Reproductive System, Male
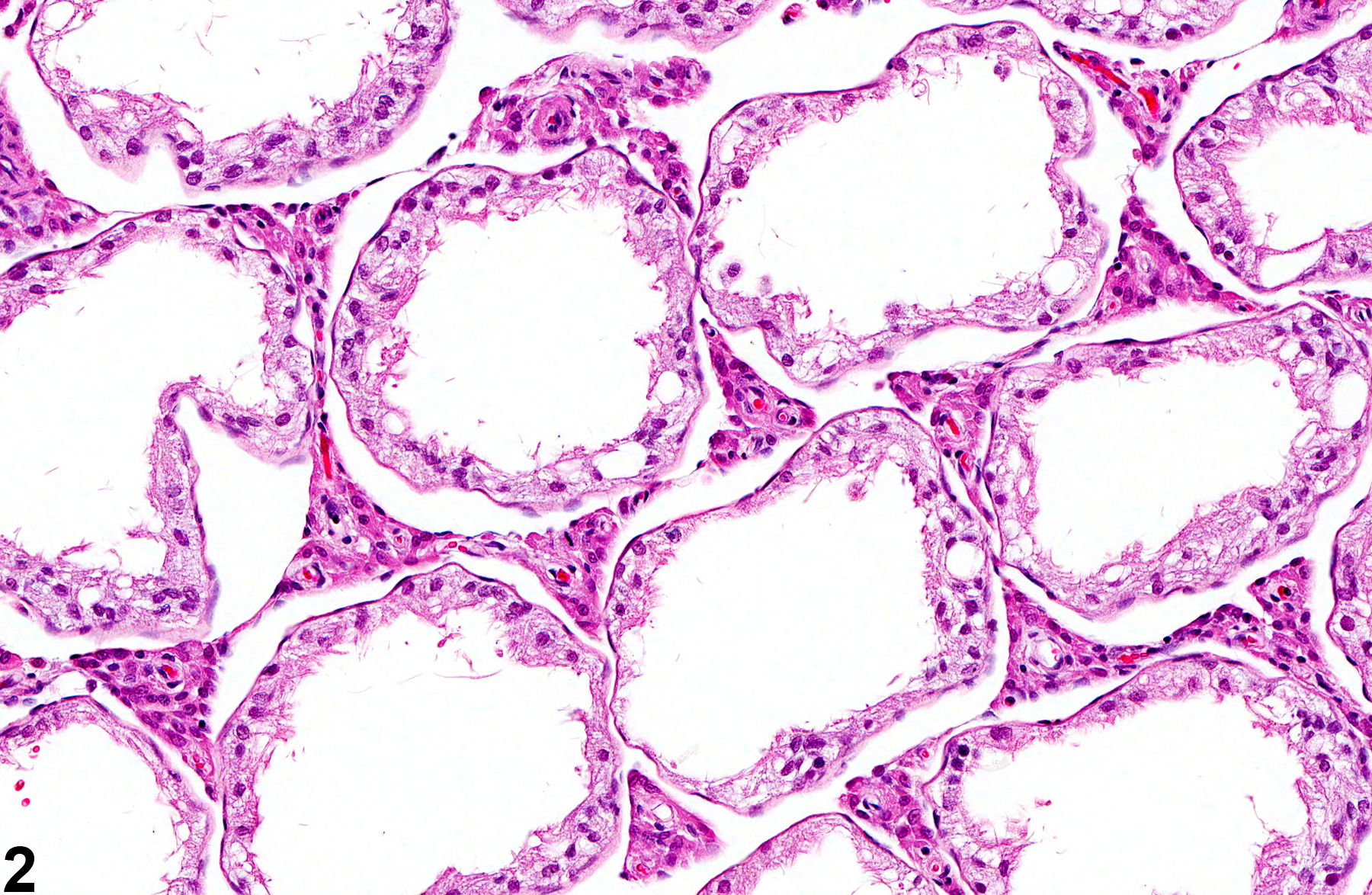
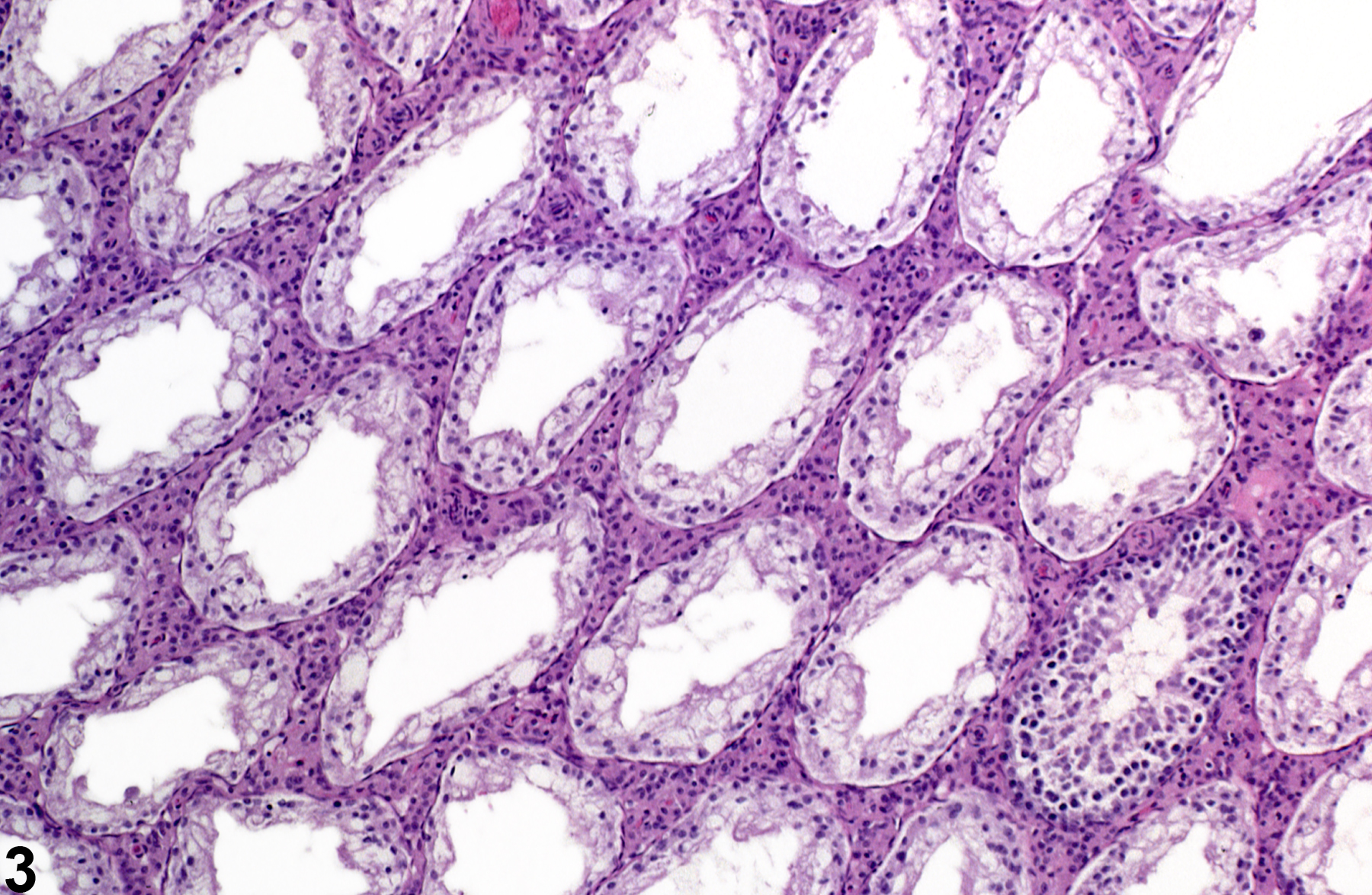
Testis, Germinal Epithelium - Atrophy
Narrative
If the tubular lumens are dilated, the atrophy is likely caused by pressure (i.e. pressure atrophy) fromprolonged and severe tubular dilation (also see ) The change may be unilateral or bilateral and focal, multifocal, or diffuse and occurs as an occasional incidental finding in mice and rats at all ages, with the incidence increasing with age. Germinal epithelial atrophy is an end-stage lesion and is generally preceded, or accompanied by, seminiferous tubule degeneration. Depending on severity, the affected testes may be macroscopically flaccid and reduced in size and weight. Severe, diffuse germinal epithelial atrophy is often irreversible. Scattered seminiferous tubules with germinal cell atrophy can be seen as an aging change in rats.
Dixon D, Heider K, Elwell MR. 1995.Incidence of nonneoplastic lesions in historical control male and female Fischer-344 rats from 90-day toxicity studies. Toxicol Pathol 23:338-348.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/7659956Nolte T, Harleman JH, Jahn W. 1995. Histopathology of chemically induced testicular atrophy in rats. Exp Toxicol Pathol 47:267-286.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/8855122Radovsky A, Mitsumori K, Chapin RE. 1999. Male reproductive tract. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 381-407.
