Endocrine System
Adrenal Gland - Pigment
Narrative
Lipofuscin refers to heterogeneous mixtures of intracytoplasmic, usually intralysosomal, nondegradable residues of lipid and protein oxidation/polymerization (as well as other moieties, such as advanced glycosylated end products) that accumulate in various somatic cells due to aging or other factors. Excessive and/or temporally inappropriate adrenal lipofuscin pigment aggregations (compared with age-matched controls) can also be a feature of cortical cell degeneration and/or atrophy (due to various causes) or can result from dietary imbalances (e.g., vitamin E deficiency) or administration of certain exogenous chemicals.
In mice, lipofuscin pigment-laden adrenal cortical cells occur most often as late-stage features or sequelae to physiologic regression of the X-zone. Thus, they are common incidental findings in aging mice. The pigment-laden cell aggregates are located primarily in the region adjacent to the corticomedullary junction formerly occupied by the X-zone. Milder, less temporally advanced cases consist of scattered small foci of pigment-laden cells, but as the animal ages the cell clusters often enlarge and can coalesce into circumferential bands surrounding the medulla. Macrophages containing similar lipofuscin and/or hemosiderin can be interspersed among the affected cortical cells.
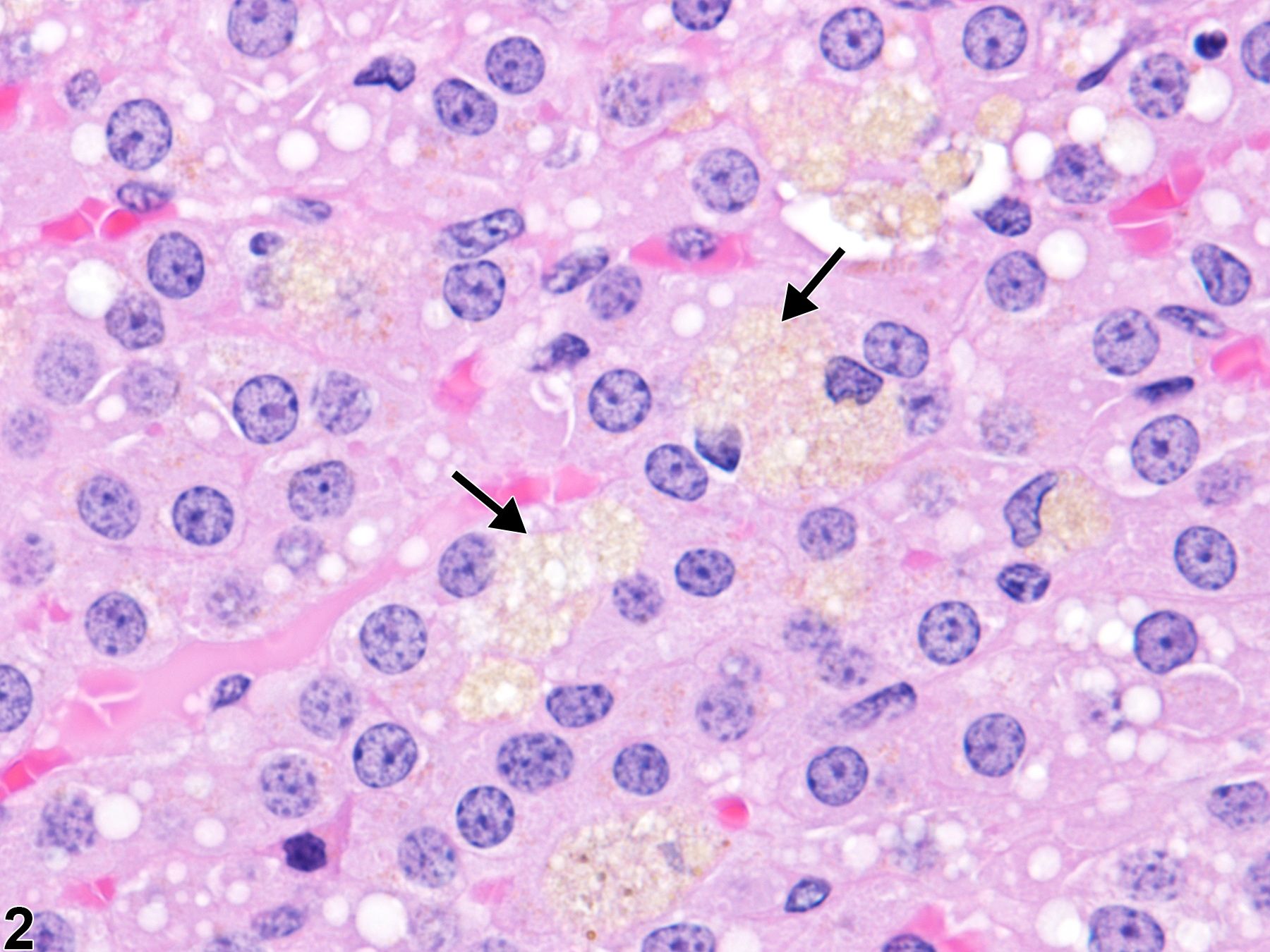
In rats, cells in the adrenal cortex containing lipofuscin pigment are also a common age-related incidental change. Even though rats do not have an X-zone, the pigment-laden cells tend to be most common in the inner cortex, usually as single cells or very small clusters (Figure 1 and Figure 2), unlike the large, prominent aggregates often seen in mice.
