Reproductive System, Male
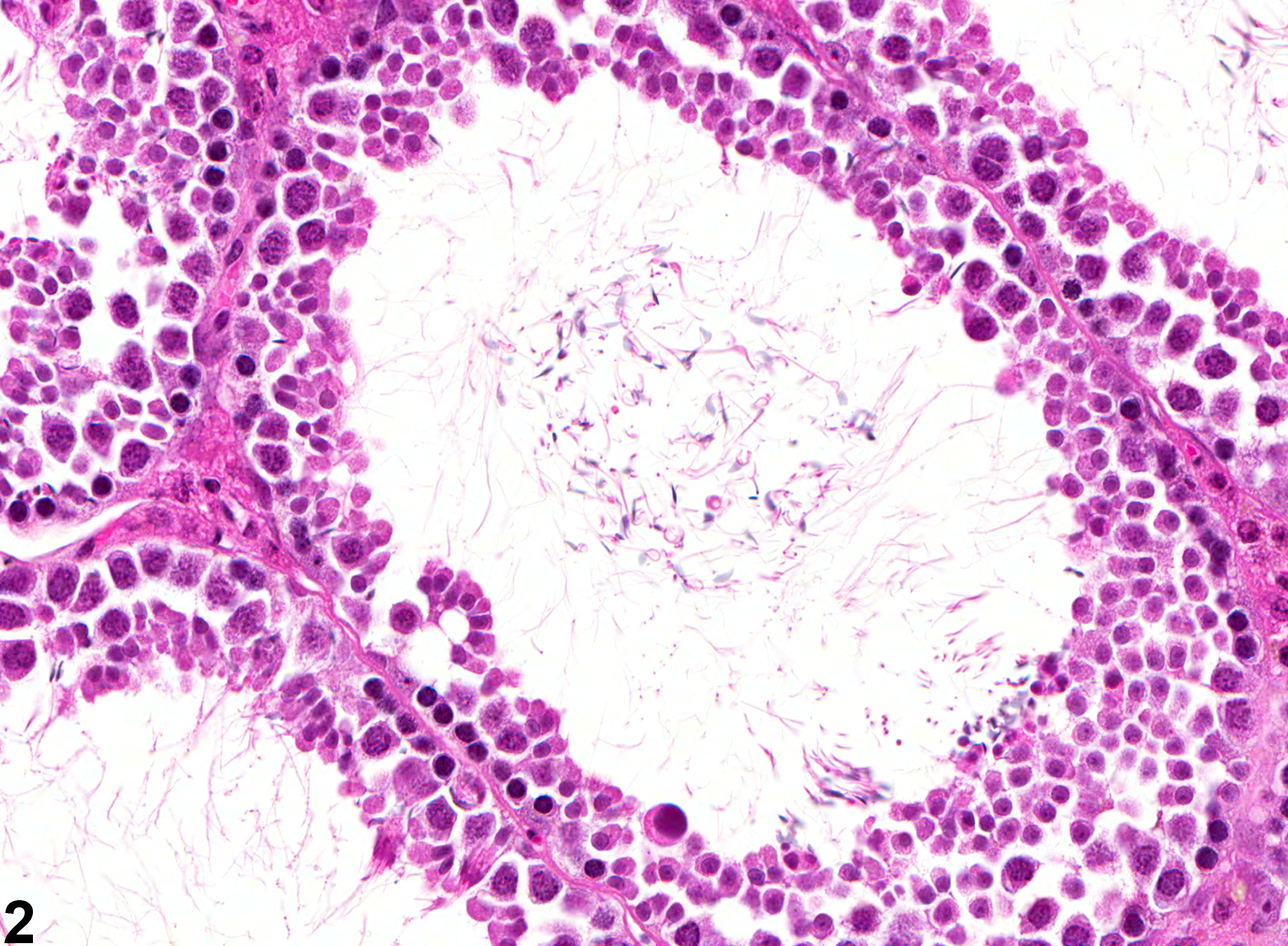
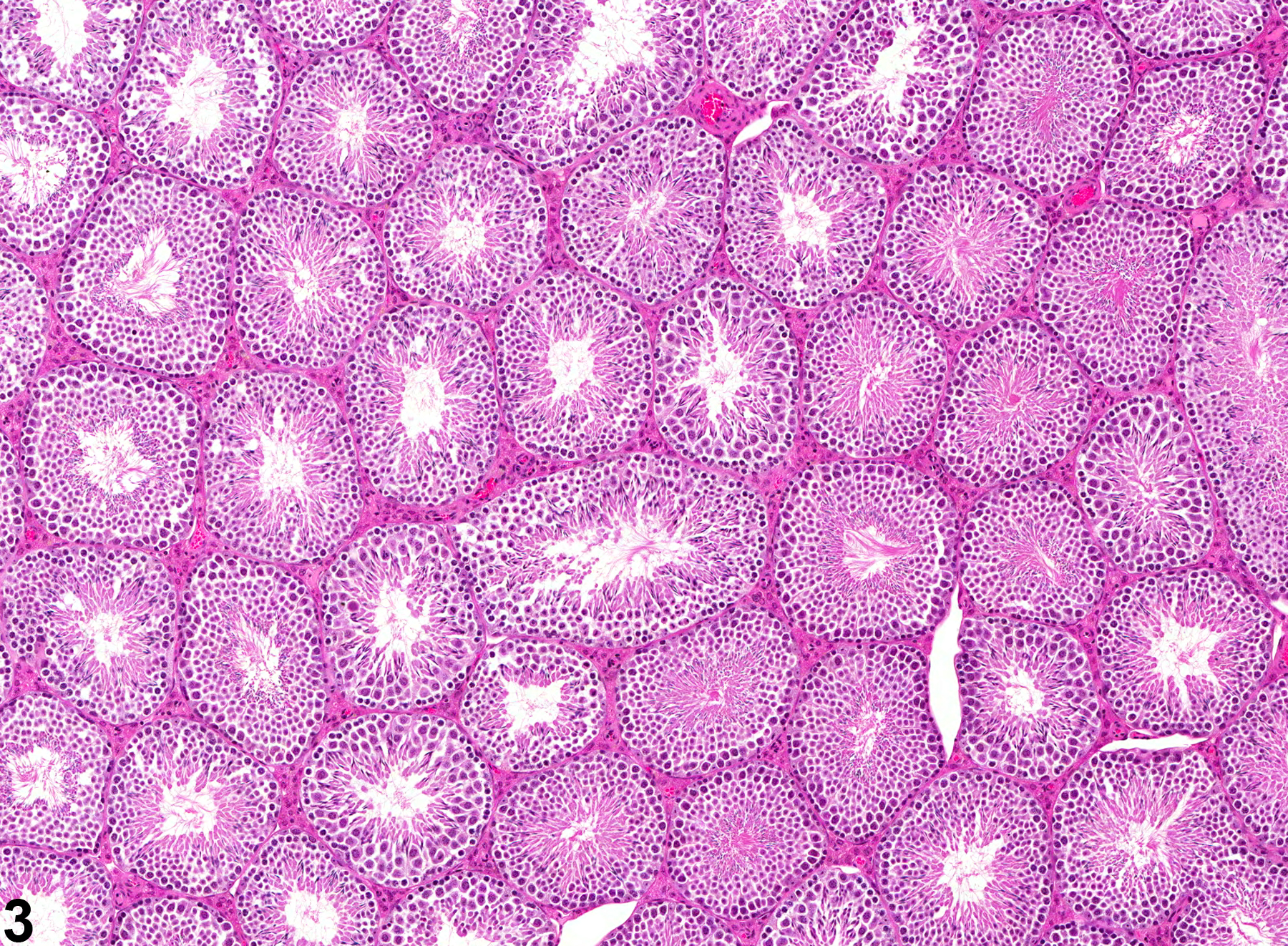
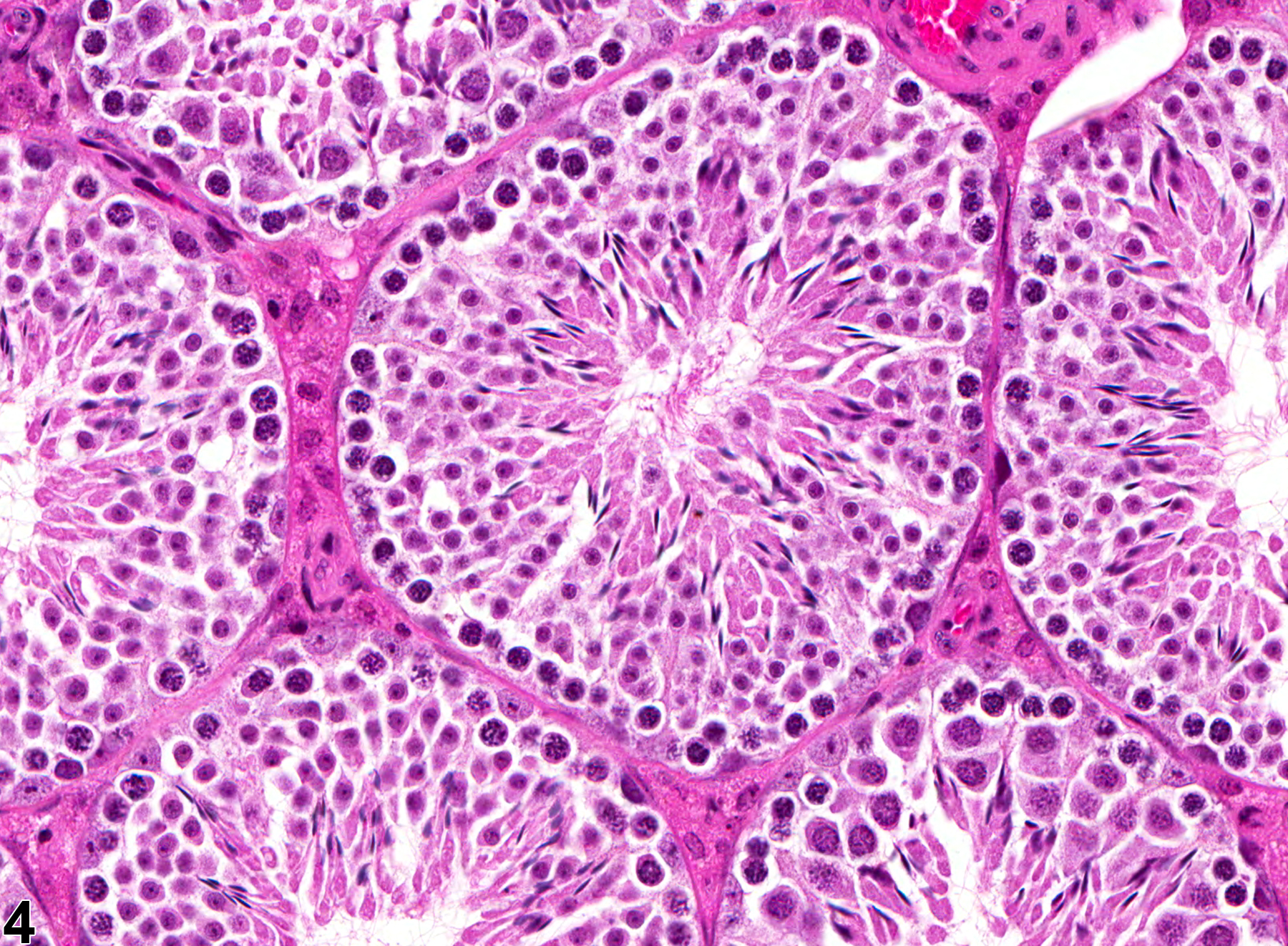
Testis, Seminiferous Tubule - Dilation
Narrative
Creasy DM. 2001. Pathogenesis of male reproductive toxicity. Toxicol Pathol 29:64-76.
Full Text: http://tpx.sagepub.com/content/29/1/64.full.pdfCreasy DM. 2012. Reproduction of the rat, primate, dog and pig. In: Background Lesions in Laboratory Animals: A Colour Atlas (McKinnes E, ed). Saunders Elselvier, Edinburgh. 101-122.
Abstract: http://www.sciencedirect.com/science/book/9780702035197Eddy EM, Washburn TF, Bunch DO, Goulding EH, Gladen BC, Lubahn DB, Korach KS. 1996. Targeted disruption of the estrogen receptor gene in male mice causes alteration of spermatogenesis and infertility. Endocrinology 137:4796-4805.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/8895349Hess RA. 1998. Effects of environmental toxicants on the efferent ducts, epididymis and fertility. J Reprod Fertil Suppl 53:247-259.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/10645284Hess RA. 2002. The efferent ductules: Structure and function. In: The Epididymis: From Molecules to Clinical Practice (Robaire B, Hinton BT, eds). Kluwer /Plenum, New York, 49-80.
