Hepatobiliary System
Liver - Necrosis
Narrative
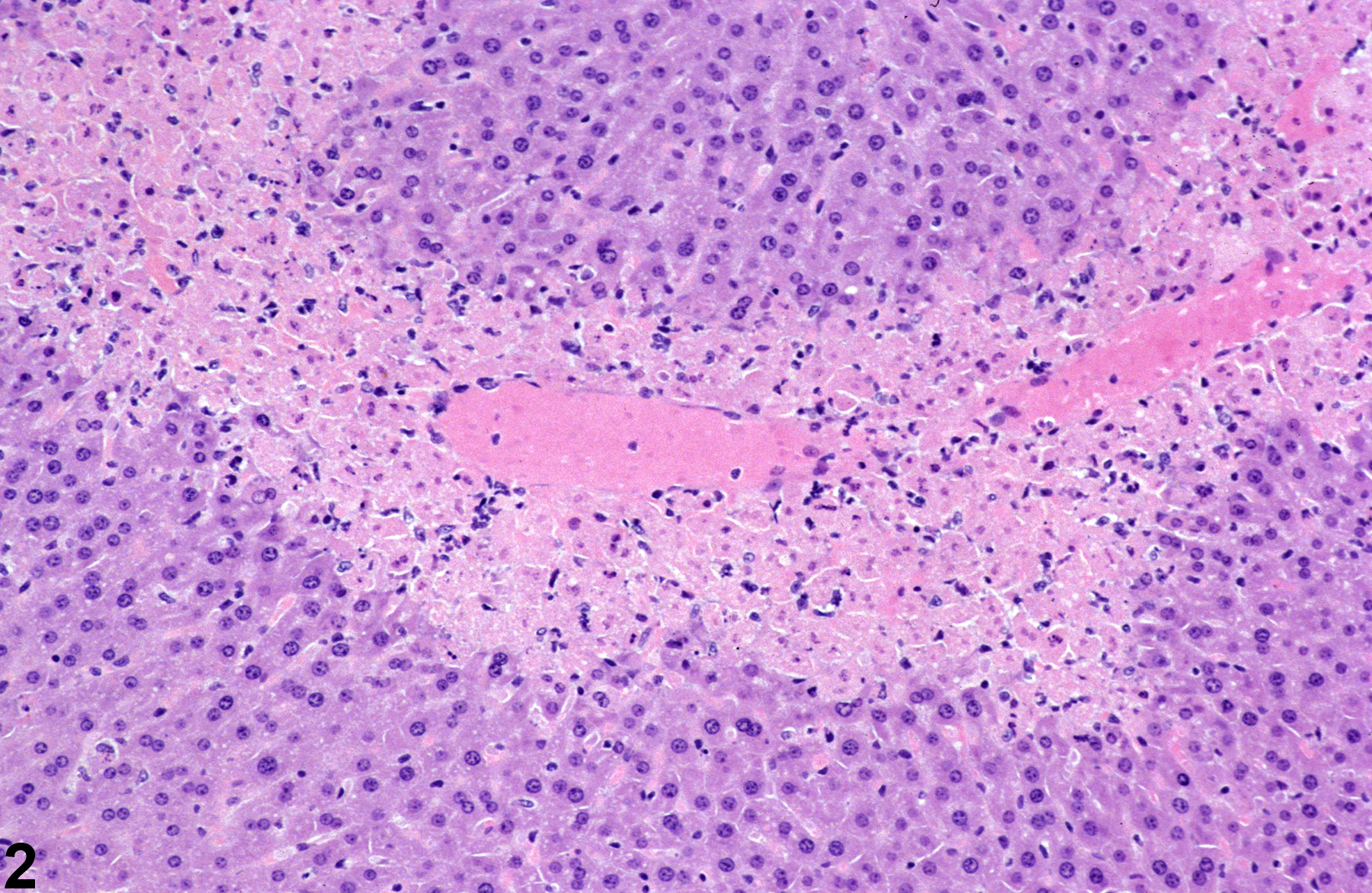
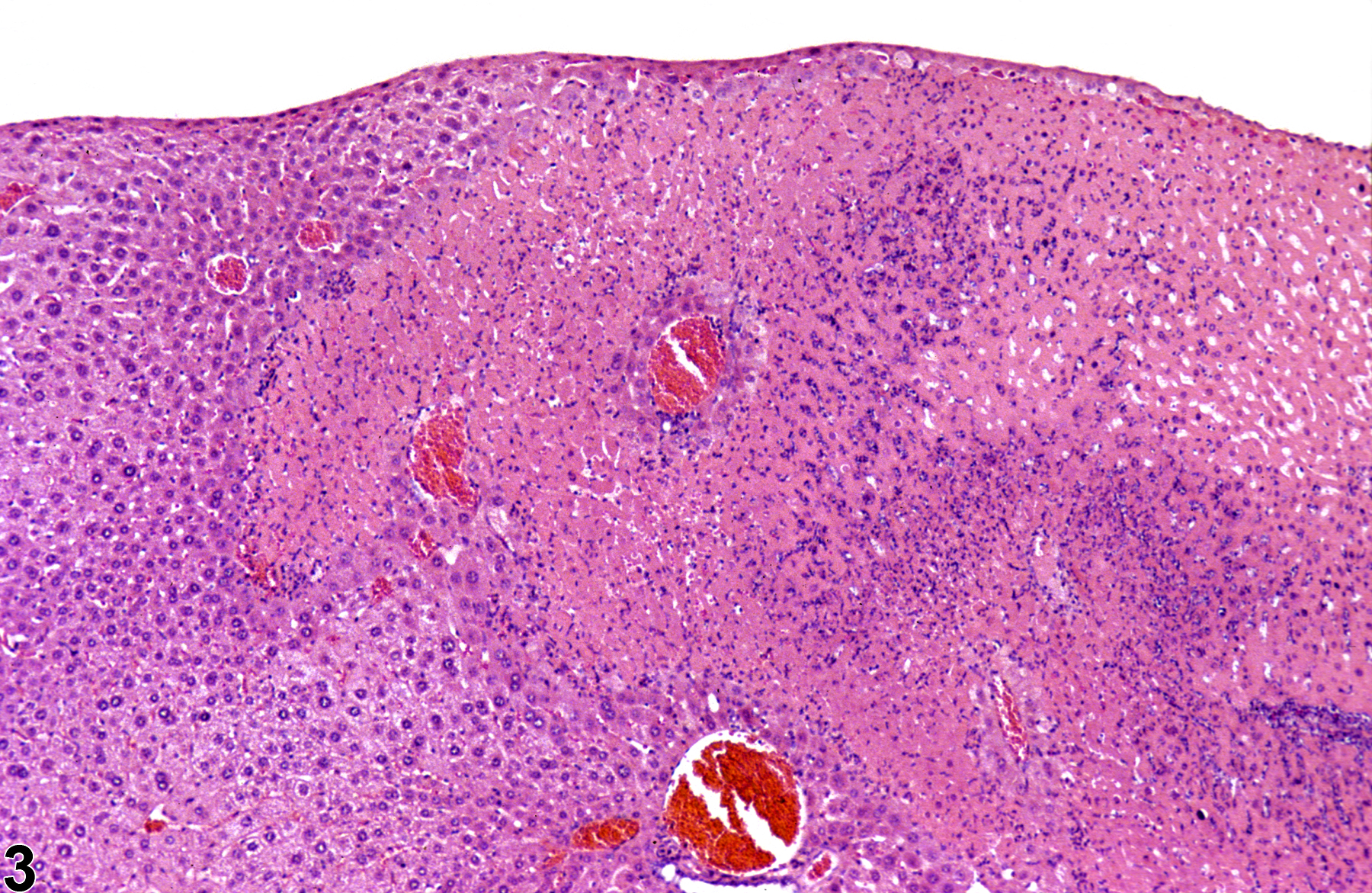
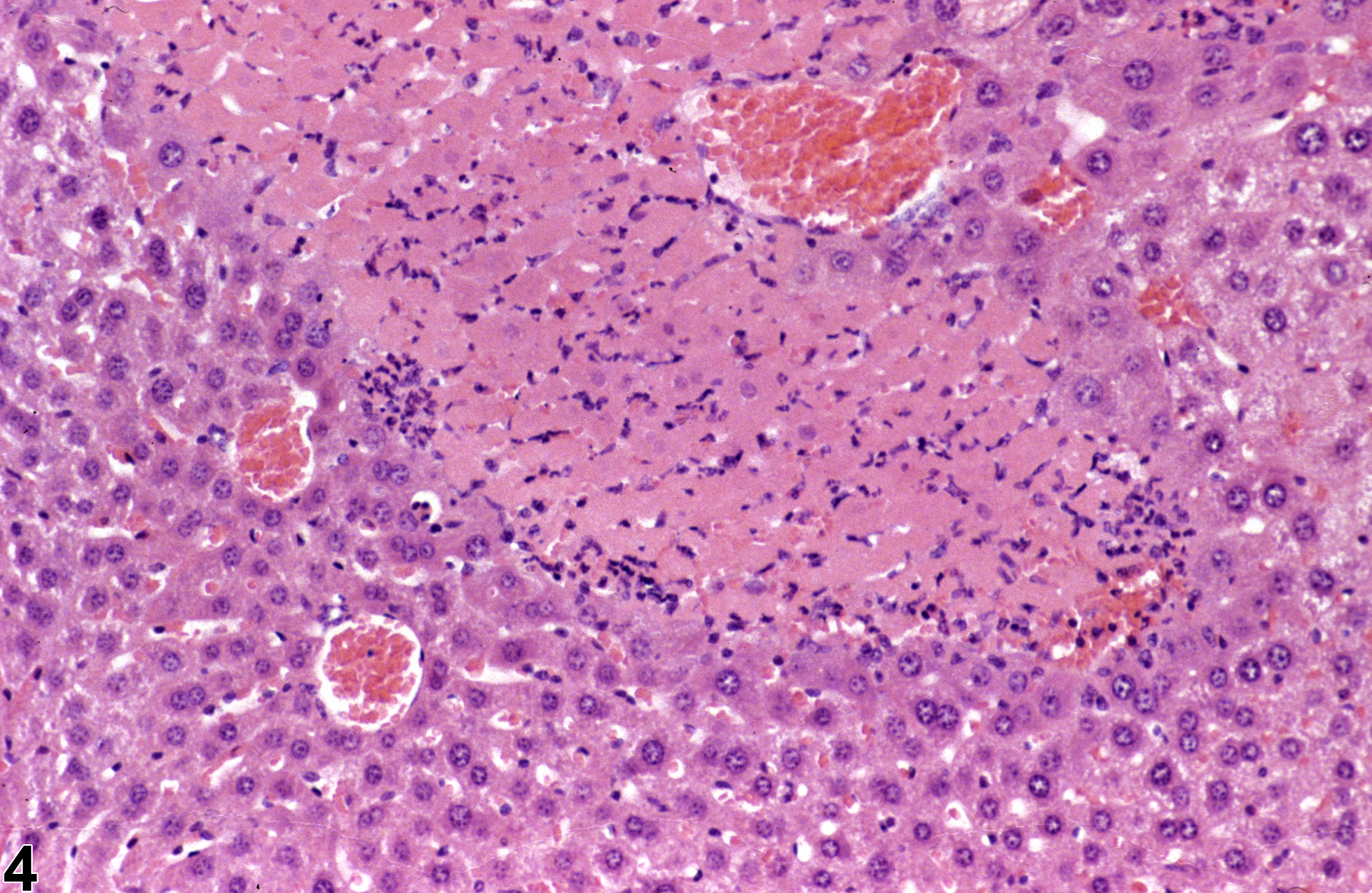
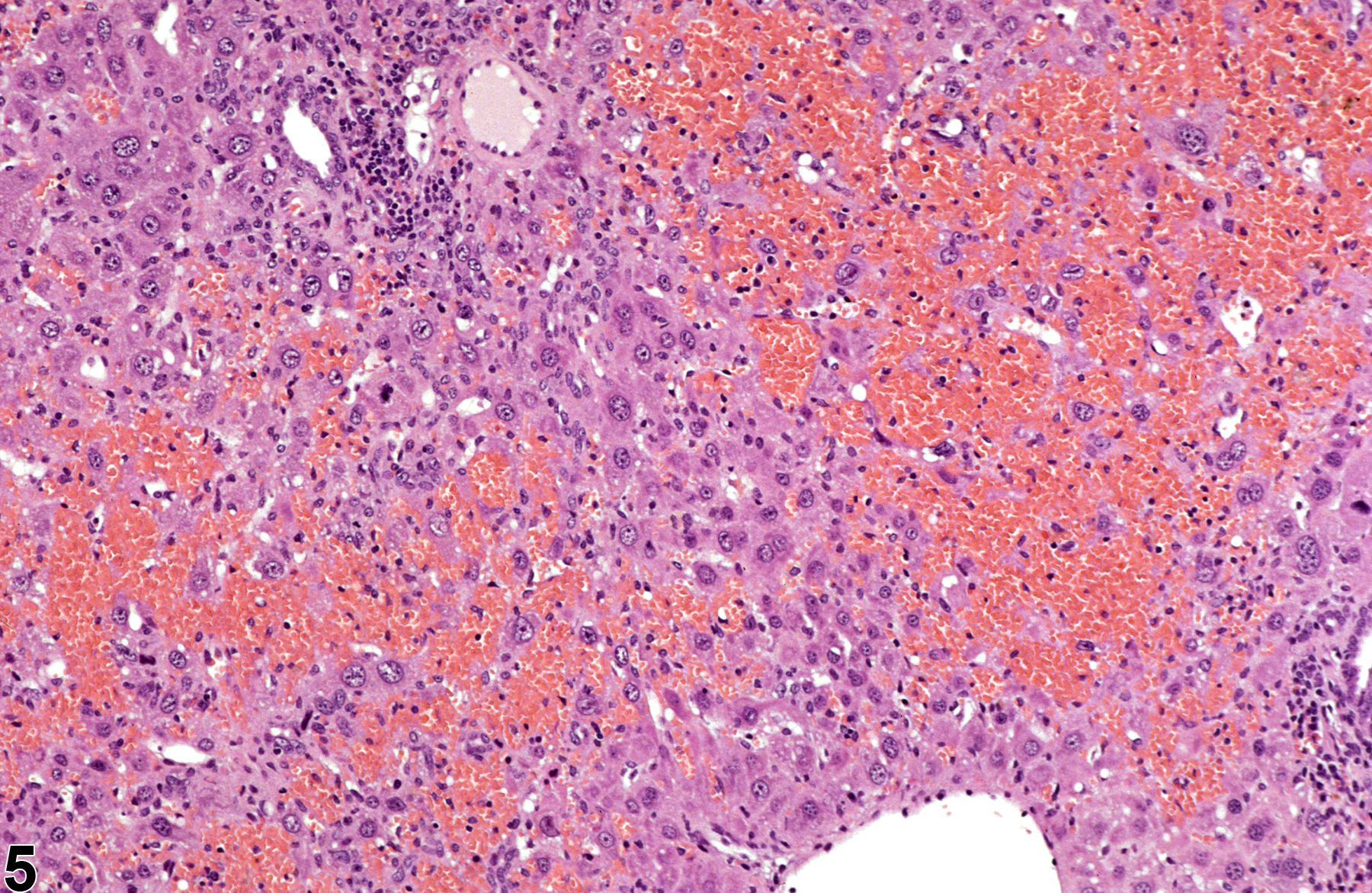
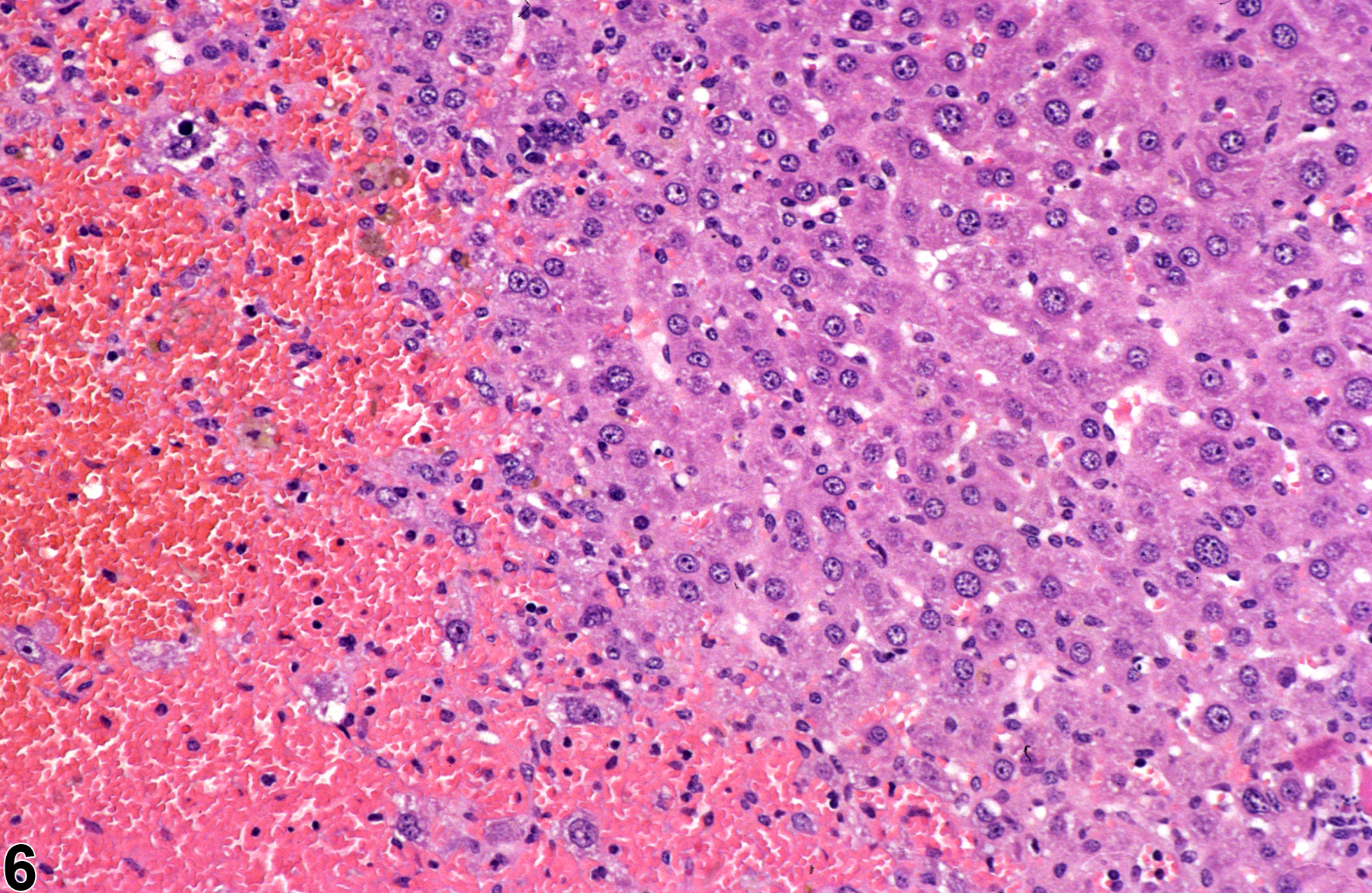
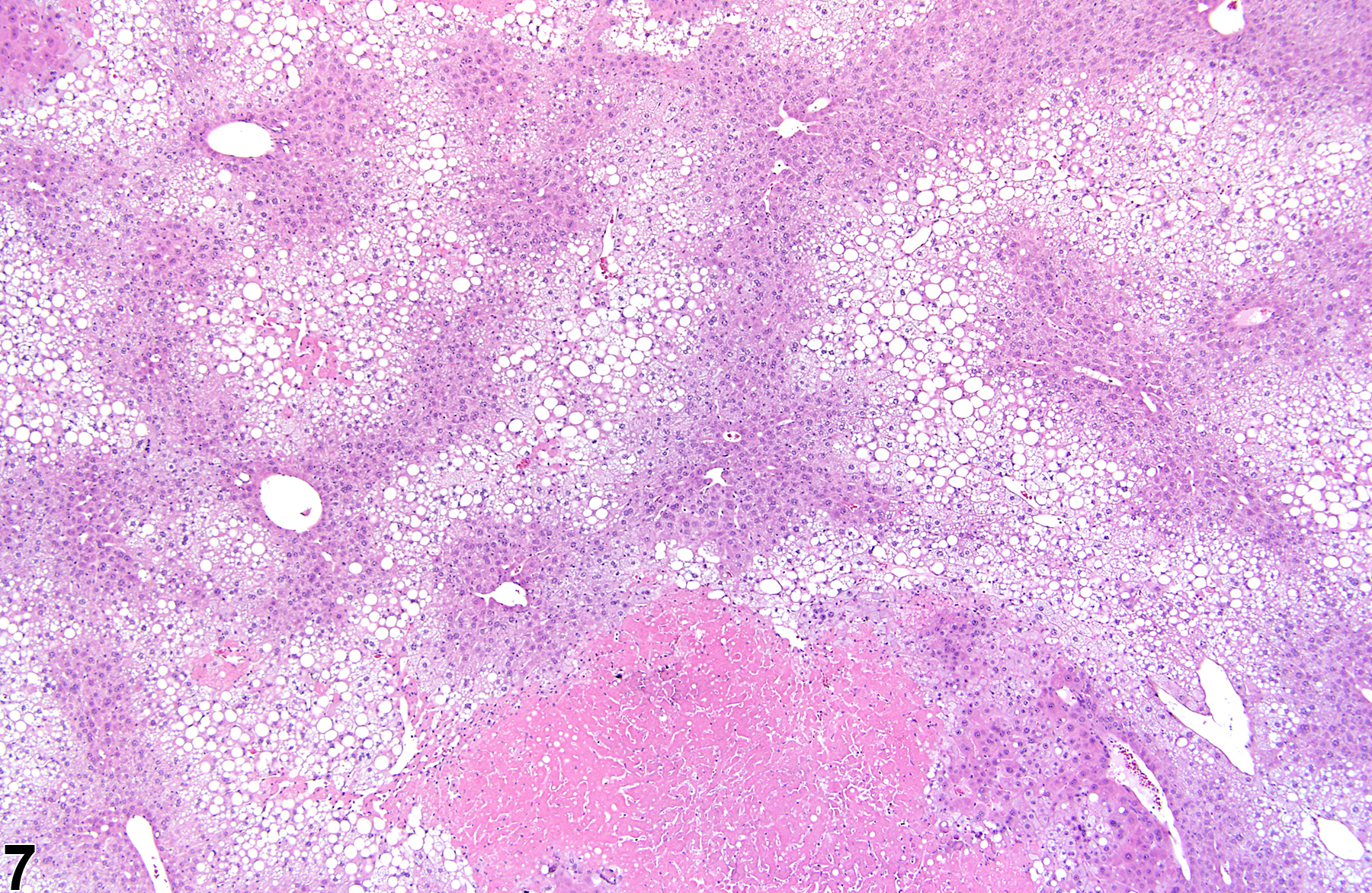
