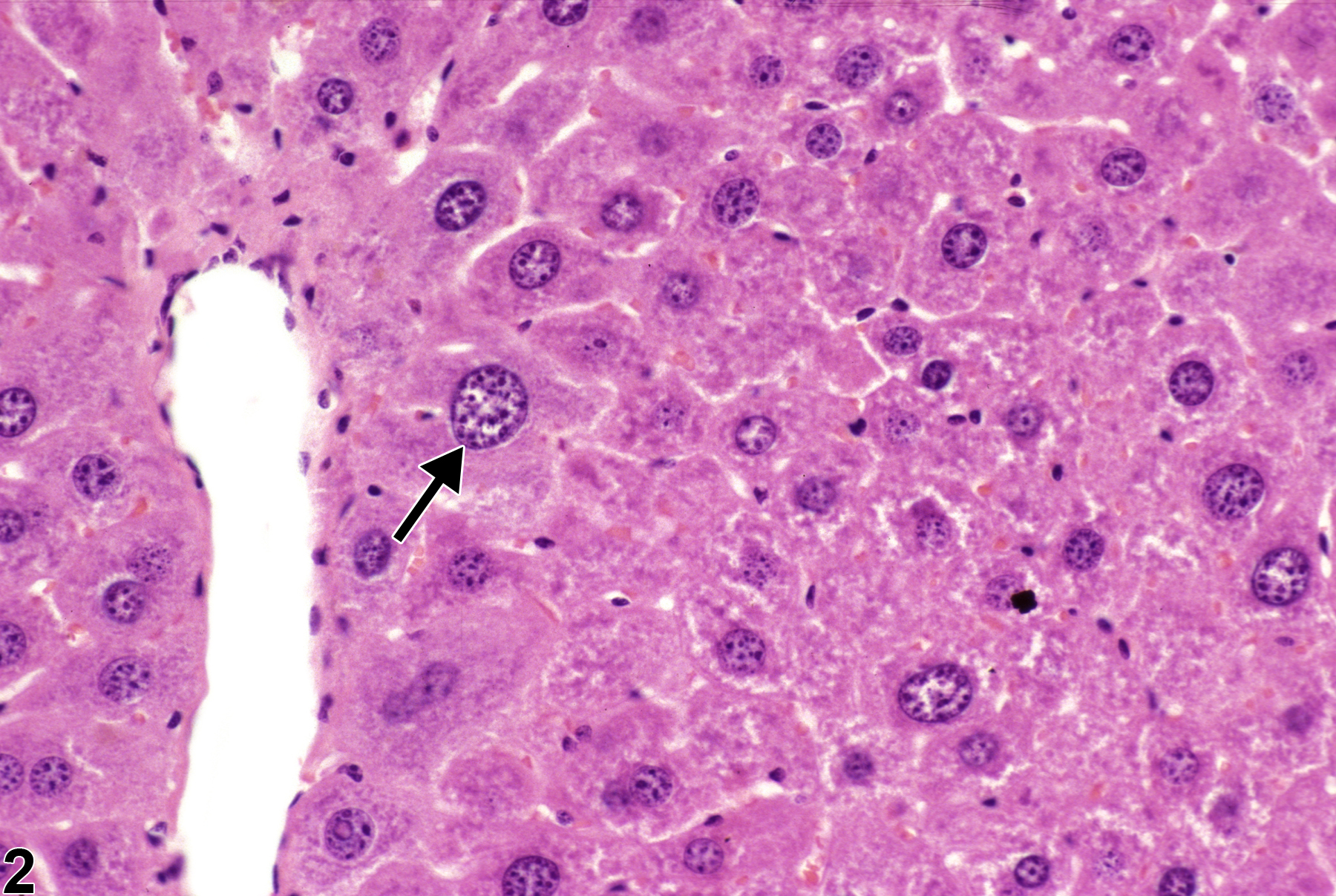
Hepatobiliary System
Liver, Hepatocyte - Karyomegaly
Narrative
Epstein CJ. 1967. Cell size, nuclear content, and the development of polyploidy in the mammalian liver. Proc Natl Acad Sci USA 57:327-334.
Full Text: http://www.pnas.org/content/57/2/327.full.pdfHarada T, Enomoto A, Boorman GA, Maronpot RR. 1999. Liver and gallbladder. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 119-183.
Inamdar NB. 1958. Development of polyploidy in mouse liver. J Morphol 103:65-90.
Abstract: http://onlinelibrary.wiley.com/doi/10.1002/jmor.1051030104/abstractJames J. 1977. The genesis of polyploidy in rat liver parenchymal cells. Eur J Cell Biol 15:410-419.
