Cardiovascular System
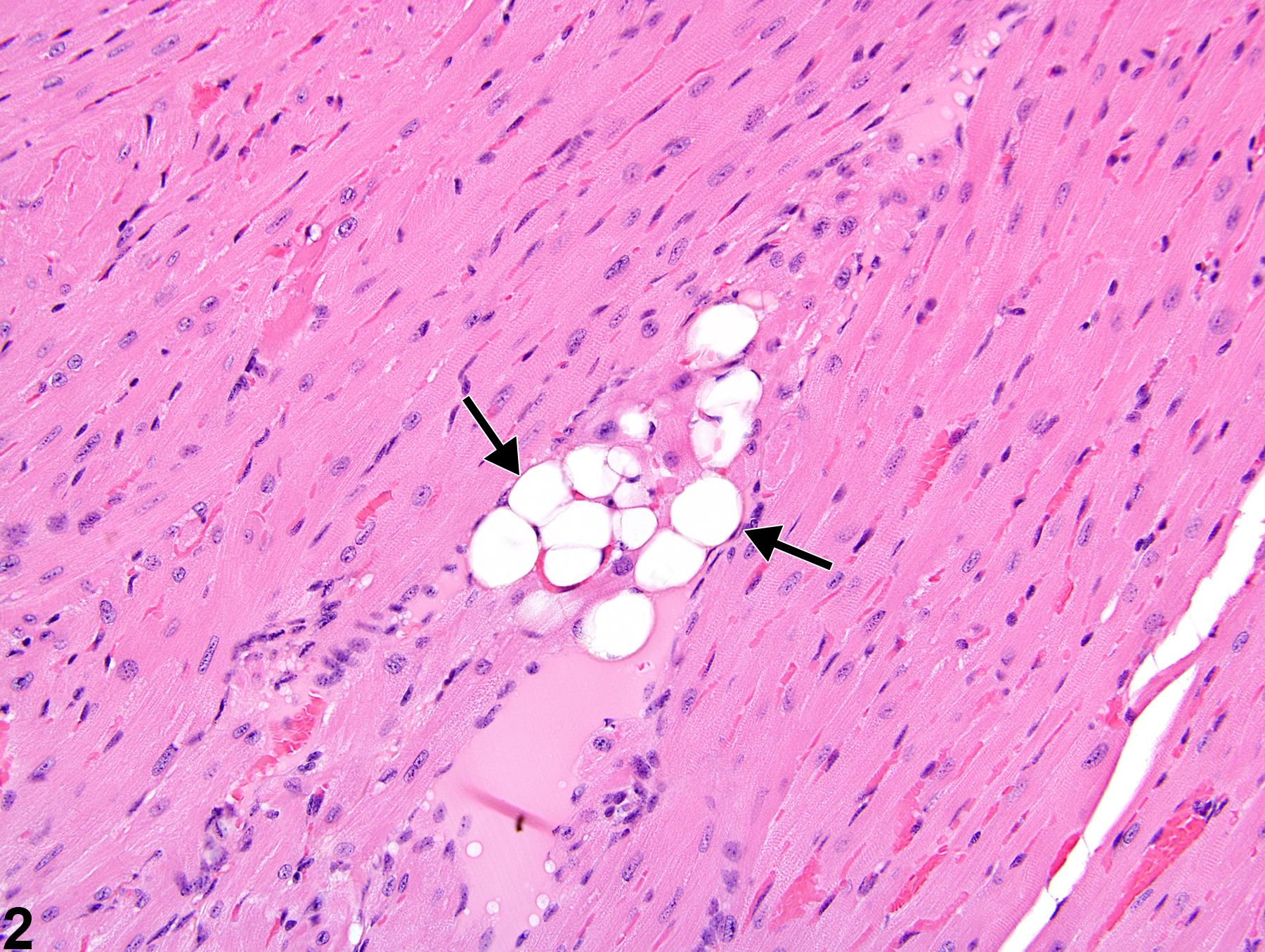
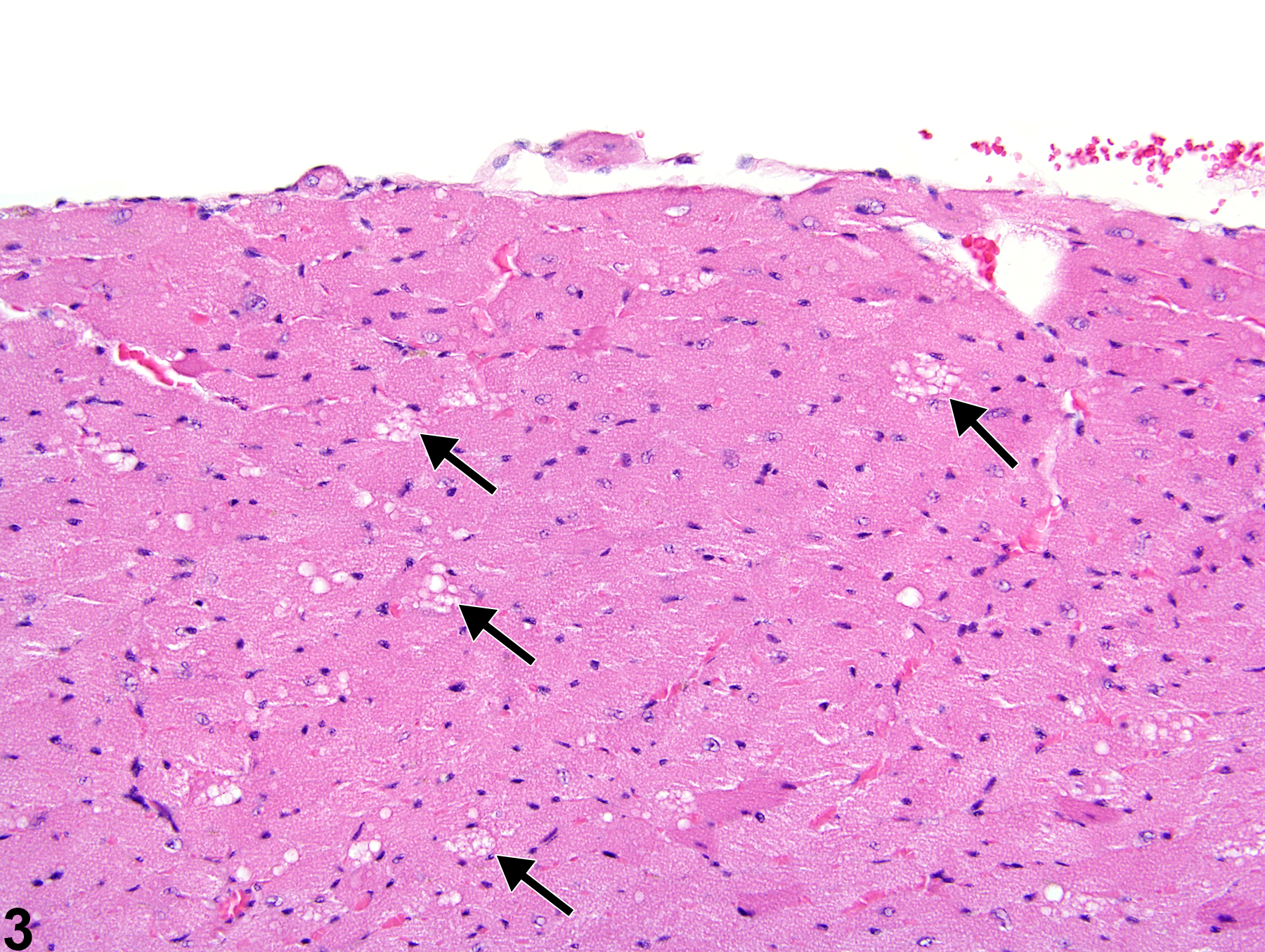
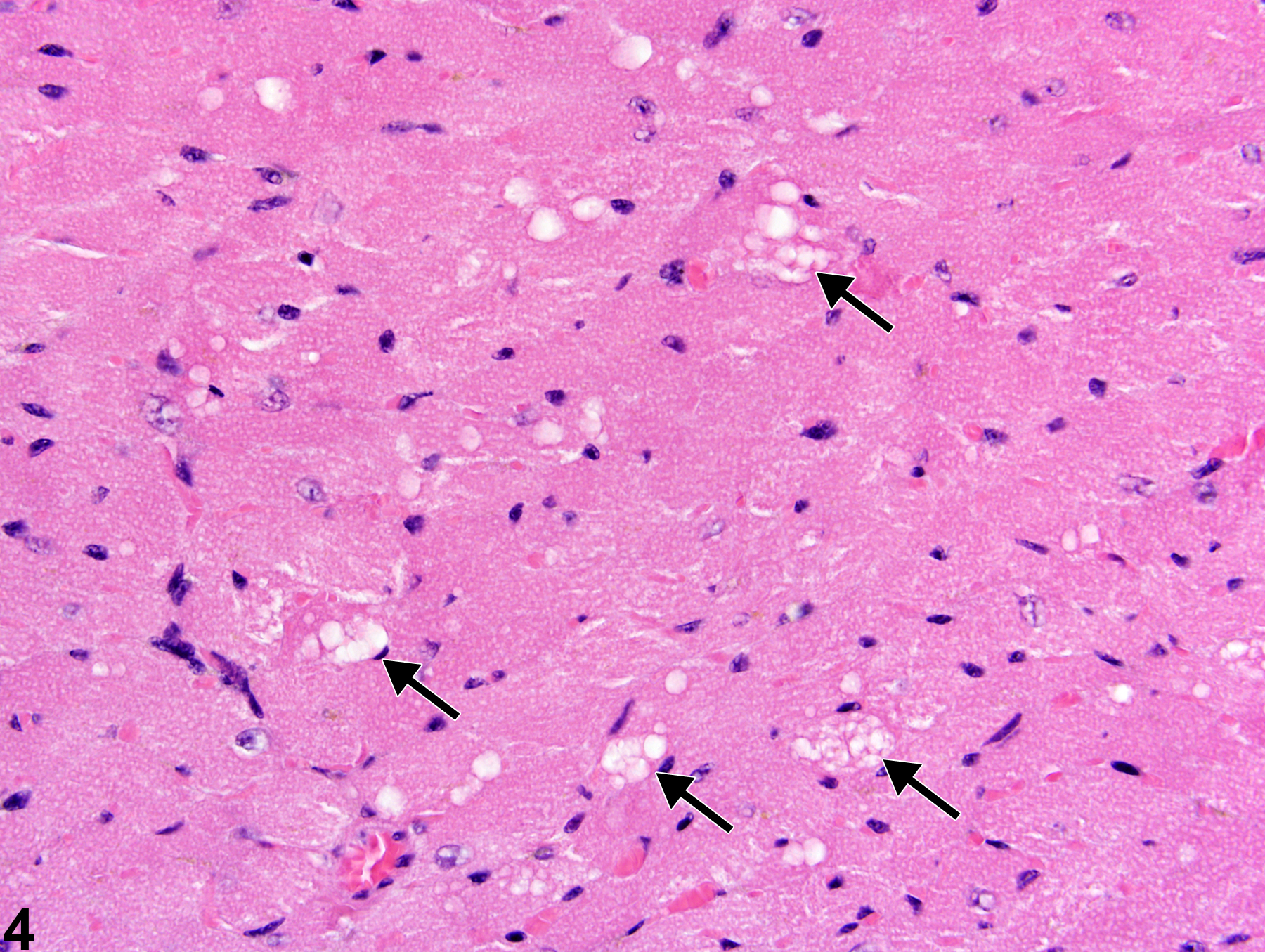
Heart, Myocardium - Vacuolation, Cytoplasmic
Narrative
Dunnick J, Johnson JA, Horton J, Nyska A. 2004. Bis(2-chloroethoxy)methane-induced mitochondrial and myofibrillar damage: Short-term time-course study. Toxicol Sci 81:243-252.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15201436Dunnick JK, Lieuallen W, Moyer C, Orzech D, Nyska A. 2004. Cardiac damage in rodents after exposure to bis(2-chloroethoxy)methane. Toxicol Pathol 32:309-317.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15204972Jokinen MP, Boyle M, Lieuallen WG, Johnson CL, Malarkey DE, Nyska A. 2011. Morphologic aspects of rodent cardiotoxicity in a retrospective evaluation of National Toxicology Program studies. Toxicol Pathol 39(5):850-860.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/21747121Jokinen MP, Lieuallen WG, Johnson CL, Dunnick J, Nyska A. 2005. Characterization of spontaneous and chemically induced cardiac lesions in rodent model systems: The National Toxicology Program experience. Cardiovasc Toxicol 5:227-244.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16046796Rahman A, Kessler A, More N, Sikic B, Rowden G, Woolley P, Schein PS. 1980. Liposomal protection of adriamycin-induced cardiotoxicity in mice. Cancer Res 40(5):1532-1537.
