Reproductive System, Male
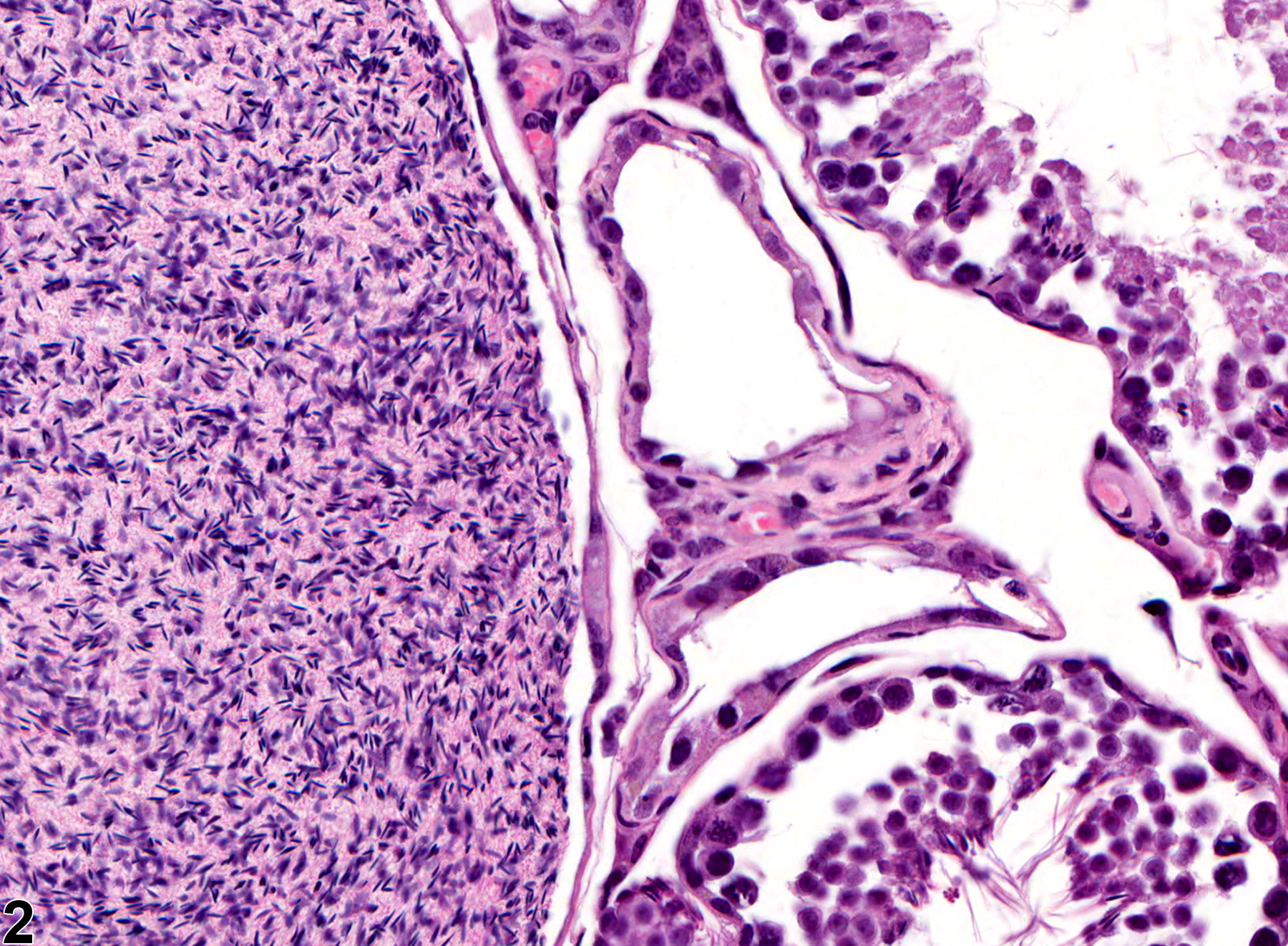
Testis - Spermatocele
Narrative
Gordon LR, Majka JA, Boorman GA. 1996. Spontaneous nonneoplastic and neoplastic lesions and experimentally induced neoplasms of the testes and accessory sex glands. In: Pathobiology of the Aging Mouse, Vol 1 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 421-441.