Abstract: http://link.springer.com/article/10.1007%2FBF00482955Lu MH, Hinson WG, He D, Turturro A, Hart RW. 1993. Hepatic nuclear ploidy distribution of dietary-restricted mice. Environ Health Perspect 101:229-233.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/8013412National Toxicology Program. 1993. NTP TR-394. Toxicology and Carcinogenesis Studies of Acetaminophen (CAS No. 103-90-2) in F344 Rats and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr394.pdfStyles JA. 1993. Measurement of ploidy and cell proliferation in the rodent liver. Environ Health Perspect 101(suppl 5):67-71.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1519461/Thoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full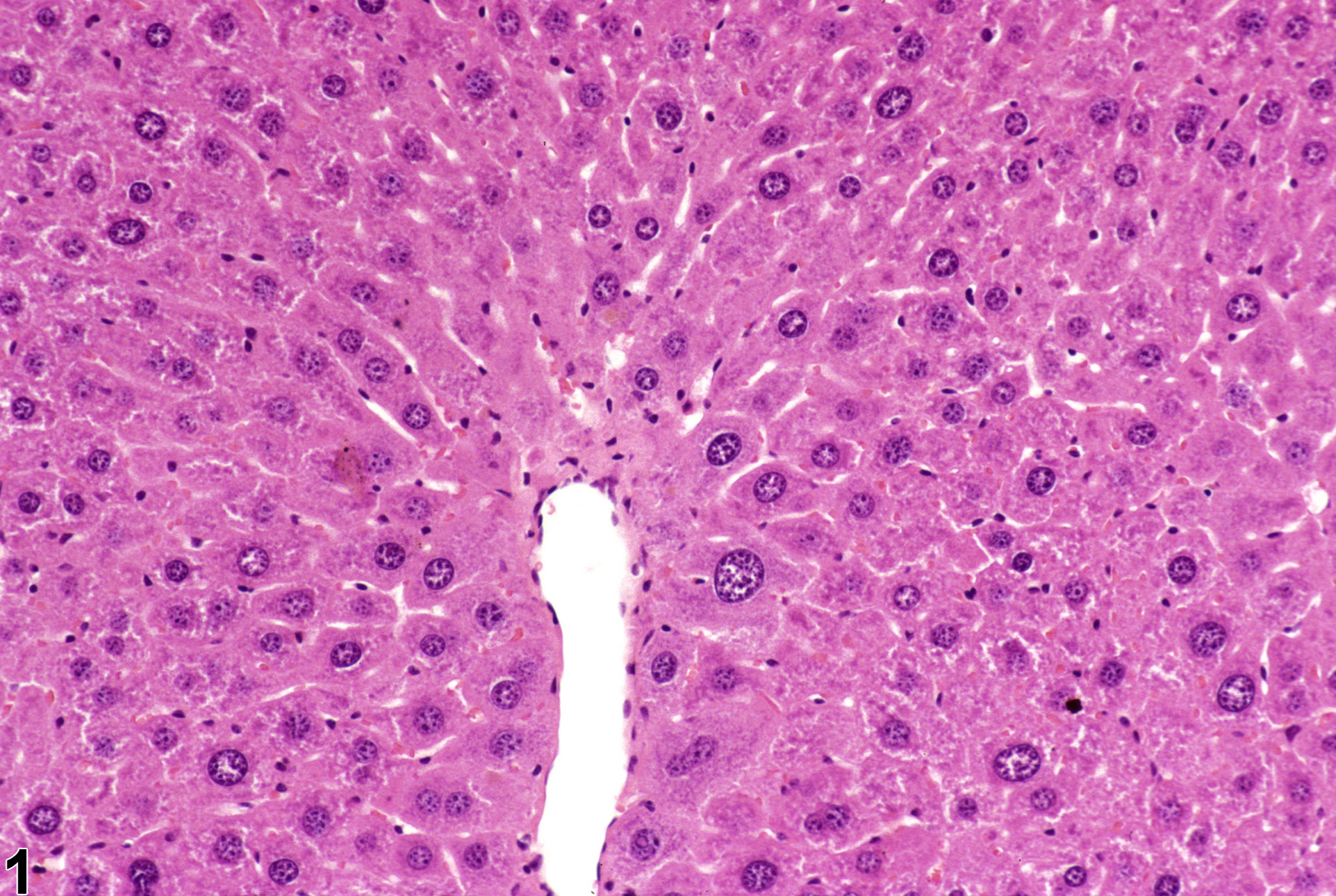
Karyomegaly in a female B6C3F1 mouse from a subchronic study.