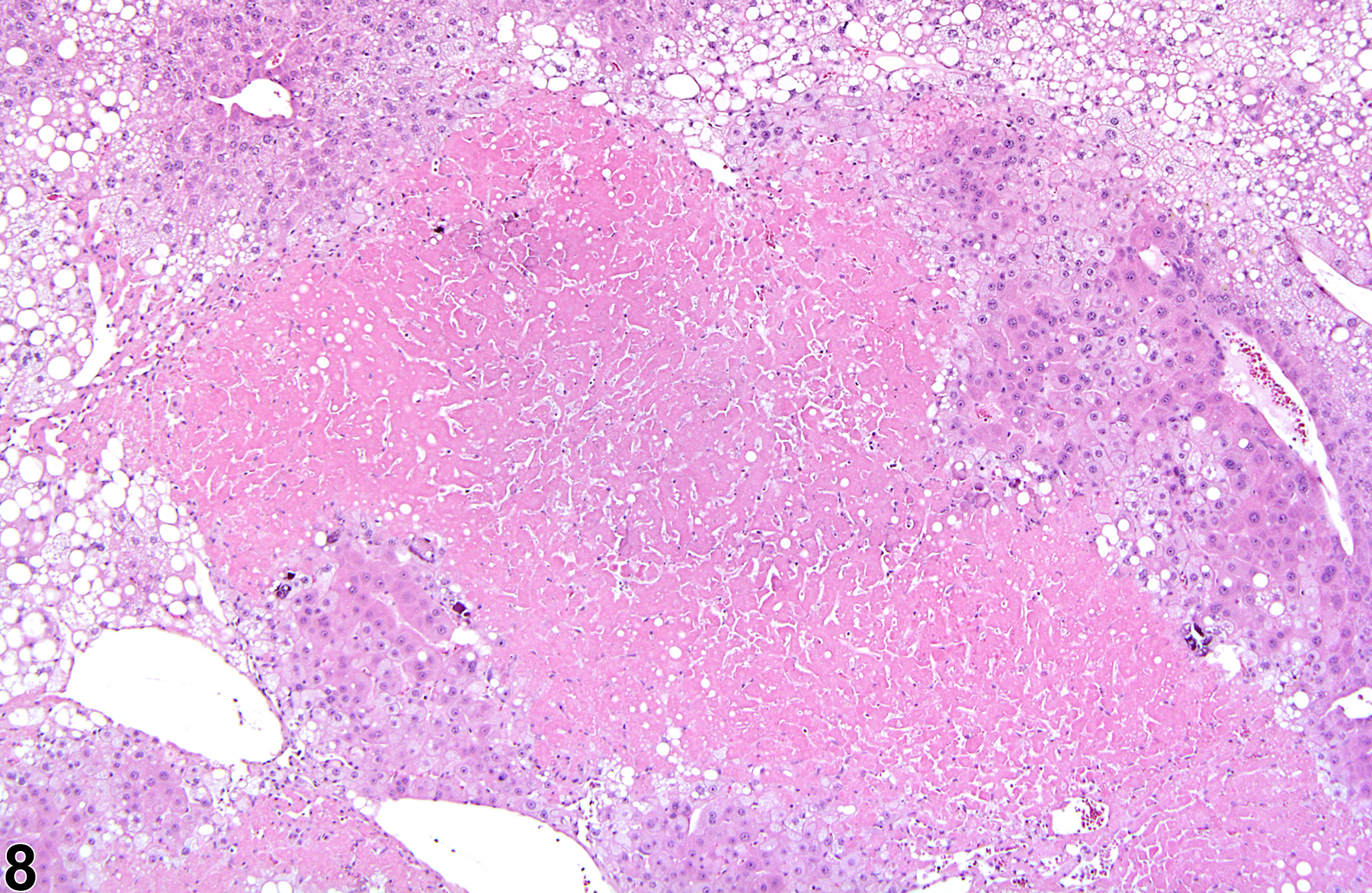
Figure 1 and Figure 2 represent sharply demarcated centrilobular necrosis with loss of hepatocyte cytologic detail. Figure 3 and Figure 4 represent irregular patchy areas of coagulation necrosis with early infiltration