Dunn TB. 1970. Normal and pathologic anatomy of the adrenal gland of the mouse, including neoplasms. J Natl Cancer Inst 44:1323-1389.
Abstract: http://jnci.oxfordjournals.org/content/44/6/1323.abstractHadjiconstantinou M, Tijoe S, Alho H, Miller C, Neff NH. 1987. 1-Methyl-4-phenyl-1,2,3,6-tetrahydro-pyridine (MPTP) accelerates the accumulation of lipofuscin in mouse adrenal gland. Neurosci Lett 83:1-6.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/3502018Hamlin MH, Banas DA. 1990. Adrenal gland. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 501-518.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Moore NA, Callas G. 1975. Observations on the fine structure of propylthiouracil-induced "brown degeneration" in the zona reticularis of mouse adrenal cortex. Anat Rec 183:293-302.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/1239202National Toxicology Program. 2013. NTP TR-578. Toxicology and Carcinogenesis Studies of Gingko biloba Extract in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/37193Nyska A, Maronpot RR. 1990. Adrenal gland. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 509-536.
Parker GA, Valerio MG. 1983. Lipogenic pigmentation: Adrenal cortex, rat. In: Monographs on the Pathology of Laboratory Animals: Endocrine System (Jones TC, Mohr U, Hunt RD, eds). Springer, Berlin, 64-66.
Abstract: http://www.springer.com/medicine/pathology/book/978-3-642-64649-2Reichel W. 1968. Lipofuscin pigment accumulation and distribution in five rat organs as a function of age. J Gerontol 23:71-78.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/5689399Rosol TJ, Yarrington JT, Latendresse J, Capen CC. 2001. Adrenal gland: Structure, function, and mechanisms of toxicity. Toxicol Pathol 29:41-48.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/11215683Samorajski T, Ordy JM. 1967. The histochemistry and ultrastructure of lipid pigment in the adrenal glands of aging mice. J Gerontol 22:253-267.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/6028494Shimokawa I, Higami Y, Horiuchi S, Iwasaki M, Ikeda T. 1988. Advanced glycosylation end products in adrenal lipofuscin. J Gerontol 53A:B49-B51.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/9467422Terman A, Bruk UT. 2004. Lipofuscin. Intl J Biochem Cell Biol 36:1400-1404.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15147719Torrey CE, Wall HG, Campbell JA, Kwanyuen P, Hoivik DJ, Miller RT, Allen JS, Jayo MJ, Selinger K, Savina PM, Santostefano MJ. 2005. Evaluation of the carcinogenic potential of clofibrate in the FVB/Tg.AC mouse after oral administration: Part I. Int J Toxicol 24:313-325.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16257851Ward JM, Reznik-Schüller H. 1980. Morphological and histochemical characteristics of pigments in aging F344 rats. Vet Pathol 17:678-685.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/7423828Weglicki WB, Reichel W, Nair P. 1968. Accumulation of lipofuscin-like pigment in the rat adrenal gland as a function of vitamin E deficiency. J Gerontol 23:469-475.
