Immune System
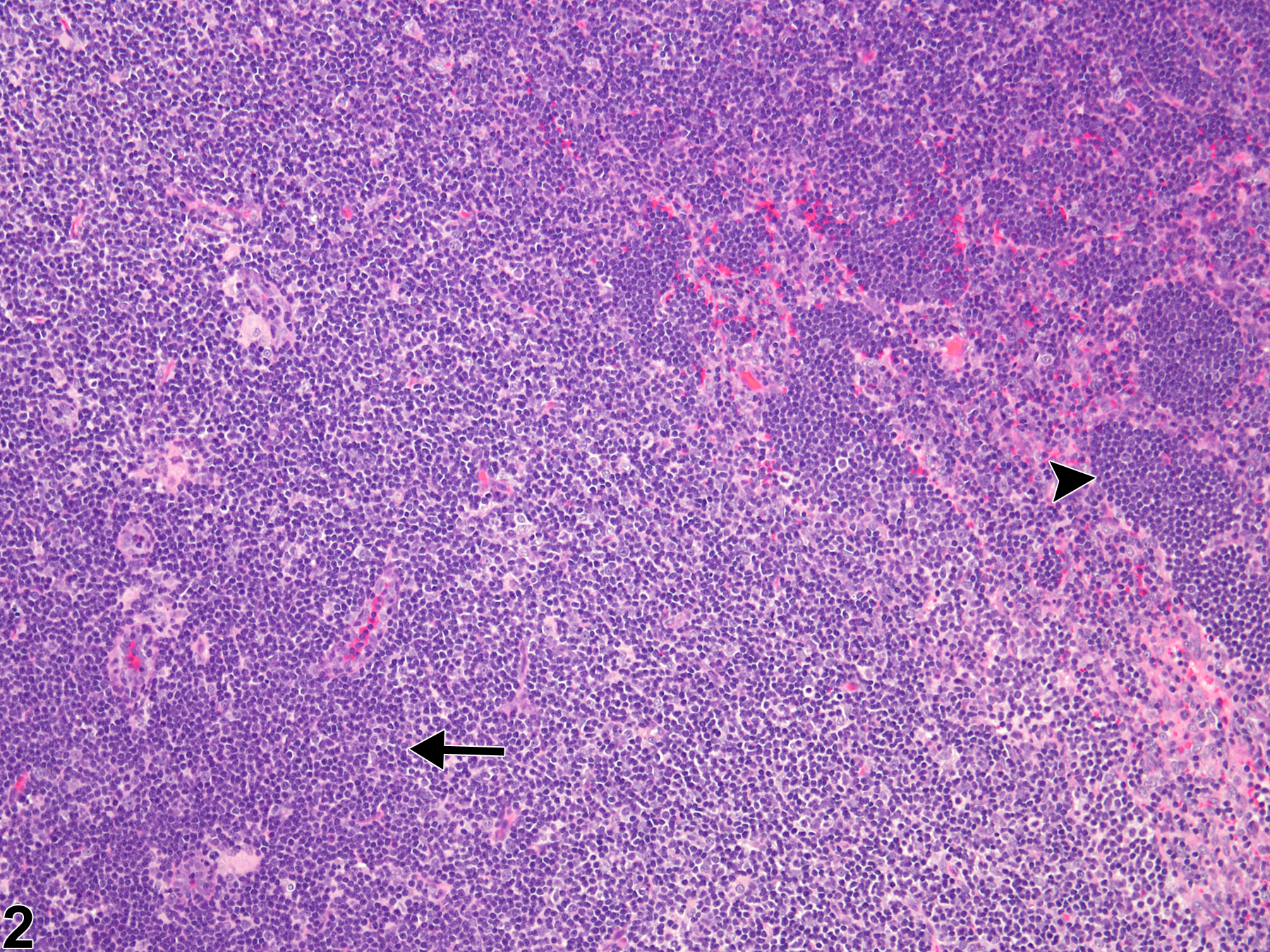
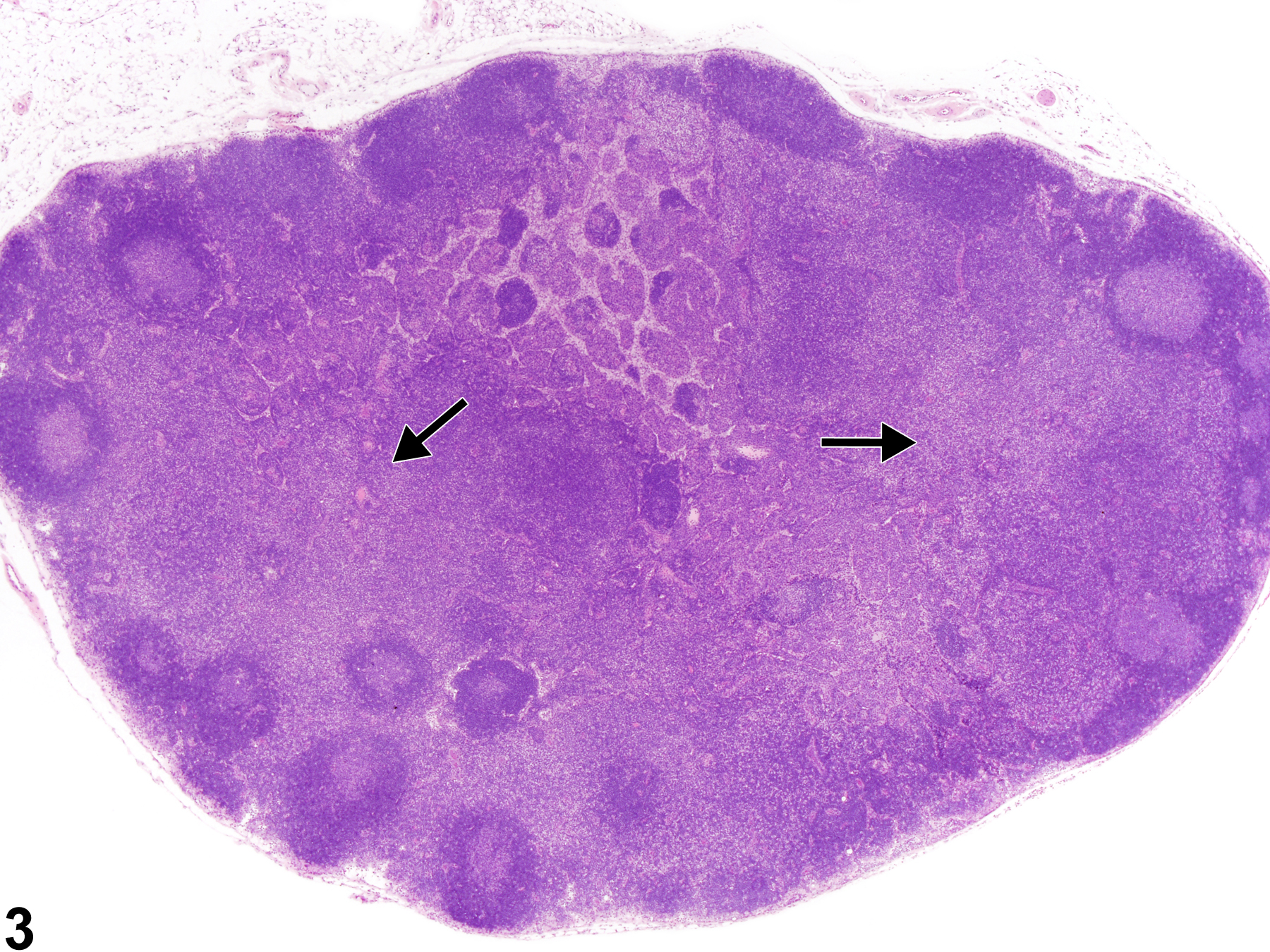
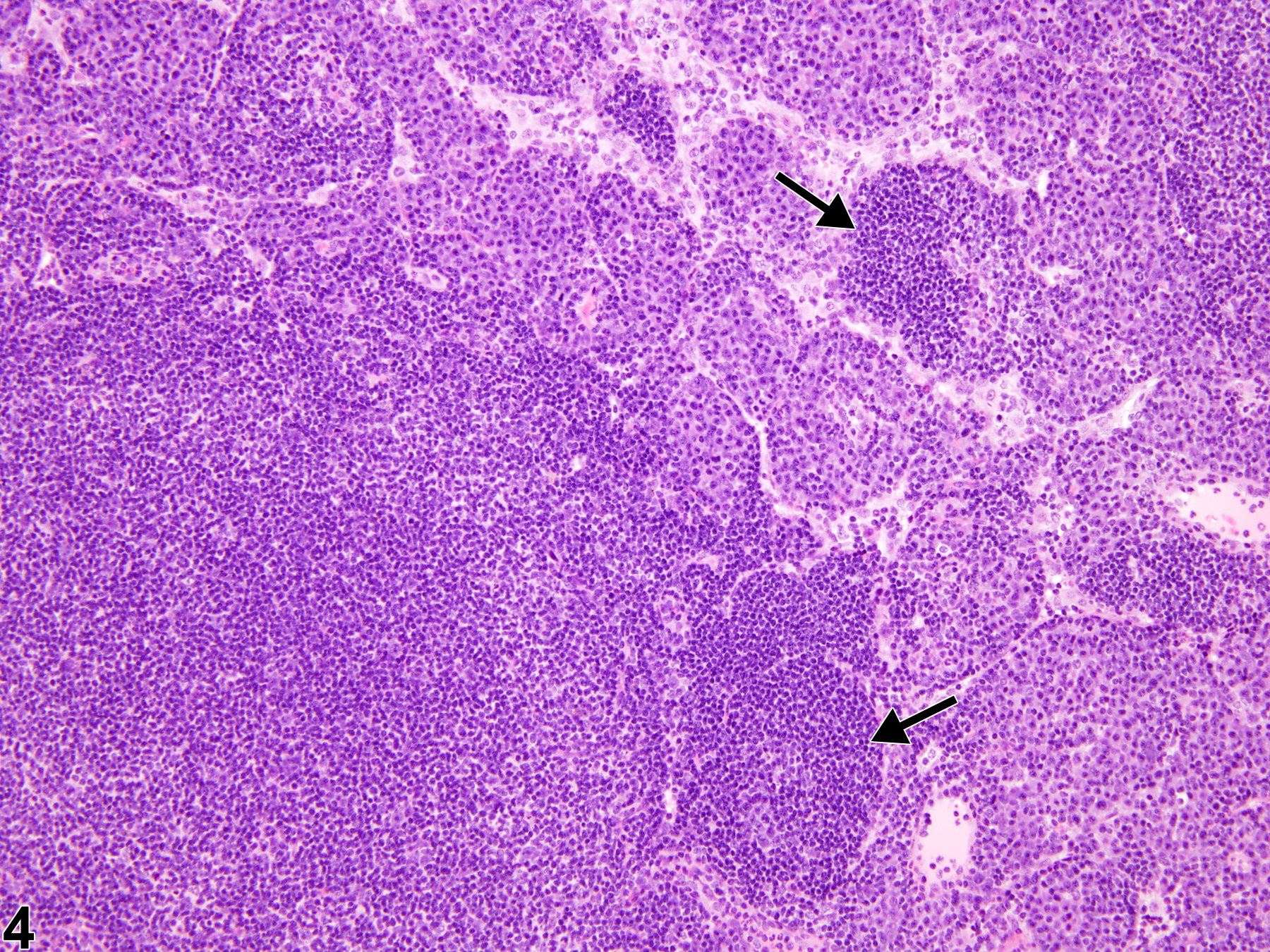
Lymph Node - Hyperplasia, Lymphocyte
Narrative
Elmore SA. 2006. Enhanced histopathology of the lymph nodes. Toxicol Pathol 34:634-647.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1783683/Elmore SA. 2006. Histopathology of the lymph nodes. Toxicol Pathol 34:425-454.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1892634/Frith CH, Ward JM, Chandra M, Losco PE. 2000. Non-proliferative lesions of the hematopoietic system in rats. In: Guides for Toxicologic Pathology.TP/ARP/AFIP, Washington, DC.
Full Text: https://www.toxpath.org/docs/SSNDC/HematopoieticNonprolifRat.pdfNational Toxicology Program. 2008. NTP TR-546. Toxicology and Carcinogenesis Studies of Sodium Dichromate Dihydrate (CAS No. 7789-12-0) in F344/N Rats and B6C3F1 Mice (Drinking Water Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/29323Stefanski SA, Elwell MR, Stromberg PC. 1990. Spleen, lymph nodes, and thymus. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 369-394.
Ward JM, Mann PC, Morishima H, Frith CH. 1999. Thymus, spleen, and lymph nodes. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, Vienna, IL, 333-360.
