Reproductive System, Male
Prostate
Narrative
General
The male reproductive system tissues include testes, epididymides, the various accessory sex glands, and the penis. These tissues can exhibit direct and indirect (usually hormonal) pleotrophic pathologic responses, often with a change in one reproductive tissue accompanied by changes in other reproductive tissues. The majority of the male reproductive tissues are paired, allowing identification of unilateral versus bilateral responses. Artifacts are rare and typically reflect the use of less than ideal fixatives for testes (modified Davidson's fixative is preferred) or physical damage, including drying out of tissues, during the weighing process prior to fixation.
Evaluation of male reproductive system tissues should be correlated with wet tissue weights when possible. Substantial changes in weight, particularly for testes and for the combined seminal vesicles and coagulating glands, will typically have a morphologic counterpart that should be documented. Prostate weights, particularly specific prostate lobe weights, are dependent upon the consistency of gross dissection and are variable. Identification of morphologic changes in the whole prostate or in specific lobes that correlate with tissue weight changes can be challenging. During evaluation of paired tissues, documentation of unilateral versus bilateral changes may help in identifying a systemic response. As a general guideline for paired tissues, if both are involved, the diagnosis should be qualified as bilateral and the severity grade based on the more severely affected tissue.
In evaluation of the male reproductive system, if potential treatment-related increases in changes occur, they should be described in the pathology narrative and correlated with other potential lesions in reproductive and endocrine tissues. Secondary changes such as metaplasia, degeneration, or apoptosis that accompany a primary lesion are usually not diagnosed unless warranted by virtue of their severity.
A final general concern relates to a variety of spontaneous background changes in testes, epididymides, and accessory sex glands, many of which can be exacerbated by treatment. The amount, tinctorial properties, and physical features of prostatic acinar secretions, for example, change significantly as rodents age or become less sexually active. Consequently, a consistent severity-grading scheme should be used in documenting commonly occurring lesions in male reproductive tissues if treatment-related exacerbation of common background changes is suspected.
Evaluation of Prostate
Prostatic acinar tissue is fundamentally similar in the dorsal, lateral, and ventral lobes but with tinctorial differences reflecting different biochemical constituents of secretions and differing staining protocols among laboratories. There is also variability in the amount of secretion both within and between specific lobes. In addition, the single-cell lining of the prostatic acini varies in height depending upon the degree of secretory activity. The challenge in evaluating the prostate is reliable identification of specific lobes. Specific lobe identification is highly dependent upon obtaining a section that clearly shows their relationship to one another and how they encircle the urethra (Figure 1). The ventral prostate is the largest lobe, is most easily separated from the rest of the prostate for weighing, and consists of varying-sized acini with homogeneous or flocculent secretion. The dorsal and lateral lobes are smaller than the ventral lobe, are adjacent to one another, have morphologically similar acini, and are often considered together as the dorsolateral lobe since they are more difficult to separate by gross dissection. Because the different prostate lobes have biochemically differing secretions, different physiologic functions, and differing responses to toxic challenge, they are unique enough to warrant their separate identification, if possible, when documenting lesions. The ability to do this will depend upon the orientation of the section and the amount of tissue present for examination.
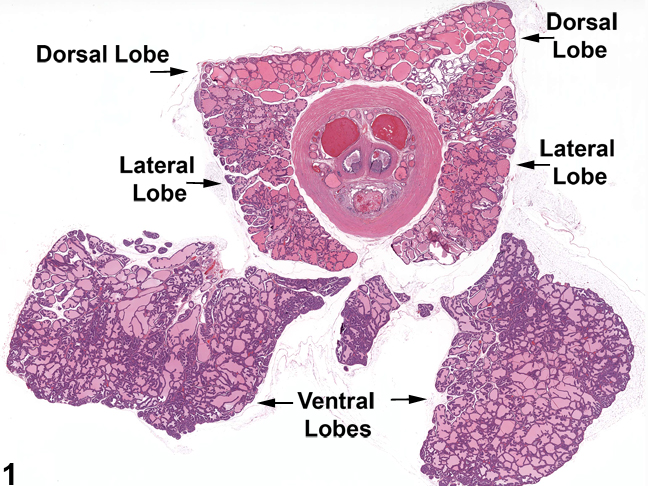
Figure 1. Low power view of the normal prostate gland showing the orientation of the different lobes.
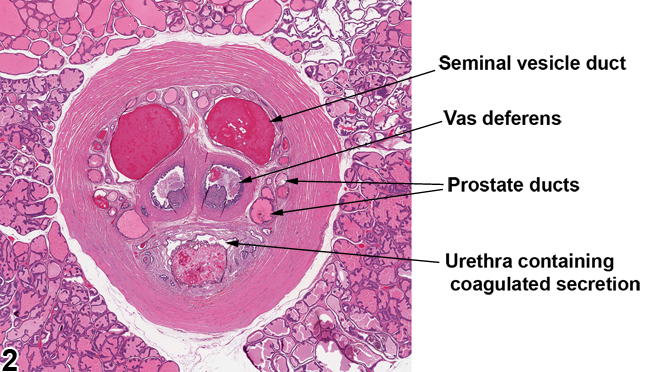
Figure 2. This higher magnification of a portion of Figure 1 shows the how the ducts of a normal prostate as well as the seminal vesicles empty into the proximal urethra. This collection of ducts is surrounded by a thick band of smooth muscle.