of inflammatory cells. There is no distinctive lobular pattern to this necrosis. Figure 5 and Figure 6 represent an example of necrosis characterized by loss of hepatocytes and replacement with erythrocytes. This is an example of hemorrhagic necrosis. Figure 7 and Figure 8 represent focal necrosis associated with fatty change. The patches of necrosis (see Figure 8, bottom center) are hypereosinophilic, and the fatty change is centrilobular with extension well into midlobular areas.
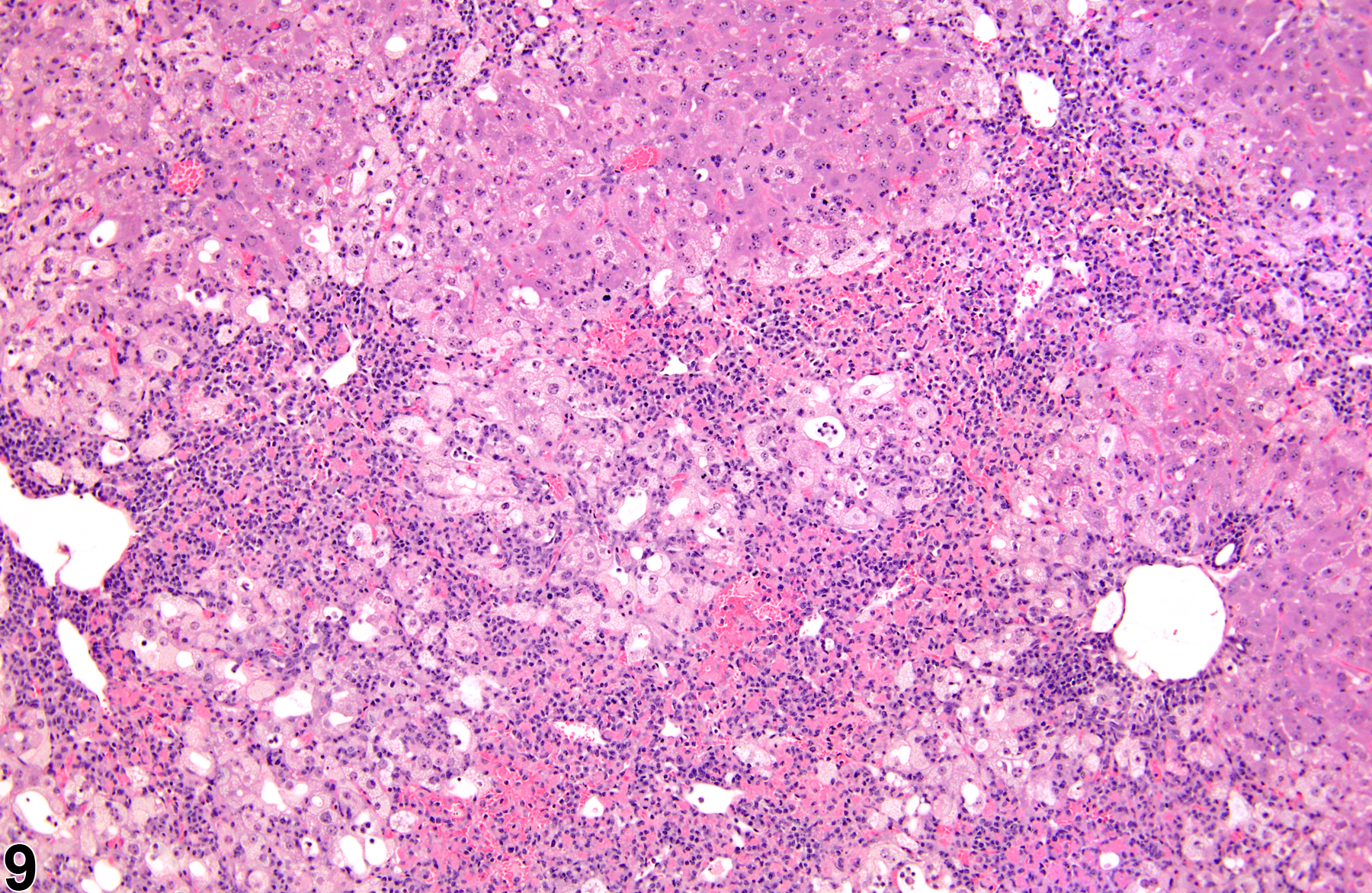
