Immune System
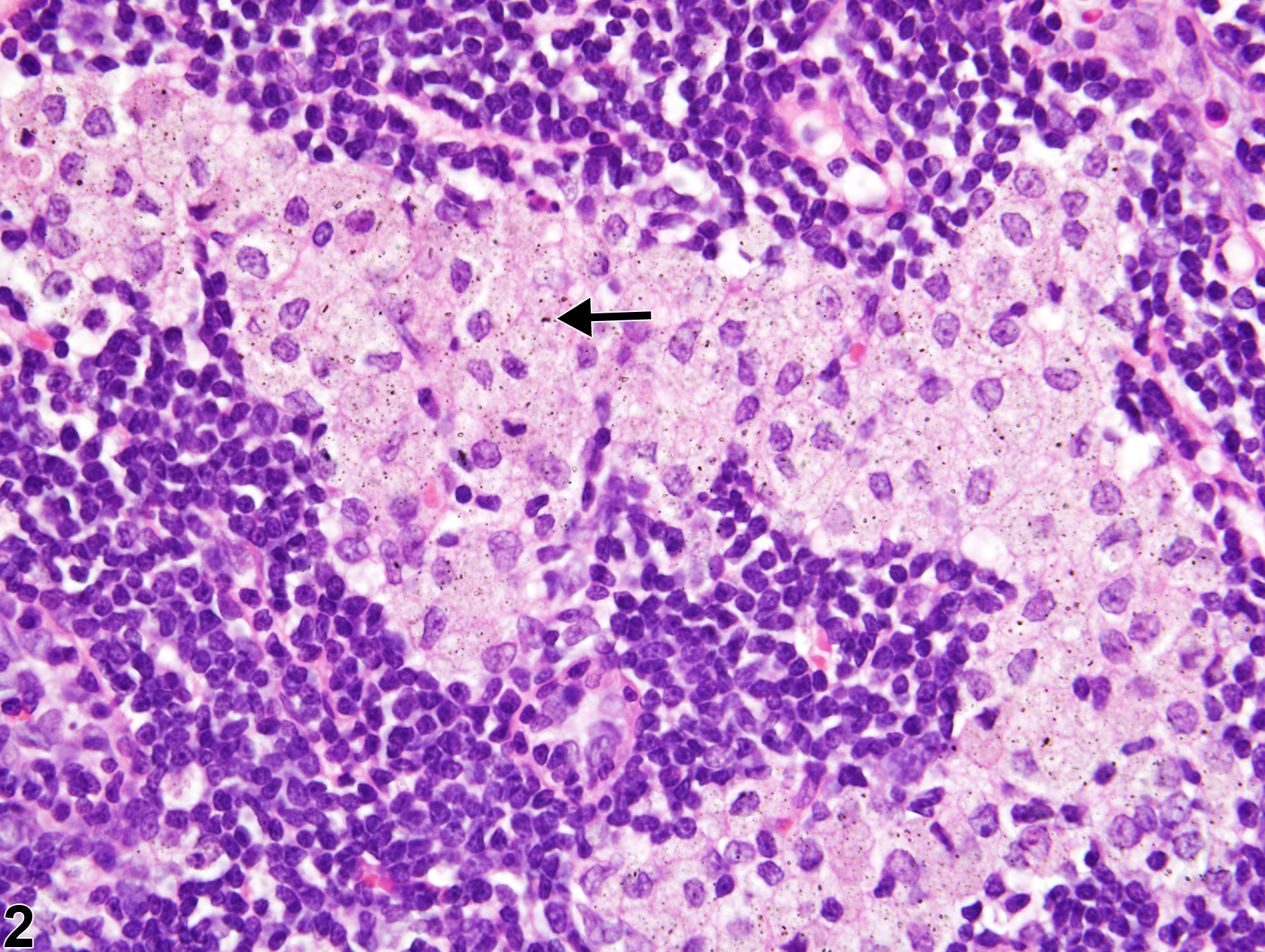
Lymph Node - Inflitration, Cellular, Histiocyte
Narrative
Elmore SA. 2006. Enhanced histopathology of the lymph nodes. Toxicol Pathol 34:634-647.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1783683/Elmore SA. 2006. Histopathology of the lymph nodes. Toxicol Pathol 34:425-454.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1892634/Frith CH, Ward JM, Brown RH, Tyler RD, Chandra M, Stromberg PC. 1996. Proliferative lesions of the hematopoietic and lymphatic systems in rats. HL-1. In: Guides for Toxicologic Pathology. STP/ARP/AFIP, Washington, DC.
Frith CH, Ward JM, Chandra M, Losco PE. 2000. Non-proliferative lesions of the hematopoietic system in rats. In: Guides for Toxicologic Pathology.TP/ARP/AFIP, Washington, DC.
