Hepatobiliary System
Liver - Inflammation
Narrative
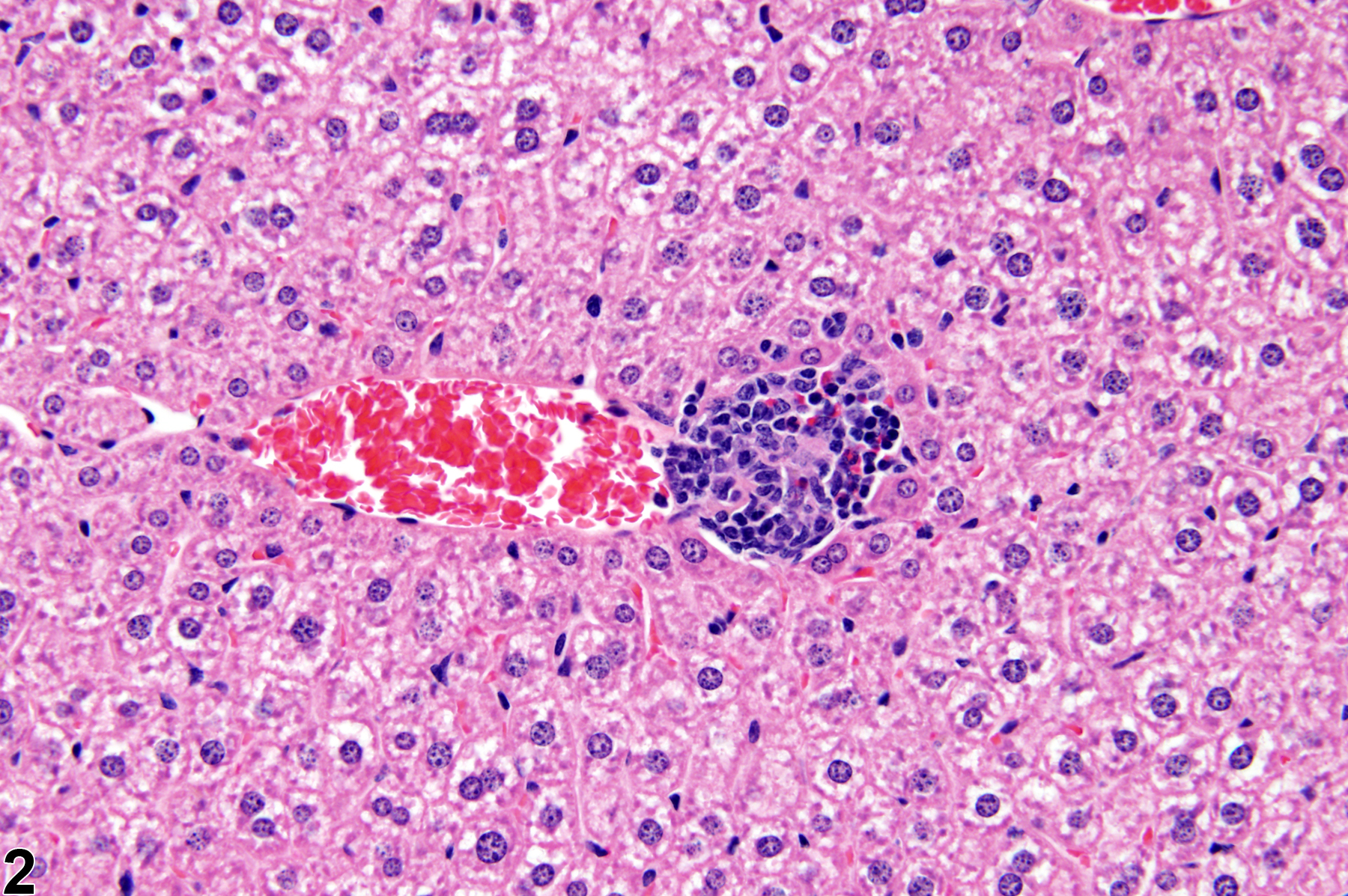
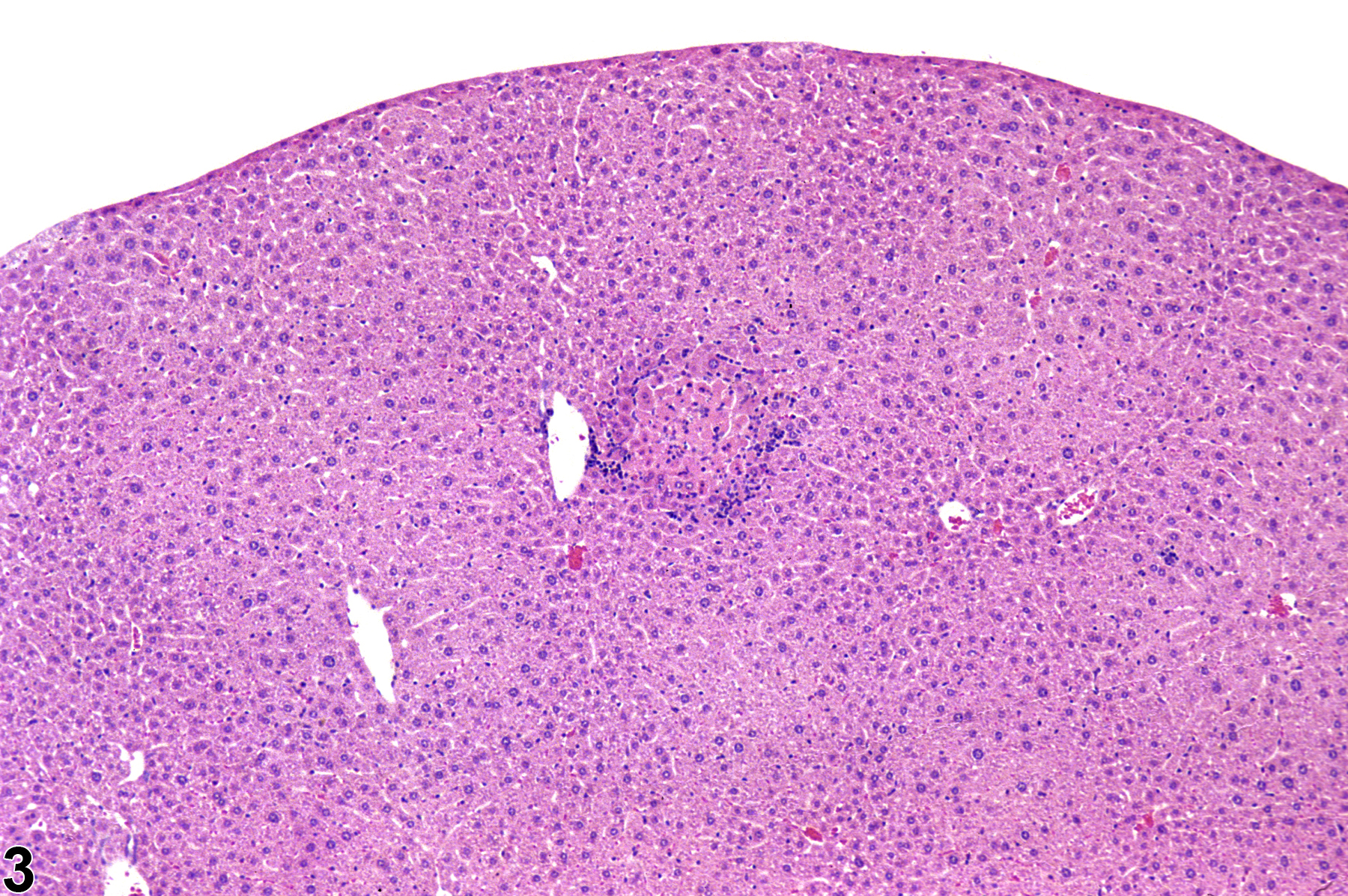
