Endocrine System
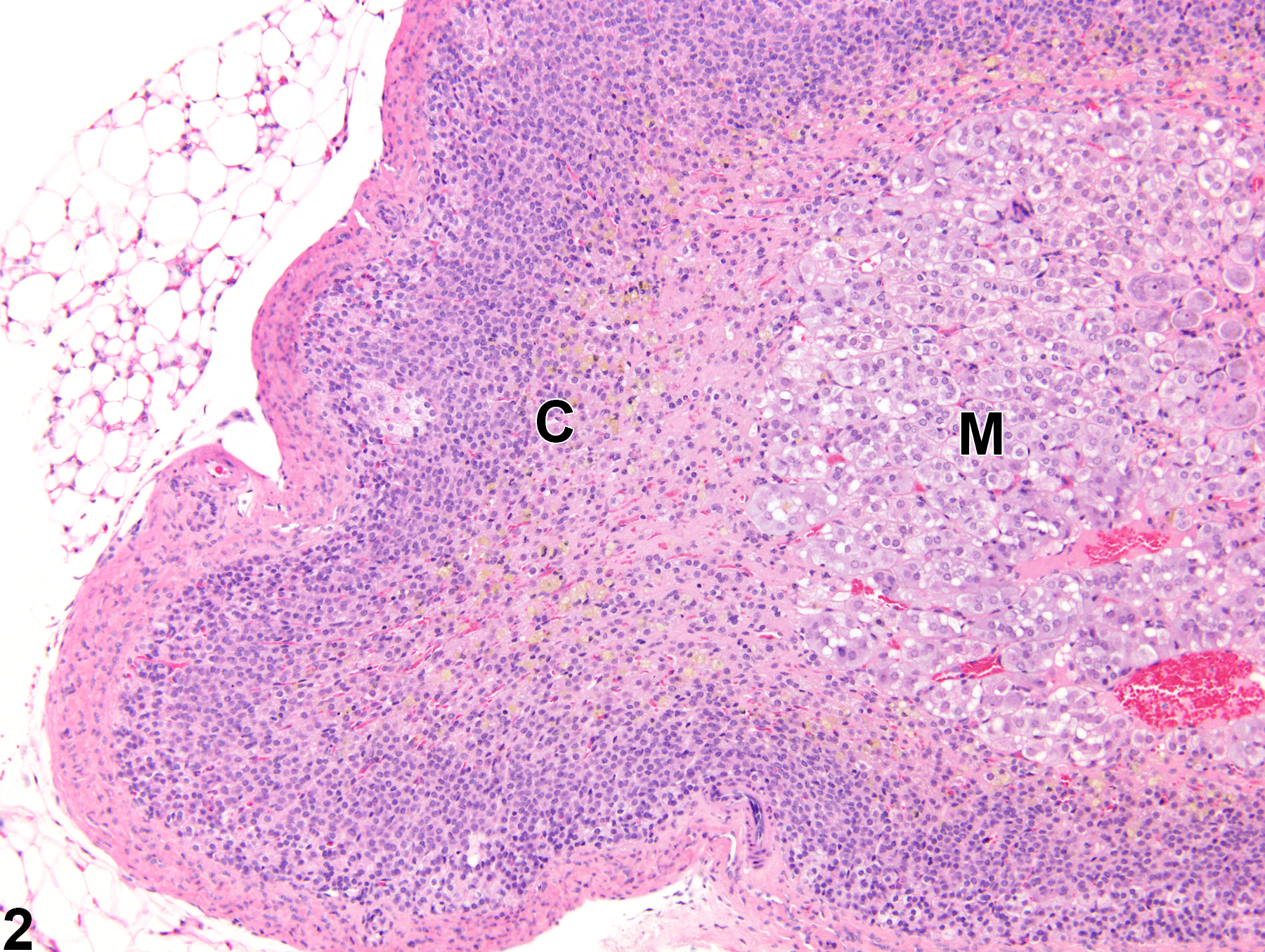
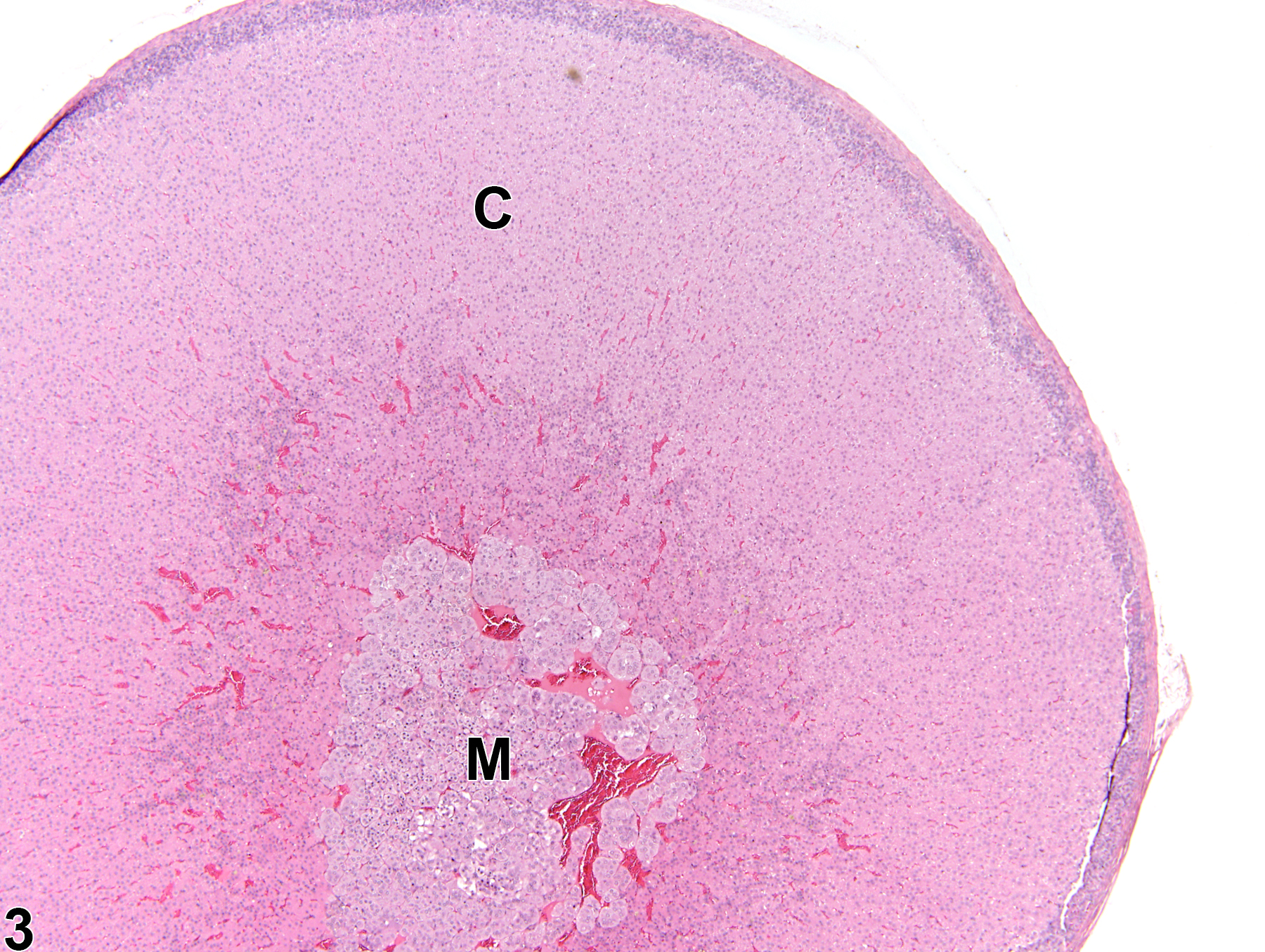
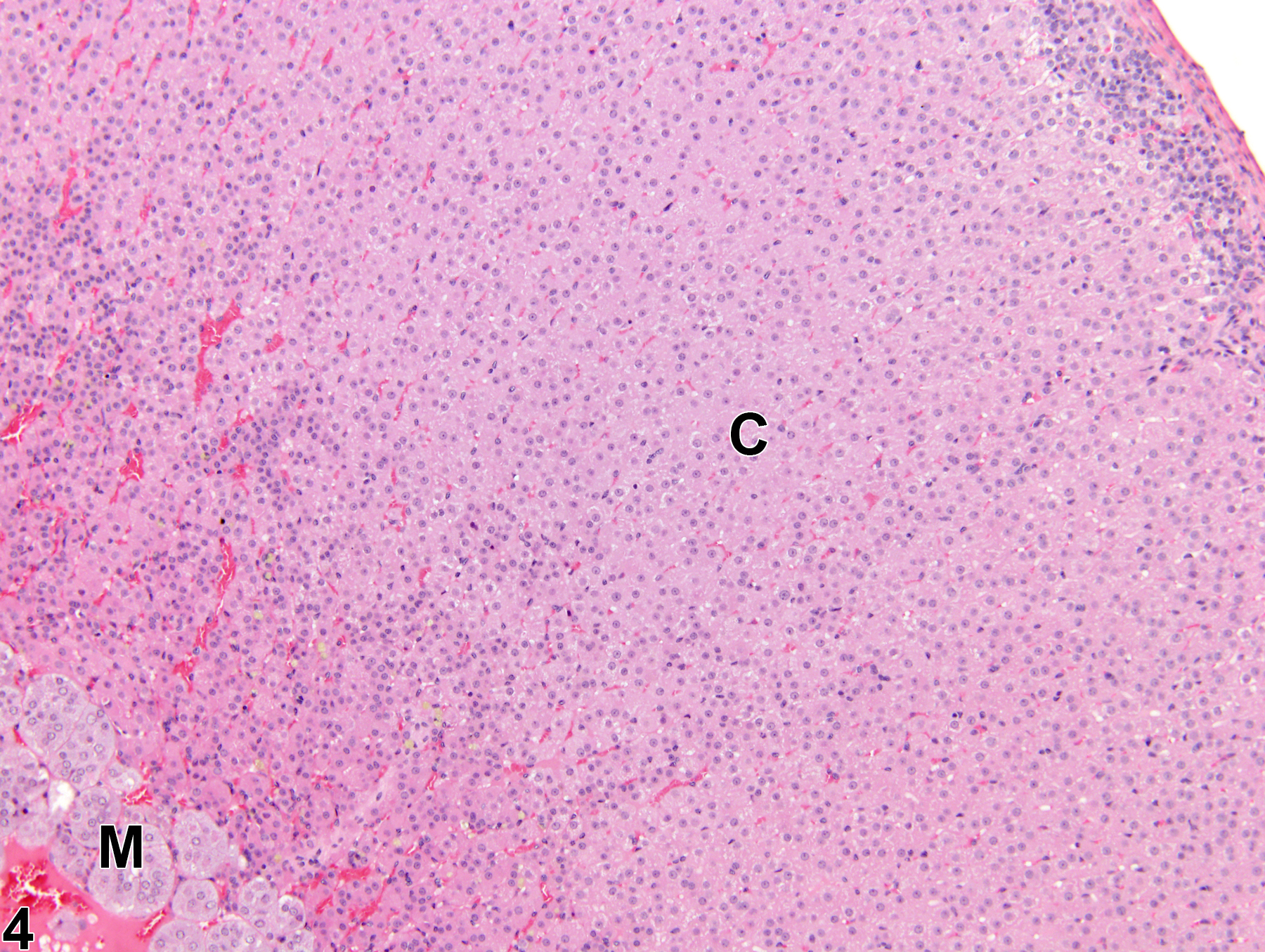
Adrenal Gland, Cortex - Atrophy
Narrative
Causes of adrenal cortical atrophy in rats and mice include any factor that increases levels of glucocorticoids, such as exogenous administration or endogenous hypersecretion by a functional adrenocortical neoplasm in the same or contralateral gland, which secondarily results in decreased adrenocorticotropic hormone (ACTH) levels. Atrophy can also result from more direct ACTH deficiency, such as may occur with abnormal pituitary function. Cortical atrophy can also be a direct effect of exogenous toxicants that interfere with normal adrenocortical steroidogenesis and/or the physiologic effects of the renin-angiotensin system on the adrenal. Because of the complex physiologic interactions of hypothalamus, pituitary, thyroid, and gonads with the adrenal gland, other exogenous toxicants and experimental manipulations that directly damage these tissues or modulate their secretory functions can result in secondary effects in the adrenal cortex, including atrophy. Cortical atrophy may also be a sequela to other pathologic processes, such as degeneration or necrosis, and may also be concurrent with cytoplasmic vacuolization or pigment accumulation.
Conran RM, Nickerson PA. 1982. Atrophy of the zona fasciculata in the adrenal cortex of thyroparathyroidectomized rats: A quantitative study. Am J Anat 164:133-143.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/6285687Dunn TB. 1970. Normal and pathologic anatomy of the adrenal gland of the mouse, including neoplasms. J Natl Cancer Inst 44:1323-1389.
