Musculoskeletal System
Skeletal Muscle - Atrophy
Narrative
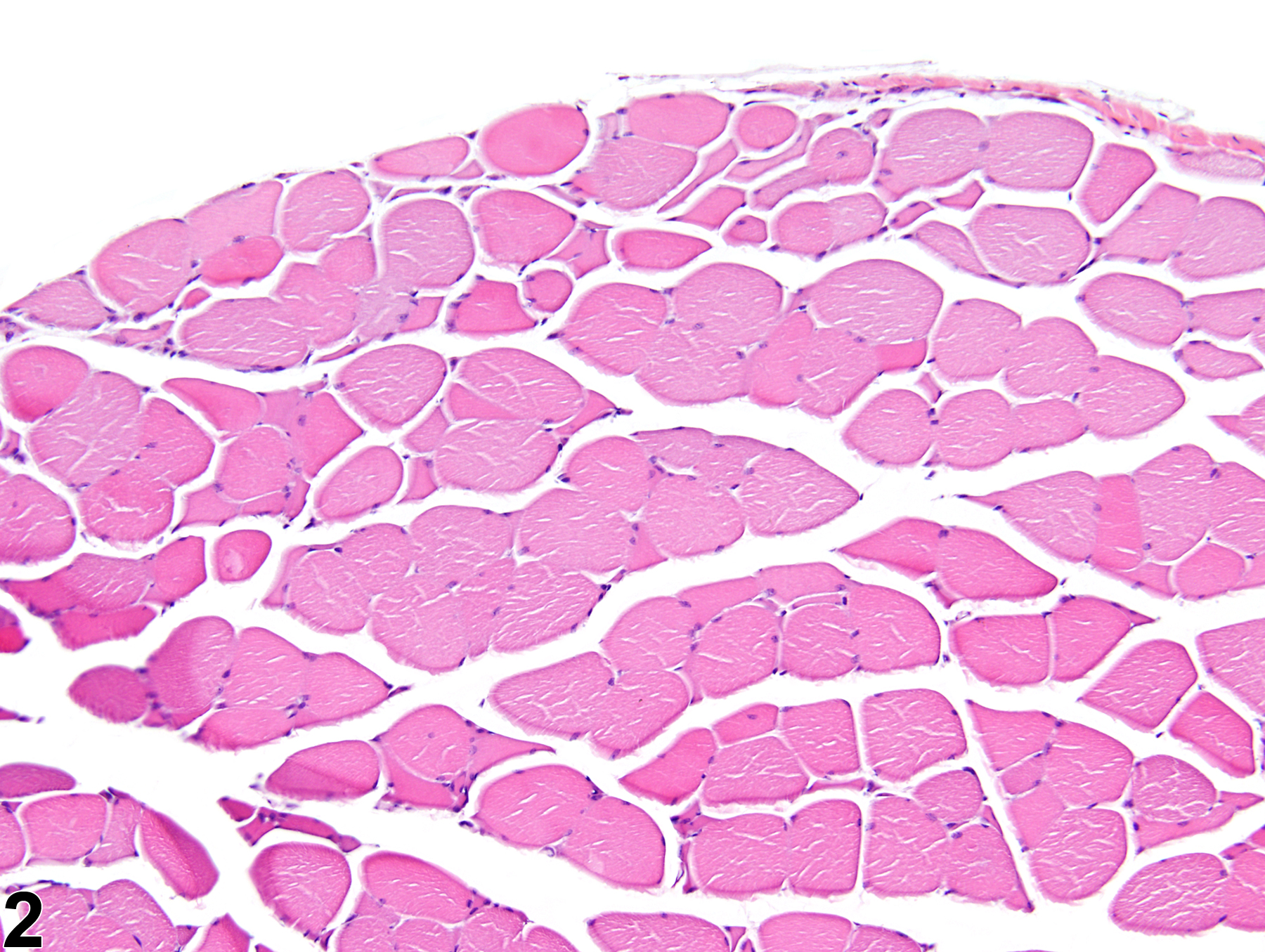
The main histologic feature of skeletal muscle atrophy is a reduction in myofiber diameter (Figure 1 and Figure 2). Affected myofibers are often rounded to angular with hypereosinophilic sarcoplasm. In denervation atrophy, the characteristic histologic feature is that of compressed angular myofibers with crowded nuclei. Atrophy due to nonneurogenic causes, such as that induced by the administration of myotoxic xenobiotics, often lacks these fibers and is instead accompanied by traditional myopathic changes, such as necrotic or hyalinized myofibers, split or fragmented myofibers, and myofibers with central nuclei. Spontaneous atrophy of aging rodents (often referred to as hind leg myopathy) is histologically characterized by decreased myofiber size and number; increased myofiber size variation; increased accumulation of degenerative inclusion bodies, lipofuscin, and lipid droplets; and increased connective tissue. Angulated myofibers are often present, suggesting a possible role of spinal degeneration within this process. However, spinal nerve lesions are not consistently observed in all affected animals; the pathogenesis remains unclear. This condition is predominately noted in the hind limbs of affected rats. It has been described in several strains of rats, including Wistar and Sprague-Dawley, but has not been recorded in the F344 rat.
Atrophy can uniformly affect myofibers or selectively target specific muscle fiber types. Type II fibers are selectively affected when atrophy is associated with disuse, cachexia, or malnutrition. Chronic administration of corticosteroids in rats results in the selective atrophy of type II fast-twitch fibers. Conversely, type I fibers are selectively targeted during experimentally induced thyrotoxicosis.
Berridge BR, Van Vleet JF, Herman E. 2013. Cardiac, vascular, and skeletal muscle systems. In: Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed (Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R, Mahler MW, eds). Elsevier, Amsterdam, 1635-1665.
