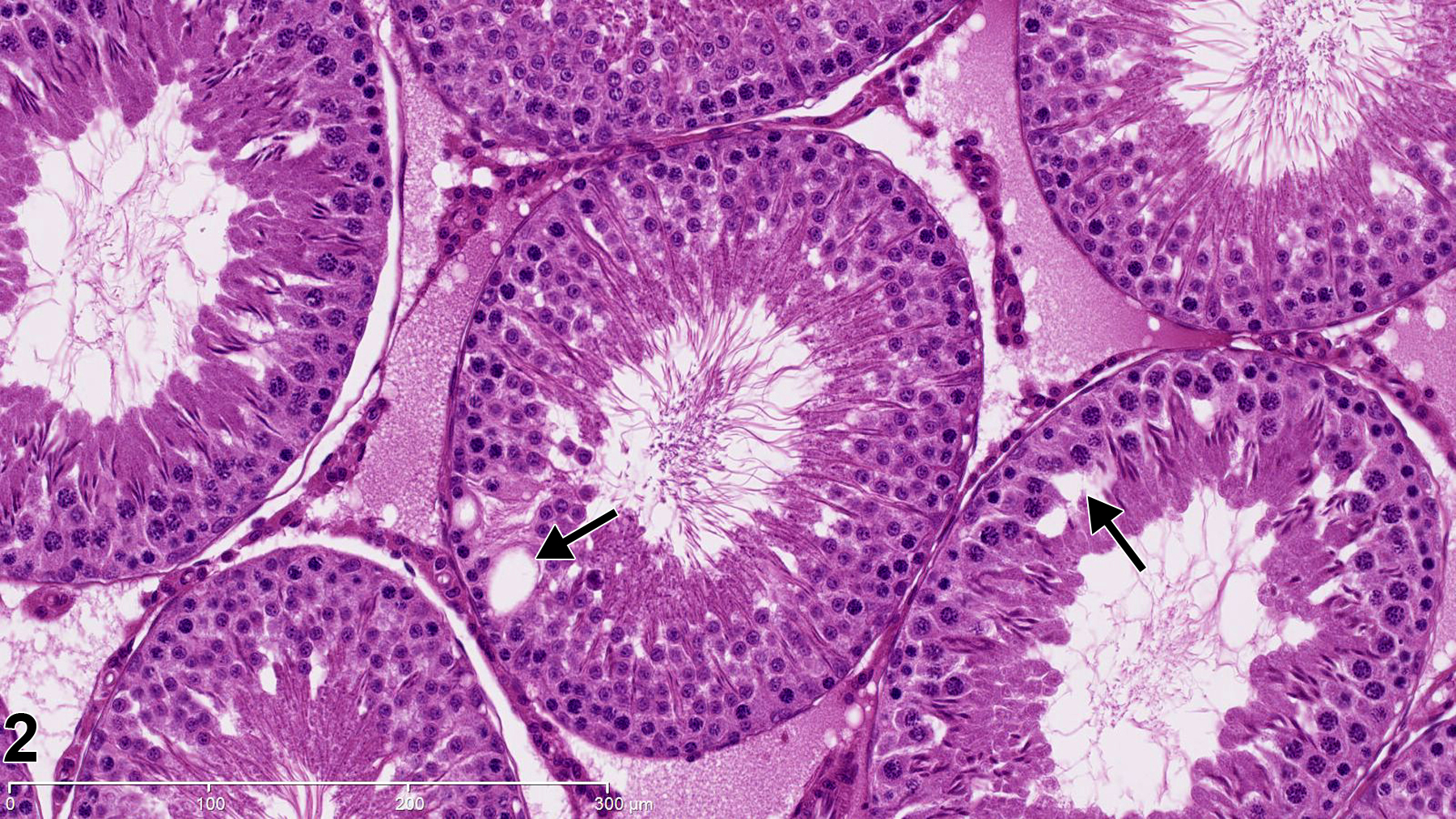
Reproductive System, Male
Testis, Seminiferous Tubule - Vacuolation
Narrative
Creasy DM. 2001. Pathogenesis of male reproductive toxicity. Toxicol Pathol 29:64-76.
