Reproductive System, Male
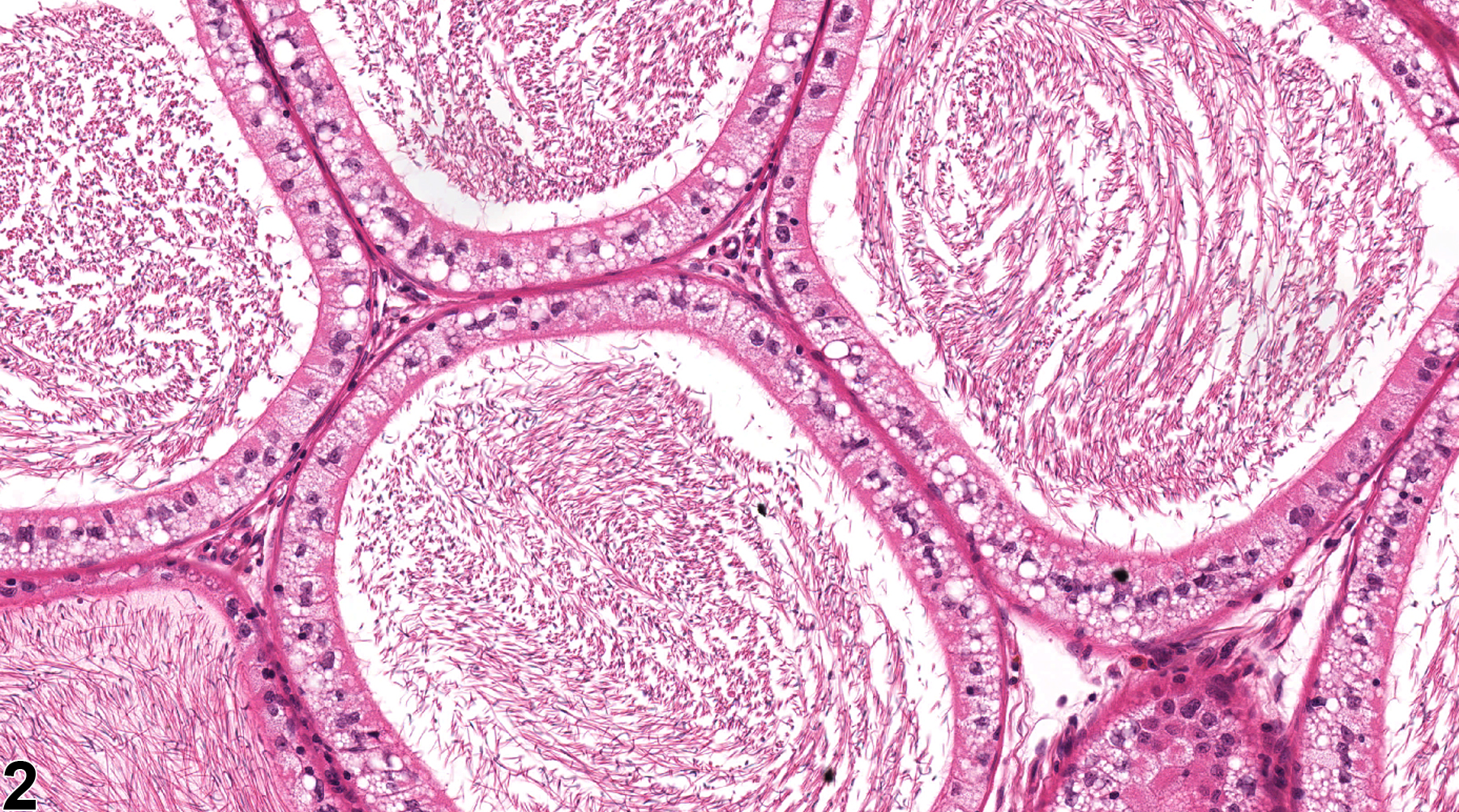
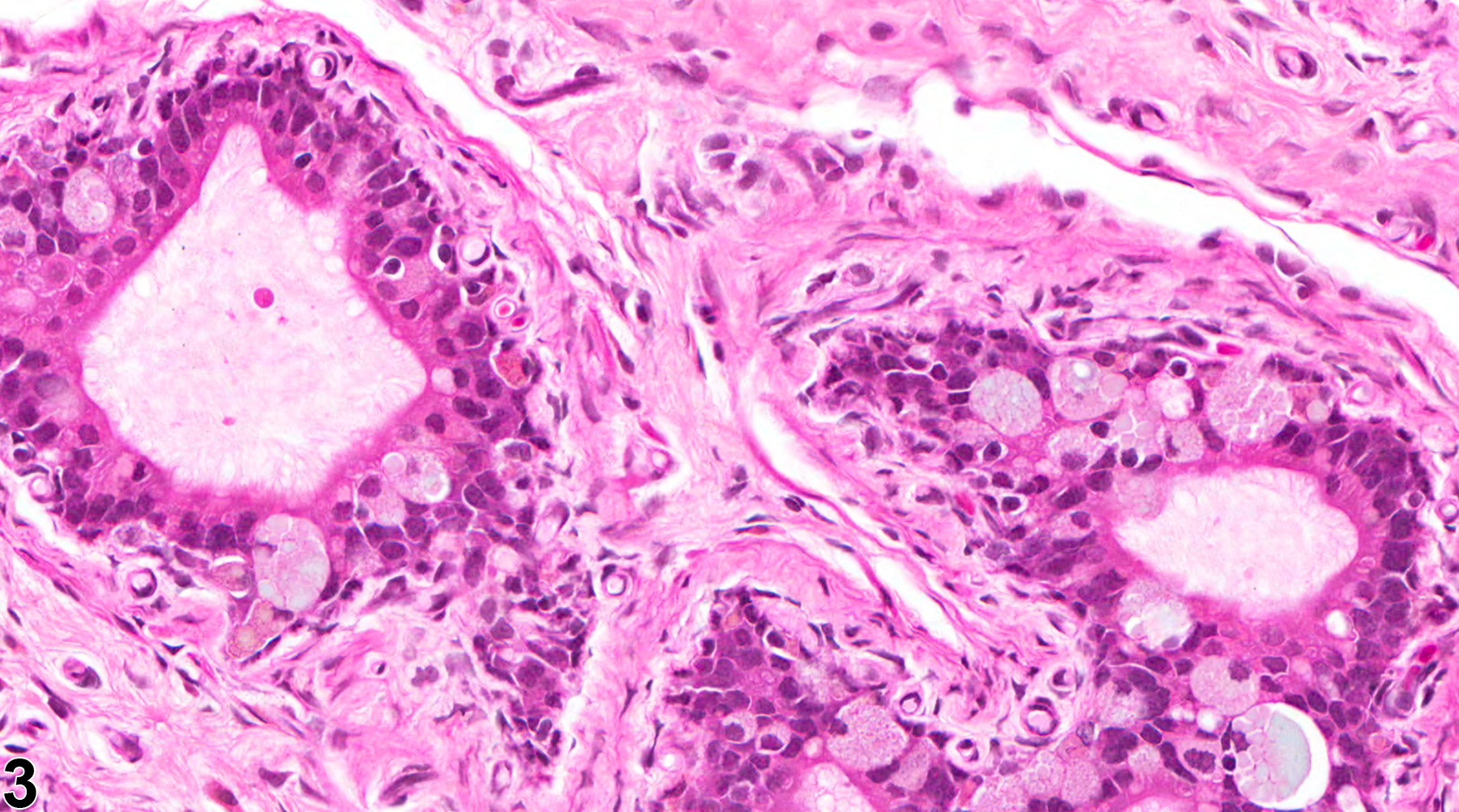
Epididymis, Epithelium - Vacuolation
Narrative
Boorman GA, Chapin RE, Mitsumori K. 1990. Testis and epididymis. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 405-418.
Haines DC, Chattopadhyay S, Ward JM. 2001. Pathology of aging B6;129 mice. Toxicol Pathol 29:653-661.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11794381Robaire B. 2002. Aging of the epididymis. In: The Epididymis: From Molecules to Clinical (Robaire B, Hinton BT, eds). New York, Kluwer/Plenum, 285-296.
Abstract: http://www.springer.com/medicine/urology/book/978-0-306-46684-7Robaire B, Hinton BT, eds. 2002. The Epididymis: From Molecules to Clinical.
Abstract: http://www.springer.com/medicine/urology/book/978-0-306-46684-7Rudmann DG, McNerney ME, Vandereide SL, Schemmer JK, Eversole RR, Vonderfecht SL. 2004. Epididymal and systemic phospholipidosis in rats and dogs treated with the dopamine D3 selective antagonist PNU-177864. Toxicol Pathol 32:326-332.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/15204974Stebbins KE, Bond DM, Novilla MN, Reasor MJ. 2002. Spinosad insecticide: Subchronic and chronic toxicity and lack of carcinogenicity in CD-1 mice. Toxicol Sci 65:276-287.