Abstract: http://catalog.hathitrust.org/Record/008994685Itoh M, Li XQ, Miyamoto K, Takeuchi Y. 1999. Degeneration of the seminiferous epithelium with aging is a cause of spermatoceles. Int J Androl 22:91-96.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/10194640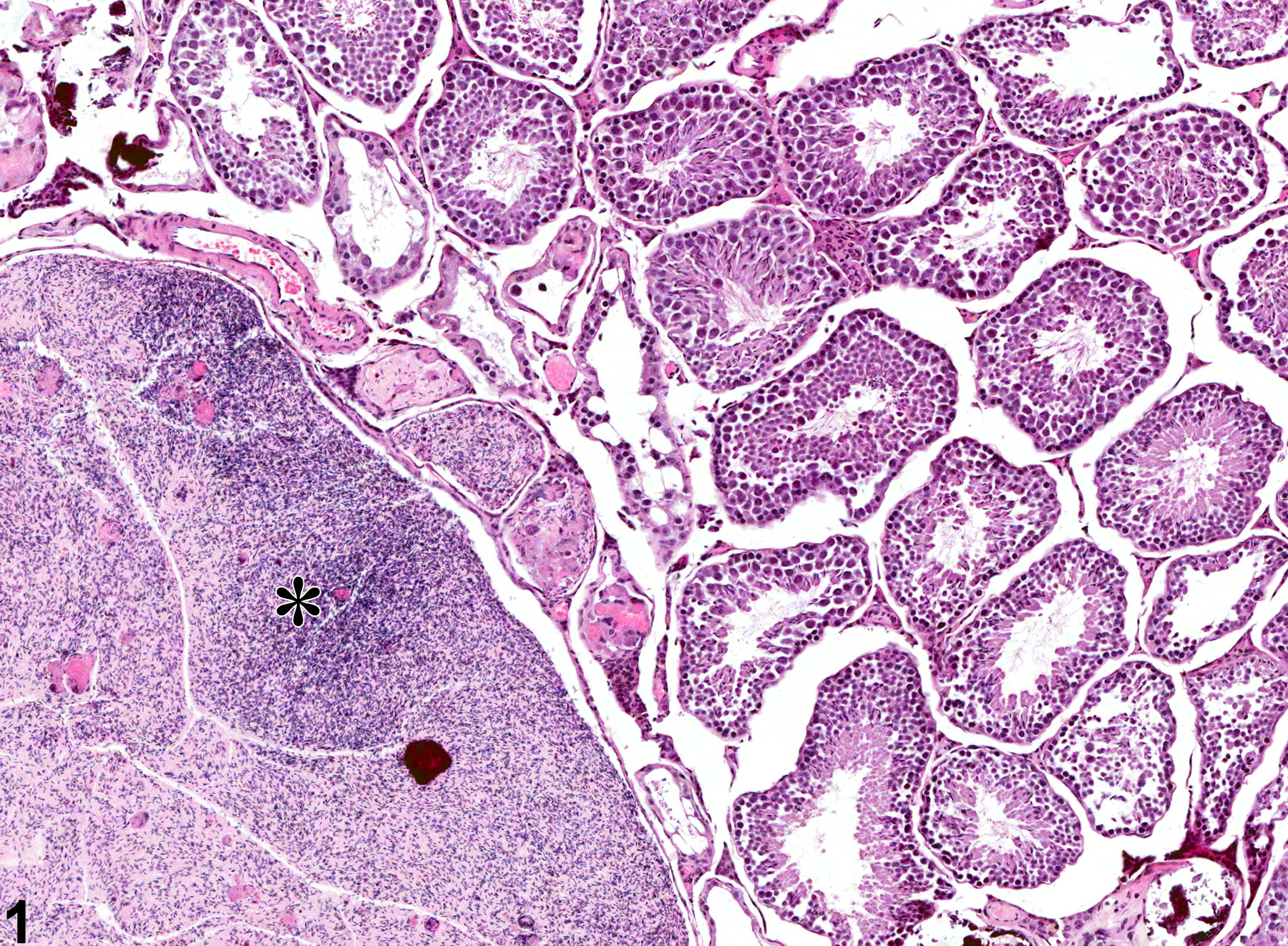
Testis - Spermatocele in a male B6C3F1 mouse from a chronic study. There is a large spermatocele in the testis (asterisk).