Reproductive System, Male
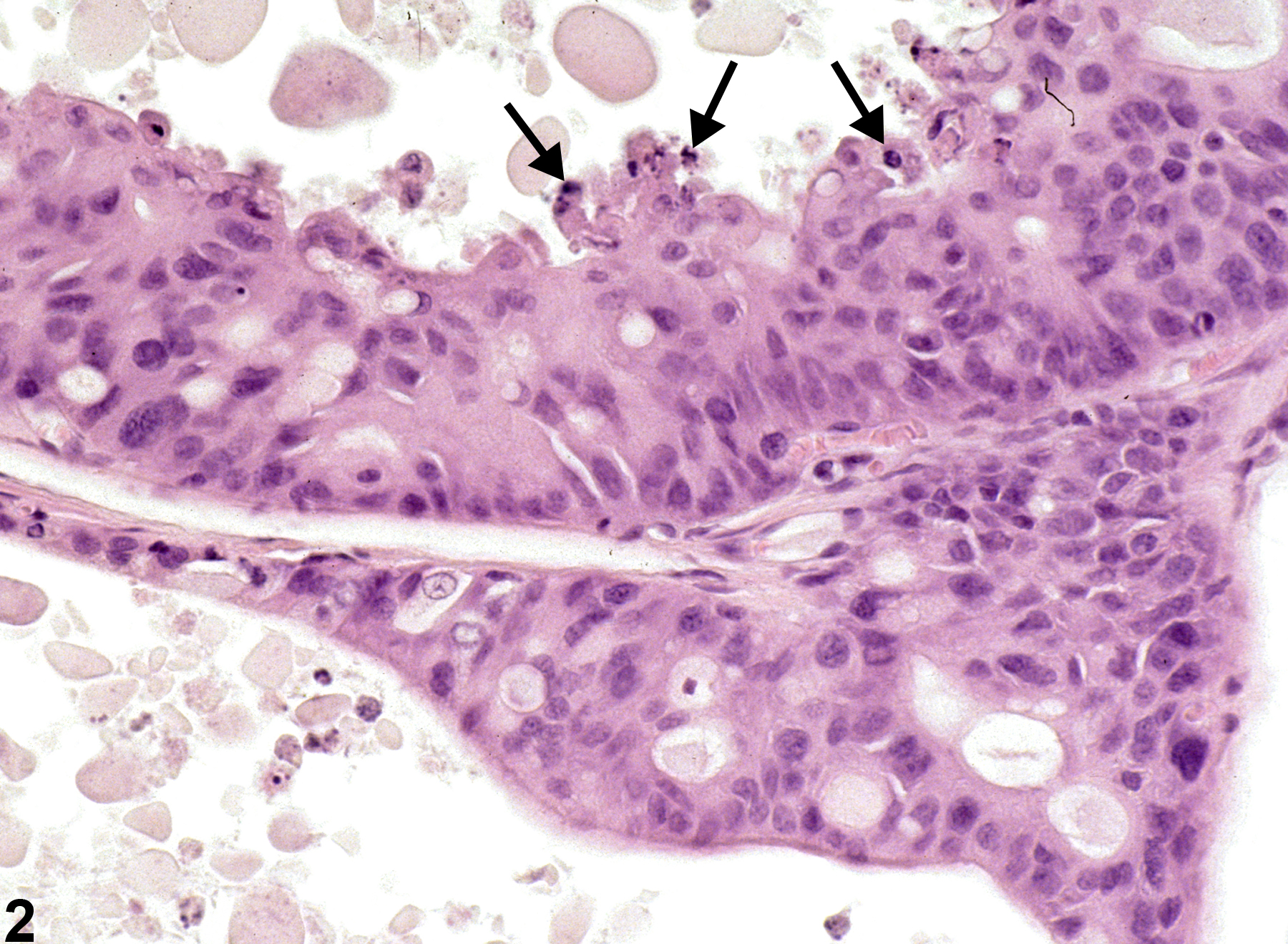
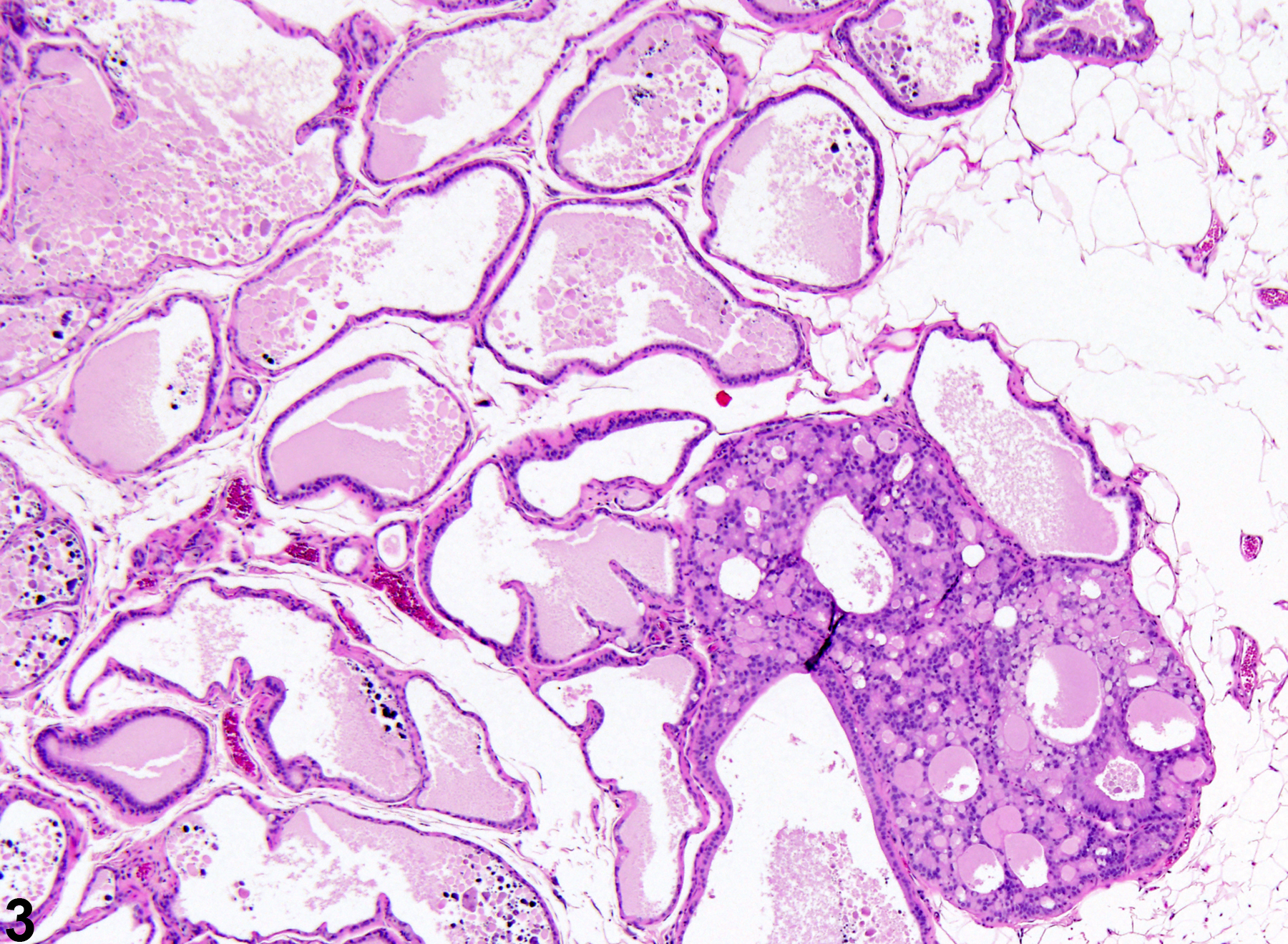
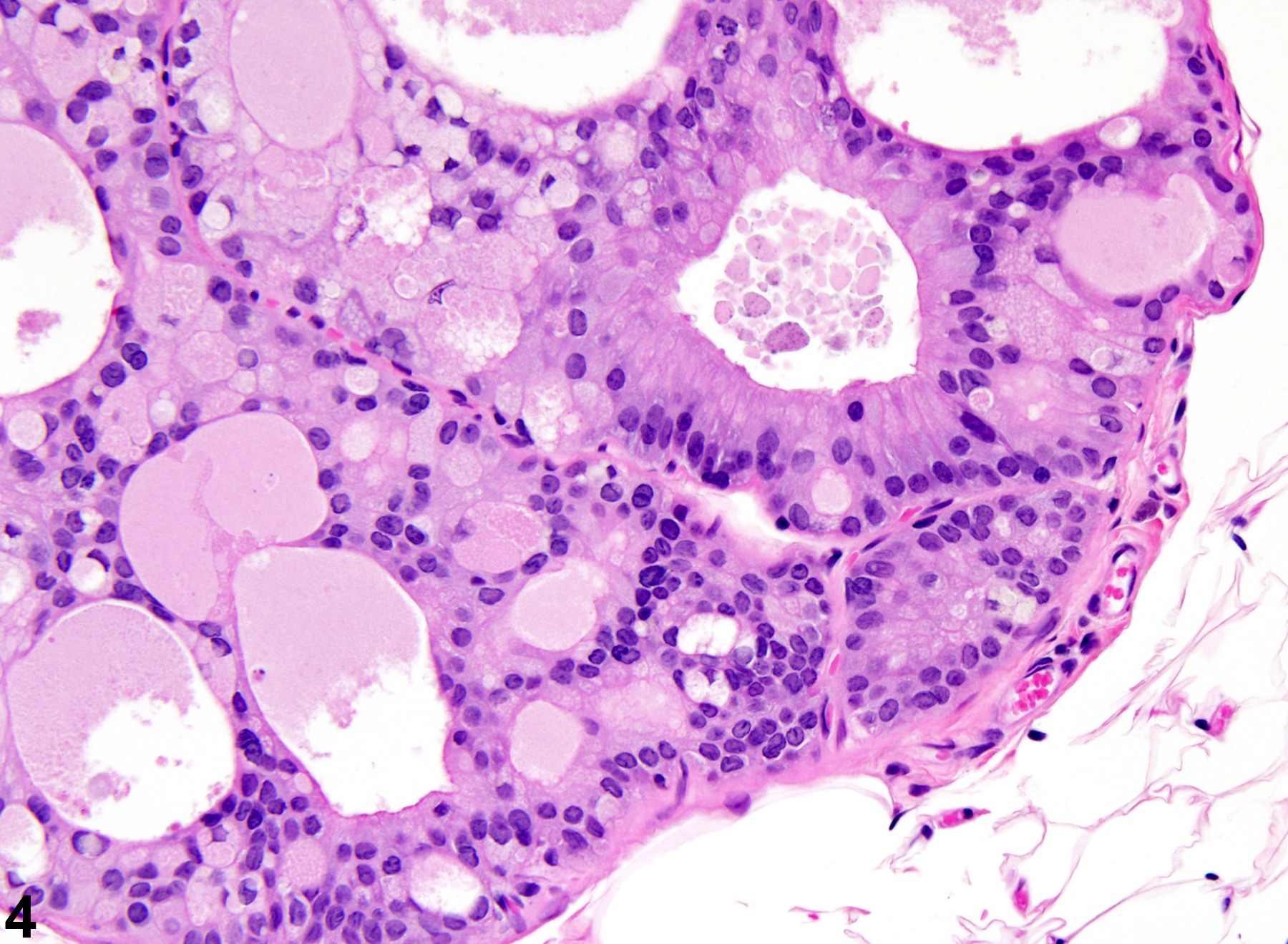
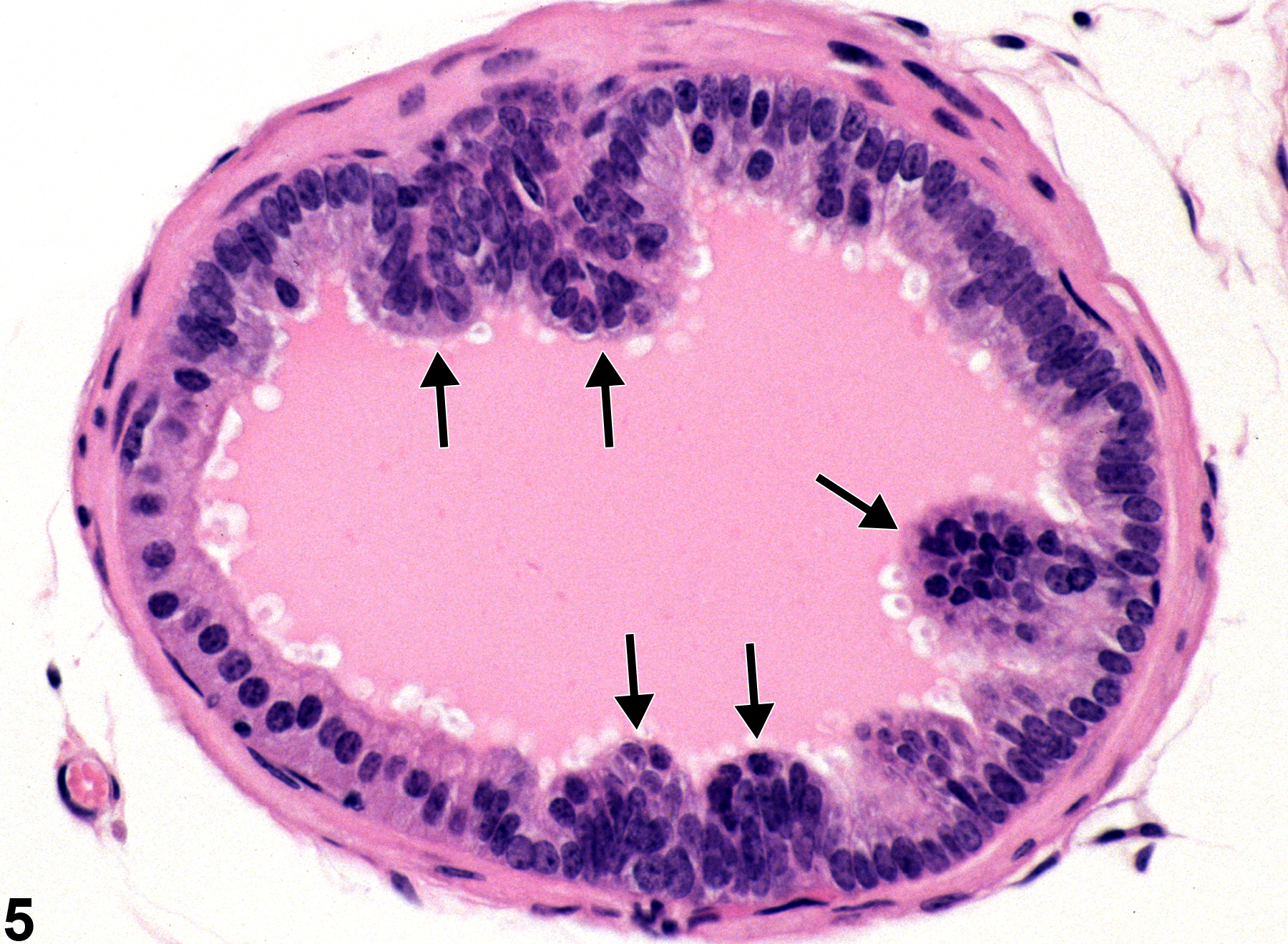
Prostate, Epithelium - Hyperplasia
Narrative
Banerjee PP, Banerjee S, Lai JM, Strandberg JD, Zirkin BR, Brown TR. 1998. Age-dependent and lobe-specific spontaneous hyperplasia in the brown Norway rat prostate. Biol Reprod 59:1163-1170.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/9780323Boorman GA, Elwell MR, Mitsumori K. 1990. Male accessory sex glands, penis, and scrotum. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 419-428.
Abstract: http://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Bosland MC. 1992. Lesions in the male accessory glands and penis. In: Pathobiology of the Aging Rat, Vol 1 (Mohr U, Dungworth DL, Capen CC, eds). ILSI Press, Washington, DC, 443-467.
Abstract: http://catalog.hathitrust.org/Record/008994685Golomb E, Kruglikova A, Dvir D, Parnes N, Abramovici A. 1998. Induction of atypical prostatic hyperplasia in rats by sympathomimetic stimulation. Prostate 34:214-221.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/9492850Gordon LR, Majka JA, Boorman GA. 1996. Spontaneous nonneoplastic and neoplastic lesions and experimentally induced neoplasms of the testes and accessory sex glands. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 421-441.