Abstract: http://cancerres.aacrjournals.org/content/40/5/1532Schmitz FJ, Rosen P, Reinauer H. 1995. Improvement of myocardial function and metabolism in diabetic rats by the carnitine palmitoyl transferase inhibitor Etomoxir. Horm Metab Res 27(12):515-522.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/8750778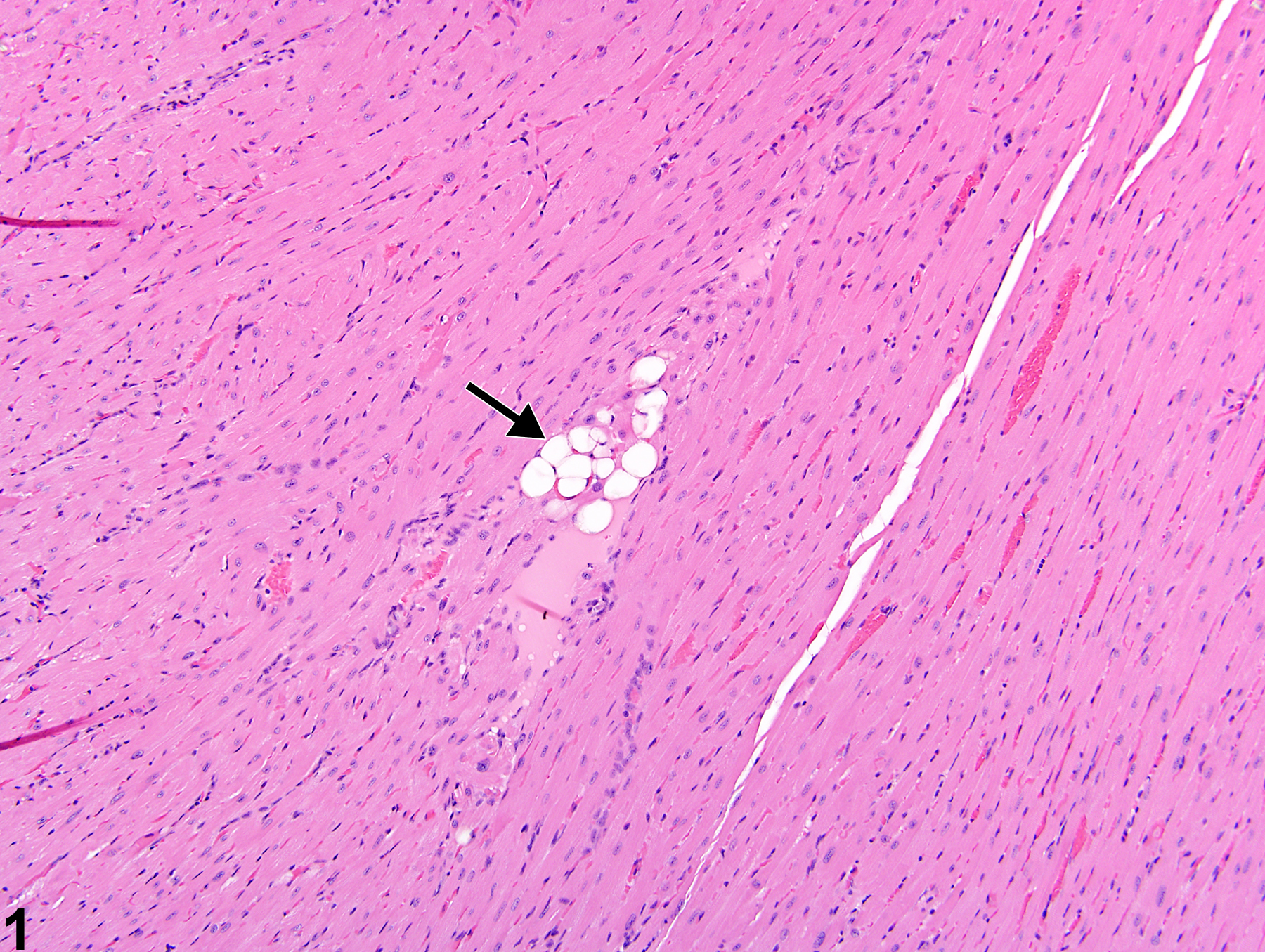
Heart, Myocardium - Vacuolation, Cytoplasmic in a female B6C3F1/N mouse from a subchronic study. A focal area of cytoplasmic vacuolation of myofibers is present (arrow).