Endocrine System
Adrenal Gland - Amyloid
Narrative
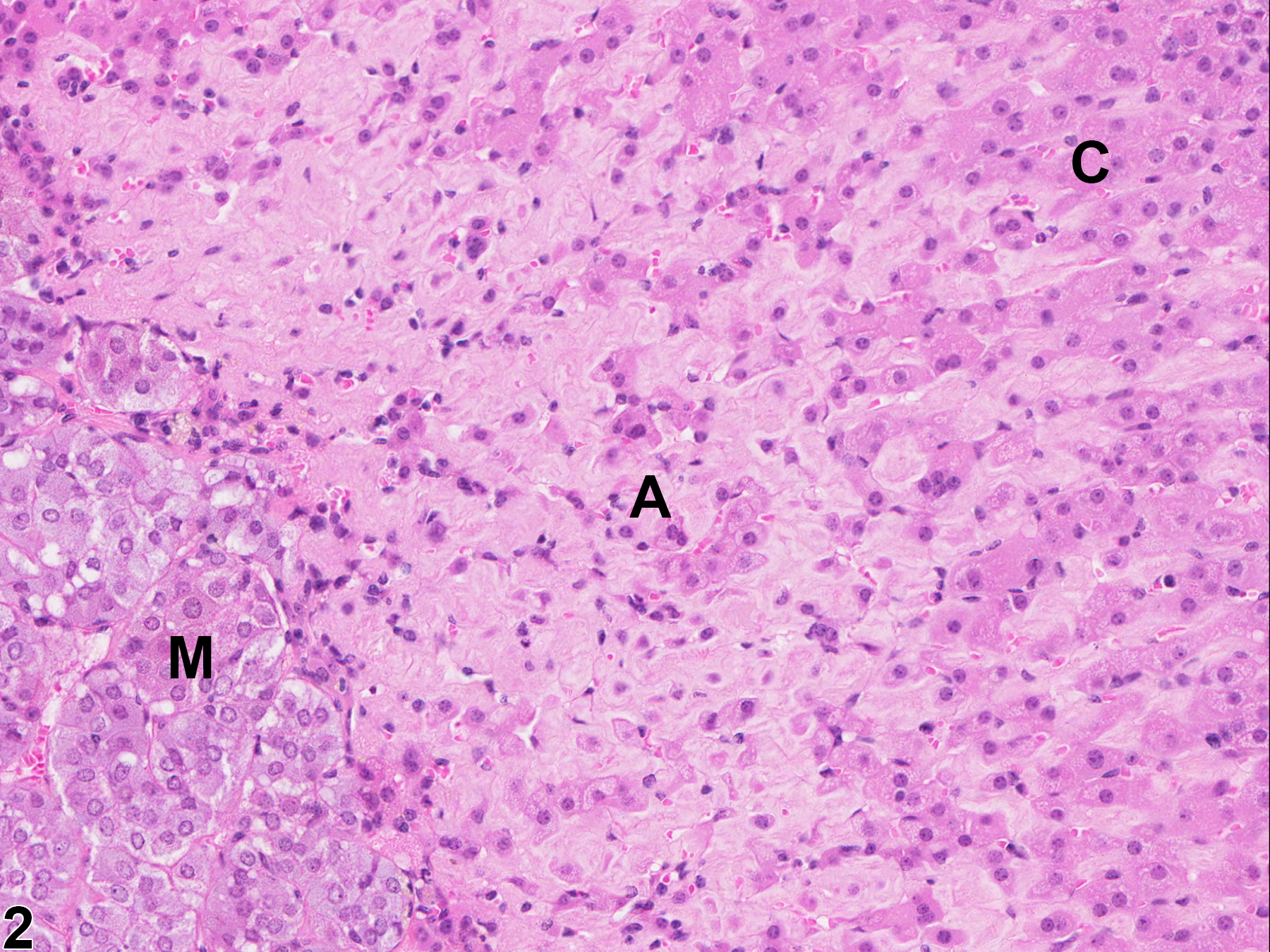
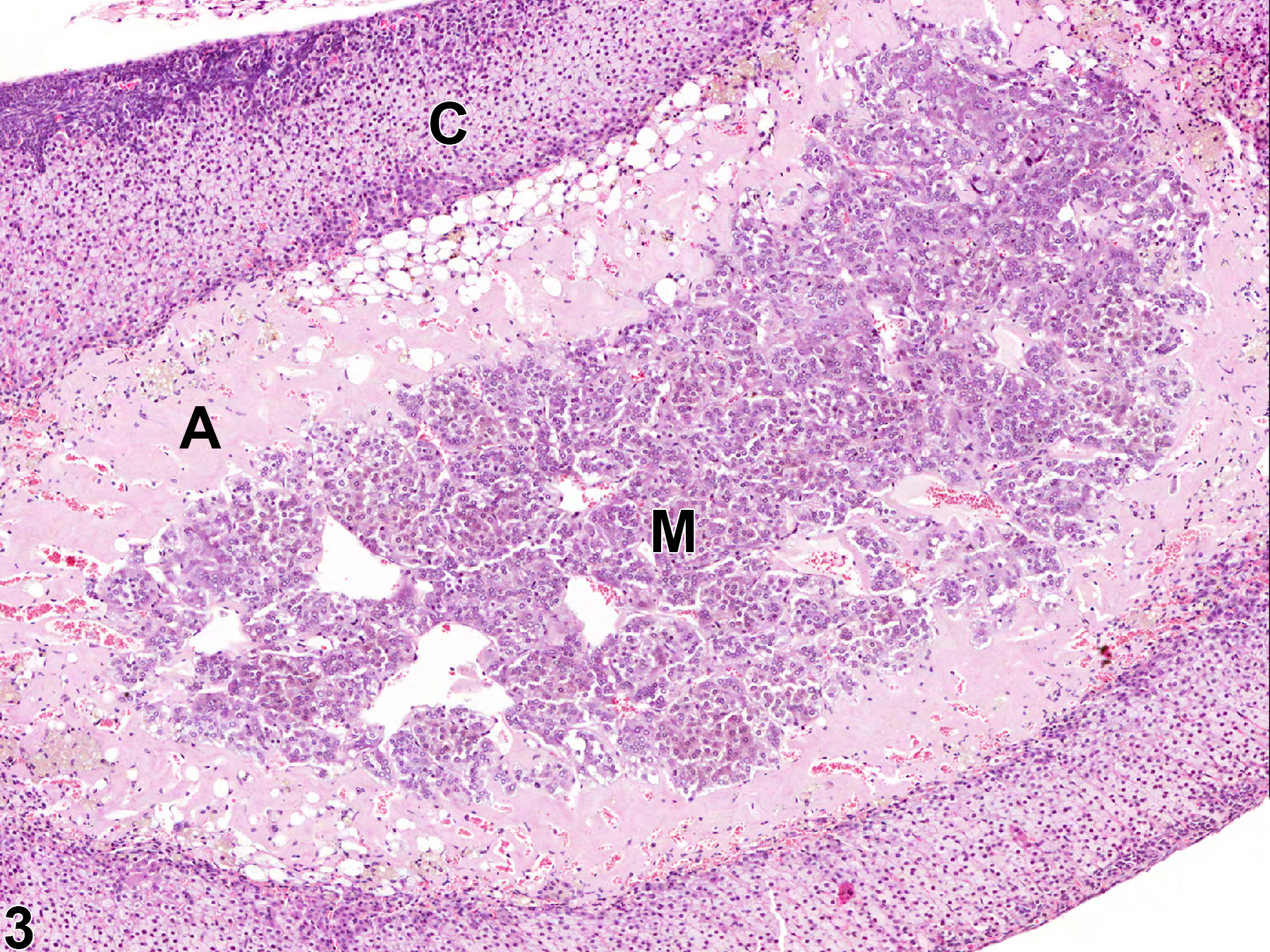
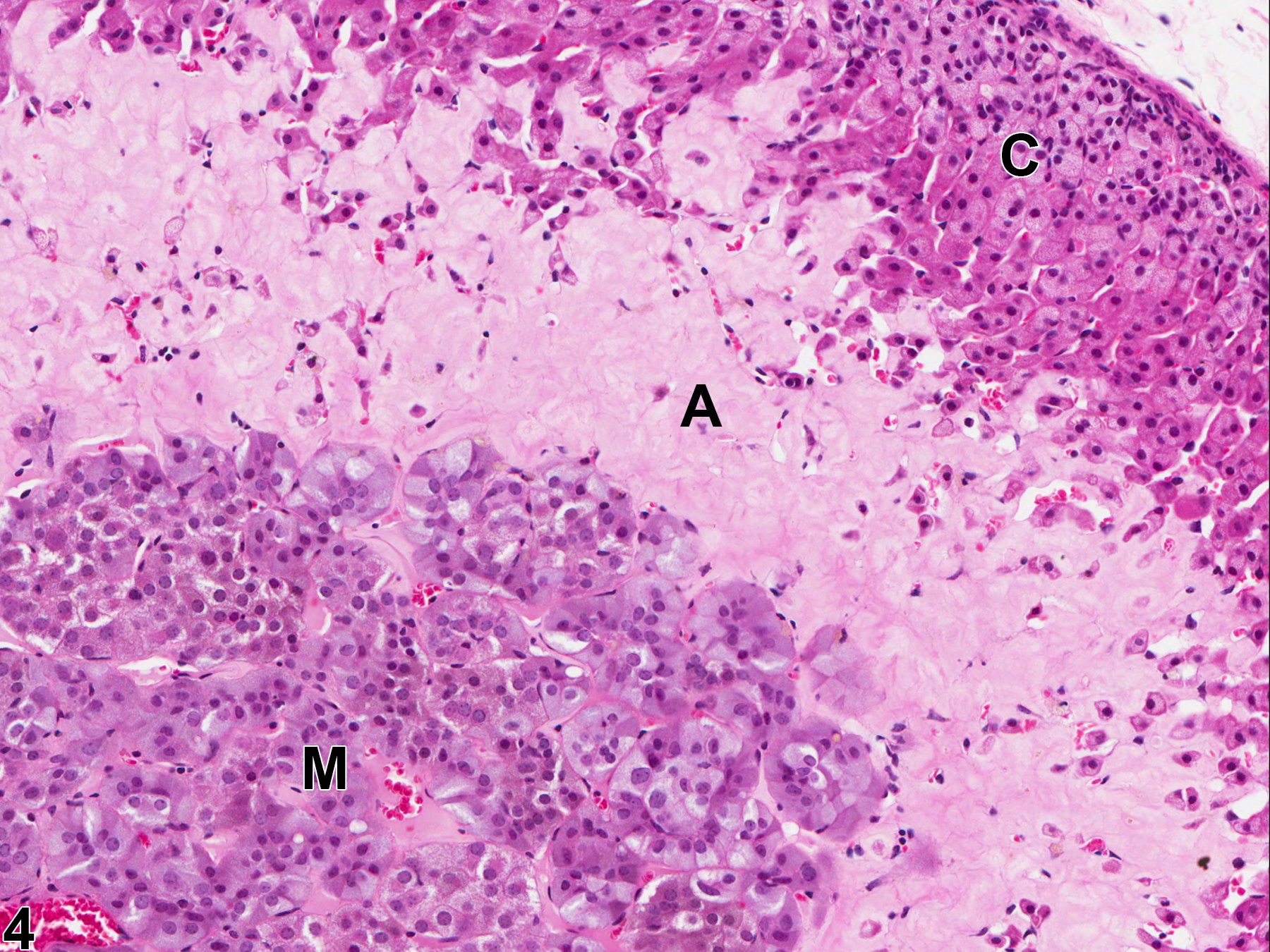
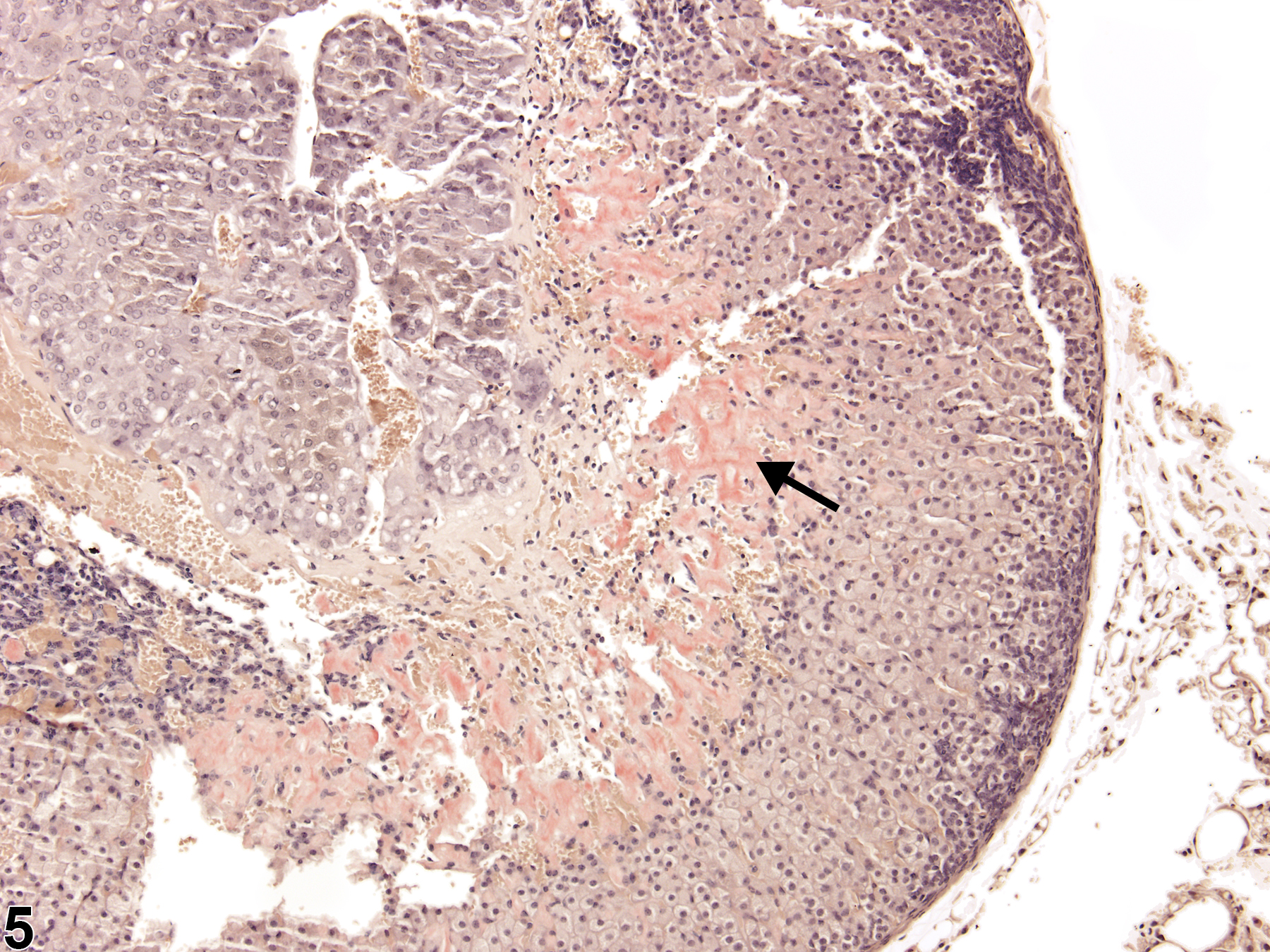
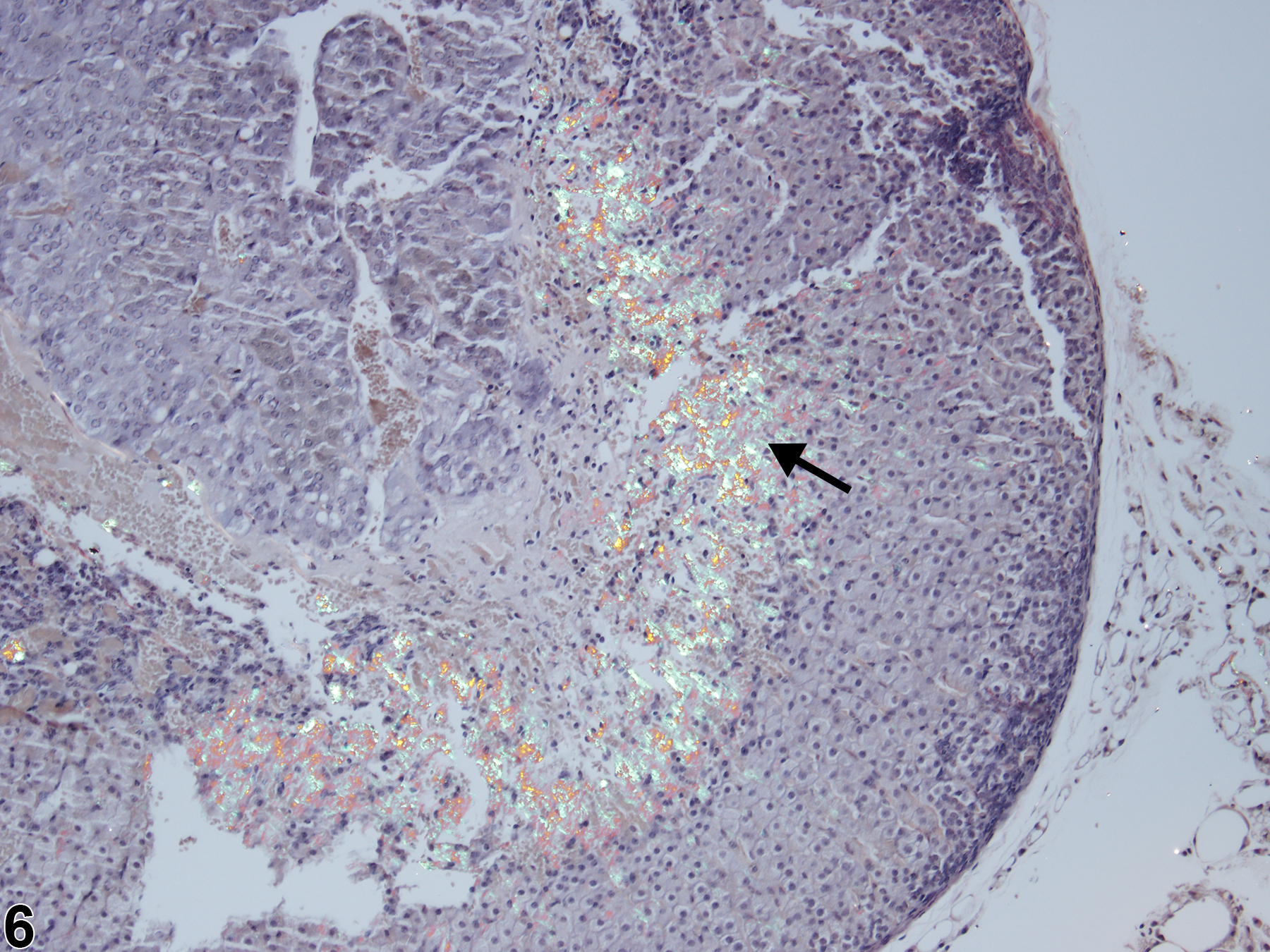
With hematoxylin and eosin (H&E) stain, amyloid deposits appear as homogeneous, amorphous, pale eosinophilic material (Figure 1, Figure 2, Figure 3, and Figure 4). Special histochemical stains are often used to distinguish amyloid from fibrous connective tissue, fibrin, and other substances. One of the