Hepatobiliary System
Liver - Focus
Narrative
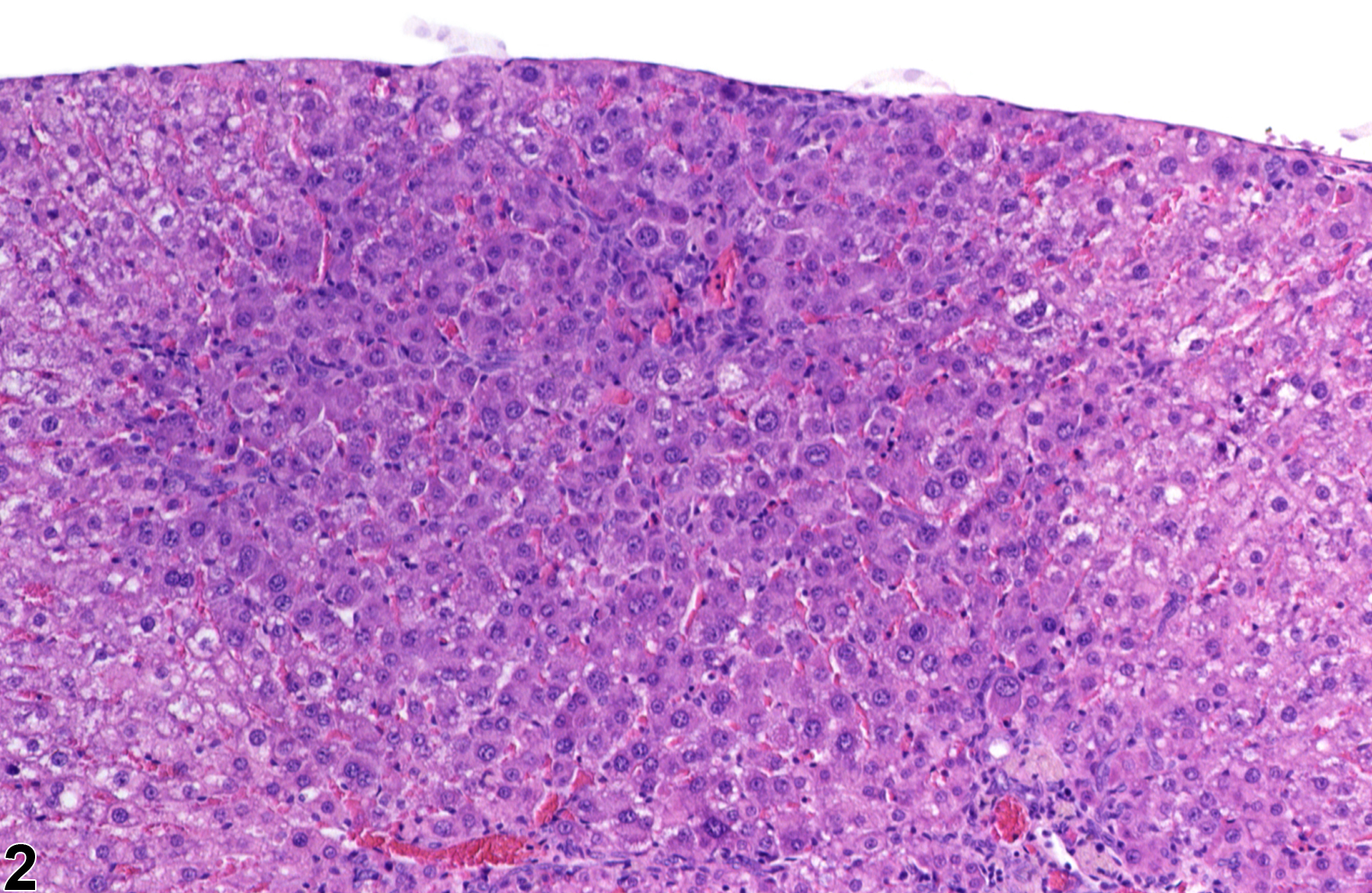
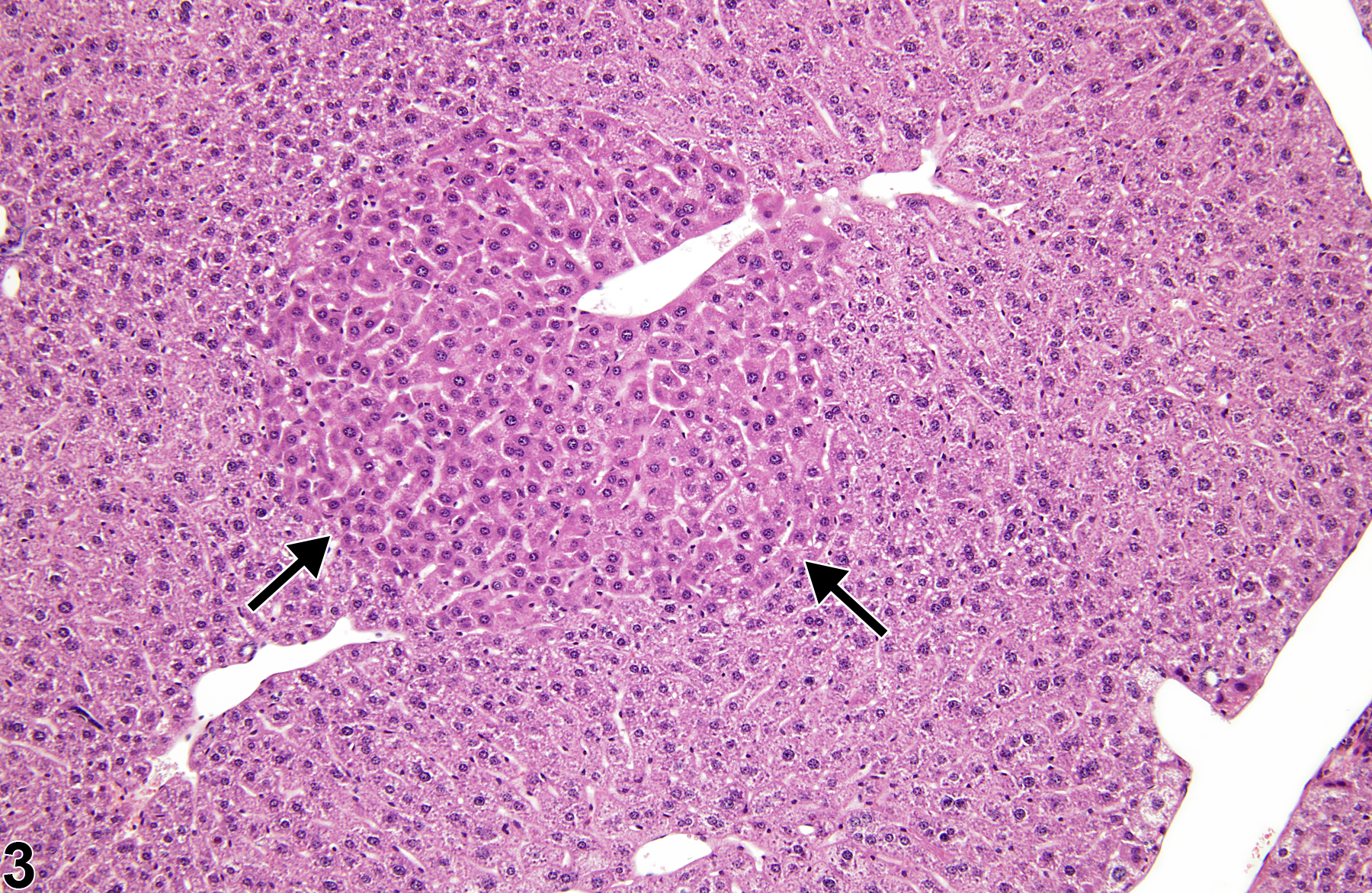
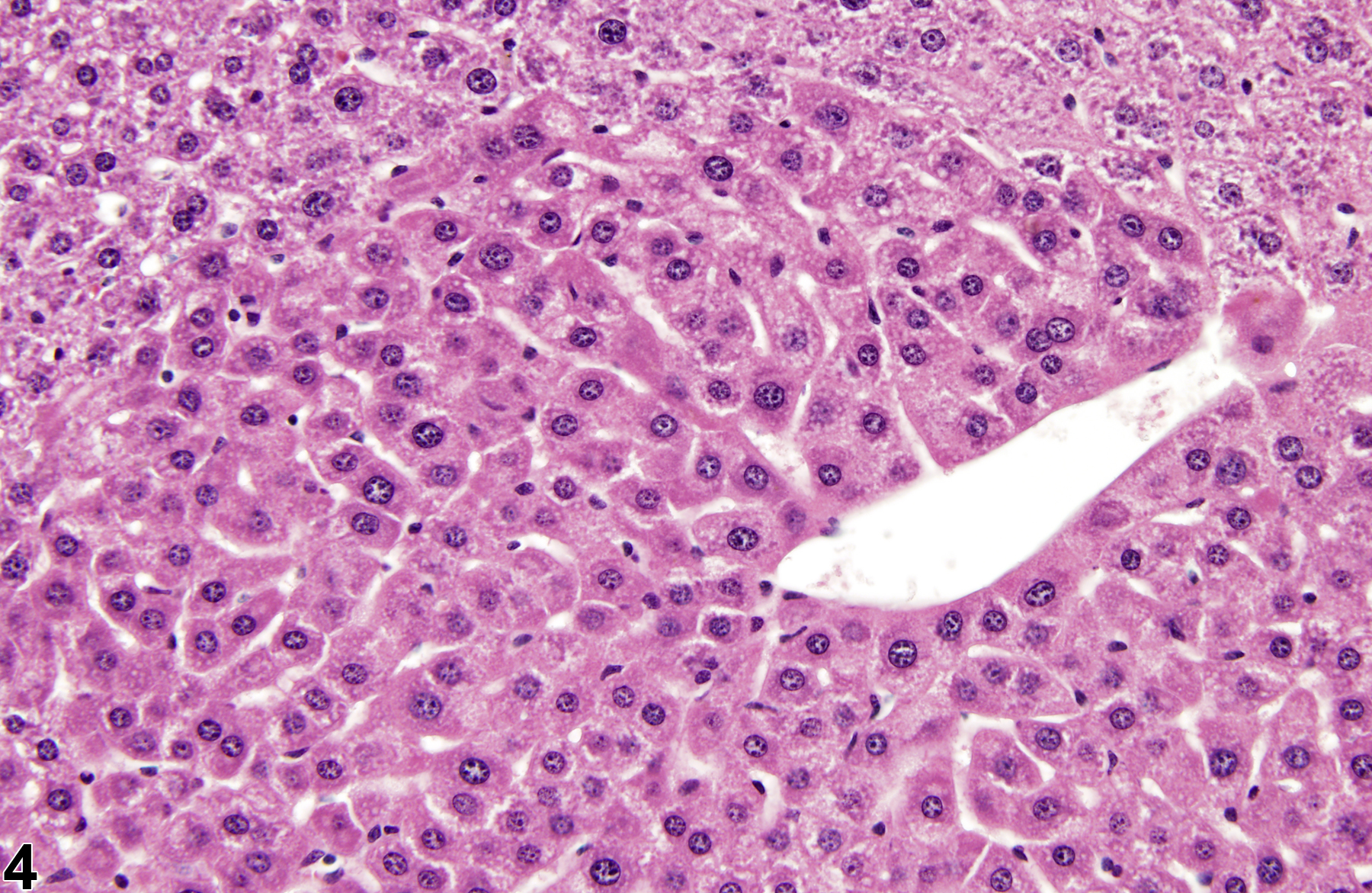
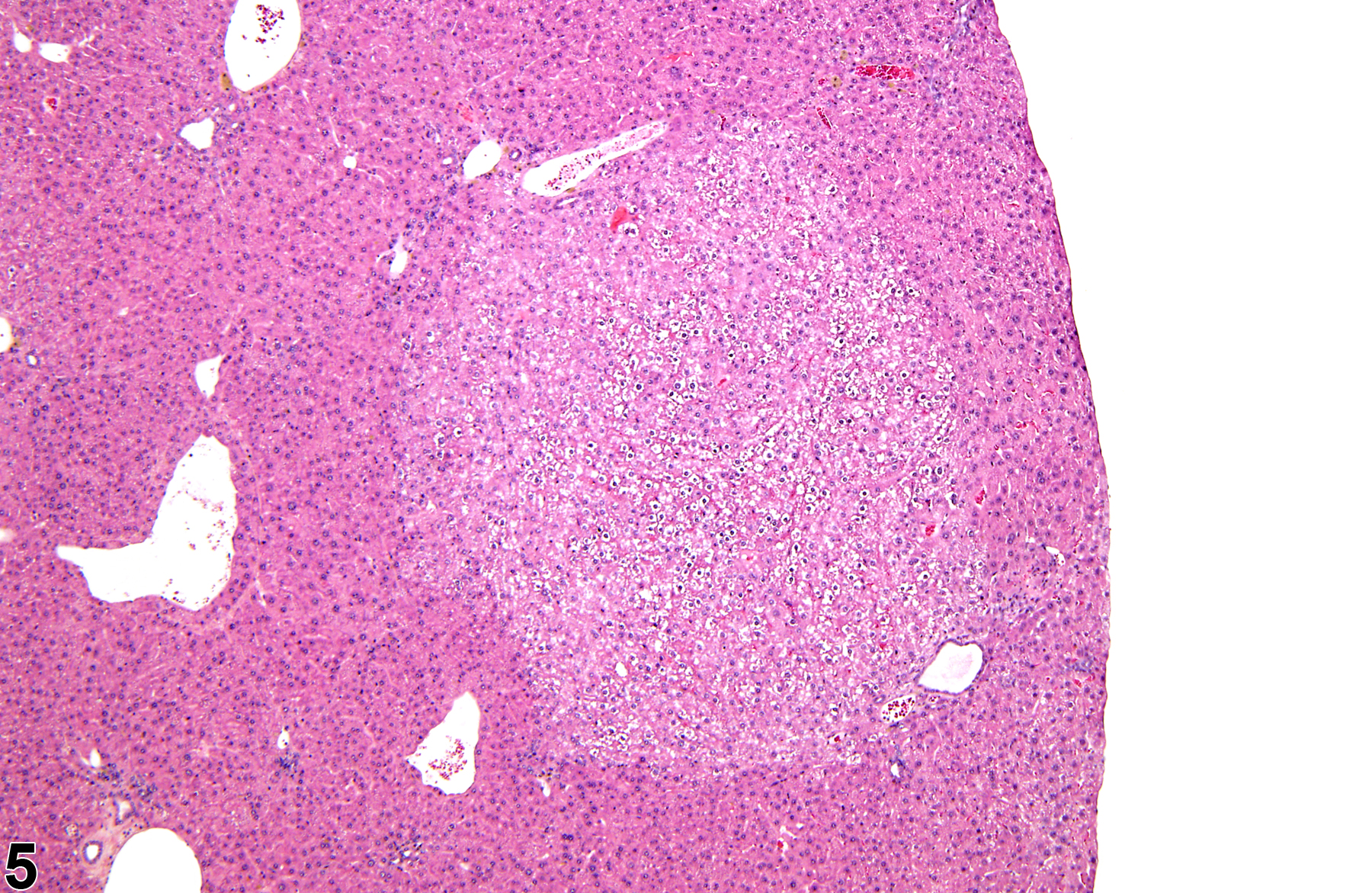
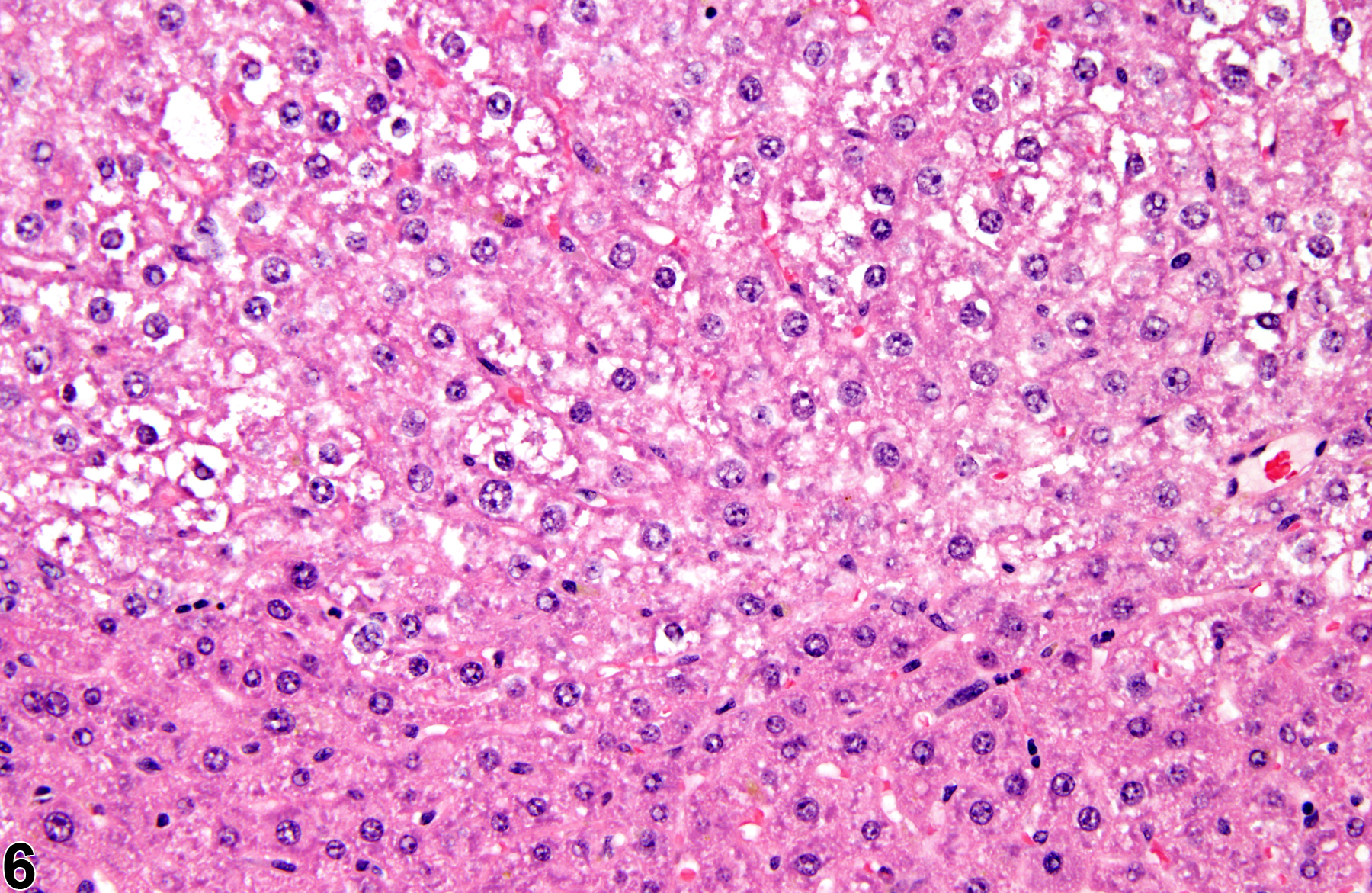
