Reproductive System, Male
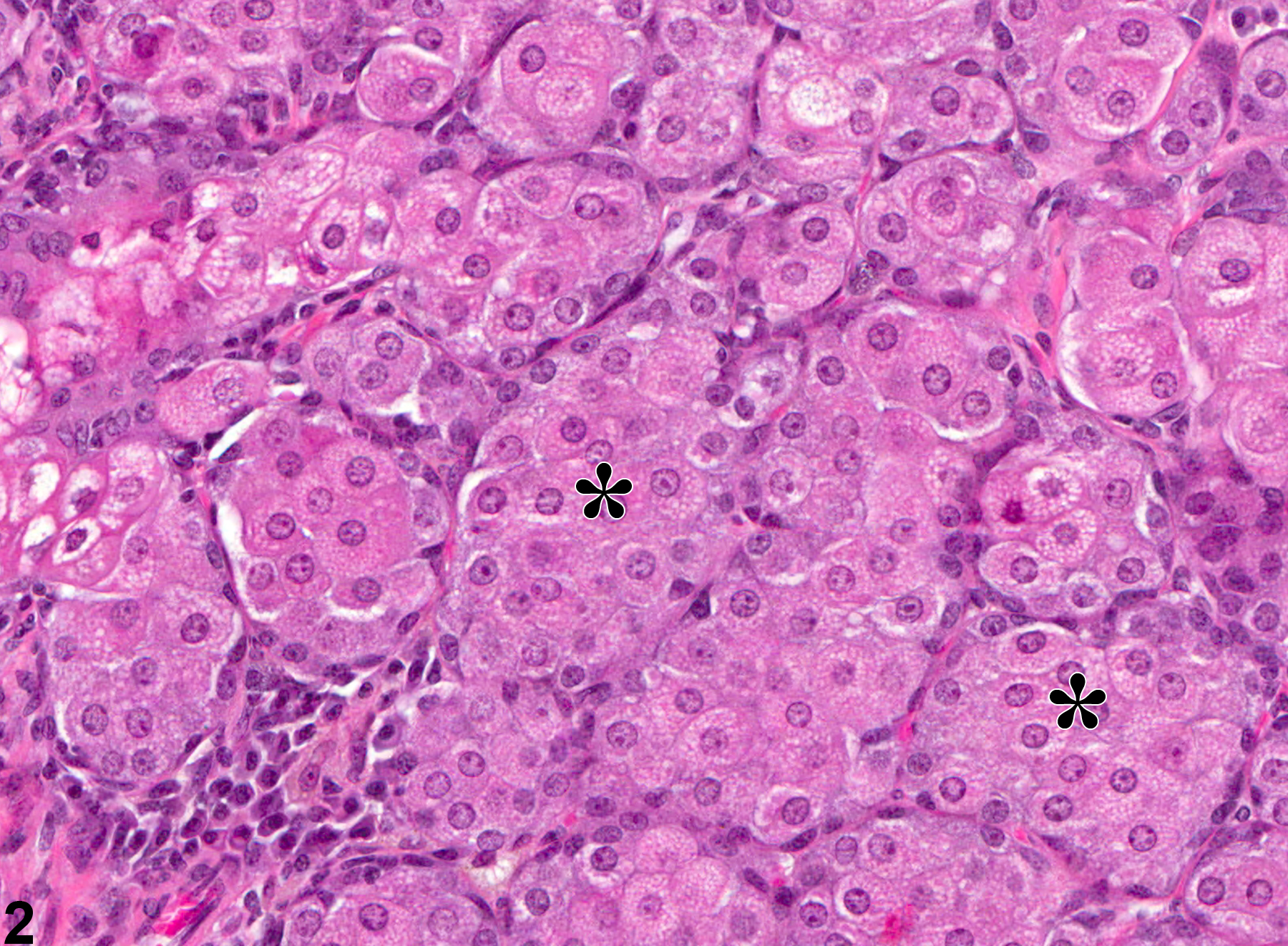
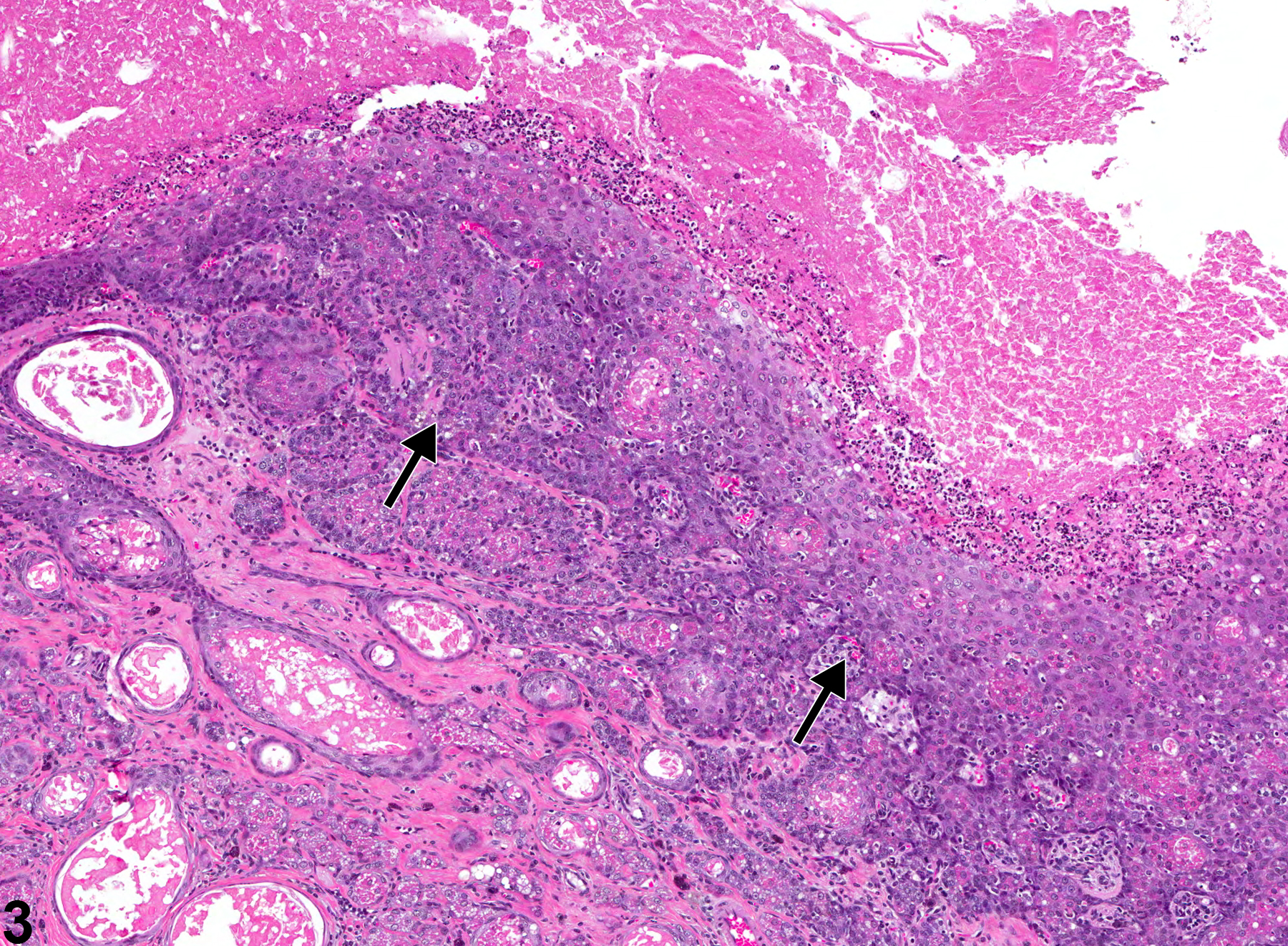
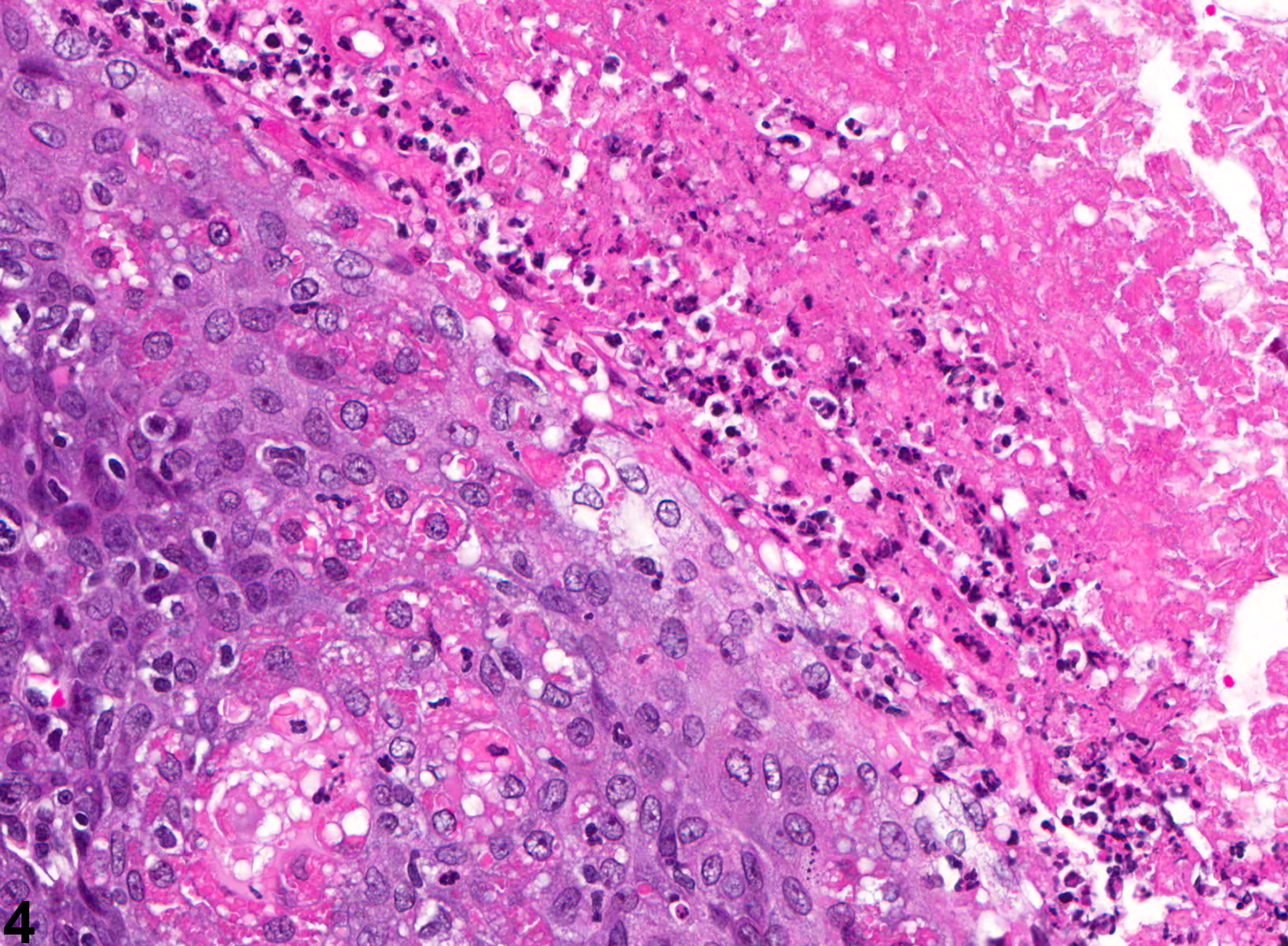
Preputial Gland - Hyperplasia
Narrative
Acinar hyperplasia is considered preneoplastic and should be differentiated from adenomas. In acinar hyperplasia, the acini appear larger than normal, with some crowding. Preputial glandular adenomas are more circumscribed and are associated with compression of adjacent tissue, and cellular organization with acini may be irregular. Likewise, ductular hyperplasia should be distinguished from squamous cell papillomas. The latter may form papillae supported by connective tissue cores and may have interconnecting cords of proliferative squamous epithelium.
Boorman GA, Elwell MR, Mitsumori K. 1990. Male accessory sex glands, penis, and scrotum. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 419-428.
Abstract: http://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Gordon LR, Majka JA, Boorman GA. 1996. Spontaneous nonneoplastic and neoplastic lesions and experimentally induced neoplasms of the testes and accessory sex glands. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 421-441.
Abstract: http://catalog.hathitrust.org/Record/008994685Haines DC, Eustis SL. 1990. Specialized sebaceous glands. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 279-293.
Abstract: http://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Reznik G, Ward JM. 1981. Morphology of hyperplastic and neoplastic lesions in the clitoral and preputial gland of the F344 rat. Vet Pathol 18:228-238.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/7467083Rudmann D, Cardiff R, Chouinard L, Goodman D, Kuttler K, Marxfeld H, Molinolo A, Treumann S, Yoshizawa K. 2012. Proliferative and nonproliferative lesions of the rat and mouse mammary, Zymbal's, preputial, and clitoral glands. Toxicol Pathol 40:7S-39S.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/22949413Seely JC, Boorman GA. 1999. Mammary gland and specialized sebaceous glands. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 613-635.