Takano H, Kazuhiro ABE. 1987. Age-related histologic changes in the adult mouse. Testis. Arch Histol Jpn 50:533-544.
Abstract: https://doi.org/10.1679/aohc.50.533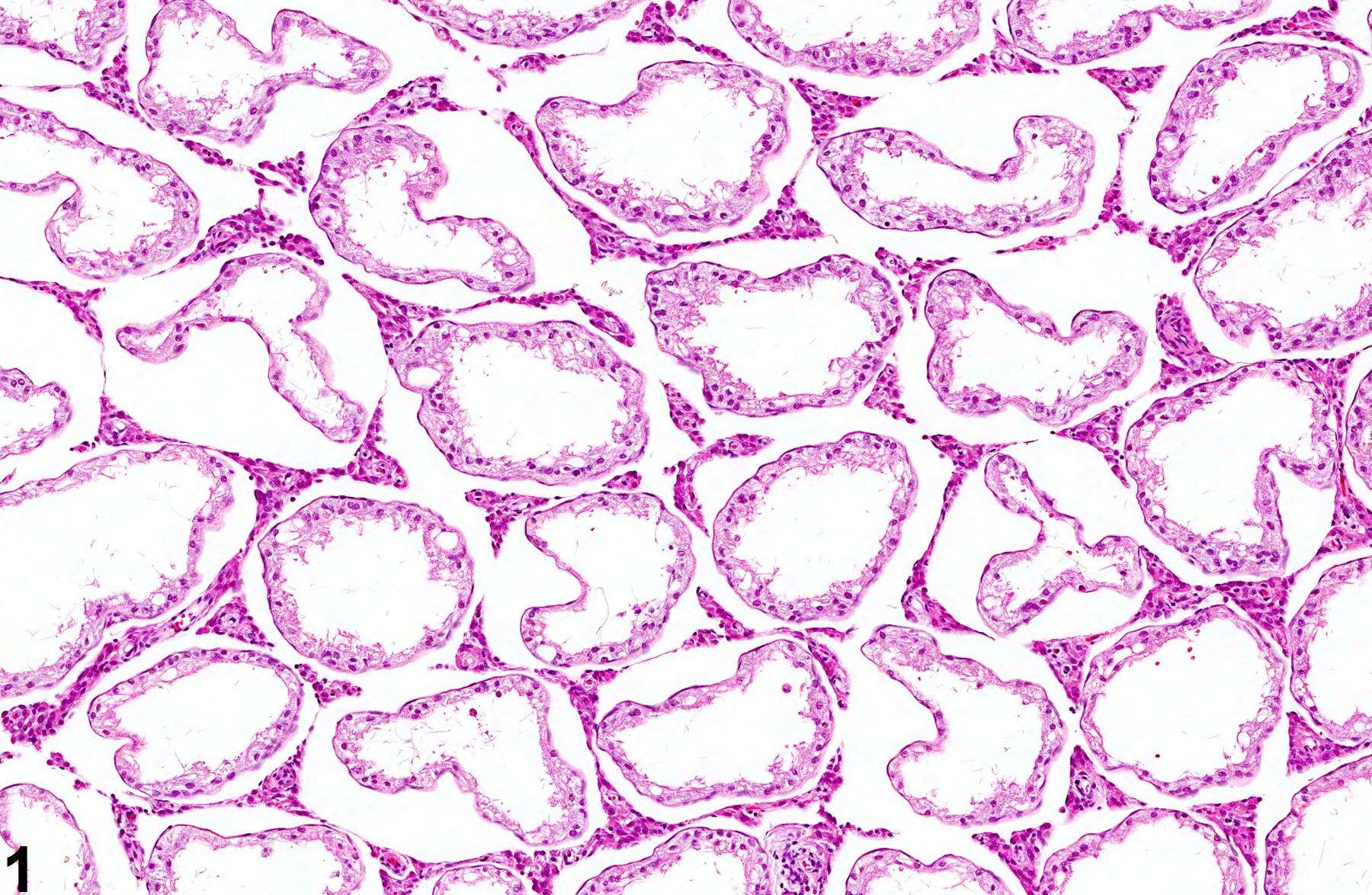
Testis, Germinal epithelium - Atrophy in a male F344/N rat from a subchronic study. Seminiferous tubules are lined only by Sertoli cells.