Reproductive System, Male
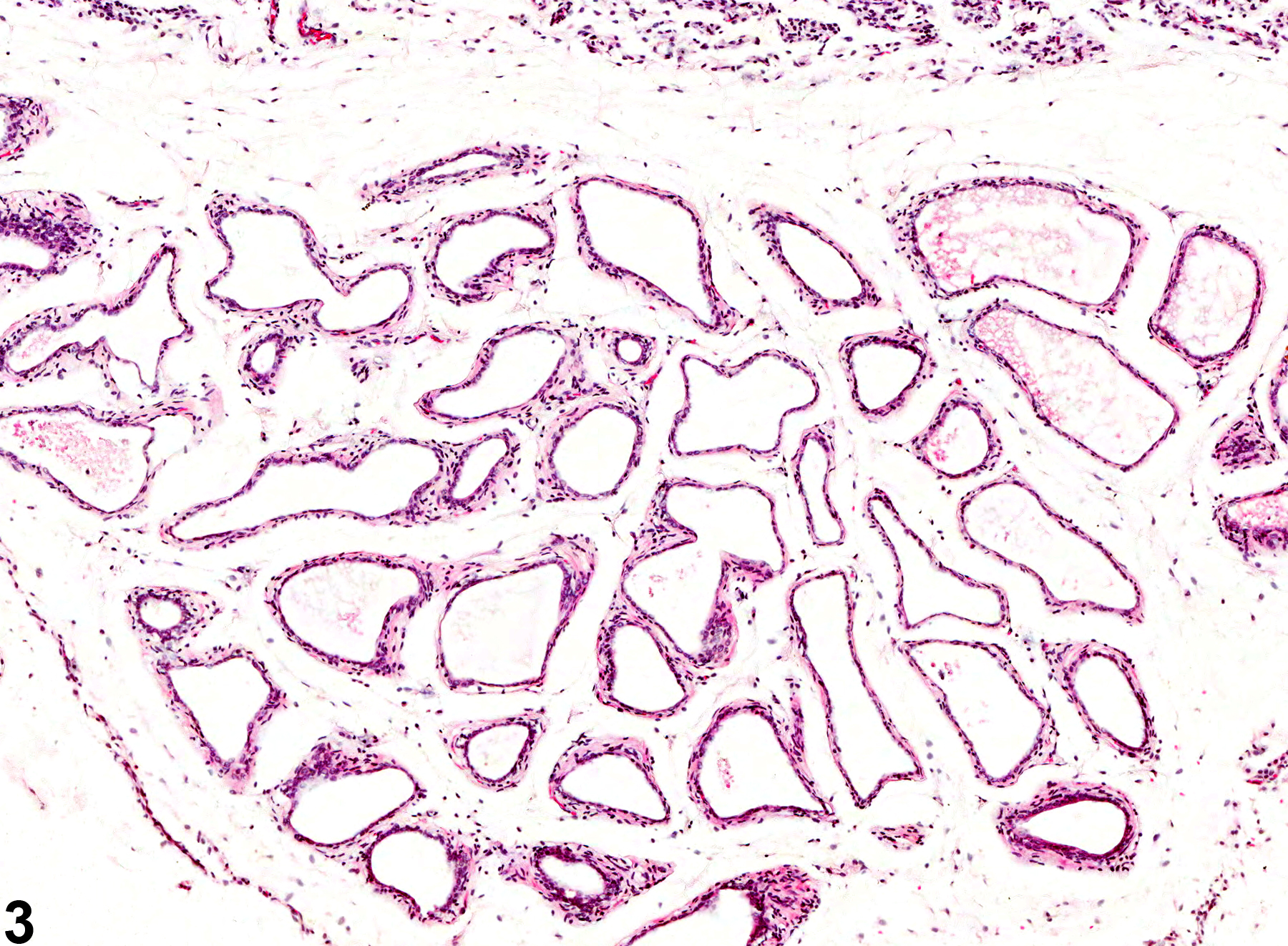
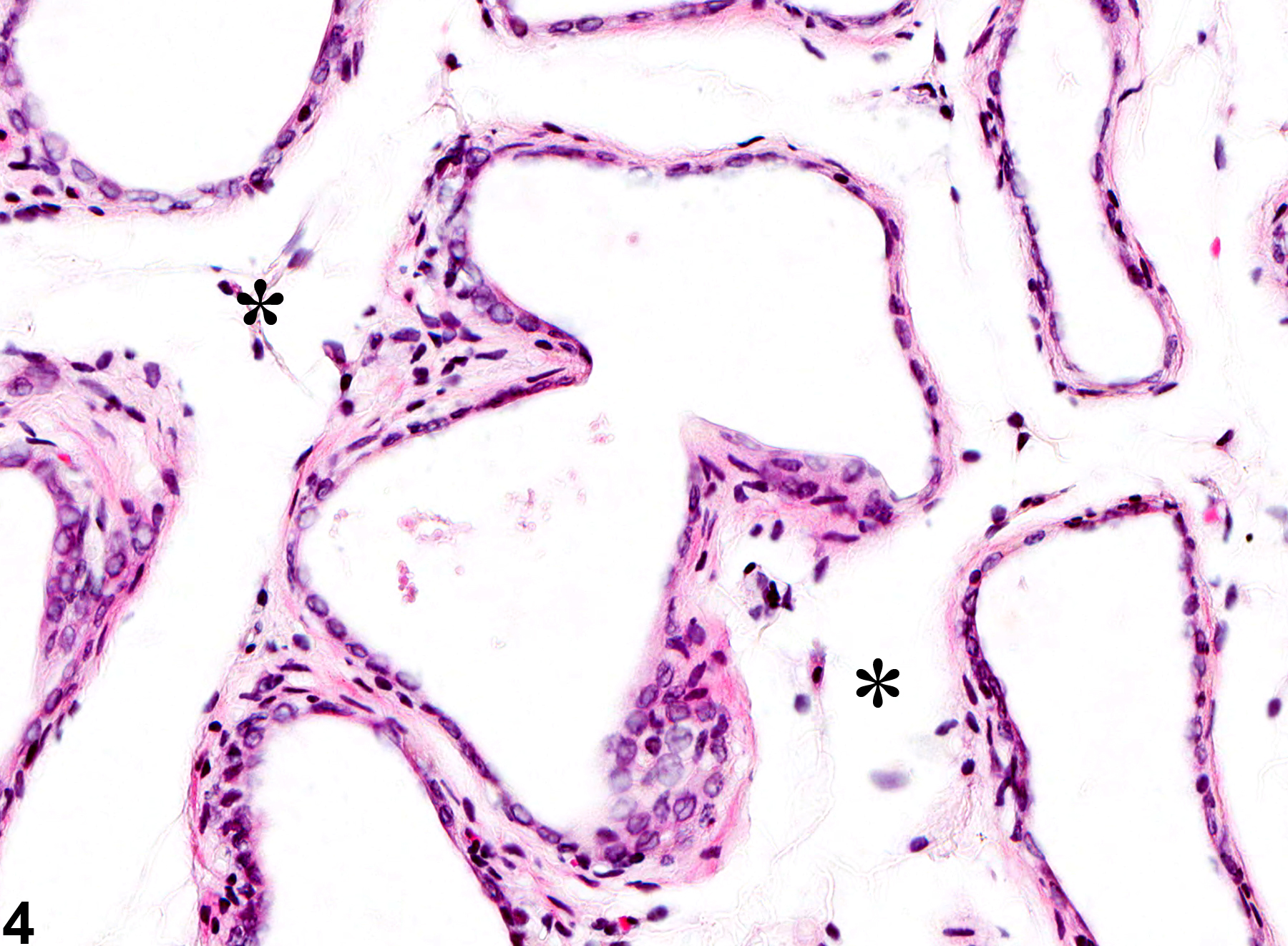
Prostate, Acinus - Atrophy
Narrative
Estrogens decrease the volume of the glandular epithelium and increase the fibromuscular stroma in both ventral prostate and seminal vesicle. Deficiency of the Egr family of zinc finger transcription factors such as Egr4 and Egr1, as in Egr4-Egr1 double-mutant mice, leads to atrophy of prostate, testis, epididymis, and seminal vesicle. Spontaneous prostatic atrophy is an age-related lesion.
Boorman GA, Elwell MR, Mitsumori K. 1990. Male accessory sex glands, penis, and scrotum. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 419-428.
Abstract: http://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Bosland MC. 1992. Lesions in the male accessory glands and penis. In: Pathobiology of the Aging Rat, Vol 1 (Mohr U, Dungworth DL, Capen CC, eds). ILSI Press, Washington, DC, 443-467.
Abstract: http://catalog.hathitrust.org/Record/008994685Gao W, Kearbey JD, Nair VA, Chung K, Parlow AF, Miller DD, Dalton JT. 2004. Comparison of the pharmacological effects of a novel selective androgen receptor modulator, the 5α-reductase inhibitor finasteride, and the antiandrogen hydroxyflutamide in intact rats: New approach for benign prostate hyperplasia. Endocrinology 145:5420-5428.