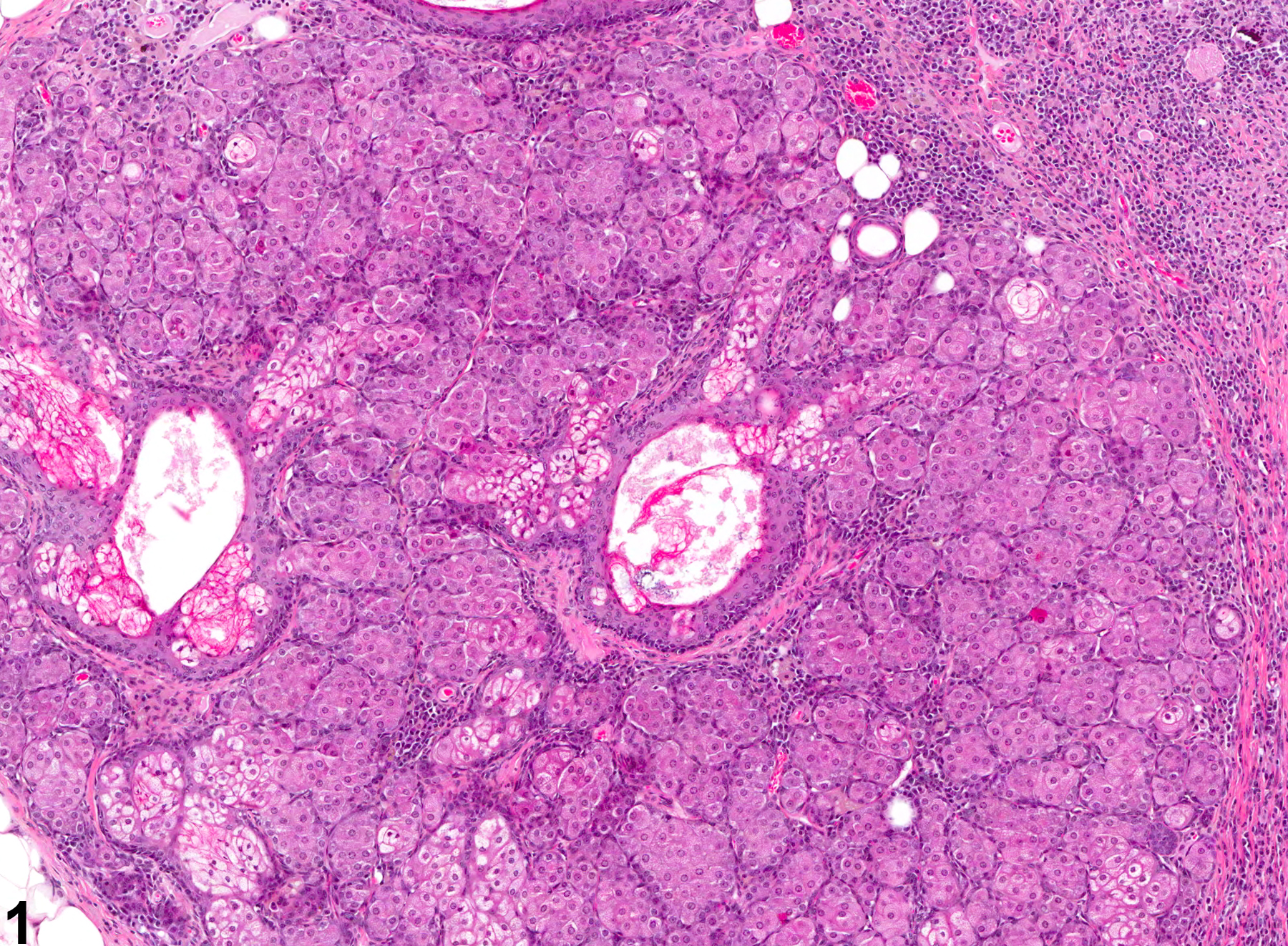
Preputial Gland, Epithelium - Hyperplasia. Hyperplasia in the preputial gland in a male F344/N rat from a chronic study.