., myofiber vacuolation, mononuclear cell infiltration, and fibrosis) or may be the predominant component in relatively early stages of spontaneous cardiomyopathy. Necrosis may also result from ischemia (infarct); these areas tend to be larger and are typically well circumscribed (Figure 6 and Figure 7). These lesions may have an inflammatory reaction at the margins and become fibrotic with time. Infarcts are caused by occlusion (e.g., by ligation or thrombosis) of a coronary artery that result in regional ischemic/coagulative necrosis of cardiomyocytes supplied by that vessel. The extent of the infarct is determined by the level at which the vessel is occluded, the volume of myocardium supplied by the vessel, and the collateral circulation to that region of the heart. There is no regional predilection for the formation of infarcts. Chemically induced models of heart infarction, without documentation of coronary vessel thrombosis, have been proposed in the literature.
Dunnick J, Johnson JA, Horton J, Nyska A. 2004. Bis(2-chloroethoxy)methane-induced mitochondrial and myofibrillar damage: Short-term time-course study. Toxicol Sci 81:243-252.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15201436Dunnick JK, Lieuallen W, Moyer C, Orzech D, Nyska A. 2004. Cardiac damage in rodents after exposure to bis(2-chloroethoxy)methane. Toxicol Pathol 32:309-317.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15204972Elmore SA, Berridge BR, Boyle MC, Cora MC, Hoenerhoff MJ, Kooistra L, Laast VA, Morrison JP, Rao D, Rinke M, Yoshizawa K. 2012. Proceedings of the 2012 National Toxicology Program Satellite Symposium. Toxicol Pathol 41(2):151-180.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/23262640Jokinen MP, Boyle M, Lieuallen WG, Johnson CL, Malarkey DE, Nyska A. 2011. Morphologic aspects of rodent cardiotoxicity in a retrospective evaluation of National Toxicology Program studies. Toxicol Pathol 39(5):850-860.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/21747121Jokinen MP, Lieuallen WG, Johnson CL, Dunnick J, Nyska A. 2005. Characterization of spontaneous and chemically induced cardiac lesions in rodent model systems: The National Toxicology Program experience. Cardiovasc Toxicol 5:227-244.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16046796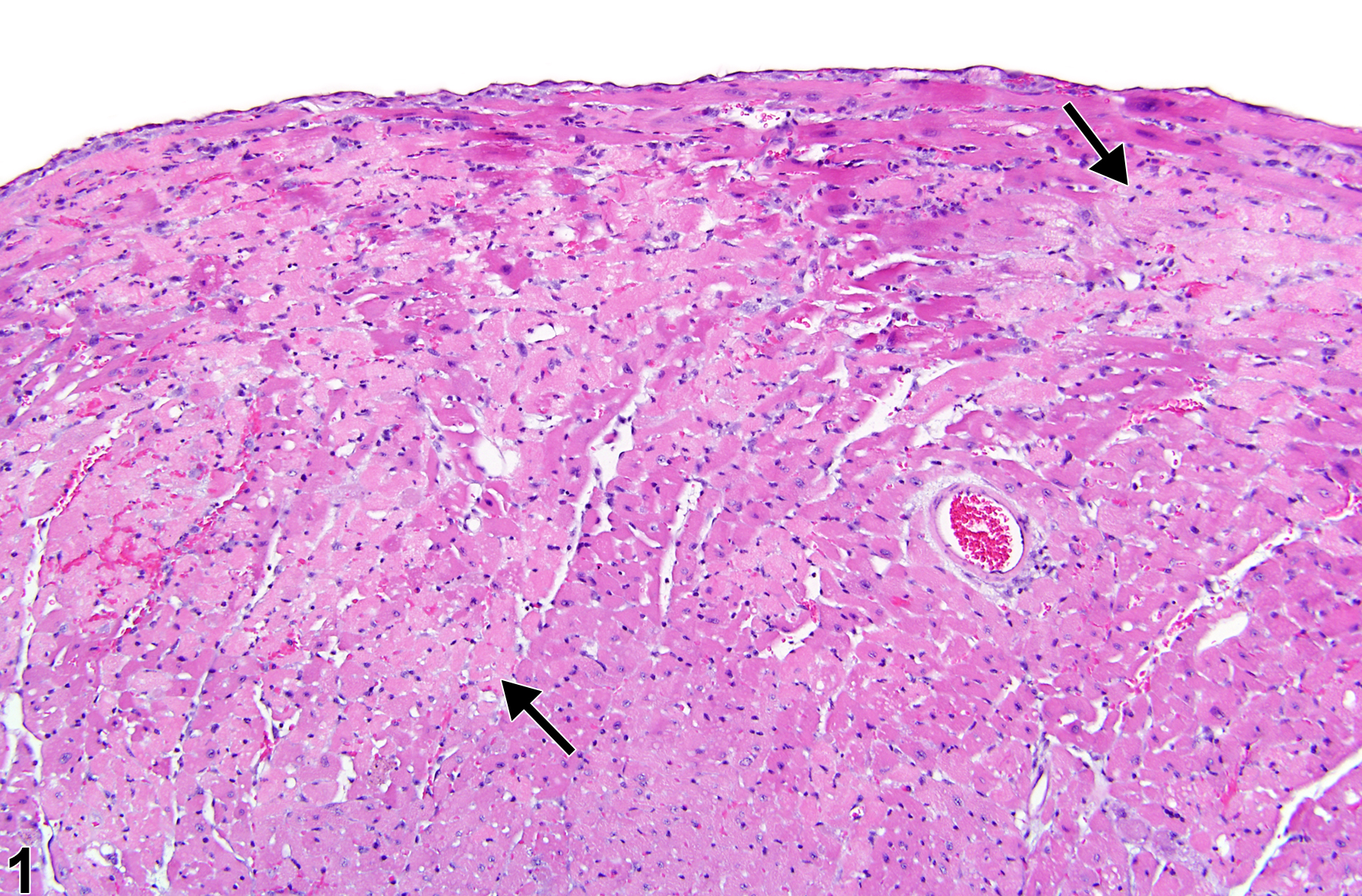
Heart, Myocardium - Necrosis in a male P16Ink4a/p19Arf (background: 80% C57BL/6, 19% 129/Sv, 1% SJL) mouse from an acute study. Myocardial necrosis (arrows) manifests as areas of pallor in the myocardium.