Full Text: https://www.toxpath.org/docs/SSNDC/HematopoieticNonprolifRat.pdfNational Toxicology Program. 2010. 39-Week Study (No. C20213) of Abrasive Blasting Agents (CAS No. Blastingsand) in Harlan Sprague Dawley Rats (Inhalation Studies). NTP, Research Triangle Park, NC.
Ward JM, Mann PC, Morishima H, Frith CH. 1999. Thymus, spleen, and lymph nodes. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, Vienna, IL, 333-360.
Ward JM, Rehg JE, Morse HC III. 2012. Differentiation of rodent immune and hematopoietic system reactive lesions from neoplasia. Toxicol Pathol 40:425-434.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/22215512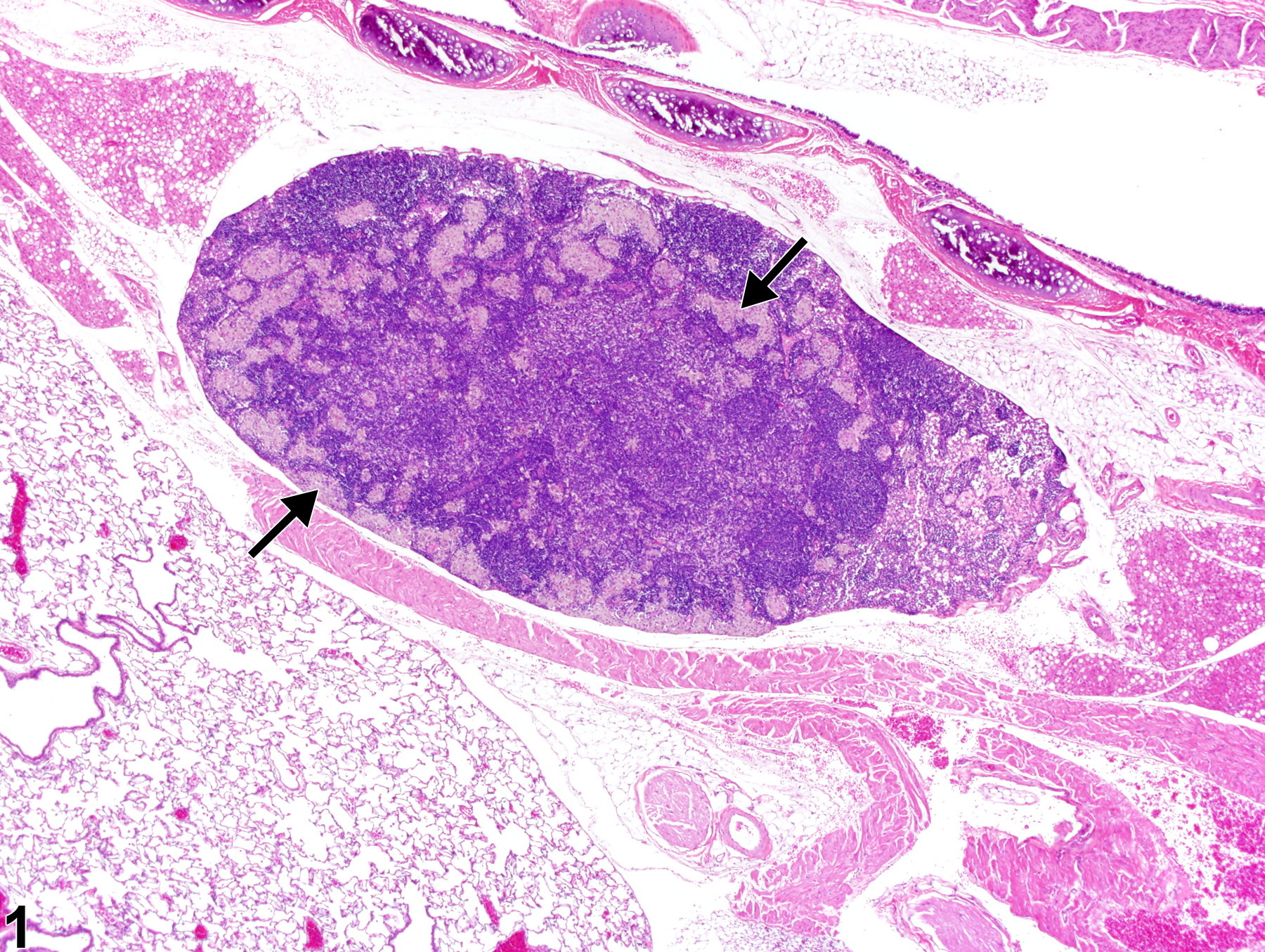
Lymph node - Infiltrate, Cellular, Histiocyte in a male Harlan Sprague Dawley rat from a subchronic study. Increased numbers of histiocytes are present within the paracortical and subcapsular sinuses (arrows).