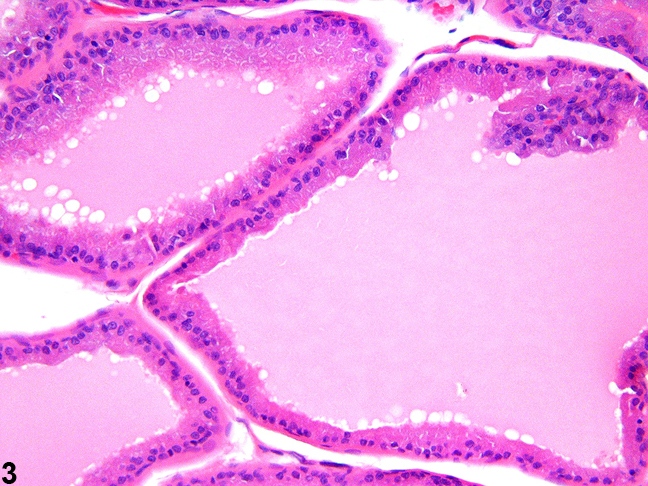
Figure 3. High magnification showing the cuboidal to columnar epithelial lining of a normal dorsal prostate.
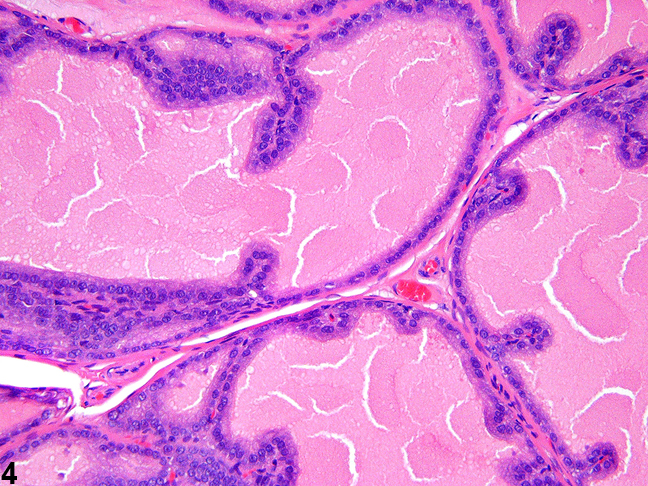
Figure 4. This high magnification of a normal lateral prostate shows the cuboidal epithelial lining of ascini with occasional papillary infoldings.
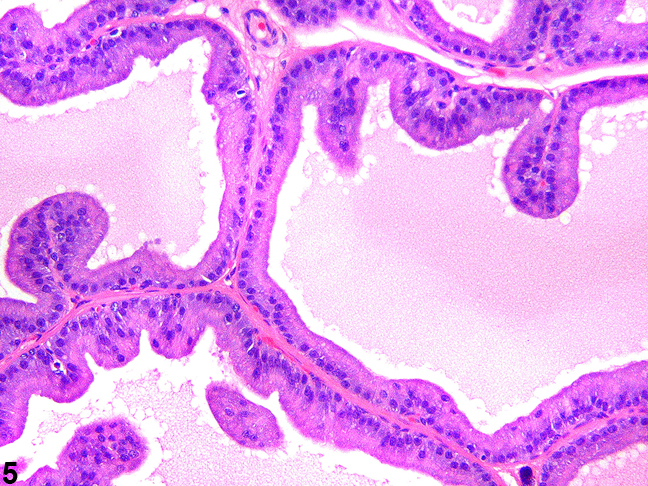
Figure 5. The tall columnar epithelial lining of the normal ventral prostate has occasional infoldings of the ascinar lining.
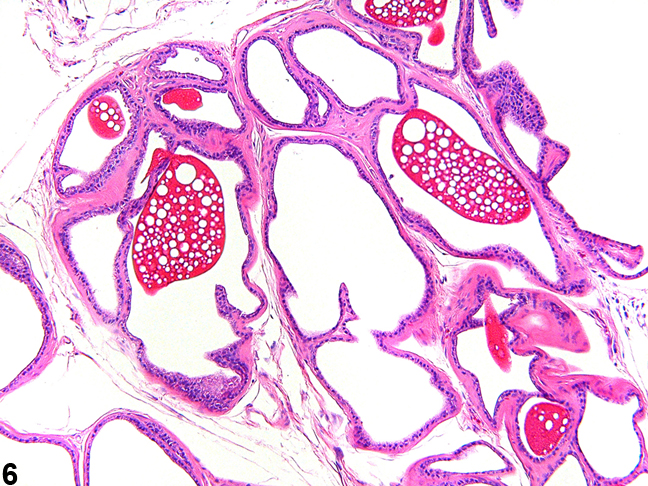
Figure 6. The normal ampullary gland is lined by flattened epithelium and contains a brightly stained secretion with characteristic vacuolation. The ampullary gland is located anterior and adjacent to the dorsal lobe of the prostate.
Creasy DM. 2001. Pathogenesis of male reproductive toxicity. Toxicol Pathol 29:64-76.
Full Text: http://tpx.sagepub.com/content/29/1/64.full.pdfCreasy DM, Foster P. 1991. Male reproductive system. In: Handbook of Toxicologic Pathology (Haschek E, Rousseaux C, eds). Academic Press, New York, 829-889.
Abstract: http://www.sciencedirect.com/science/book/9780123302151