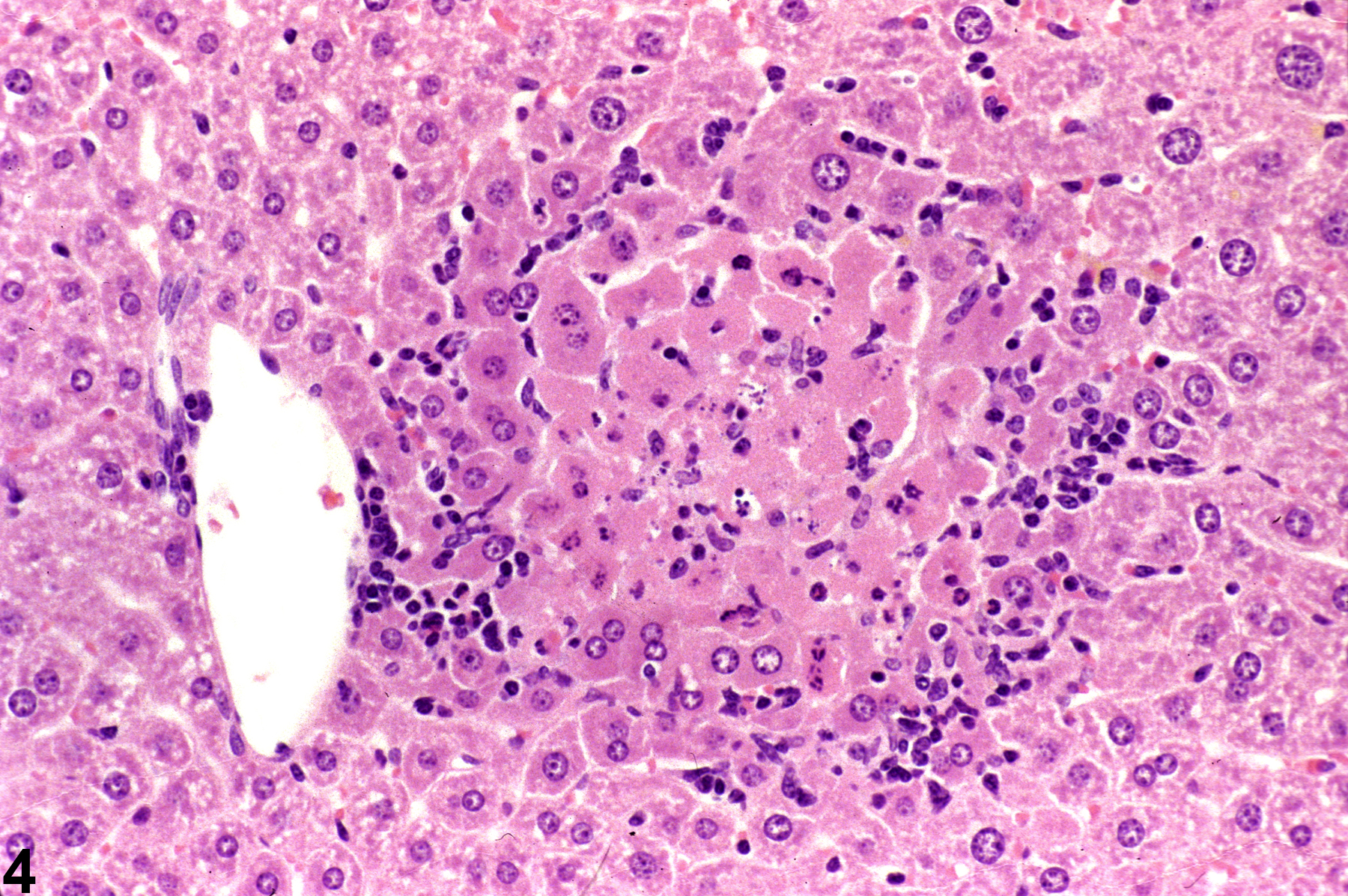
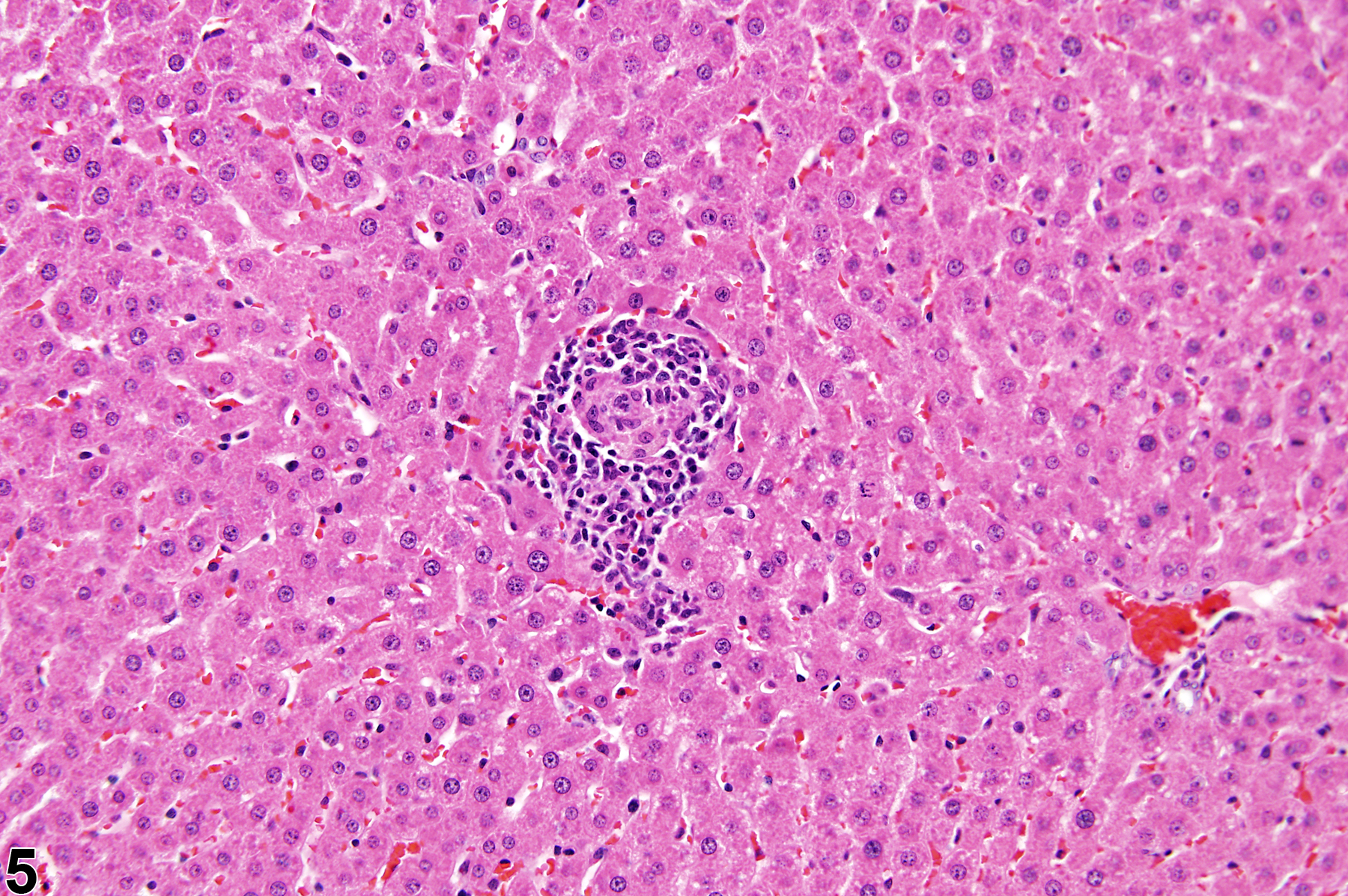
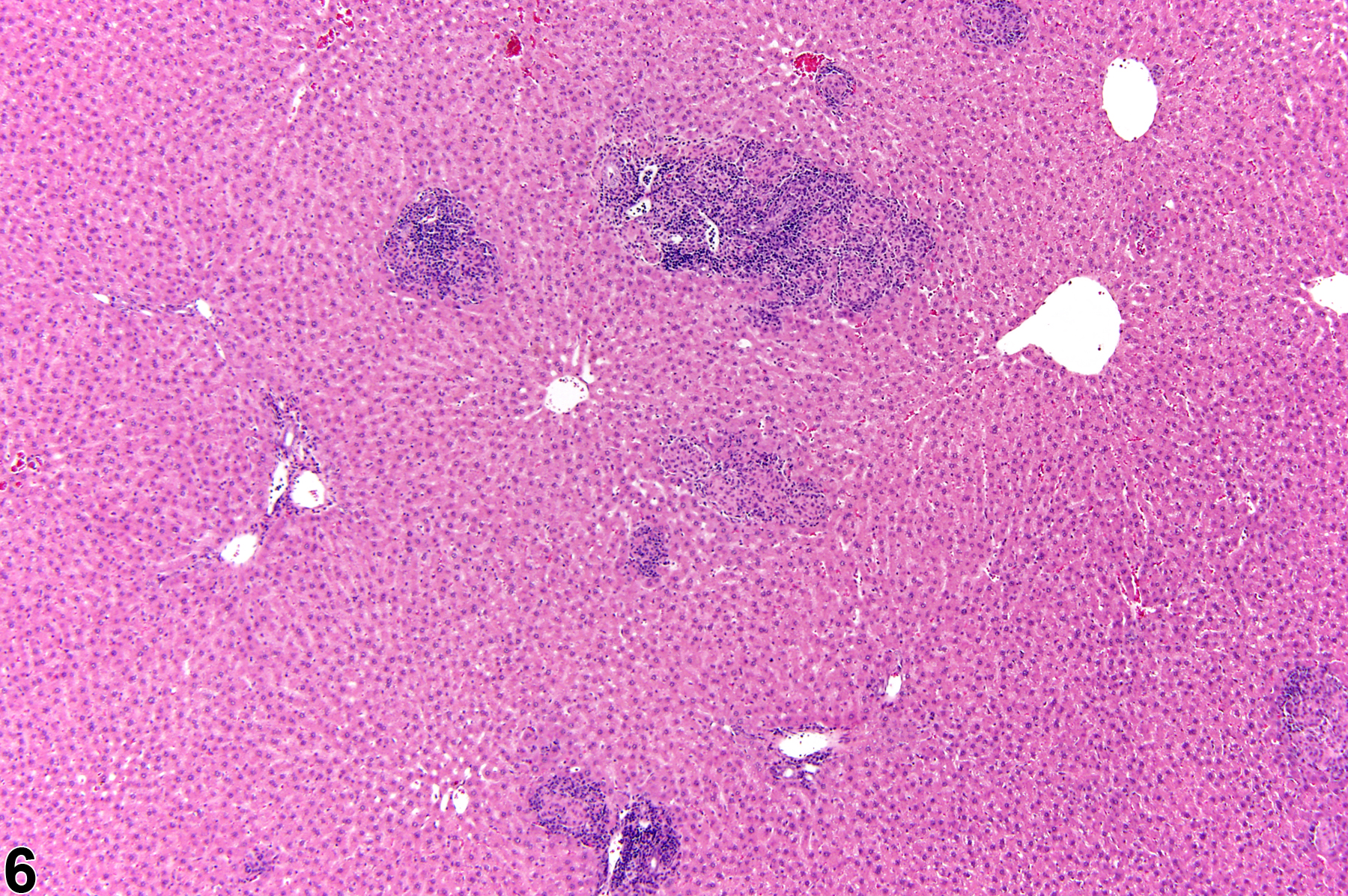
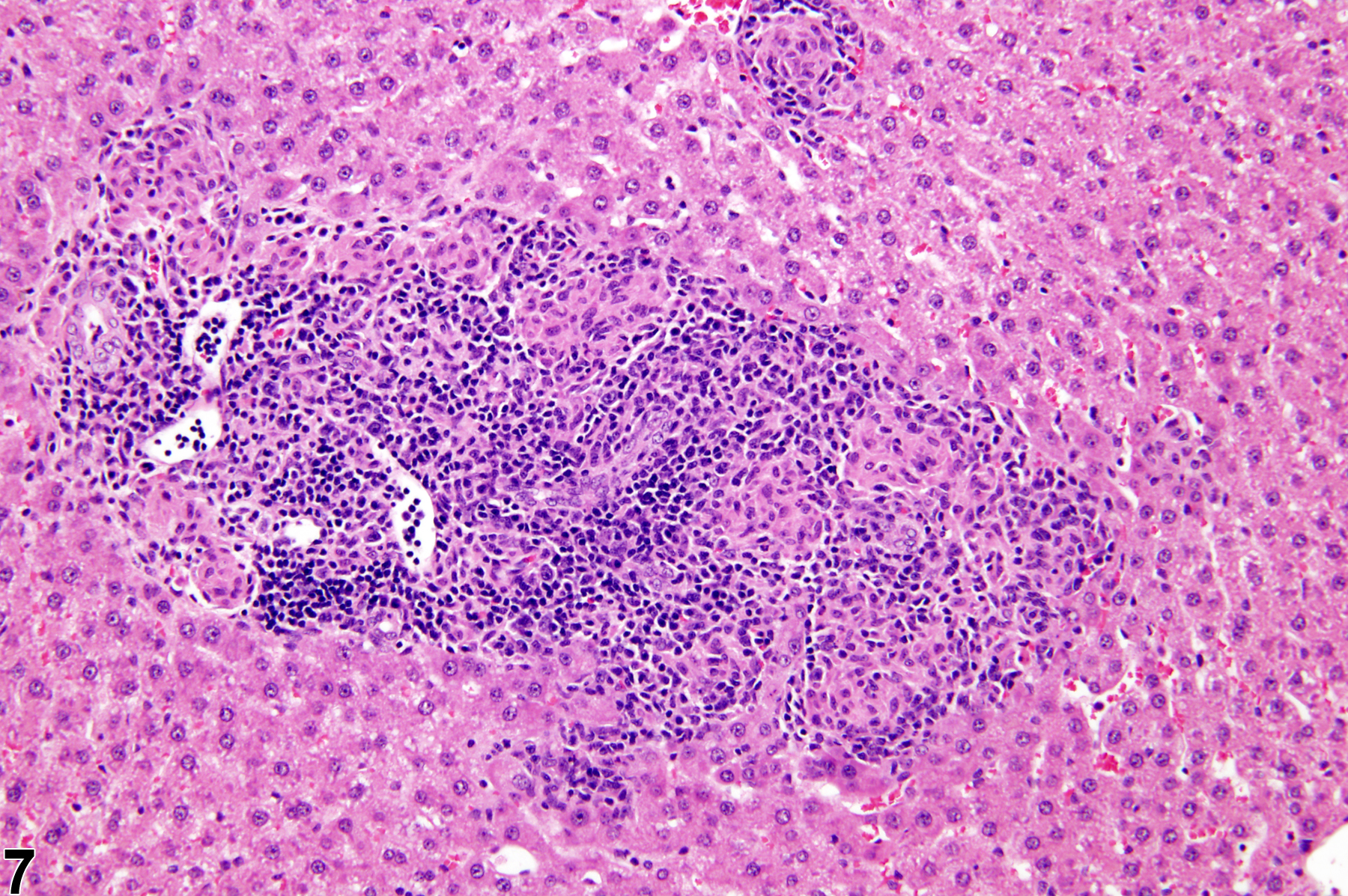
Focal inflammation is the most frequently seen inflammatory lesion of the liver in rodent toxicity studies and is a form of chronic inflammation. In chronic inflammation, lymphocytes predominate. Chronic active inflammation predominately involves lymphocytes as well but also includes a significant number of neutrophils. Both lesions may contain macrophages. However, the term “chronic inflammation” or “chronic active inflammation” may be used only if the lesion differs in some qualitative way from the above description of “inflammation, focal.”
