Endocrine System
Pancreatic Islets - Hyperplasia
Narrative
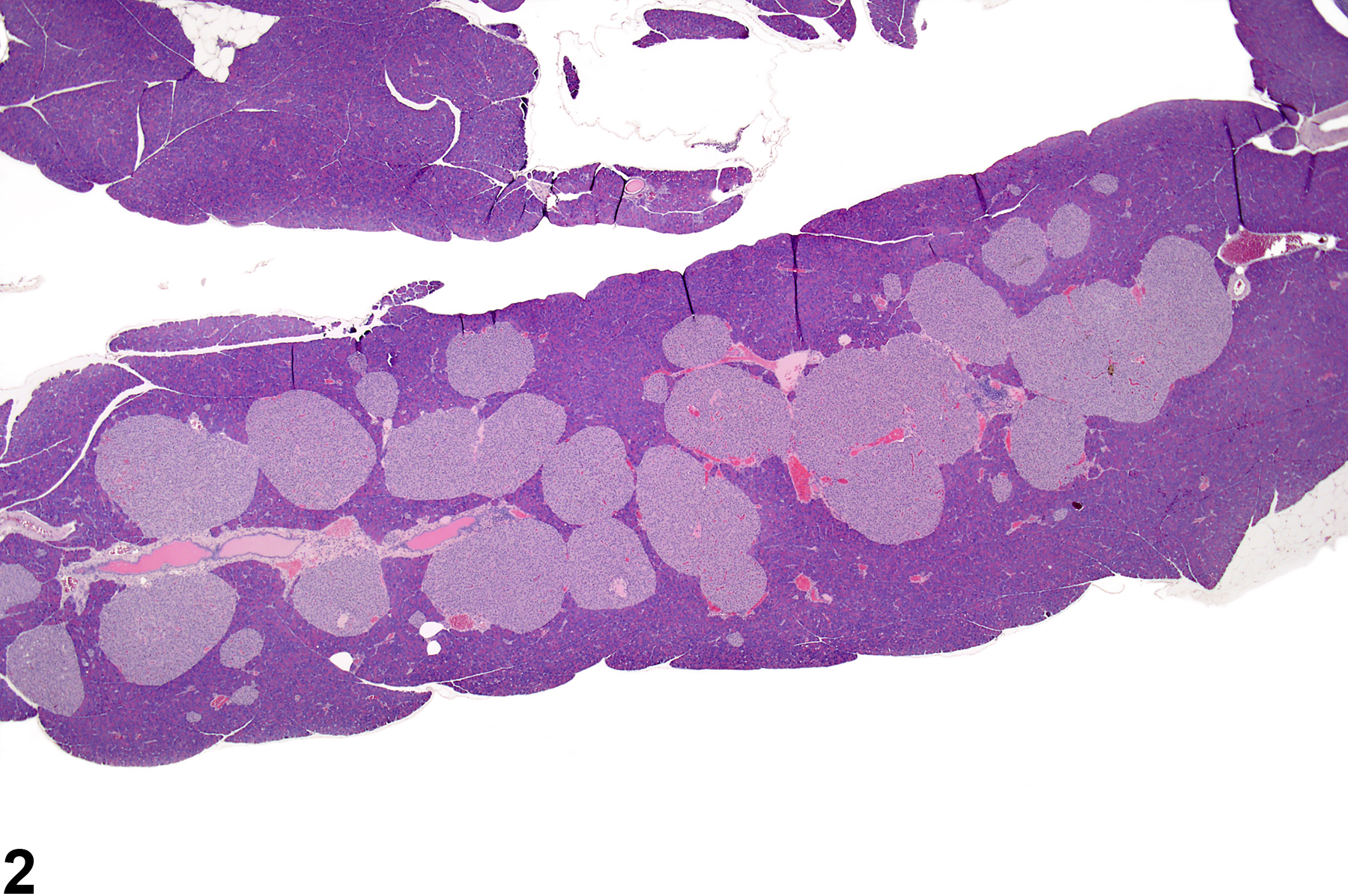
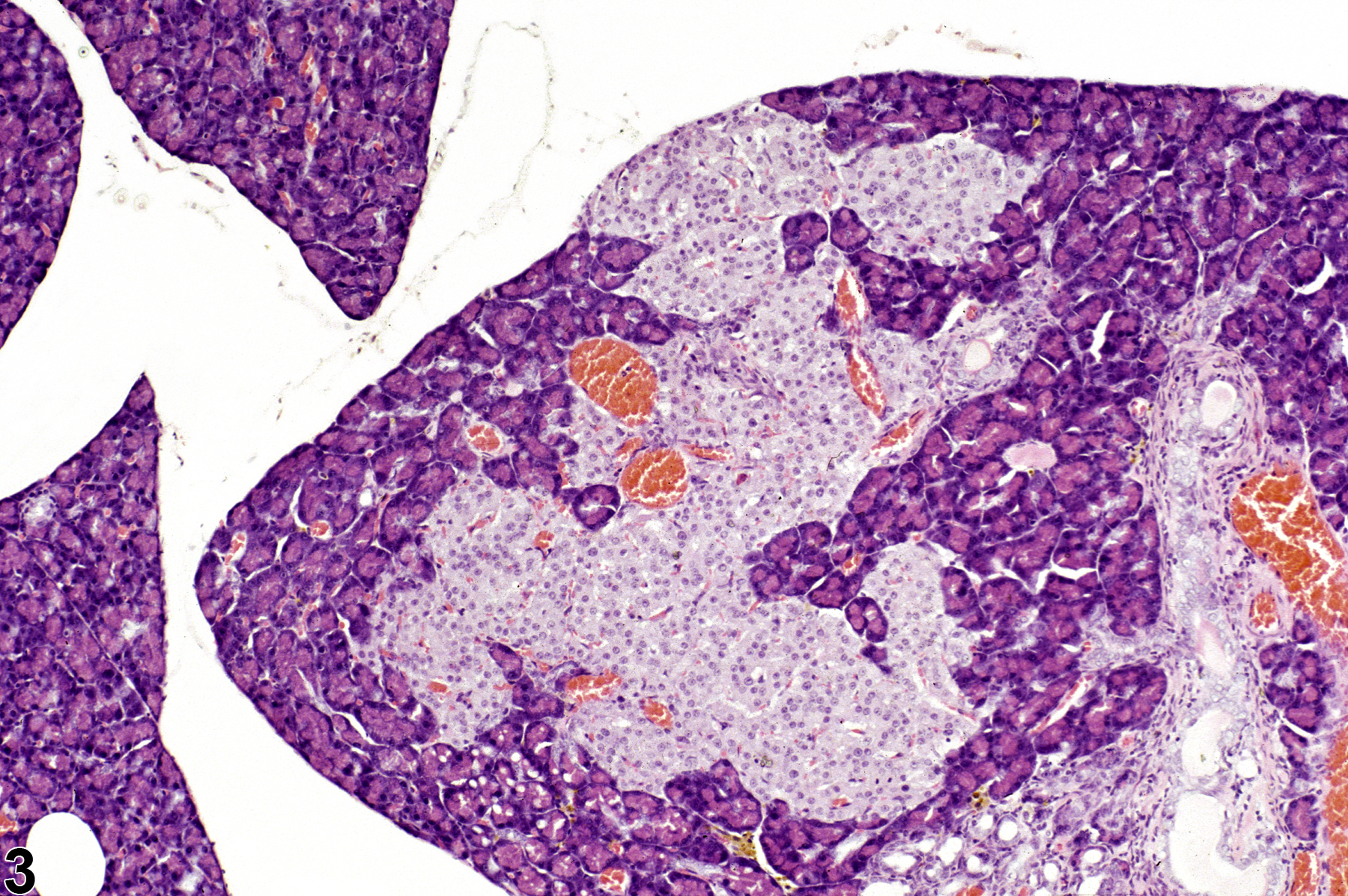
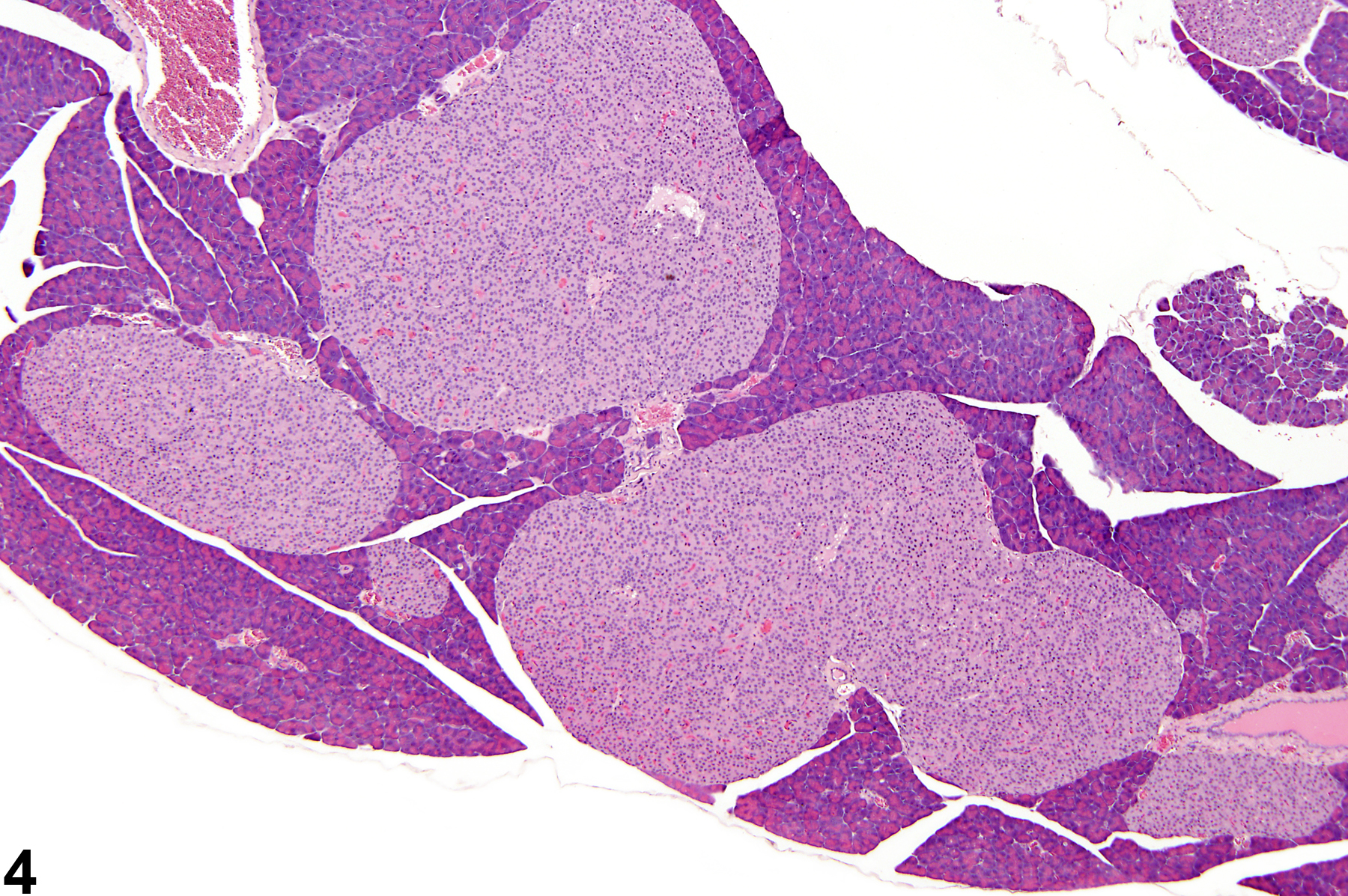
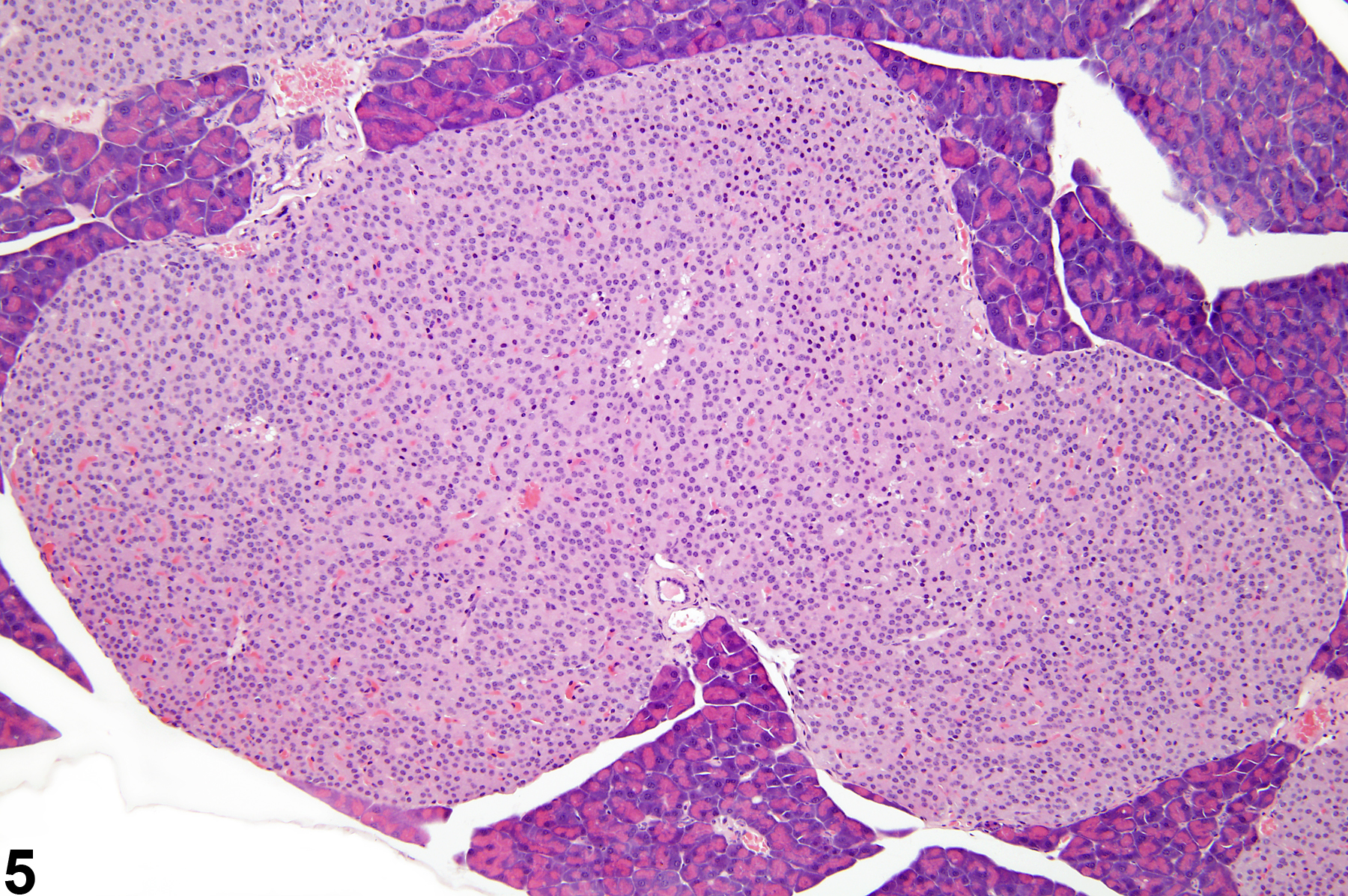