most widely used stains is Congo red. Amyloid stained with Congo red appears pale to dull red under nonpolarized light (Figure 5) but exhibits brilliant birefringence (usually "apple green") under polarized light (Figure 6). Other methods for amyloid detection include fluorescent or metachromatic stains and immunohistochemistry.
Brayton CF, Treuting PM, Ward JM. 2012. Pathobiology of aging mice and GEM: Background strains and experimental design. Vet Pathol 49:85-105.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/22215684Elghetany MT, Saleem A. 1988. Methods for staining amyloid in tissues: A review. Stain Technol 63:201-212.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/2464206Hamlin MH, Banas DA. 1990. Adrenal gland. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 501-518.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Howie AJ, Brewer DB, Howell D, Jones AP. 2008. Physical basis of colors seen in Congo red-stained amyloid in polarized light. Lab Invest 88:232-242.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/18166974National Toxicology Program. 1993. NTP TR-417. Toxicology and Carcinogenesis Studies of p-Nitrophenol (CAS No. 100-02-7) in Swiss Webster Mice (Dermal Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/7714National Toxicology Program. 2010. NTP TR-558. Toxicology and Carcinogenesis Studies of 3,3',4,4'-Tetrachloroazobenzene (TCAB) [CAS No. 14047-09-7] in Harlan Sprague-Dawley Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/33564National Toxicology Program. 2012. NTP TR-576. Toxicology and Carcinogenesis Studies of Trimethylpropane Triacrylate (Technical Grade) (CAS No. 15625-89-5) in F344/N Rats and B6C3F1/N Mice (Dermal Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/37176Nyska A, Maronpot RR. 1990. Adrenal gland. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 509-536.
Rosol TJ, Yarrington JT, Latendresse J, Capen CC. 2001. Adrenal gland: Structure, function, and mechanisms of toxicity. Toxicol Pathol 29:41-48.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11215683Sass B. 1983. Amyloidosis, adrenal, mouse. In: Monographs on the Pathology of Laboratory Animals: Endocrine System (Jones TC, Mohr U, Hunt RD, eds). Springer, Berlin, 57-59.