Full Text: http://tpx.sagepub.com/content/29/1/64.full.pdfHild SA, Reel JR, Larener JM, Blye RP. 2001. Disruption of spermatogenesis and Sertoli cell structure and function by the indenopyridine CDB-4022 in rats. Biol Reprod 65:1771-1779.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/11717140Kerr JB, Savage GN, Millar M, Sharpe RM. 1993. Response of the seminiferous epithelium of the rat testis to withdrawal of androgen: Evidence for direct effect upon intercellular spaces associated with Sertoli cell junctional complexes. Cell Tissue Res 274:153-161.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/8242704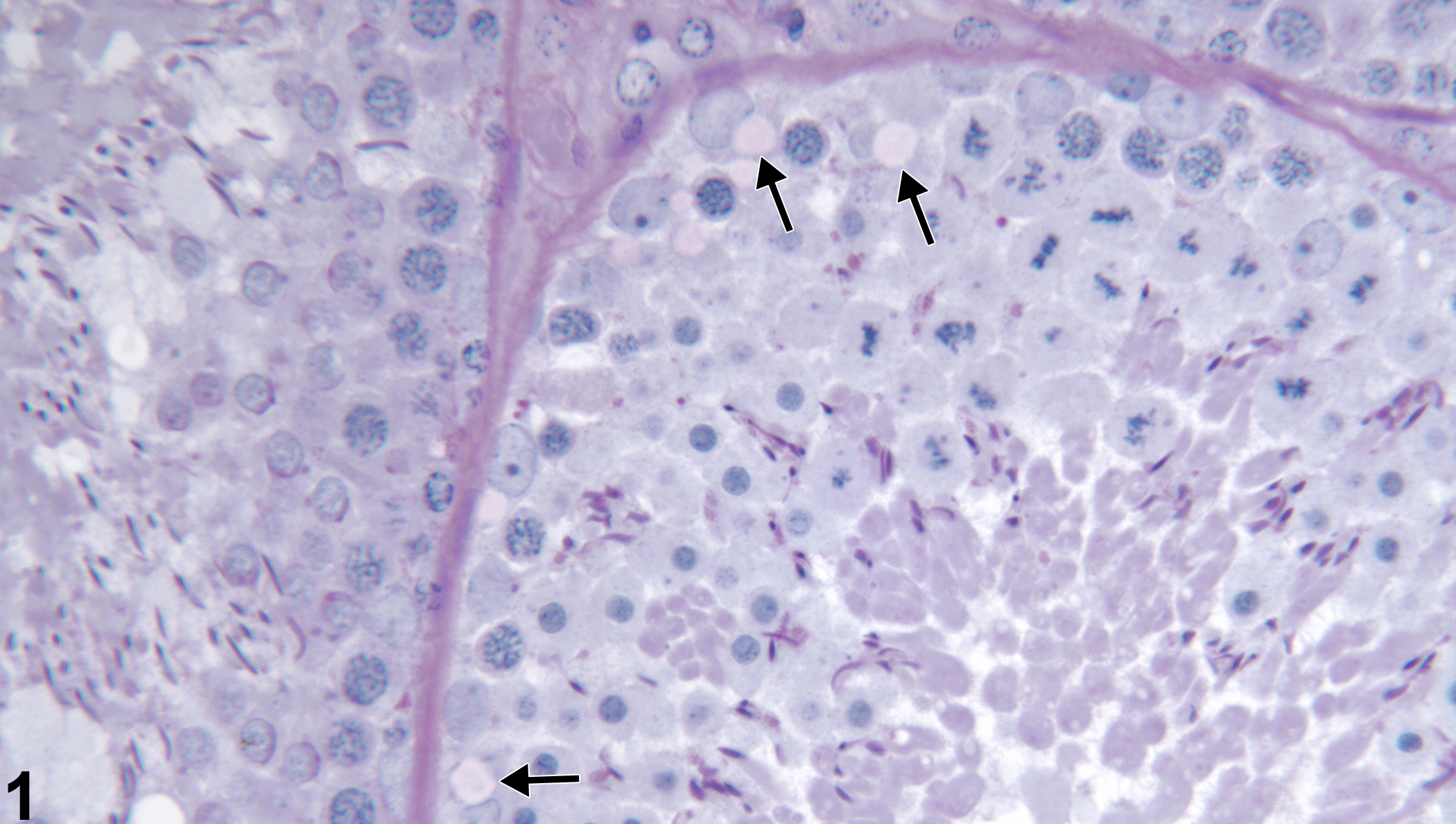
Testis, Seminiferous tubule - Vacuolation in a male Harlan Sprague-Dawley rat from a reproductive and continuous breeding study. Vacuolation in basal Sertoli cell cytoplasm (arrows). PAS stain.