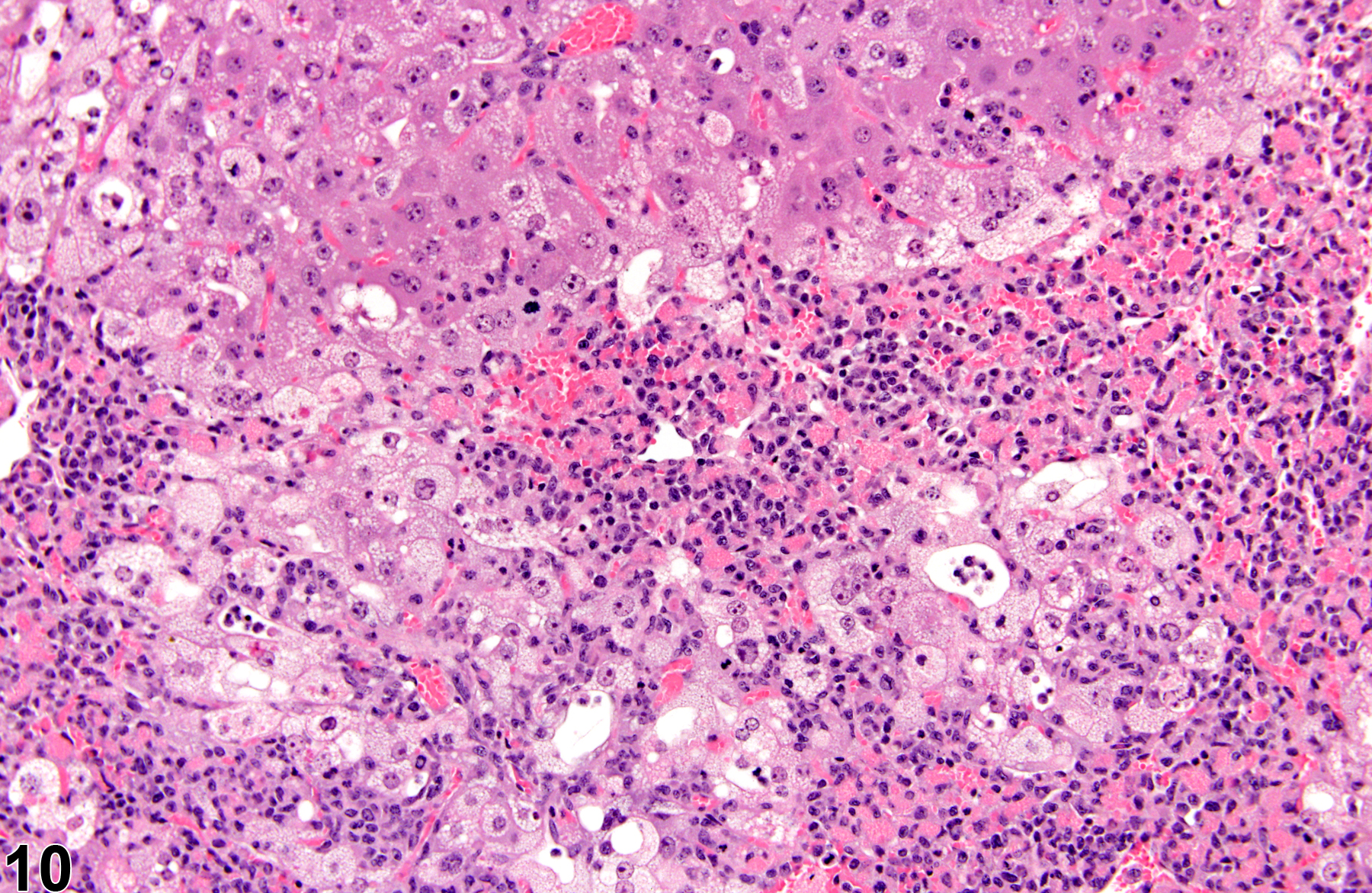
Figure 9 and Figure 10 represent a diagnosis of hepatocyte necrosis accompanied by hepatocyte degeneration, microvesicular fatty change, nuclear pyknosis, and inflammation. “Degeneration” is a term often used to indicate reversible cell or tissue damage and is considered to be a precursor to necrosis. Because the presence of the inflammatory response is extensive, an additional diagnosis of inflammation (cellular infiltrate) to capture the ongoing process with more fidelity may be appropriate.
Greaves P. 2007. Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Elsevier, Amsterdam.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Evans JG, Lake BG. 1998. The digestive system II. Hepatobiliary system. In: Target Organ Pathology (Turton J, Hooson J, eds). Taylor and Francis, London, 61-98.