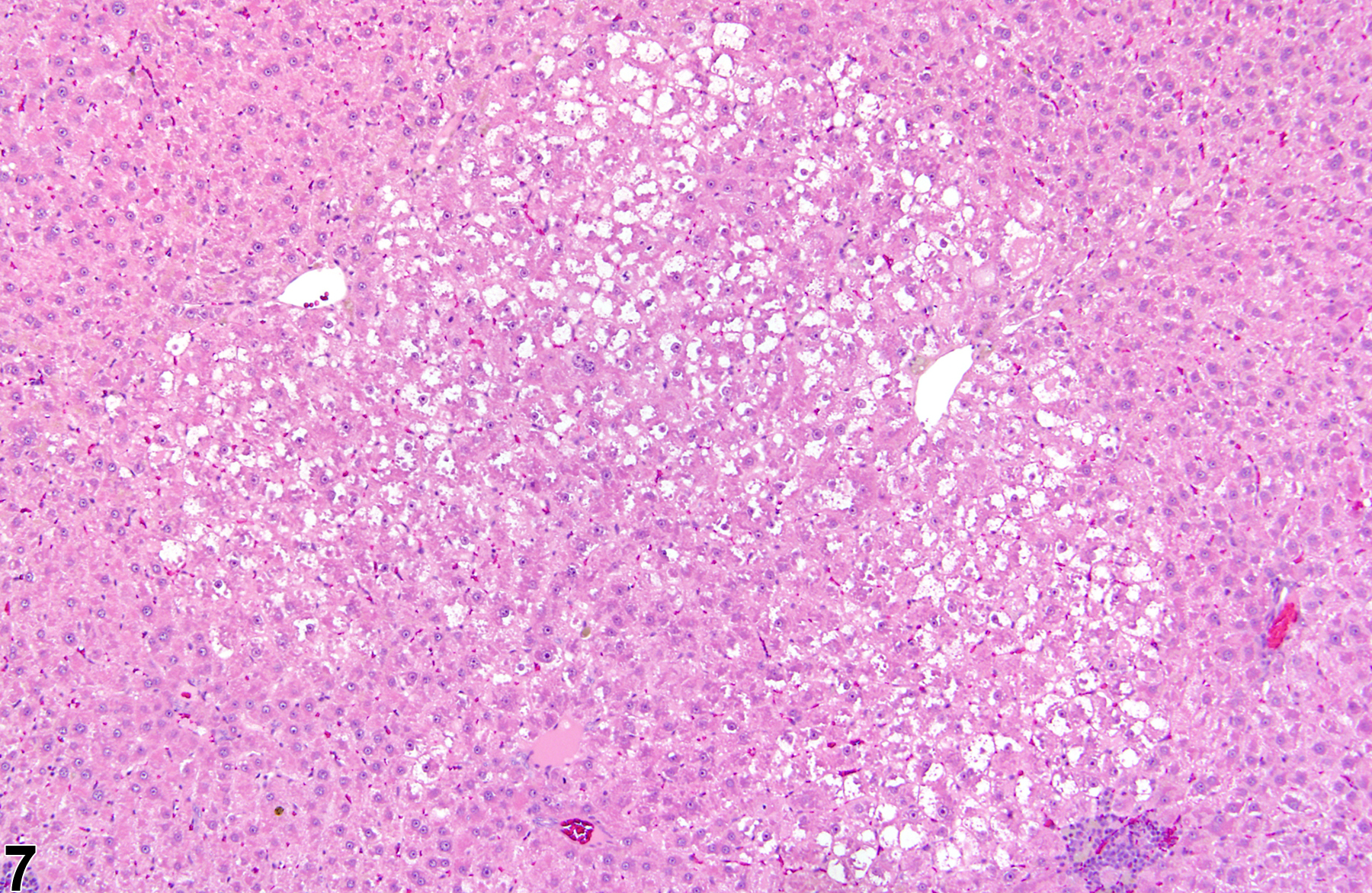
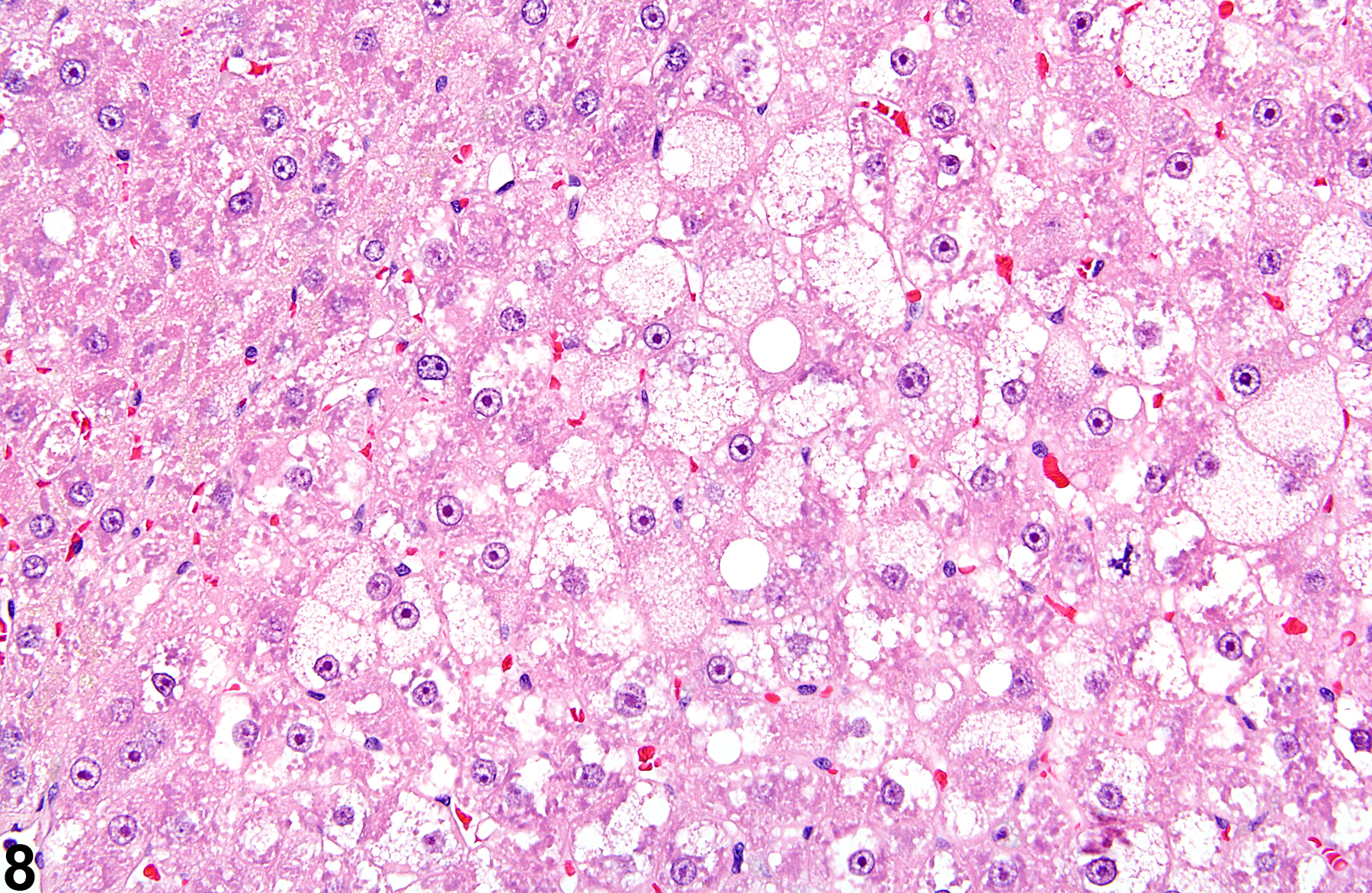
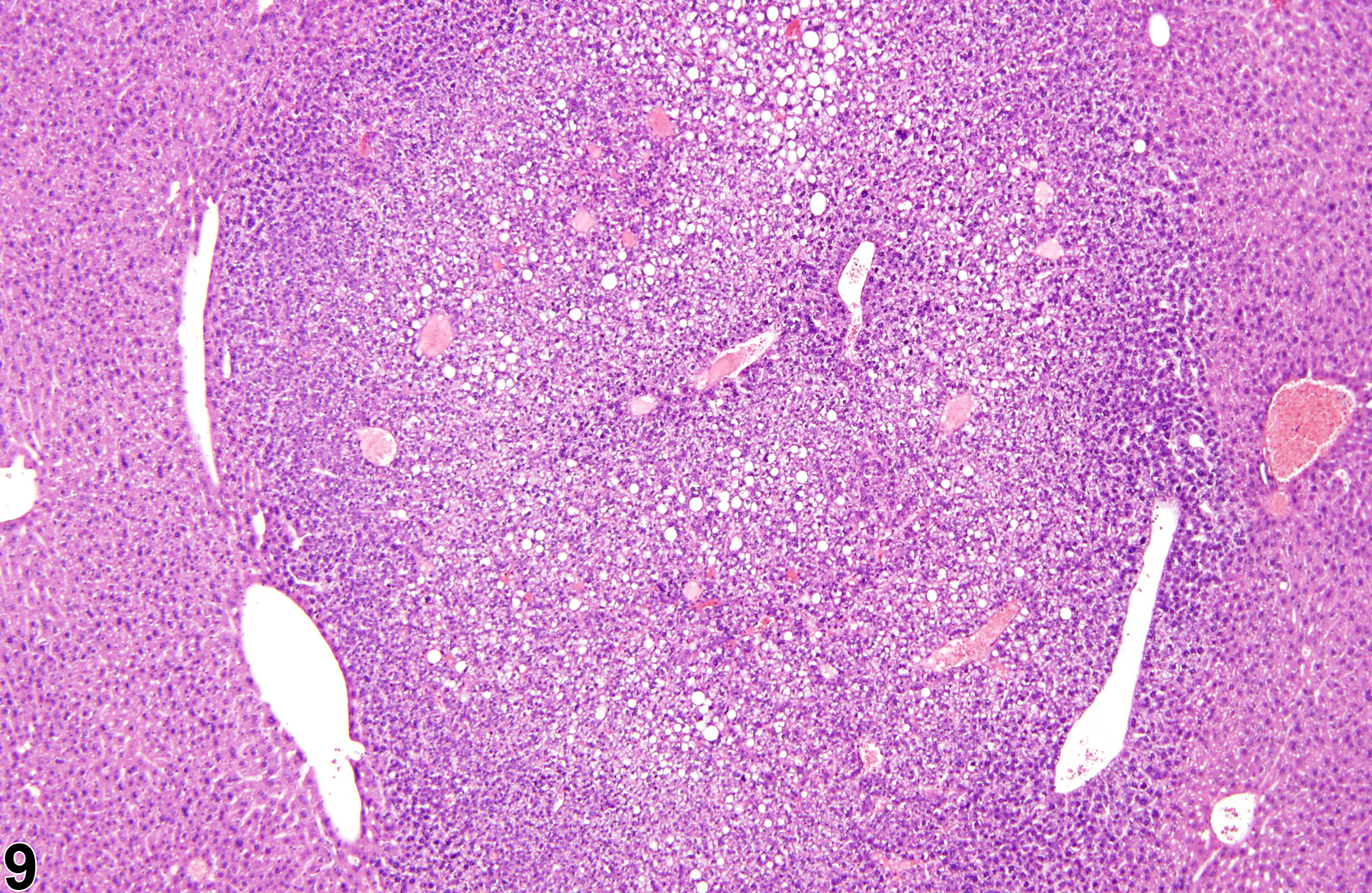
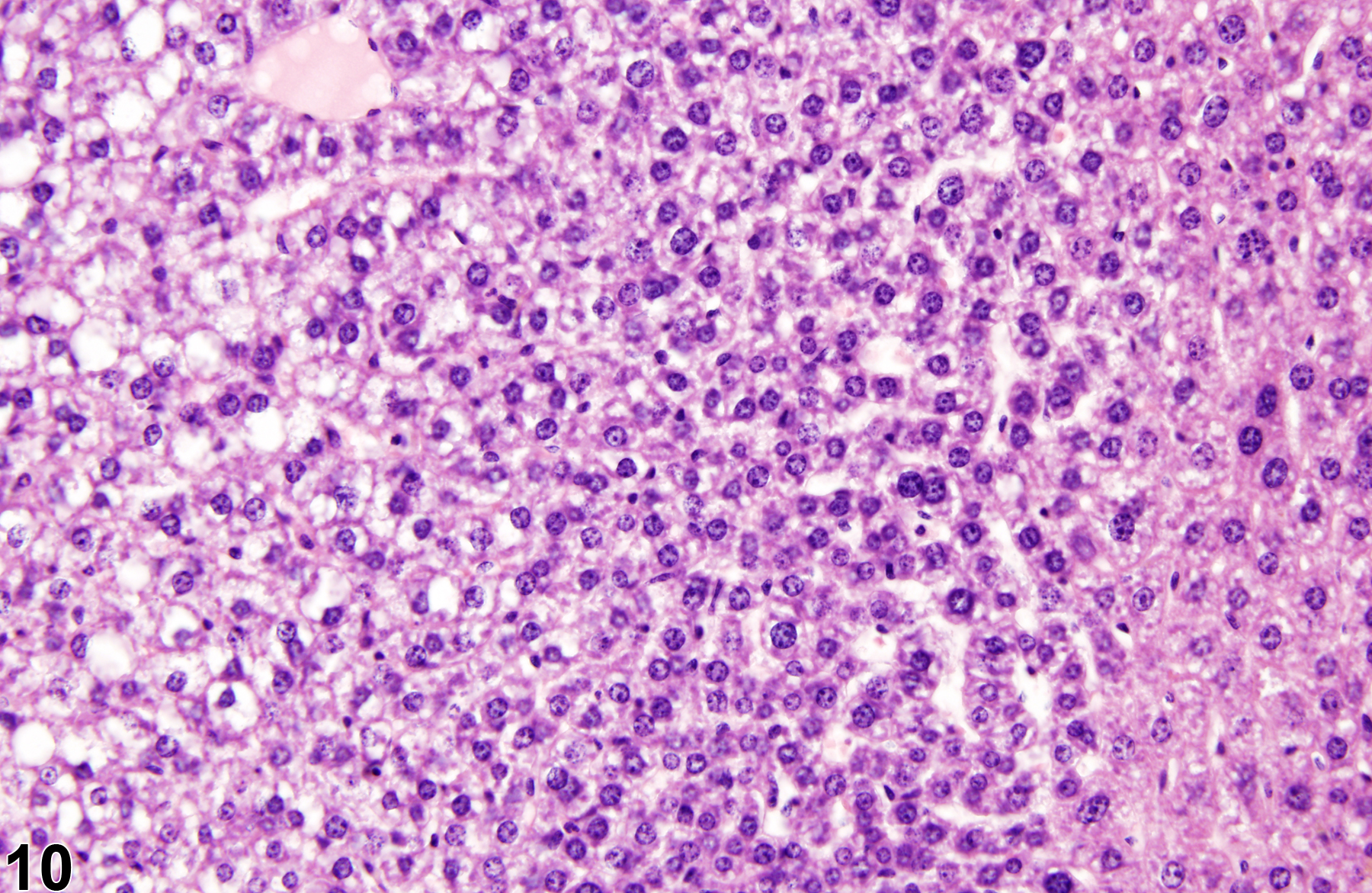
Basophilic foci predominantly stain with hematoxylin (Figure 1, arrows; Figure 2). A tigroid variant has been identified in rats but is considered a subclassification of basophilic focus. Eosinophilic foci typically stain more eosinophilic than surrounding hepatocytes and often consist of hepatocytes that are larger than the adjacent normal parenchyma (Figure 3, arrows; Figure 4). Clear cell foci have empty spaces in hepatocyte cytoplasm surrounding centrally localized nuclei. The clear spaces represent glycogen dissolved out during fixation and processing. Clear cell foci are readily identified because they stand out against surrounding parenchyma that has a more uniformly eosinophilic cytoplasm (Figure 5 and Figure 6). Some texts discuss vacuolated cell foci, but these are now diagnosed as focal fatty change and are not considered foci. Mixed cell focus is diagnosed when there is no single predominant phenotype. Figure 7 and Figure 8 represent a mixed cell focus composed of clear cells, vacuolated cells, and amphophilic cells with no single cell type constituting more than 80% of the focus cells. Figure 9 and Figure 10 present a mixed focus composed of an outer rim of basophilic cells surrounding an inner core of clear cells, each comprising an equal proportion of the cell types present.
Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Harada T, Enomoto A, Boorman GA, Maronpot RR. 1999. Liver and gallbladder. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 119-183.
Harada T, Maronpot RR, Enomoto A, Tamano S, Ward JM. 1996. Changes in the liver and gallbladder. In: Pathobiology of the Aging Mouse (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 2:207-241.
Abstract: http://catalog.hathitrust.org/Record/008994685Harada T, Maronpot RR, Morris RW, Stitzel KA, Boorman GA. 1989. Morphological and stereological characterization of hepatic foci of cellular alteration in control Fischer 344 rats. Toxicol Pathol 17:579-593.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/2483465Maronpot RR, Harada T, Murthy ASK, Boorman GA. 1989. Documenting foci of hepatocellular alteration in two-year carcinogenicity studies: Current practices of the National Toxicology Program. Toxicol Pathol 17:675-684.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/2697942National Toxicology Program. 2011. NTP TOX-82. 3-Month Toxicity Studies of Estragole (CAS No. 140-67-0) Administered by Gavage to F344/N Rats and B6C3F1 Mice. NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/st_rpts/tox082.pdfNational Toxicology Program. 2011. NTP TR-563. Toxicology and Carcinogenesis Studies of Pulegone (CAS No. 89-82-7) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr563.pdfThoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full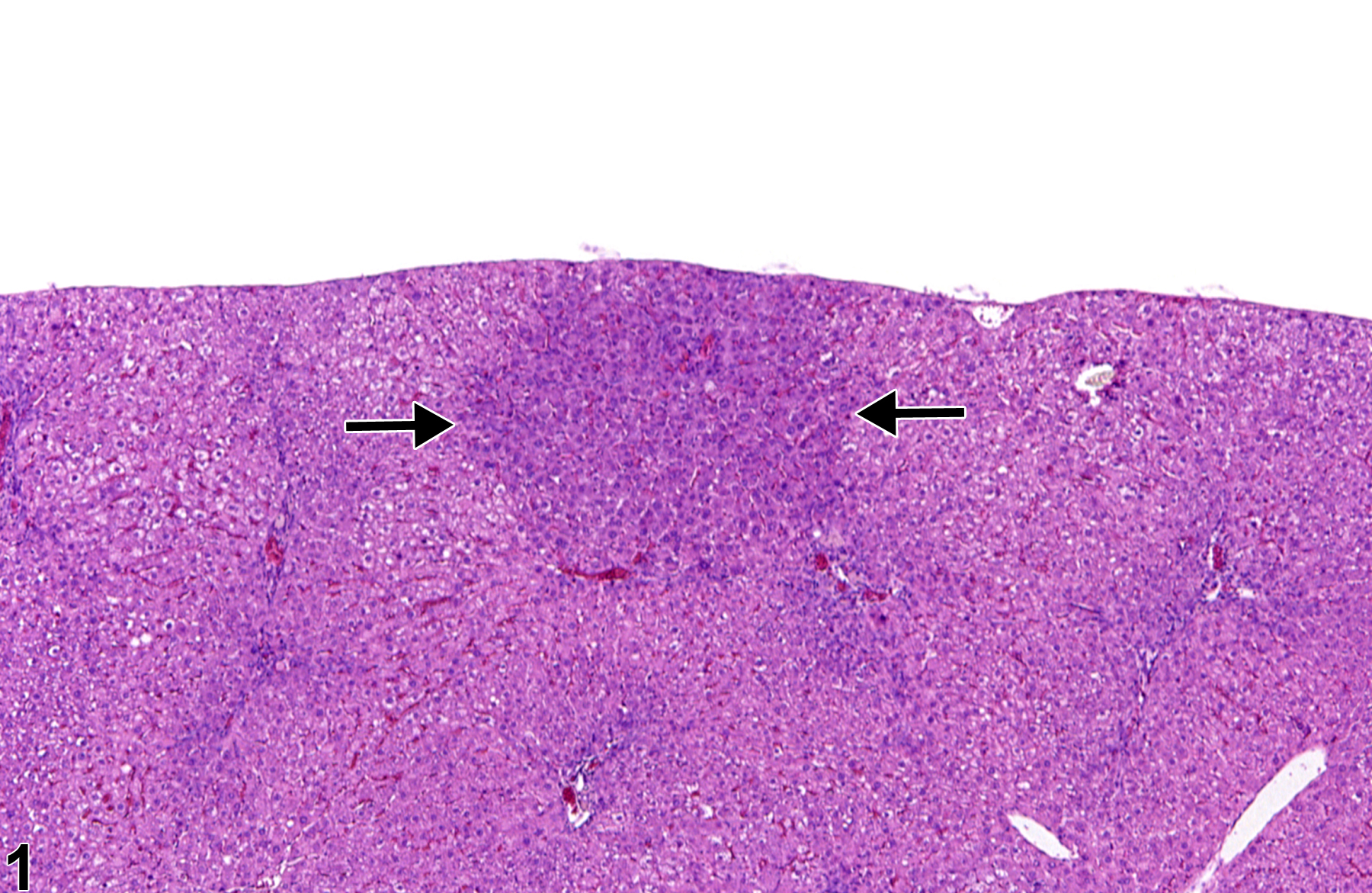
Basophilic focus (arrows) in a male F344/N rat from a subchronic study.