Abstract: http://geronj.oxfordjournals.org/content/23/4/469.extract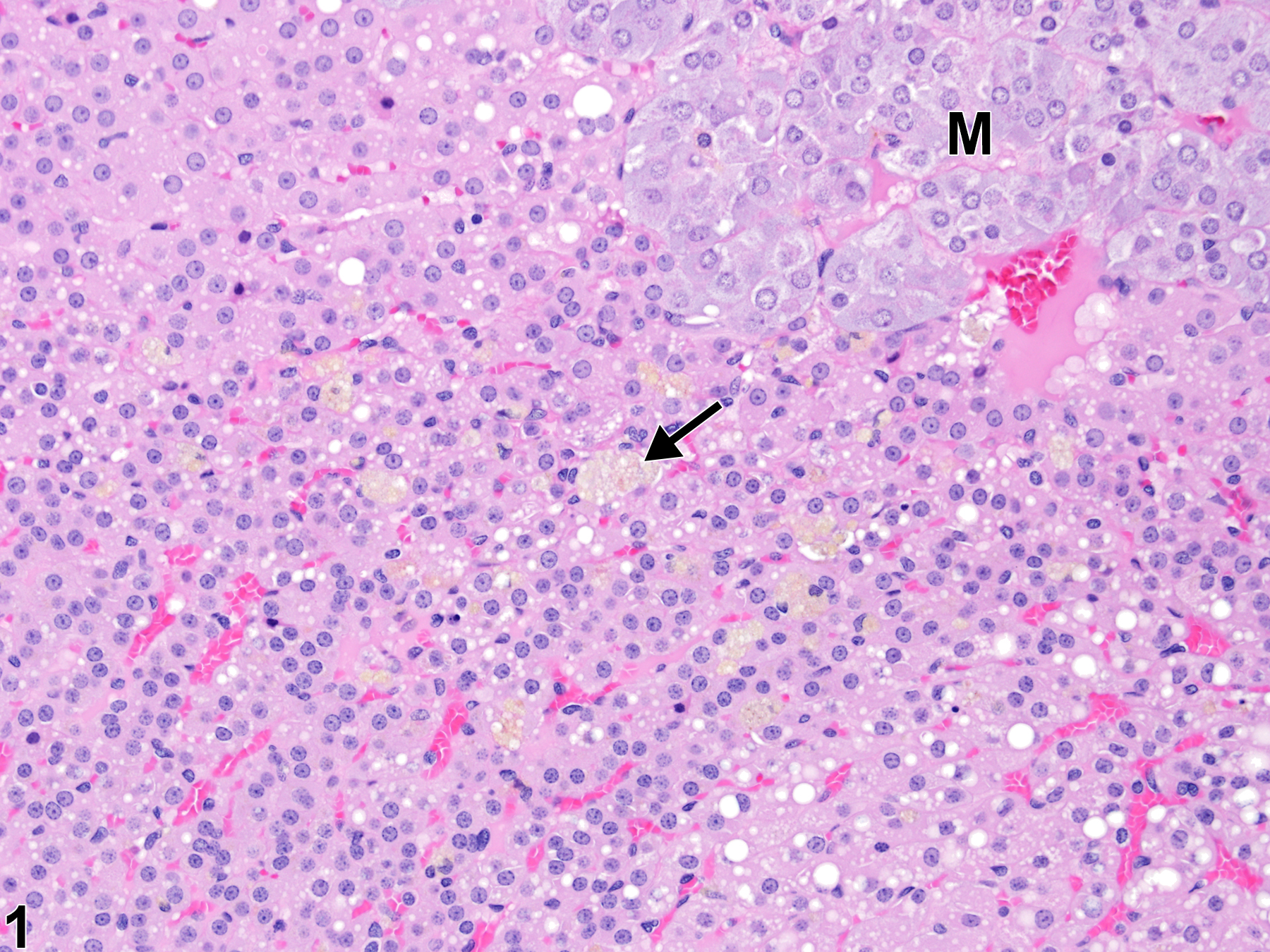
Adrenal gland, Cortex - Pigment in a male F344/N rat from a chronic study. There are scattered individual cells in the inner cortex (arrow) containing yellow-brown pigment. M = medulla.