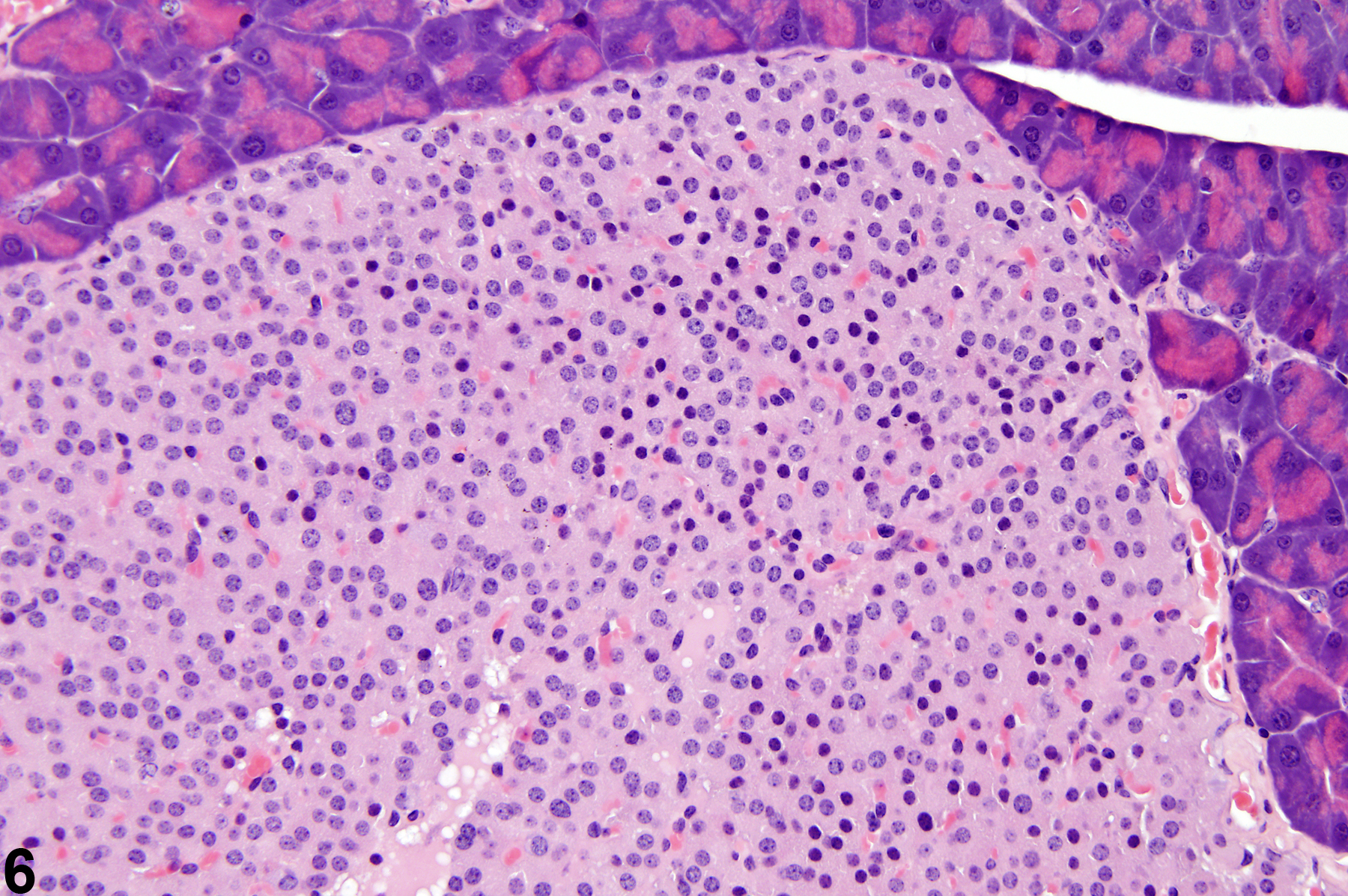
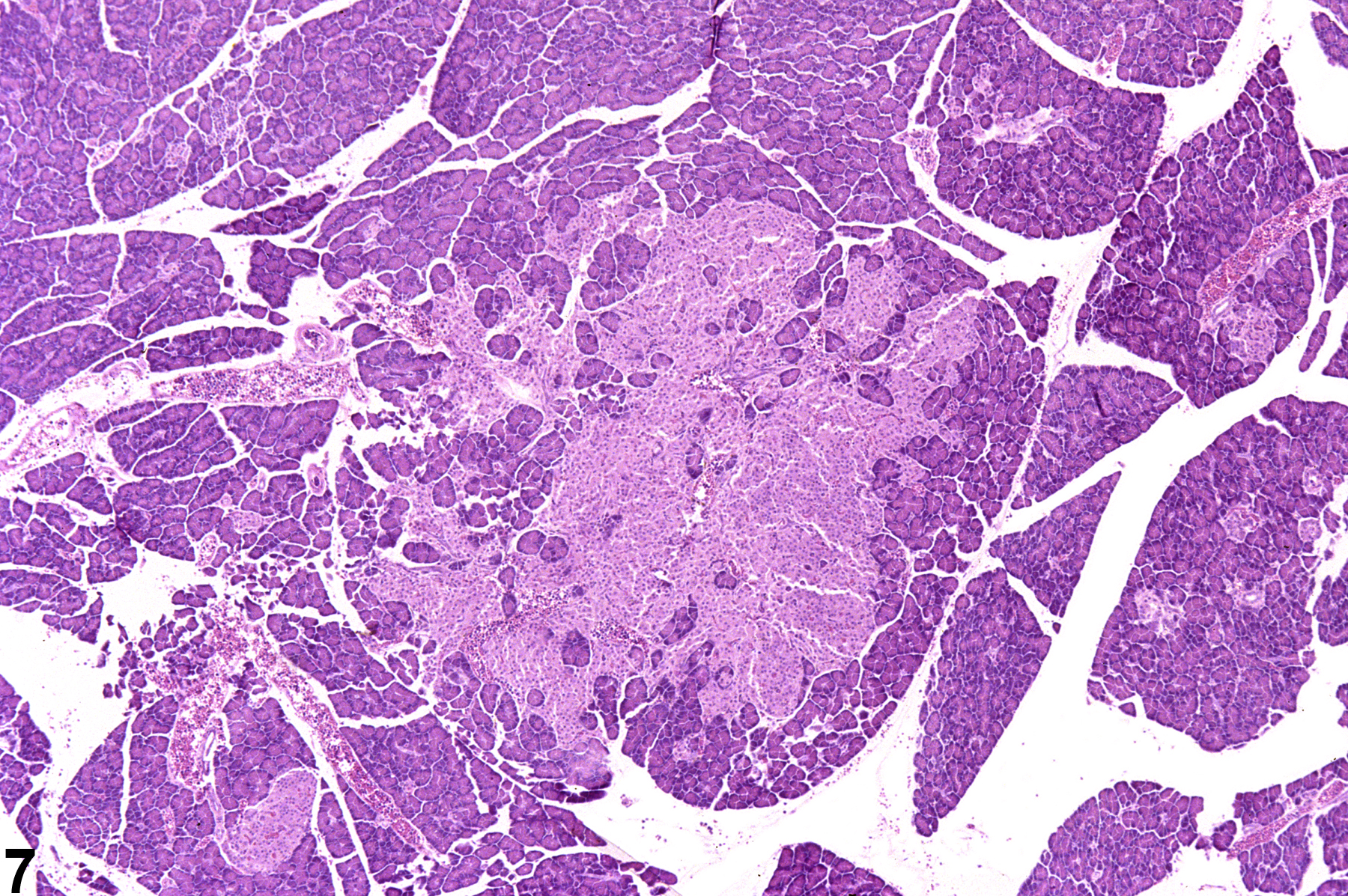
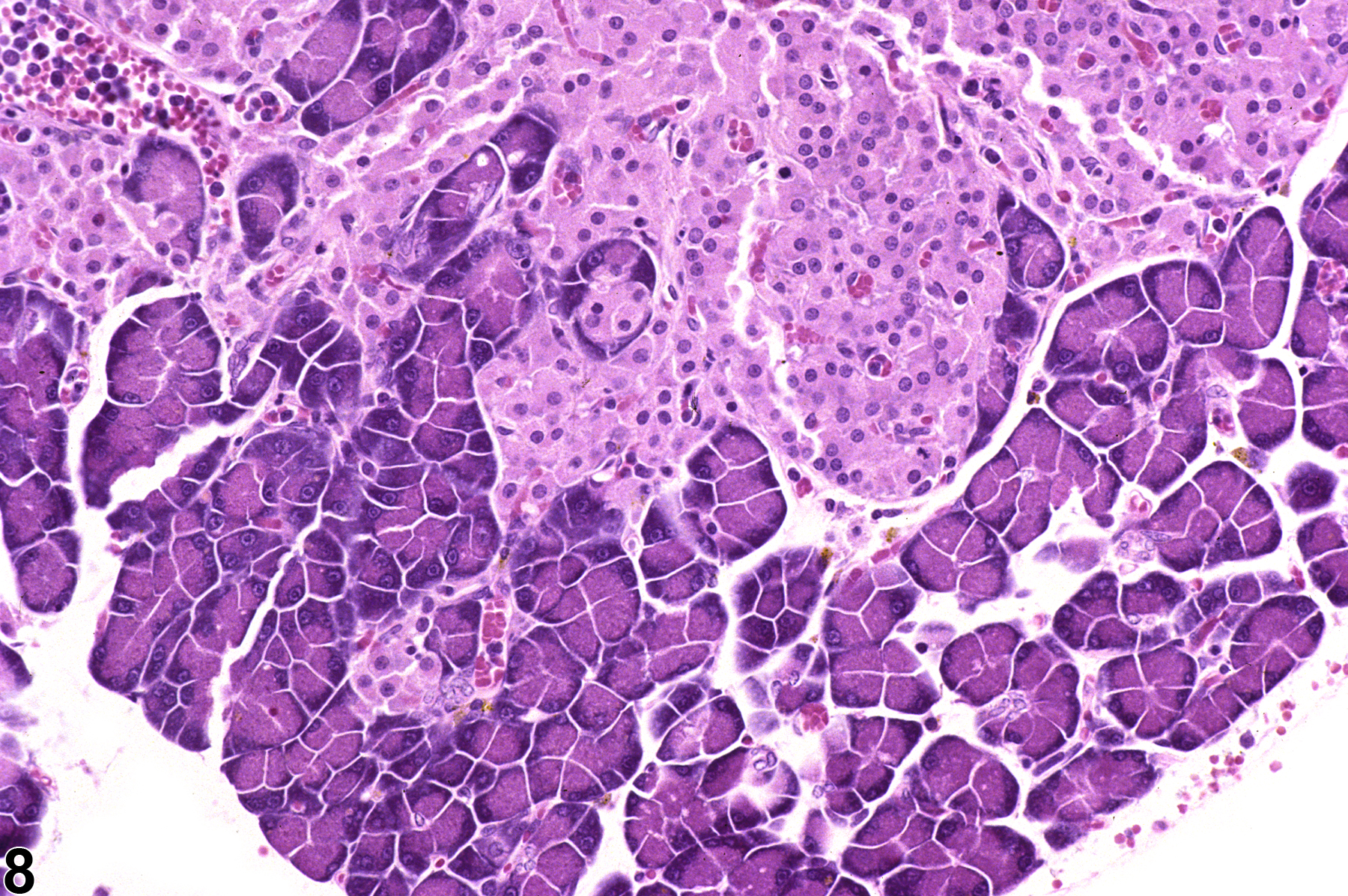
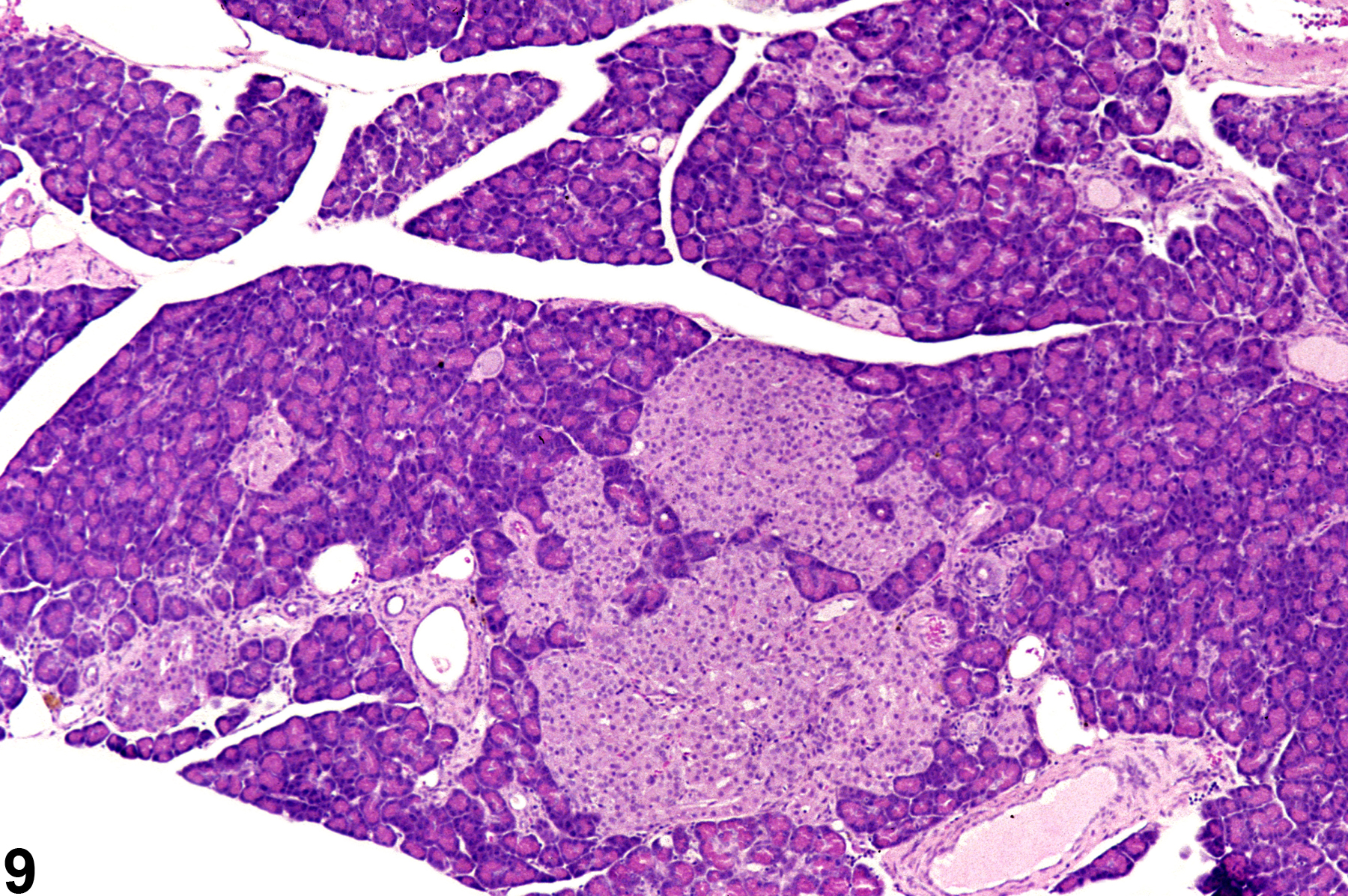
If there several islets greater than 300 micrometers in diameter, a diagnosis of islet cell hyperplasia should be made. Also, if the overall area occupied by the islets in a section of pancreas is greater than that of the controls, a diagnosis of islet cell hyperplasia may be appropriate. Affected islets may be contiguous, giving a lobular appearance (Figure 3) or distinctly separated (Figure 4). The distinction between hyperplasia and adenoma is largely influenced by evidence of compression in adenomas and lack of compression in hyperplasia (Figure 5 and Figure 6). In islet cell adenomas, there may be some cellular atypia or pleomorphism, which is not typically seen in hyperplastic lesions. Occasional acinar cells may become trapped within coalescing hyperplastic islets (Figure 7, Figure 8, and Figure 9) and should not be confused with invasion of an islet cell carcinoma.
Boorman GA, Sills RC. 1999. Exocrine and endocrine pancreas. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 185-206.
Frith CH, Sheldon WD. 1996. Hyperplasia, adenoma, and carcinoma of pancreatic islets, mouse. In: Endocrine System (Jones TC, Capen CC, Mohr U, eds). Springer, Heidelberg, 297-303.