Abstract: http://www.springer.com/medicine/urology/book/978-0-306-46684-7La DK, Johnson CA, Creasy DM, Hess RA, Baxter E, Pereira M, Snook SS. 2011. Efferent duct toxicity with secondary testicular changes in rats following administration of a novel leukotriene A4 hydrolase inhibitor. Toxicol Pathol 40:705-714.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/22552396Mecklenburg L, Ockert D, Kemkowski J, Kohler M. 2013. Selective inhibition of PDE4 in Wistar rats can lead to dilatation in the testis, efferent ducts and epididymis and subsequent formation of sperm granulomas. Toxicol Pathol 41:615-627.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/23197197Nakai M, Hess RA, More BJ, Guttroff RF, Strader LF, Linder RE. 1992. Acute and long-term effects of a single dose of the fungicide carbendazim (methyl 2-benzimidazole carbamate) on the male reproductive system in the rat. J Androl 13:507-518.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/1293130Piner J, Sutherland M, Millar M, Turner K, Newall D, Sharpe RM. 2002. Changes in vascular dynamics of the adult rat testis leading to transient accumulation of seminiferous tubule fluid after administration of a novel 5-hydroxytryptamine (5HT) agonist. Reprod Toxicol 16:141-150.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11955945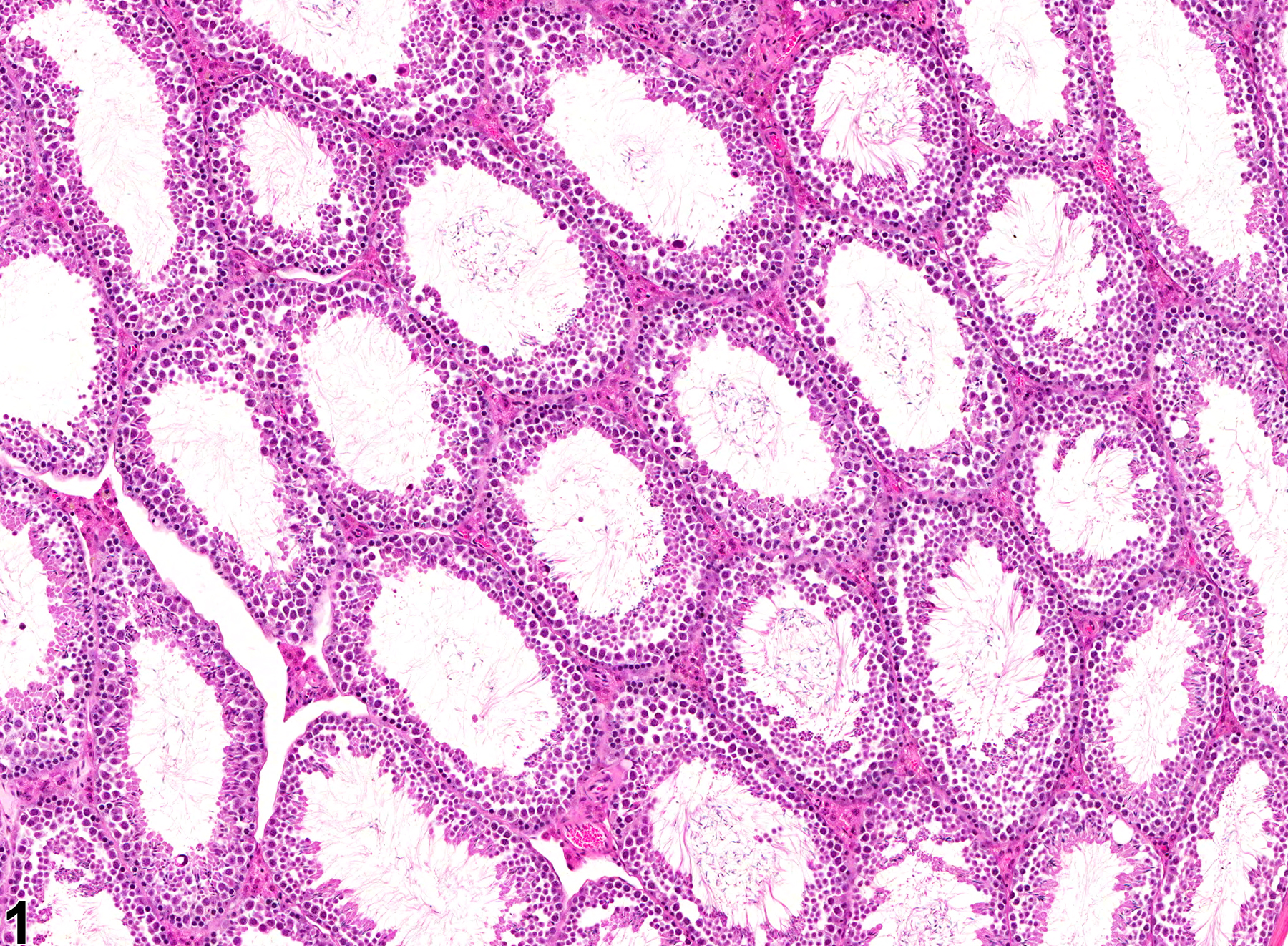
Testis, Seminiferous tubule - Dilation in a male B6C3F1 mouse from a chronic study. These seminiferous tubules are dilated.