Full Text: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2098692/Gayton F, Bellido C. Aguilar R, Lucena MC. 1986. Morphometric analysis of the rat ventral prostate and seminal vesicles during prepubertal development: Effects of neonatal treatment with estrogen. Biol Reprod 35:219-225.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/3741952Gordon LR, Majka JA, Boorman GA. 1996. Spontaneous nonneoplastic and neoplastic lesions and experimentally induced neoplasms of the testes and accessory sex glands. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 421-441.
Abstract: http://catalog.hathitrust.org/Record/008994685Greaves P. 2007. Male genital tract. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation. 3rd ed. Academic Press, San Diego, 661-716.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Quin LH, Wang XL, Tu ZH. 2001. Atrophy and apoptosis in ventral prostate of rats induced by 5 alpha-reductase inhibitor epristeride. Acta Pharmacol Sin 22:399-404.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11743885Rittmaster RS, Manning AP, Wright AS, Thomas LN, Whitefield S, Norman RW, Lazier CB, Rowden G. 1995. Evidence for atrophy and apoptosis in the ventral prostate of rats given the 5alpha-reductase inhibitor finasteride. Endocrinology 136:741-748.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/7835306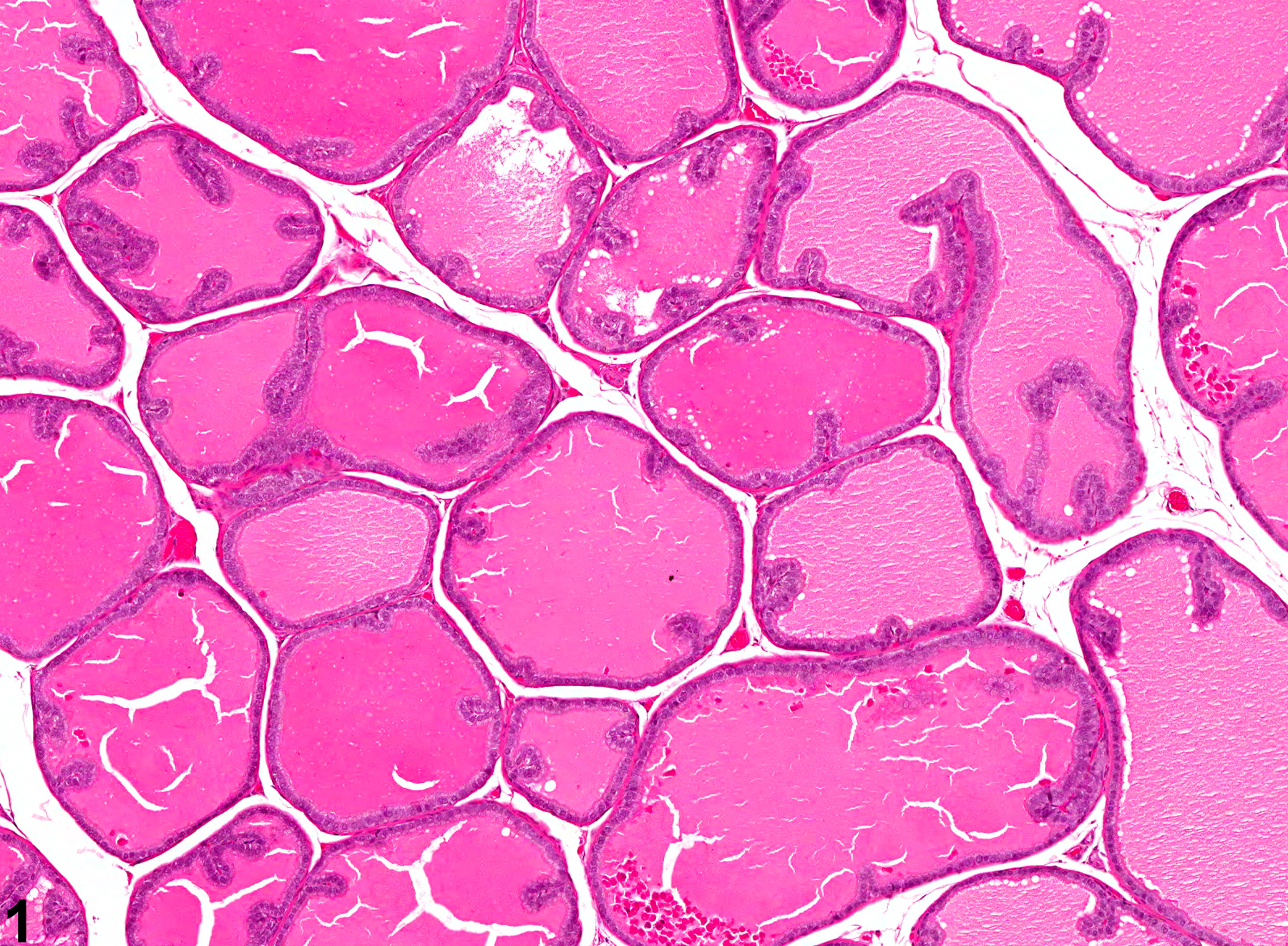
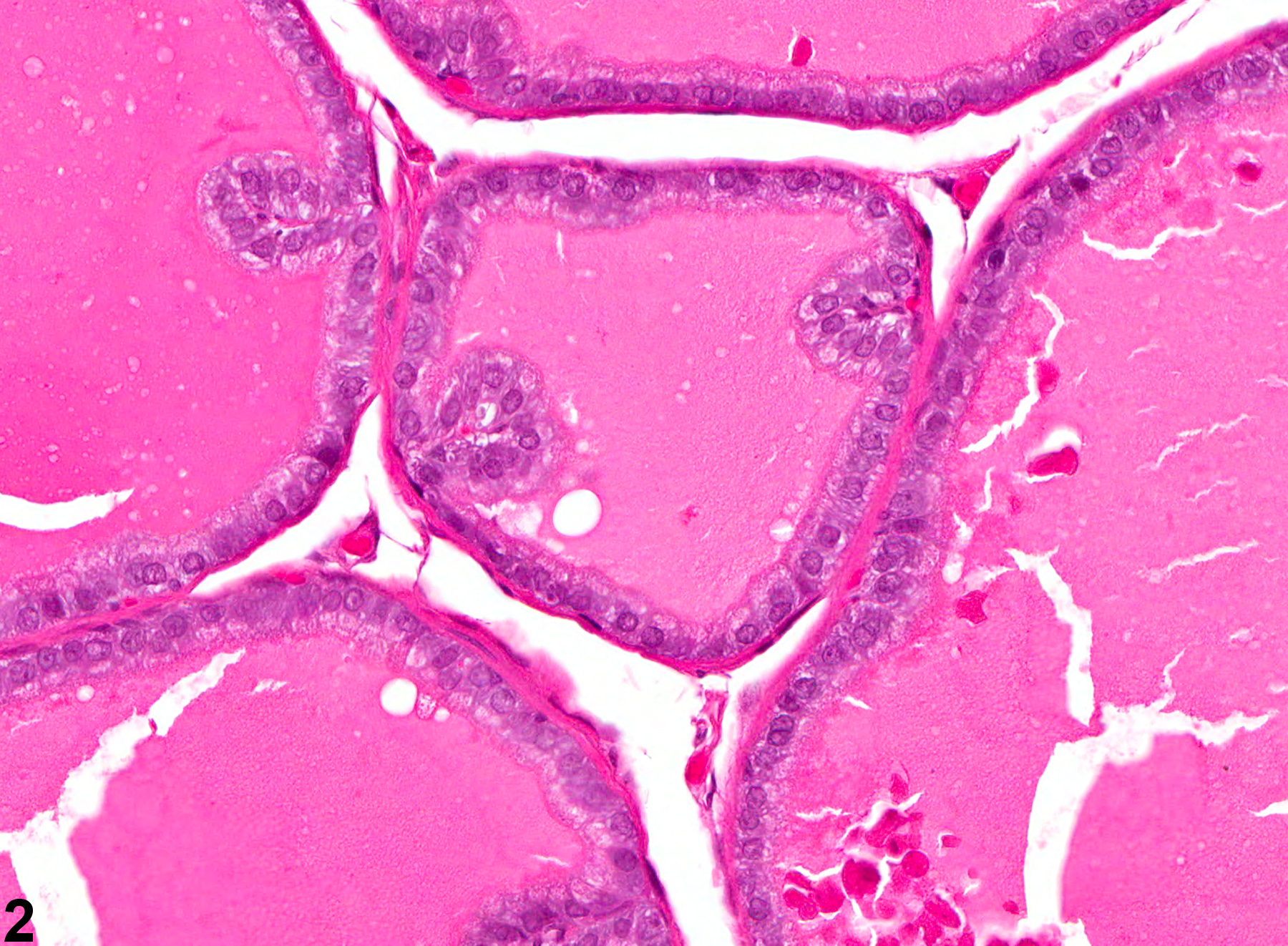
Prostate - Normal. Normal dorsolateral prostate for comparison with Figure 3 in a male F344/N rat from a subchronic study.