Ward JM, Rehg JE, Morse HC III. 2012. Differentiation of rodent immune and hematopoietic system reactive lesions from neoplasia. Toxicol Pathol 40:425-434.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/22215512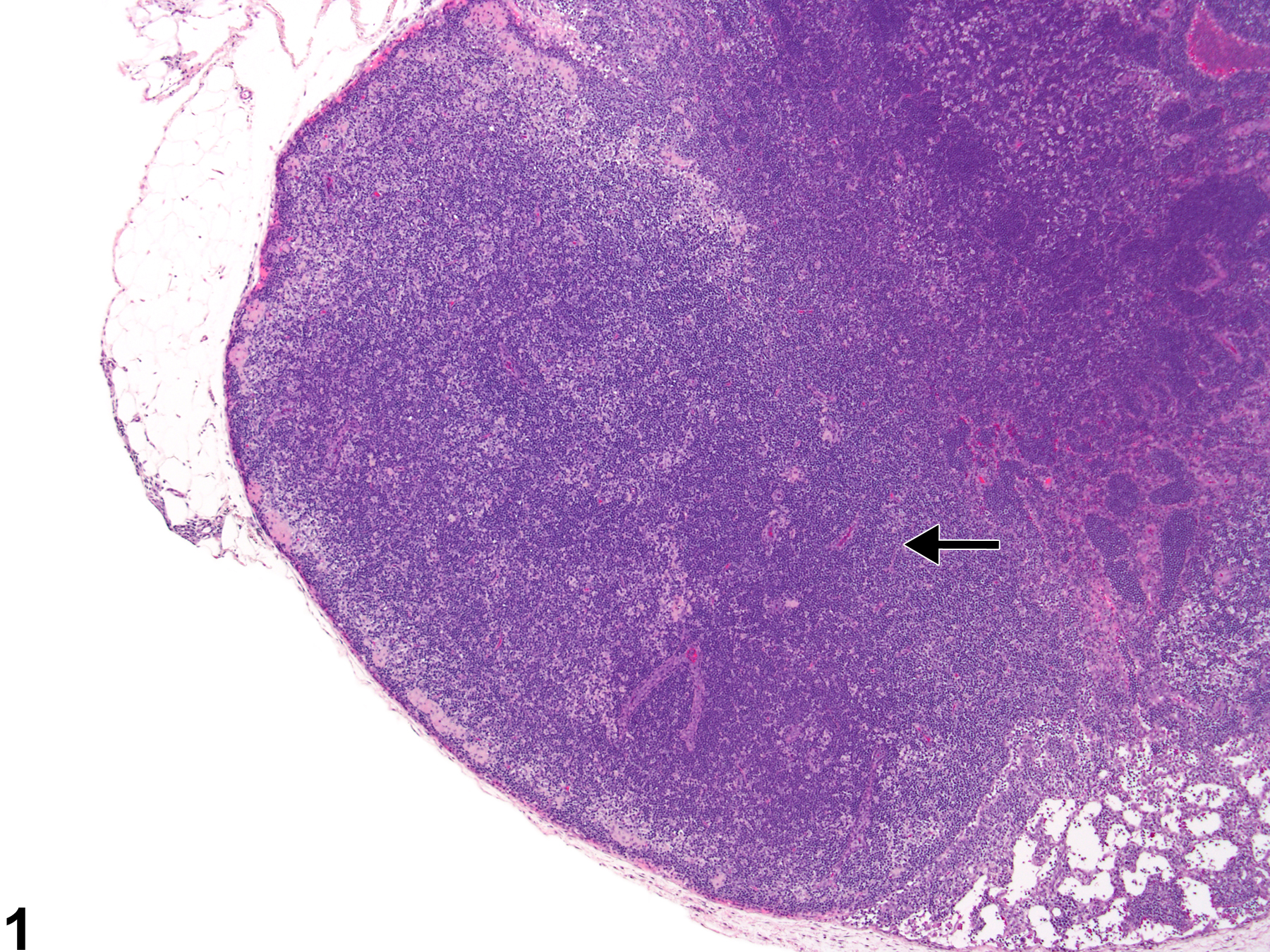
Lymph node - Hyperplasia, Lymphocyte in a male F344/N rat from a subchronic study. The lymph node paracortex is expanded by increased numbers of lymphocytes (arrow).