Hepatobiliary System
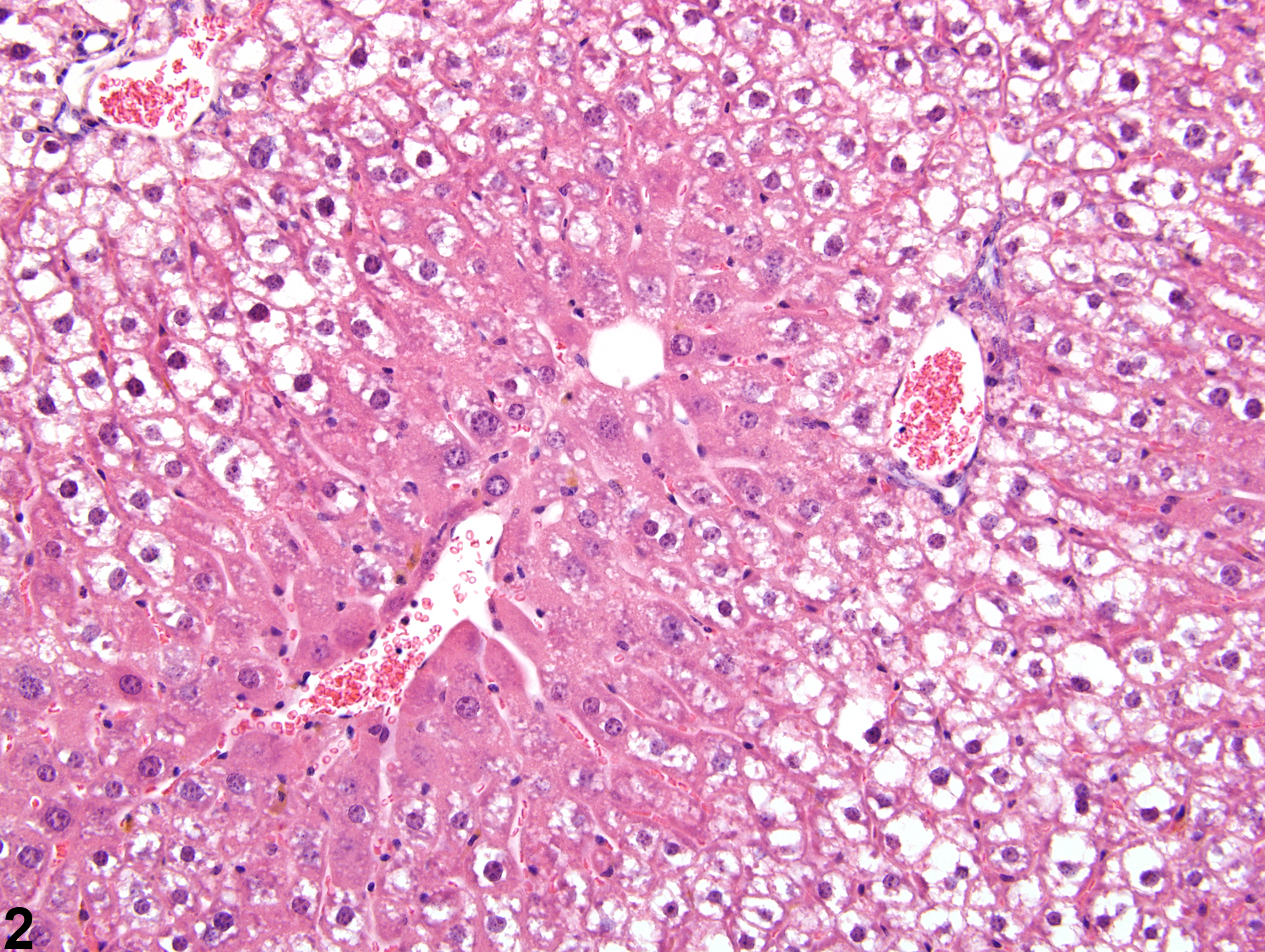
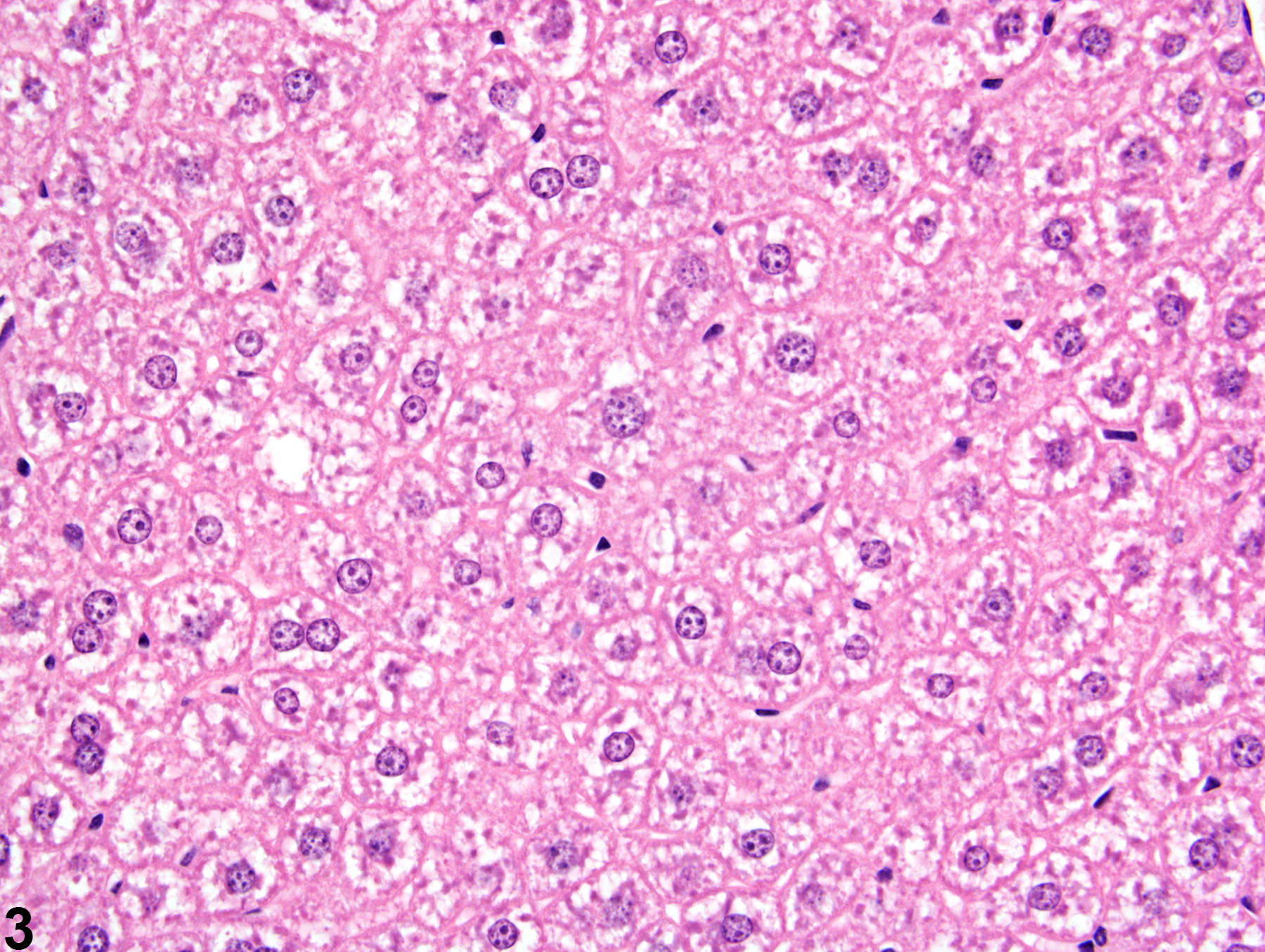
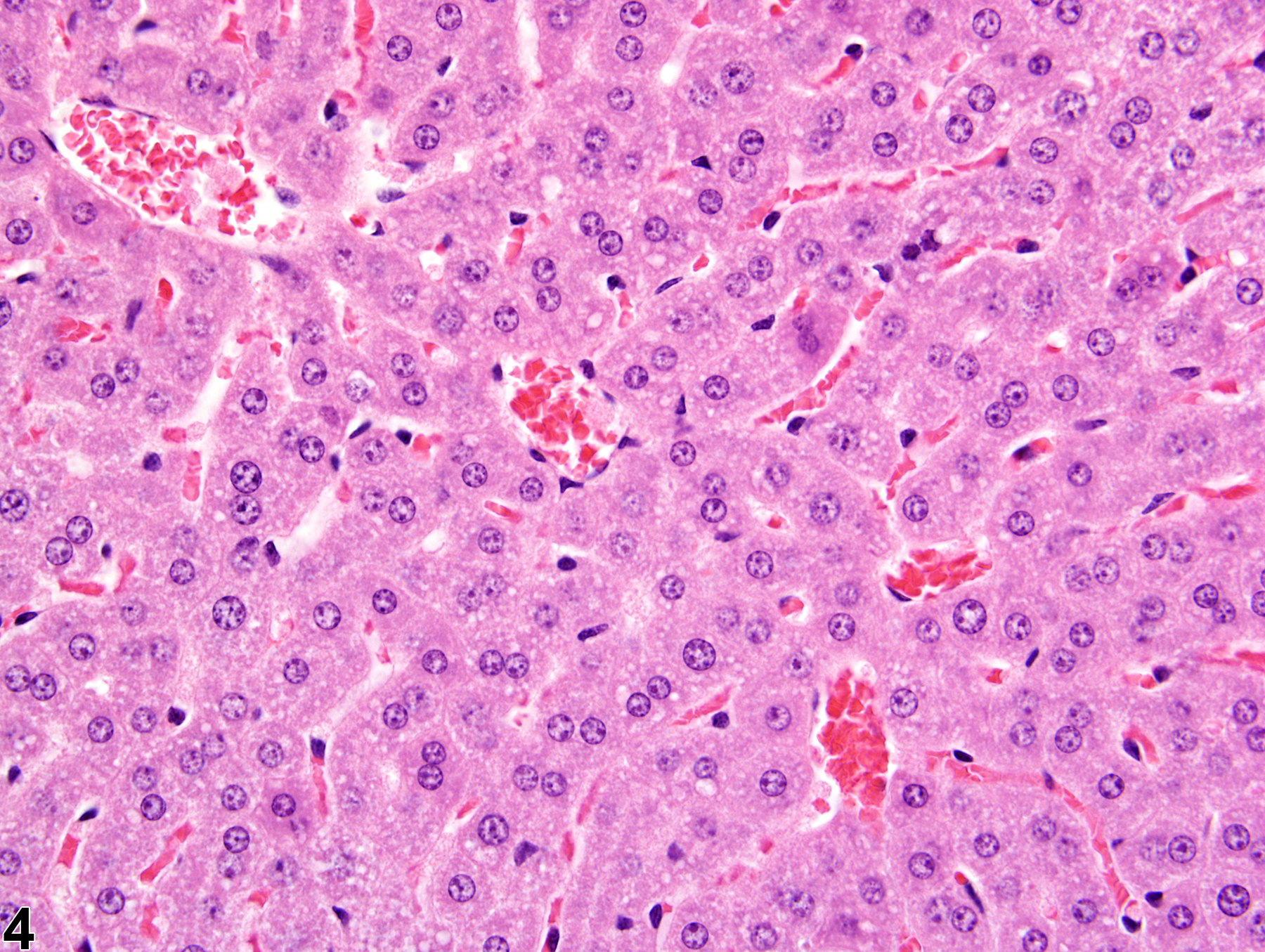
Liver, Hepatocyte - Glycogen Accumulation and Depletion
Narrative
The morphologic appearance of glycogen in hepatocytes is distinctive, allowing glycogen accumulation or depletion to be readily diagnosed in most cases. However, appropriate fixation and definitive special staining are necessary for unequivocal evidence of glycogen deposition. In some cases, particularly when differences in the amount of glycogen are accompanied by other cytoplasmic changes, glycogen accumulation or depletion may not be easily discerned.
Changes in the amount of hepatocellular glycogen are frequently accompanied by other cytoplasmic alterations, such as fatty change or cytoplasmic vacuolization, and alterations in glycogen content can be difficult to distinguish from the accompanying changes. In those situations, a diagnosis of hepatocyte cytoplasmic vacuolization is appropriate, along with a severity grade, but the morphology of the cytoplasmic changes in the hepatocytes should be thoroughly described in the pathology narrative. The pathology narrative should also include the pathologist’s opinions regarding the contents of the cytoplasmic vacuoles.
Cattley RC, Popp JA. 2002. Liver. In: Handbook of Toxicologic Pathology (Haschek WM, Rousseaux CG, Wallig MA, eds). Academic Press, San Diego, 2:187-225.
Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Evans JG, Lake BG. 1998. The digestive system II. Hepatobiliary system. In: Target Organ Pathology (Turton J, Hooson J, eds). Taylor and Francis, London, 61-98.
Greaves P. 2007. Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Elsevier, Amsterdam.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Harada T, Enomoto A, Boorman GA, Maronpot RR. 1999. Liver and gallbladder. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 119-183.