Abstract: http://jnci.oxfordjournals.org/content/44/6/1323.abstractHamlin MH, Banas DA. 1990. Adrenal gland. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 501-518.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563McEwan PE, Lindop GB, Kenyon CJ. 1996. Control of cell proliferation in the rat adrenal gland in vivo by the renin-angiotensin system. Am J Physiol (Endocrinol Metab) 34:E192-E198.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/8760097Mirsky ML, Sivaraman L, Houle C, Potter DM, Chapin RE, Cappon GD. 2011. Histologic and cytologic detection of endocrine and reproductive tract effects of exemestane in female rats treated for up to twenty-eight days. Toxicol Pathol 39:589-605.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/21540174National Toxicology Program. 2010. NTP TR-559. Toxicology and Carcinogenesis Studies of 2,3',4,4',5-Pentachlorobiphenyl (PCB 118) (CAS No. 31508-00-6) in Female Harlan Sprague-Dawley Rats (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/33539Rosol TJ, Yarrington JT, Latendresse J, Capen CC. 2001. Adrenal gland: Structure, function, and mechanisms of toxicity. Toxicol Pathol 29:41-48.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11215683Stachiwiak A, Macchi C, Nussdorfer GG, Malendowicz LK. 1995. Effects of oxytocin on the function and morphology of the rat adrenal cortex: In vitro and in vivo investigations. Res Exp Med 195:265-274.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/8578002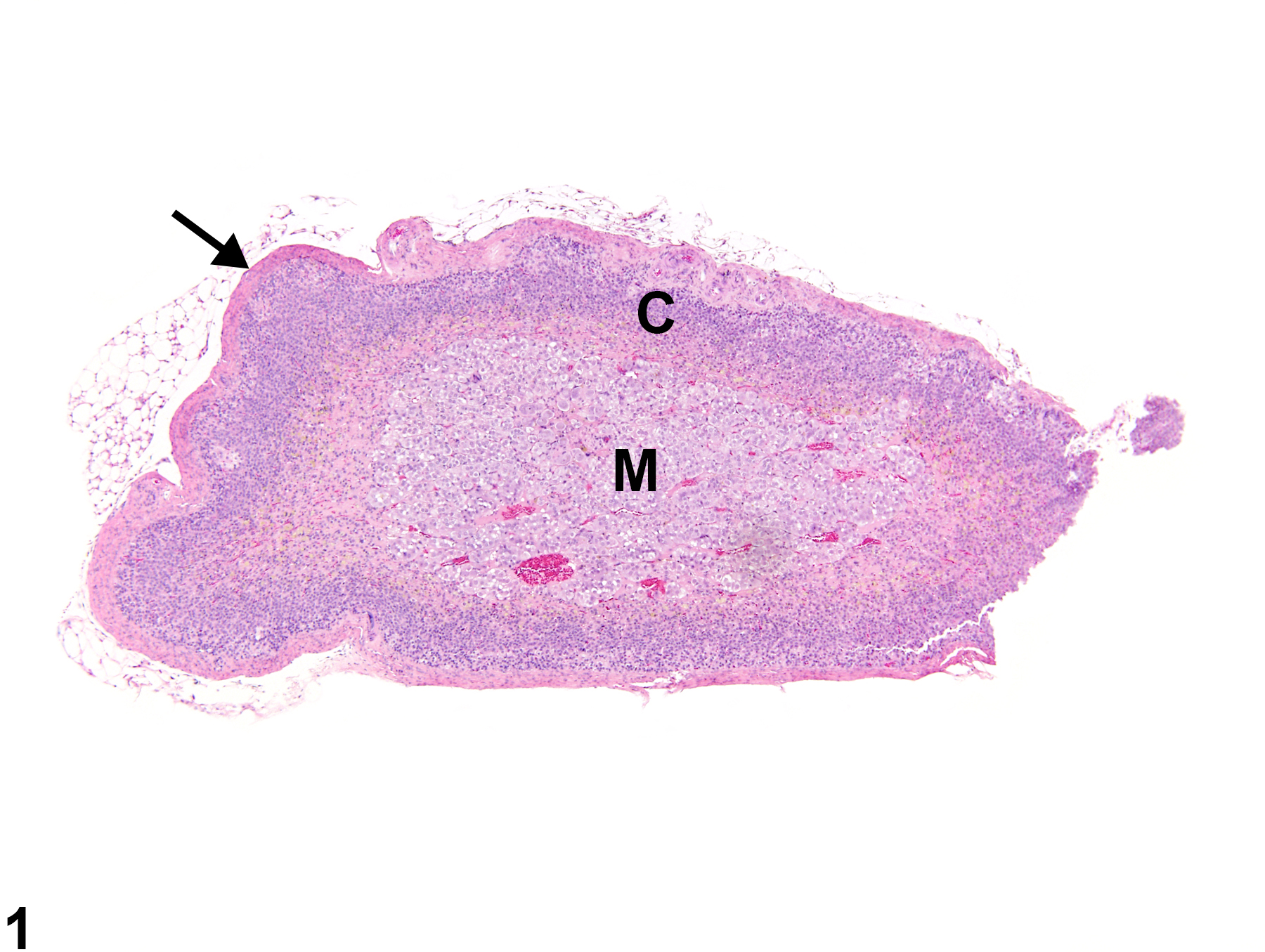
Adrenal gland, Cortex - Atrophy in a female Sprague-Dawley rat from a chronic study. The width of the cortex (C) and the overall size of the gland are decreased; the capsule (arrow) is thickened by fibrosis. M = medulla.