Greaves P. 2007. Musculoskeletal system. In: Histopathology of Preclinical Toxicity Studies, 3rd ed. Elsevier, Oxford, 160-214.
Greaves P, Seely JC. 1996. Non-proliferative lesions of soft tissues and skeletal muscle in rats, MST-1. In: Guides for Toxicologic Pathology. STP/ARP/AFIP, Washington, DC.
Greaves P, Chouinard L, Ernst H, Mecklenburg L, Pruimboom-Brees IM, Rinke M, Rittinghausen S, Thibault S, von Erichsen J, Yoshida T. 2013. Proliferative and non-proliferative lesions of the rat and mouse soft tissue, skeletal muscle, and mesothelium. J Toxicol Pathol 26(3 suppl):1S-26S.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/25035576Leininger JR. 1999. Skeletal muscle. In: Pathology of the Mouse (Maronpot R, Boorman G, Gaul BW, eds). Cache River Press, St Louis, 637-643.
McDonald MM, Hamilton BF. 1990. Bones, joints, and synovia. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman G, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 193-207.
Vahle JL, Leininger JR, Long PH, Hall DG, Ernst H. 2013. Bone, muscle, and tooth. In: Toxicologic Pathology: Nonclinical Safety Assessment (Sahota PS, Popp JA, Hardisty JF, Gopinath C, eds). CRC Press, Boca Raton, FL, 561-587.
Van Vleet JF, Valentine BA. 2007. Muscle and tendon. In: Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, 5th ed, Vol 1 (Grant MG, ed). Elsevier, Edinburgh, 185-280.
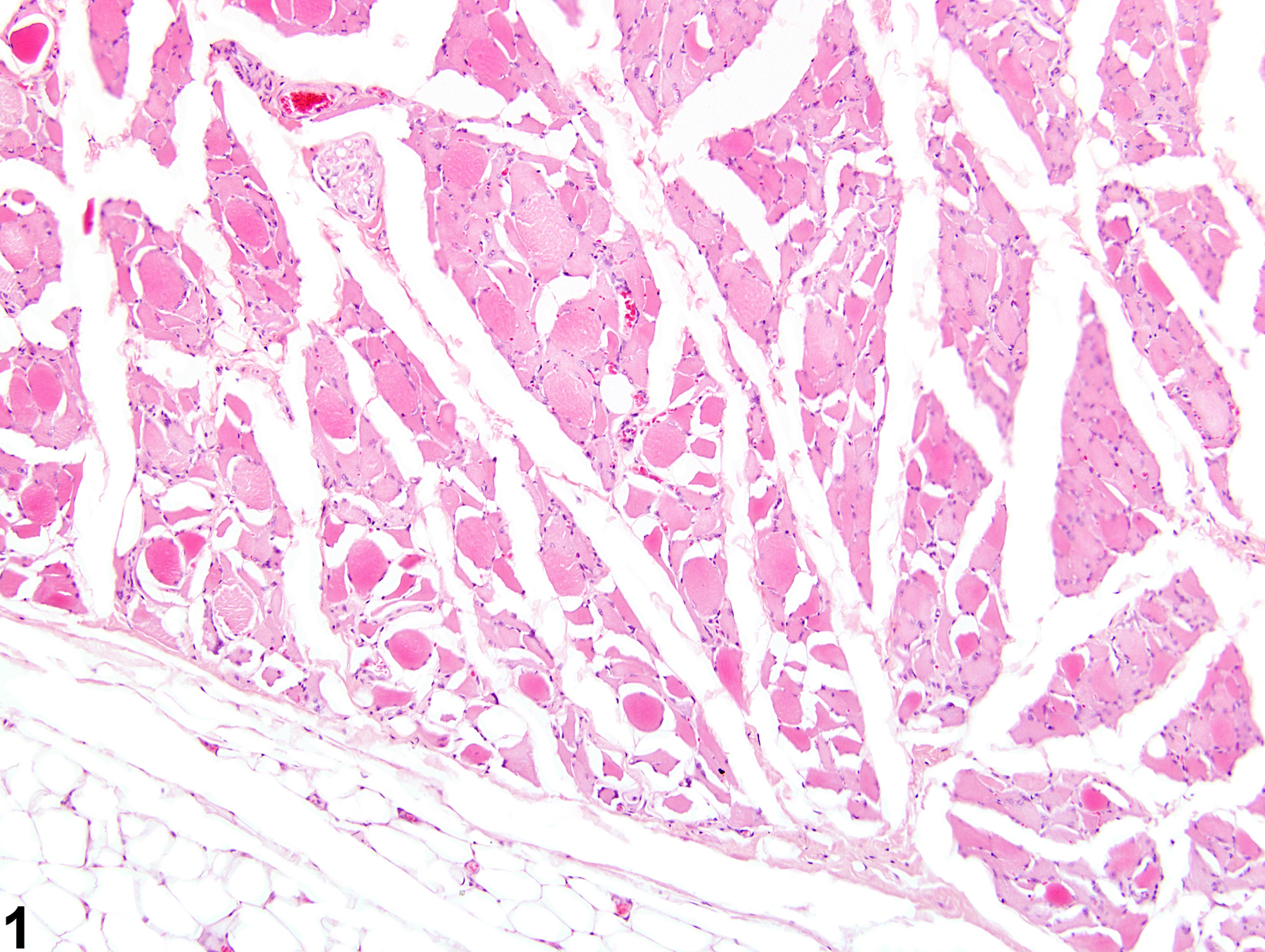
muscle - Atrophy in a male F344/N rat from a chronic study. Individual hypereosinophilic muscle fibers of reduced diameter are sometimes surrounded by clear spaces.