Hardisty JF, Brix AE. 2005. Comparative hepatic toxicity: Prechronic/chronic liver toxicity in rodents. Toxicol Pathol 33:35-40.
Full Text: http://tpx.sagepub.com/content/33/1/35.full.pdfHaschek WM, Rousseaux CG, Wallig MA. 2010. Fundamentals of Toxicologic Pathology, 2nd ed. Academic Press, San Diego, 197-235.
Thoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full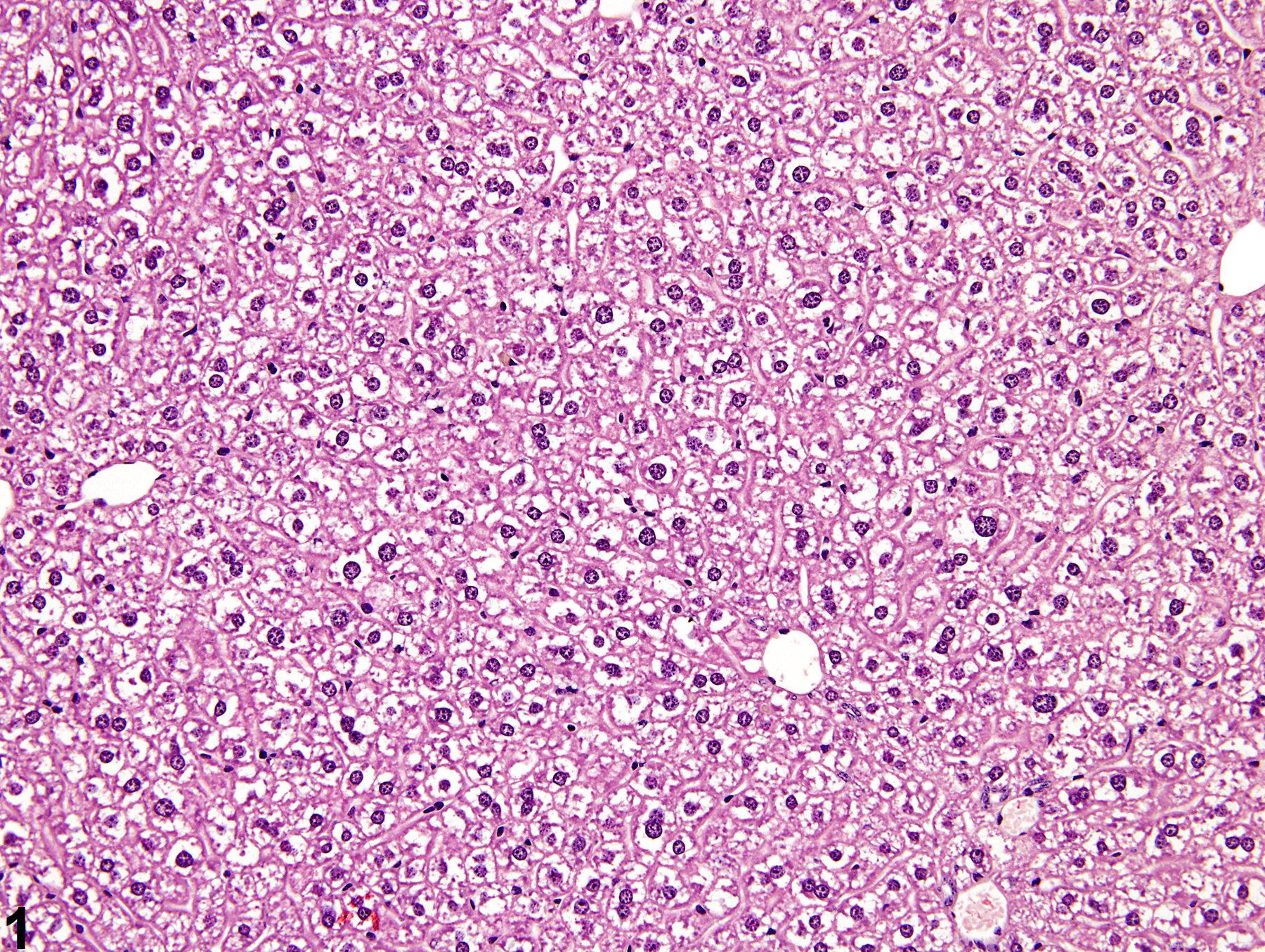
Glycogen accumulation-normal accumulation in hepatocytes in a female B6C3F1 mouse from a chronic study.