Other less common forms of inflammation include acute, suppurative, and granulomatous inflammation. In acute inflammation, neutrophils are the predominant infiltrating cell, though fewer macrophages and lymphocytes may also be present, as well as evidence of edema or hyperemia. In suppurative inflammation, neutrophils are also the predominant infiltrating cell type but they are aggregated, and many are degenerate. Cell debris from both the resident cell populations and infiltrating leukocytes may also be present in the exudate, as well as proteinaceous fluid containing fibrin, a few macrophages, occasional lymphocytes or plasma cells, and possibly an infectious agent. Grossly, these lesions would be characterized by the presence of pus. The tissue surrounding the exudate may contain fibroblasts, fibrous connective tissue, and mixed inflammatory cells, depending on the chronicity of the lesion. Granulomatous inflammation is another form of chronic inflammation whose diagnosis requires the presence of a significant number of aggregated, large, activated macrophages, epithelioid macrophages, and/or multinucleated giant cells.
The term “cellular infiltrate” has been used to describe the presence of inflammatory cells without other evidence of an inflammatory process (e.g., edema, necrosis or degeneration of cells, or evidence of vascular injury). Such infiltrates of inflammatory cells are generally quite small and may not warrant diagnosis. If significant enough to warrant documentation, accumulations of leukocytes should be diagnosed as “inflammation.” Occasionally, infiltrates of nonleukocytic cells such as mast cells are seen in the liver; these should be diagnosed as “cellular infiltrate.”
If an inflammatory lesion is present that differs in some qualitative manner (morphology, distribution, etc.) from the above description of focal inflammation, one of the other diagnostic terms described above may be used. However, because focal inflammation is such a common lesion and may be present concurrently with another type of inflammation, these lesions must be thoroughly described in the narrative so that they may be clearly differentiated from focal inflammation. The term “inflammation” should be used in reference to leukocytes, but “infiltration, cellular” may be used when nonleukocytic cells are present in the liver (e.g., mast cells).
All of these lesions should be graded based on the extent of liver involvement. Morphologic features such as the distribution pattern can be presented in the pathology narrative. Any associated changes, such as hepatocyte degeneration, necrosis, pigmentation, or vascular changes, may be diagnosed as a separate lesion if warranted by the severity of these associated lesions.
Cattley RC, Popp JA. 2002. Liver. In: Handbook of Toxicologic Pathology (Haschek WM, Rousseaux CG, Wallig MA, eds). Academic Press, San Diego, 2:187-225.
Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Evans JG, Lake BG. 1998. The digestive system II. Hepatobiliary system. In: Target Organ Pathology (Turton J, Hooson J, eds). Taylor and Francis, London, 61-98.
Greaves P. 2007. Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Elsevier, Amsterdam.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Hall WC, Ganaway JR, Rao GN, Peters RL, Allen AM, Luczak JW, Sandberg EM, Quigley BH. 1992. Histopathologic observations in weanling B6C3F1 mice and F344/N rats and their adult parental strains. Toxicol Pathol 20:146-154.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/1475576Harada T, Enomoto A, Boorman GA, Maronpot RR. 1999. Liver and gallbladder. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 119-183.
Harada T, Maronpot RR, Enomoto A, Tamano S, Ward JM. 1996. Changes in the liver and gallbladder. In: Pathobiology of the Aging Mouse (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 2:207-241.
Abstract: http://catalog.hathitrust.org/Record/008994685Hardisty JF, Brix AE. 2005. Comparative hepatic toxicity: Prechronic/chronic liver toxicity in rodents. Toxicol Pathol 33:35-40.
Full Text: http://tpx.sagepub.com/content/33/1/35.full.pdfHaschek WM, Rousseaux CG, Wallig MA. 2010. Fundamentals of Toxicologic Pathology, 2nd ed. Academic Press, San Diego, 197-235.