Abstract: http://link.springer.com/chapter/10.1007%2F978-3-642-60996-1_42Greaves P. 2007. Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Academic Press, Amsterdam, 528-537.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Riley MGI, Boorman GA, Hayashi Y. 1990. Endocrine pancreas. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 545-553.
Abstract: http://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Tucker MJ. 1998. The endocrine system. In: Target Organ Pathology (Turton J, Hooson J, eds). Taylor and Francis, London, 311-334.
Abstract: http://www.amazon.com/Target-Organ-Pathology-Basic-Text/dp/0748401571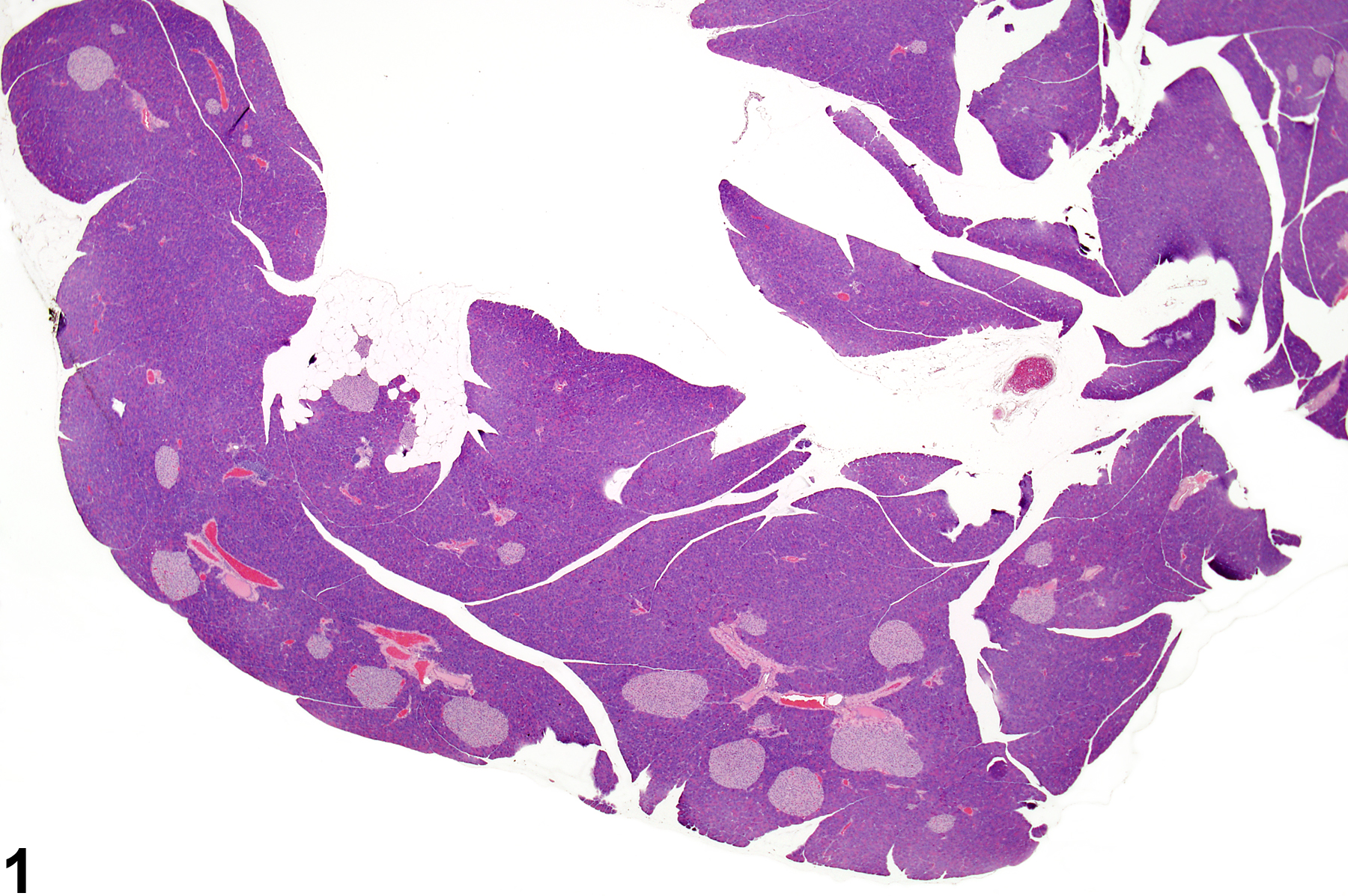
Pancreatic islet - normal. Normal islets in a control male B6C3F1 mouse from a chronic study.