Abstract: http://www.springer.com/medicine/pathology/book/978-3-642-64649-2Yarrington JT. 1996. Adrenal cortex. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). International Life Sciences Press, Washington, DC, 124-133.
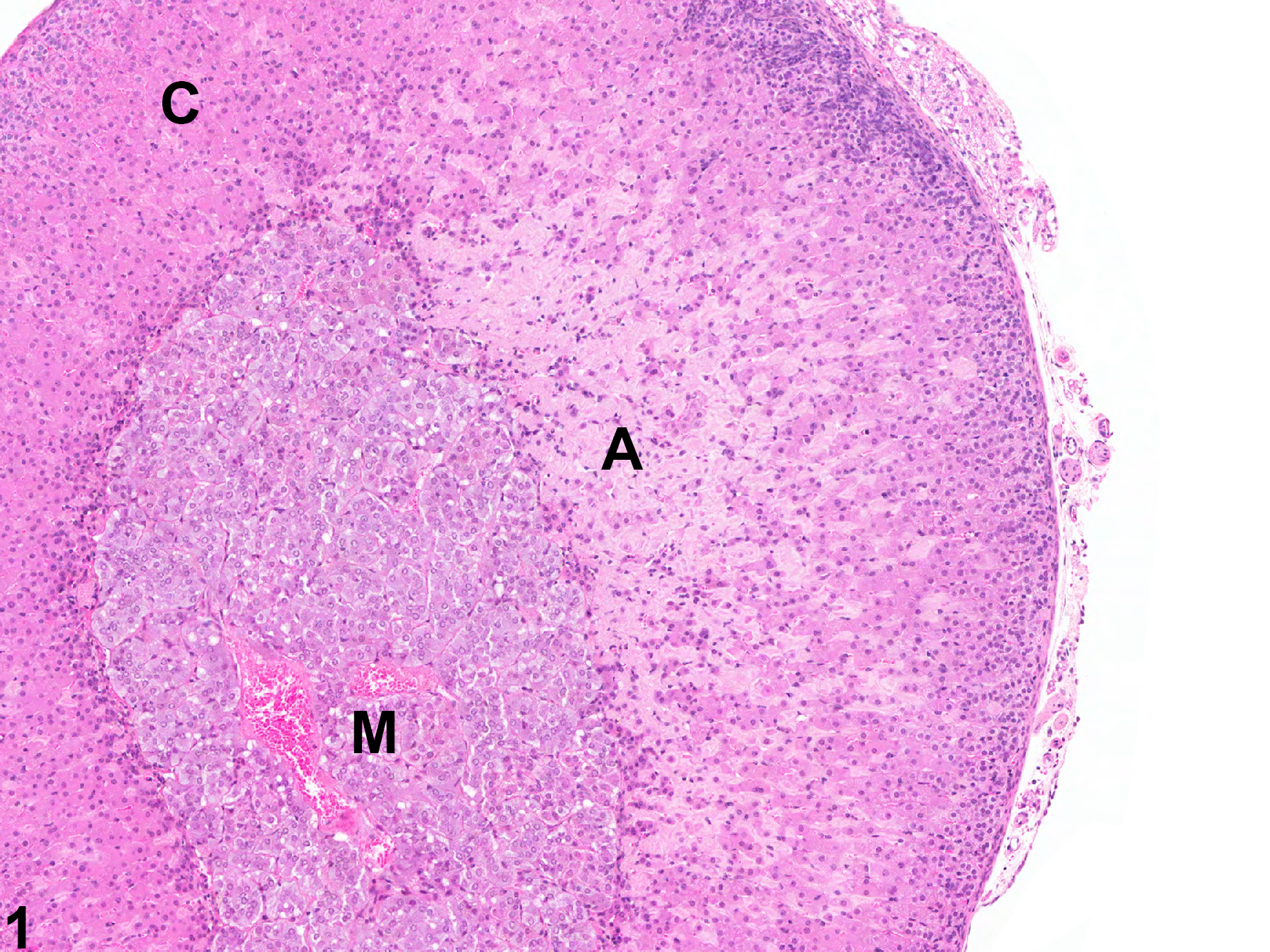
Adrenal gland, Cortex - Amyloid in a male B6C3F1/N mouse from a chronic study. There is homogeneous, amorphous, pale eosinophilic material (amyloid, A) expanding the zona fasciculata and zona reticularis; C = cortex, M = medulla.