Hepatobiliary System
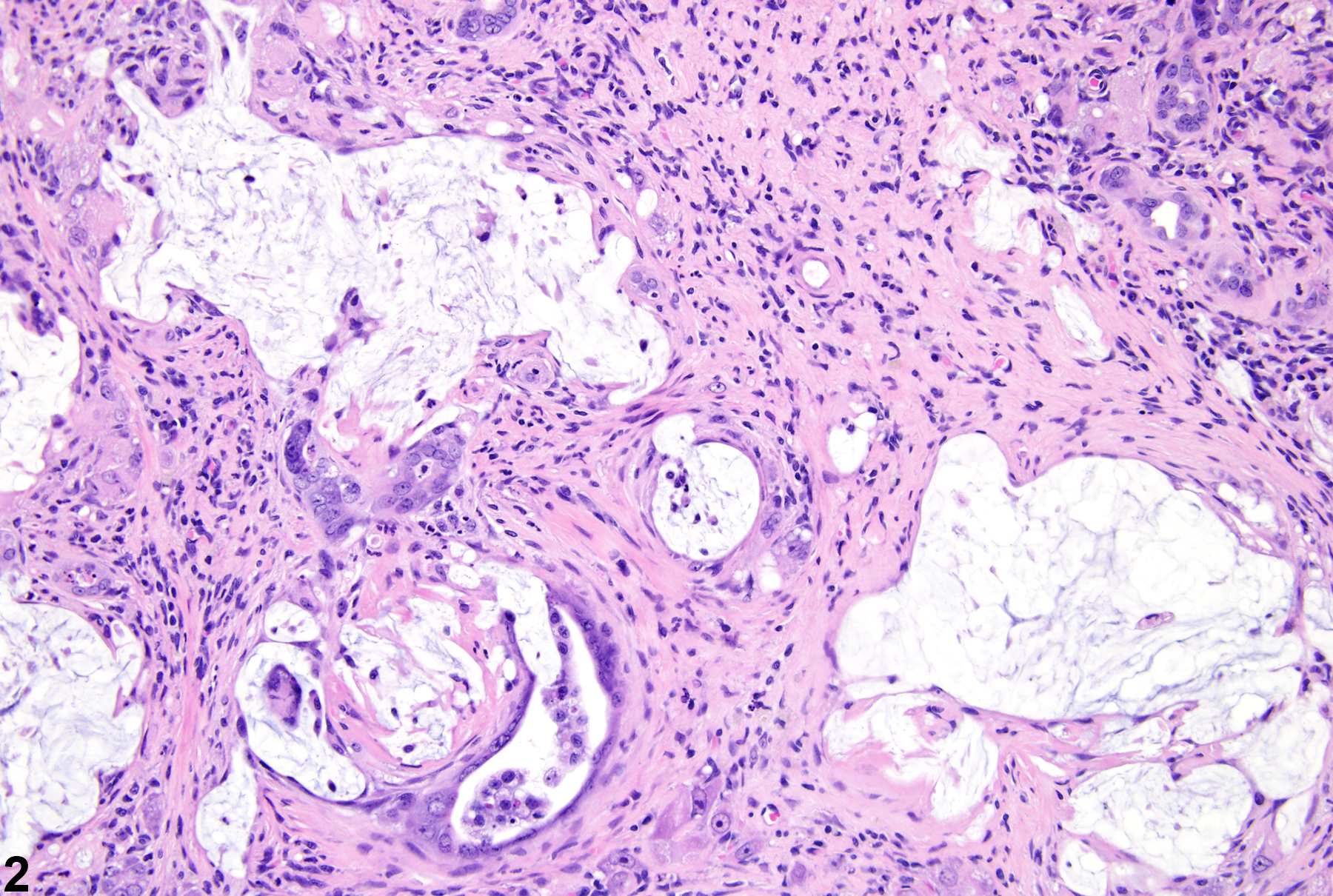
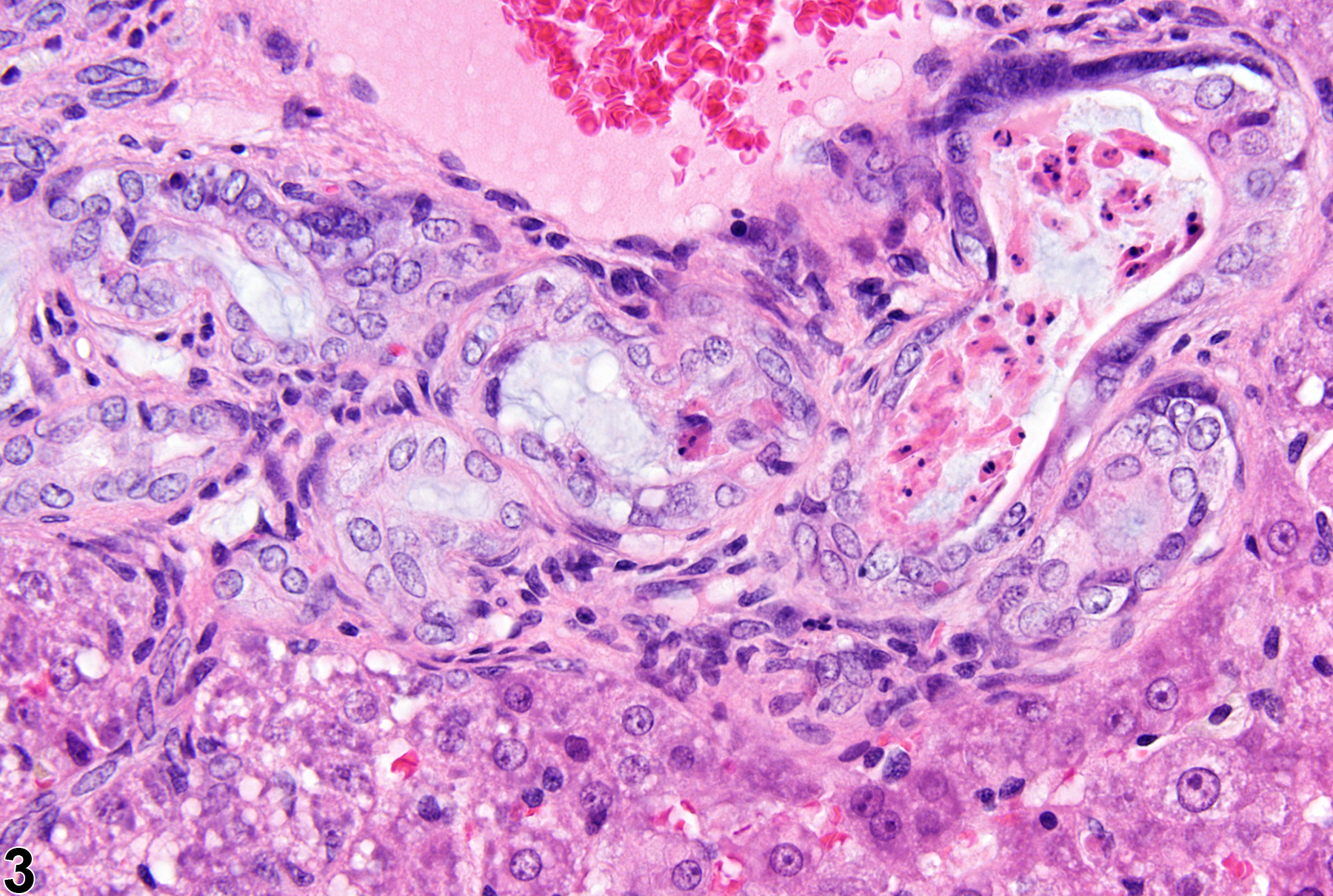
Liver - Cholangiofibrosis
Narrative
Adams ET, Auerbach S, Blackshear PE, Bradley A, Gruebbel MM, Little PB, Malarkey D, Maronpot R, McKay JS, Miller RA, Moore RR, Morrison JP, Nyska A, Ramot Y, Rao D, Suttie A, Wells MY, Willson GA, Elmore SA. 2011. Proceedings of the 2010 National Toxicology Program Satellite Symposium. Toxicol Pathol 39(1):240-266.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/21177527Bannasch P, Zerban H. 1990 Tumours of the liver. In: Pathology of Tumours in Laboratory Animals, Vol 1: Tumours of the Rat, 2nd ed. (Turusov VS, Mohr U, eds). IARC Scientific Publication No. 99. International Agency for Research on Cancer, Lyon, France, 199-240.
Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Hailey JR, Walker NJ, Sells DM, Brix AE, Jokinen MP, Nyska A. 2005. Classification of proliferative hepatocellular lesions in Harlan Sprague-Dawley rats chronically exposed to dioxin-like compounds. Toxicol Pathol 33:165-174.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15805068Kimbrough RD, Linder RE, Burse VW, Jennings RW. 1973. Adenofibrosis in the rat liver, with persistence of polychlorinated biphenyls in adipose tissue. Arch Environ Health 27:390-395.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/4356600Sirica AE. 1992. The Role of Cell Types in Hepatocarcinogenesis. CRC Press, Boca Raton, FL.
Abstract: http://www.crcpress.com/product/isbn/9780849347467Thoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full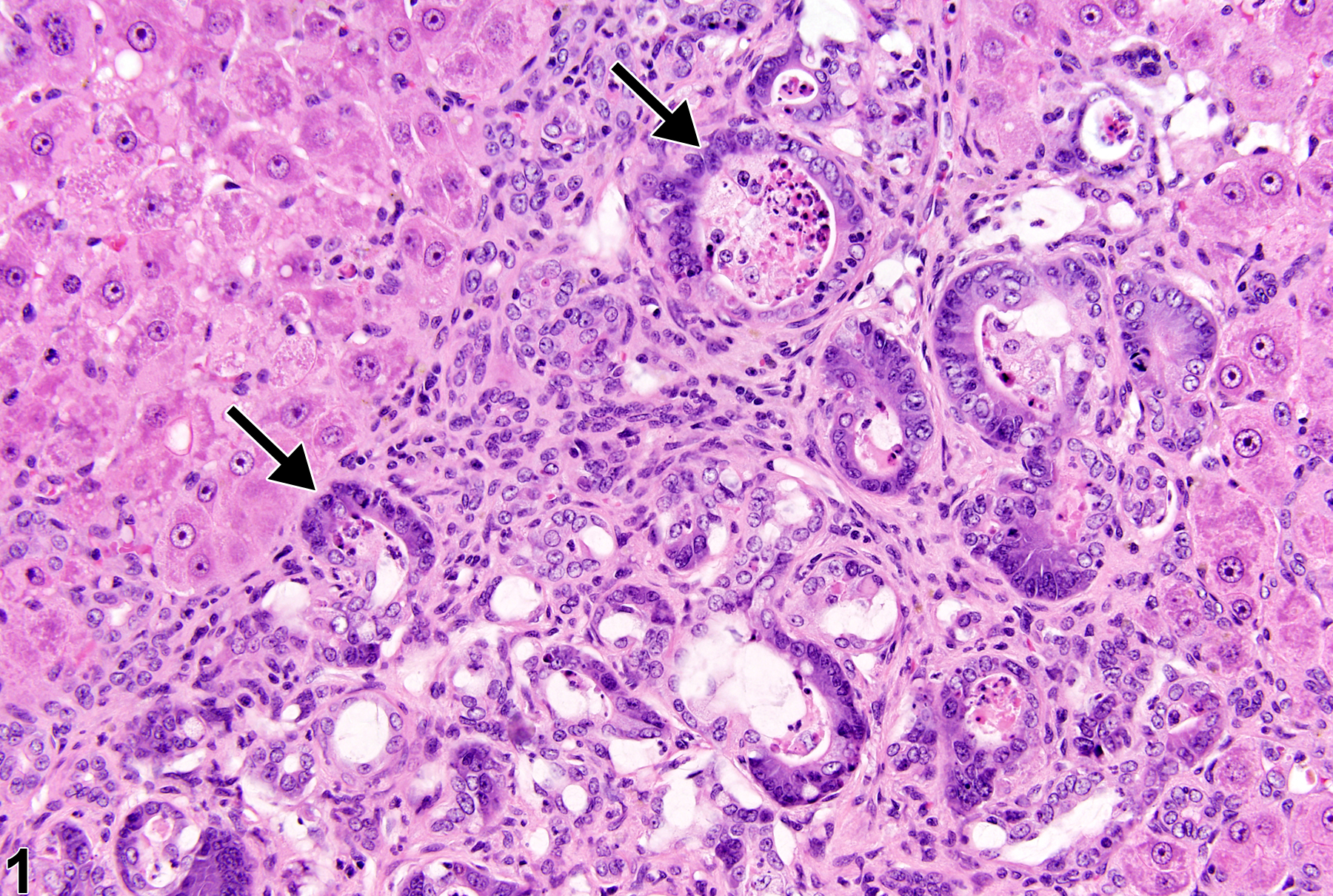
Cholangiofibrosis-arrows indicate incompletely lined proliferating bile ducts in a female Harlan Sprague-Dawley rat in a chronic study.