National Toxicology Program. 1993. NTP TR-443. Toxicology and Carcinogenesis Studies of Oxazepam (CAS No. 604-75-1) in Swiss-Webster and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr443.pdfThoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full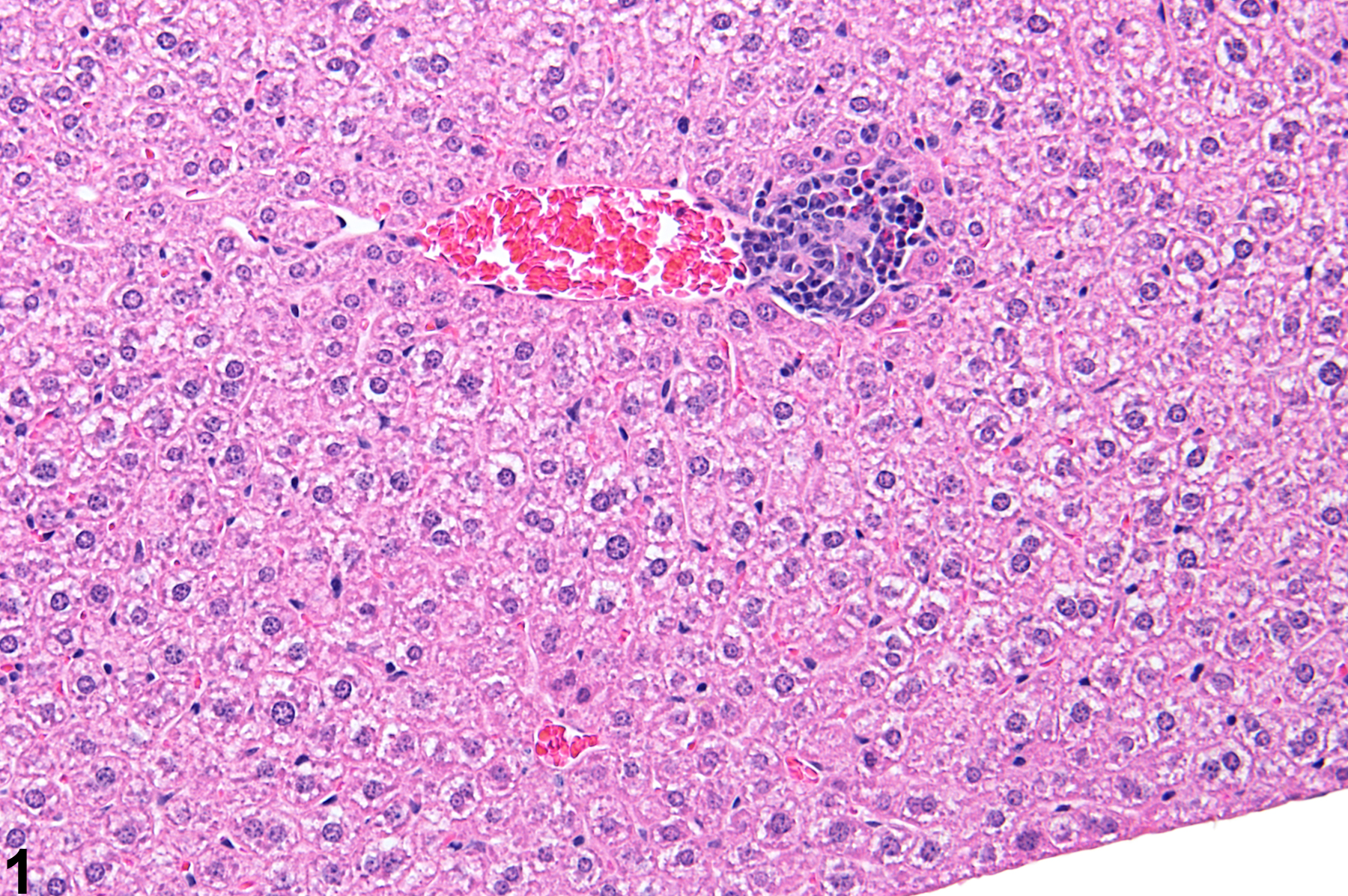
Focal inflammation in a female B6C3F1 mouse from a subchronic study.