Harada T, Enomoto A, Boorman GA, Maronpot RR. 1999. Liver and gallbladder. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 119-183.
Hardisty JF, Brix AE. 2005. Comparative hepatic toxicity: Prechronic/chronic liver toxicity in rodents. Toxicol Pathol 33:35-40.
Full Text: http://tpx.sagepub.com/content/33/1/35.full.pdfHaschek WM, Rousseaux CG, Wallig MA. 2010. Fundamentals of Toxicologic Pathology, 2nd ed. Academic Press, San Diego, 197-235.
National Toxicology Program. 1993. NTP TR-394. Toxicology and Carcinogenesis Studies of Acetaminophen (CAS No. 103-90-2) in F344 Rats and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr394.pdfNational Toxicology Program. 1998. NTP TR-468. Toxicology and Carcinogenesis Studies of Oxazepam (CAS No. 604-75-1) in Swiss-Webster and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr468.pdfNational Toxicology Program. 2012. NTP TR-571. Toxicology and Carcinogenesis Studies of Kava Kava Extract (CAS No. 9000-38-8) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Full Text: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr571.pdfThoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full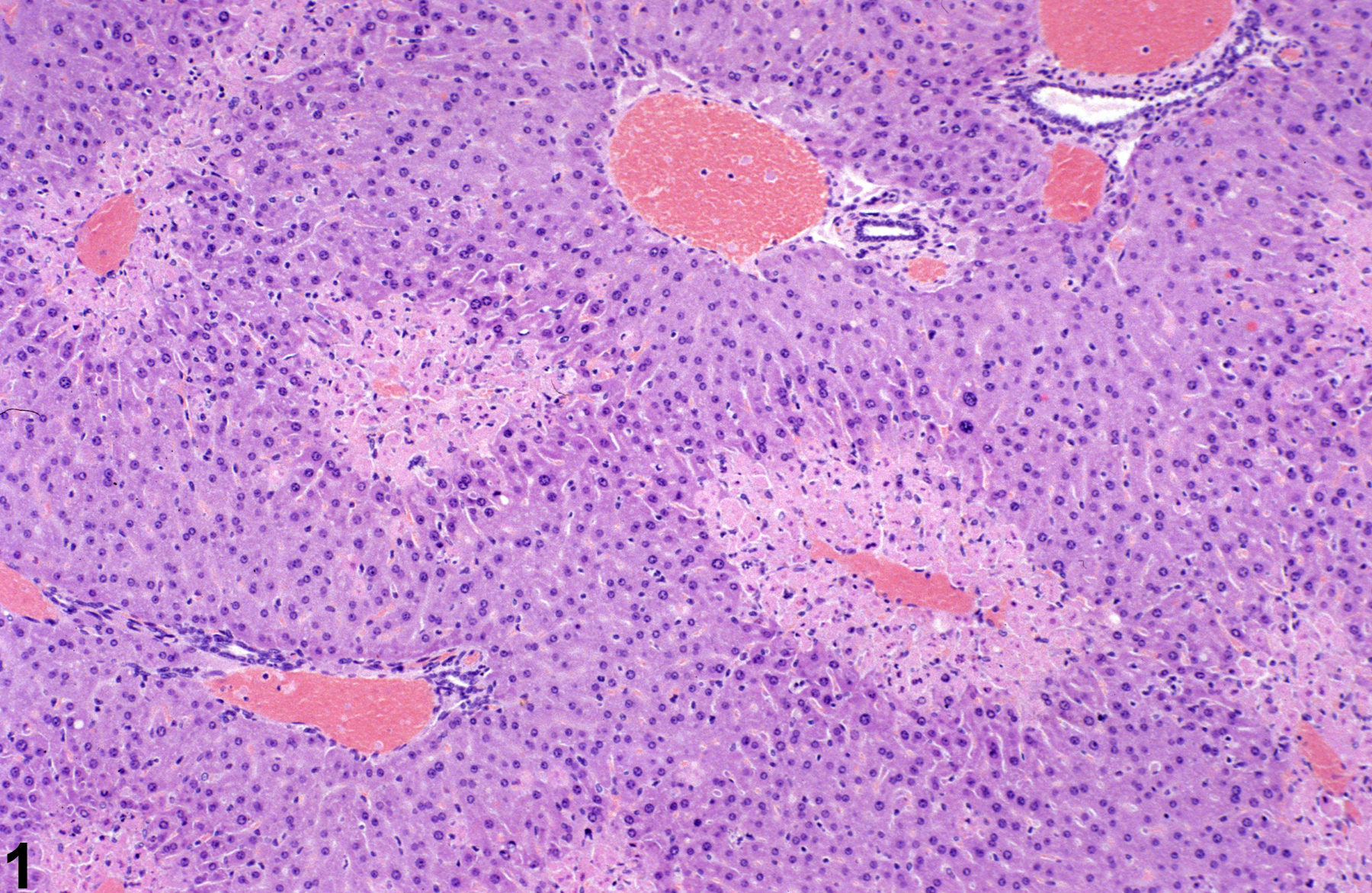
Necrosis-sharply demarcated centrilobular necrosis in a male B6C3F1 mouse from a subchronic study.