Abstract: http://catalog.hathitrust.org/Record/008994685Greaves P. 2007. Male genital tract. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation. 3rd ed. Academic Press, San Diego, 661-716.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Reznik G, Hamlin MH II, Ward JM, Stinson SF. 1981. Prostatic hyperplasia and neoplasia in aging F344 rats. Prostate 2:261-268.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/6170967Suwa T, Nyska A, Peckham JC, Hailey JR, Mahler JF, Haseman JK, Maronpot RR. 2001. A retrospective analysis of background lesions and tissue accountability for male accessory sex organs in Fischer-344 rats. Toxicol Pathol 29(4):467-478.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11560252Suwa T, Nyska A, Haseman JK, Mahler JF, Maronpot RR. 2002. Spontaneous lesions in control B6C3F1 mice and recommended sectioning of male accessory sex organs. Toxicol Pathol 30(2):228-234.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11950166Tam NN, Nyska A, Maronpot RR, Kissling G, Lomnitski L, Suttie A, Bakshi S, Bergman M, Grossman S, Ho SM. 2006. Differential attenuation of oxidative/nitrosative injuries in early prostatic neoplastic lesions in TRAMP mice by dietary antioxidants. Prostate 66:57-69.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/16114064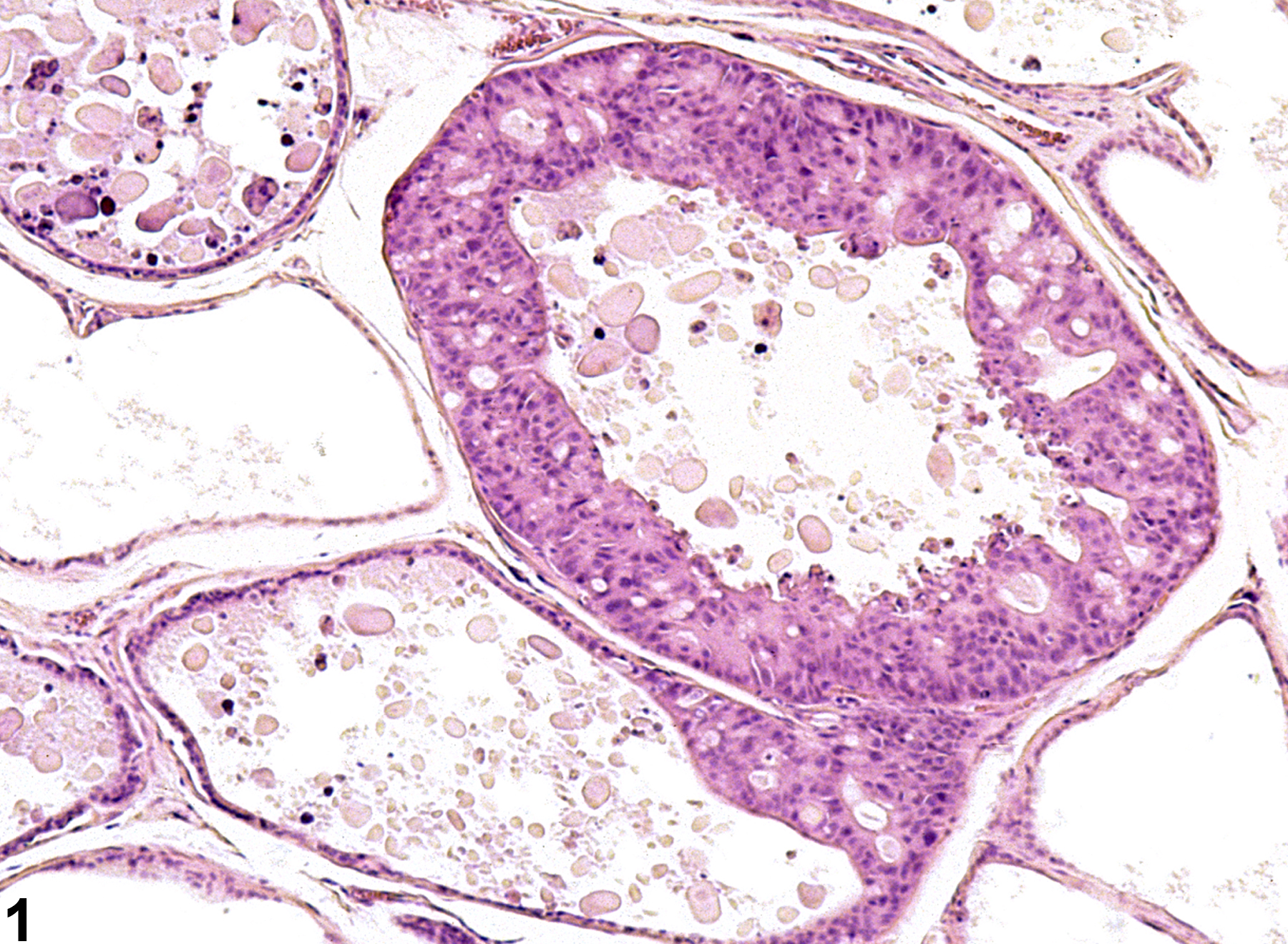
Prostate, Epithelium - Hyperplasia. Epithelial hyperplasia of the prostate in a male F344/N rat from a chronic study.