Abstract: http://toxsci.oxfordjournals.org/content/65/2/276.abstractYano BL, Bond DM, Novilla MN, McFadden LG, Reasor MJ. 2002. Spinosad insecticide: Subchronic and chronic toxicity and lack of carcinogenicity in Fischer 344 rats. Toxicol Sci 65:288-298.
Abstract: http://toxsci.oxfordjournals.org/content/65/2/288.abstract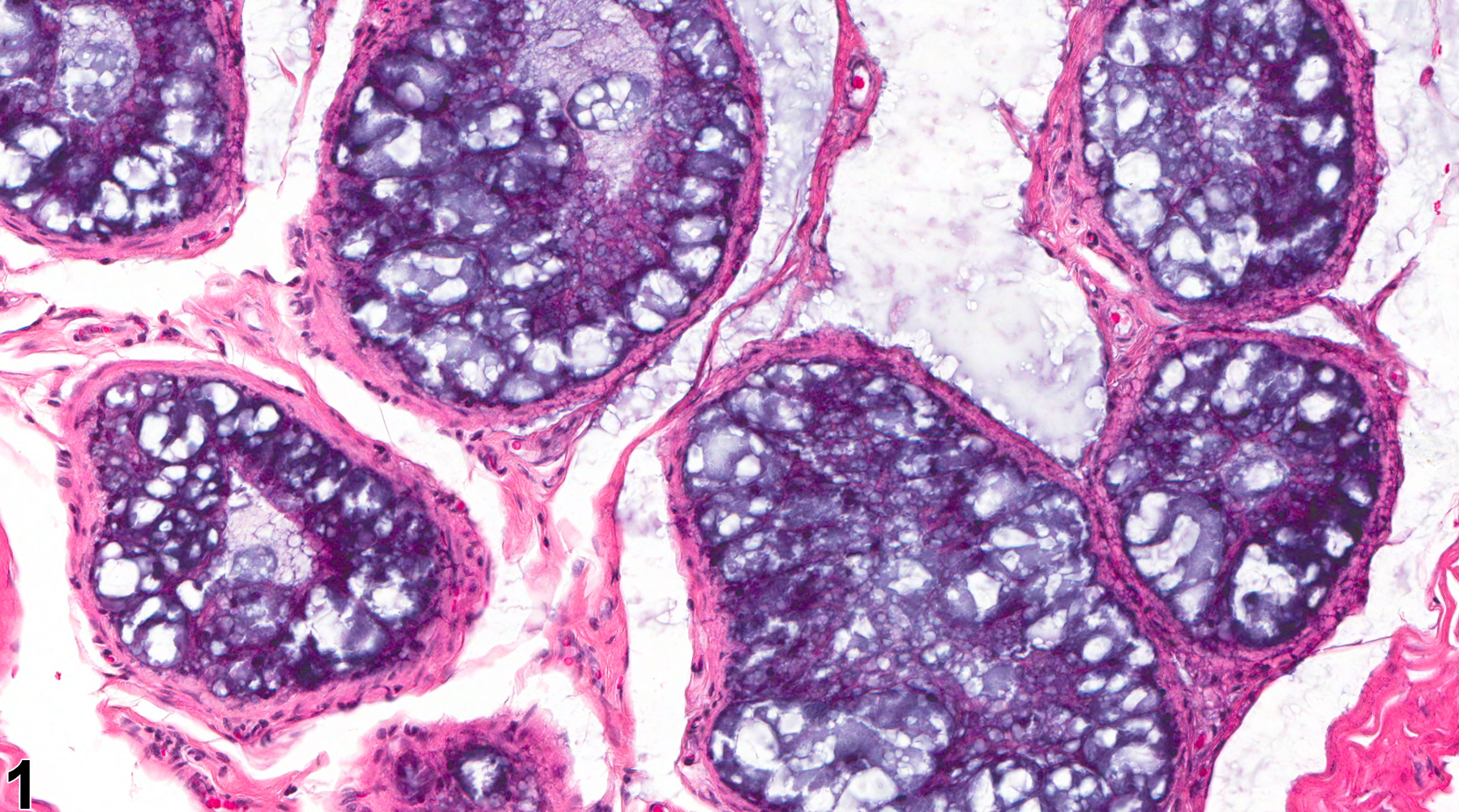
Epididymis, Epithelium - Vacuolation. Numerous macrovesicular cytoplasmic vacuoles are present in the ductal epithelium in a male F344/N rat from a chronic study.