Musculoskeletal System
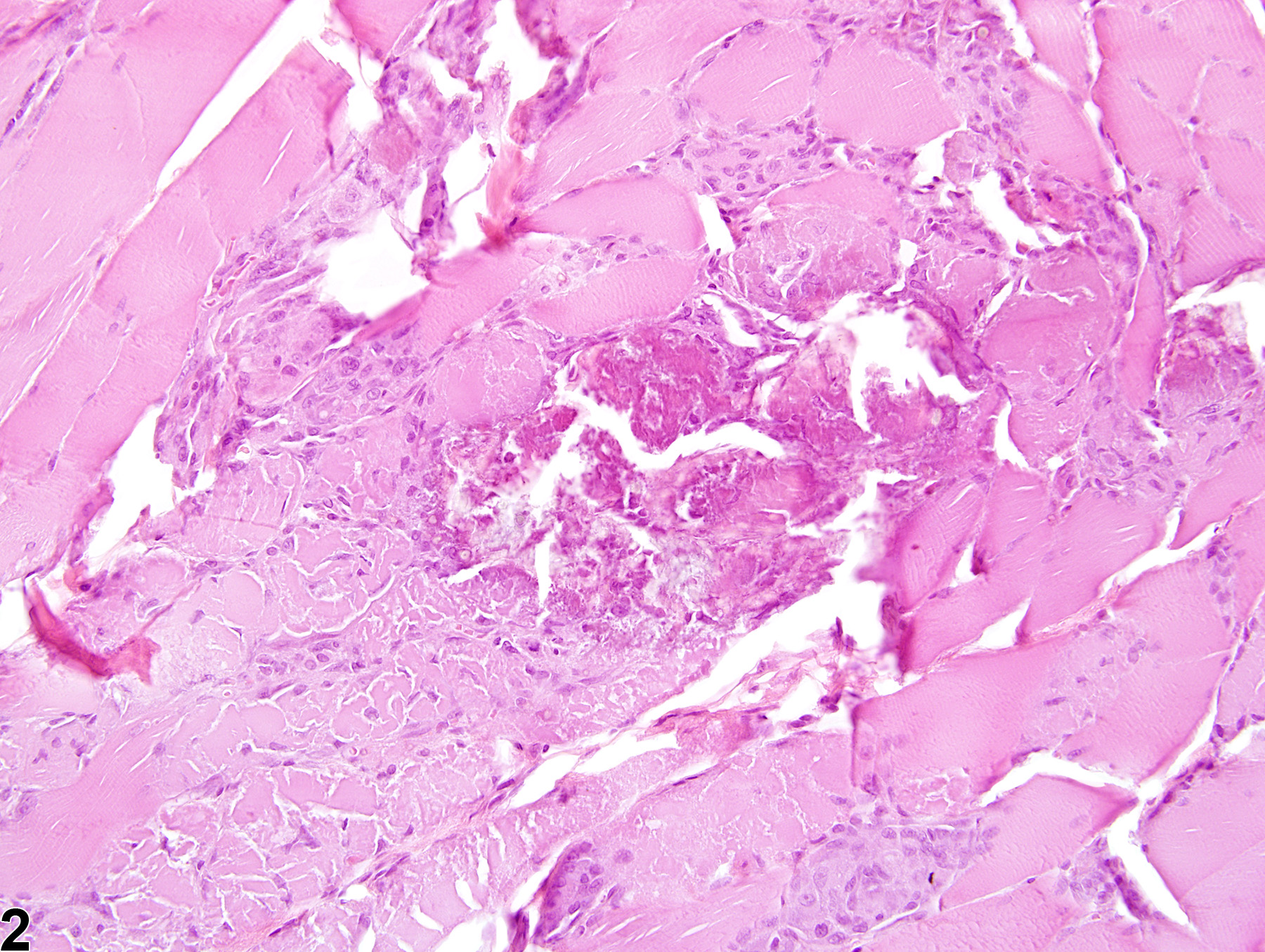
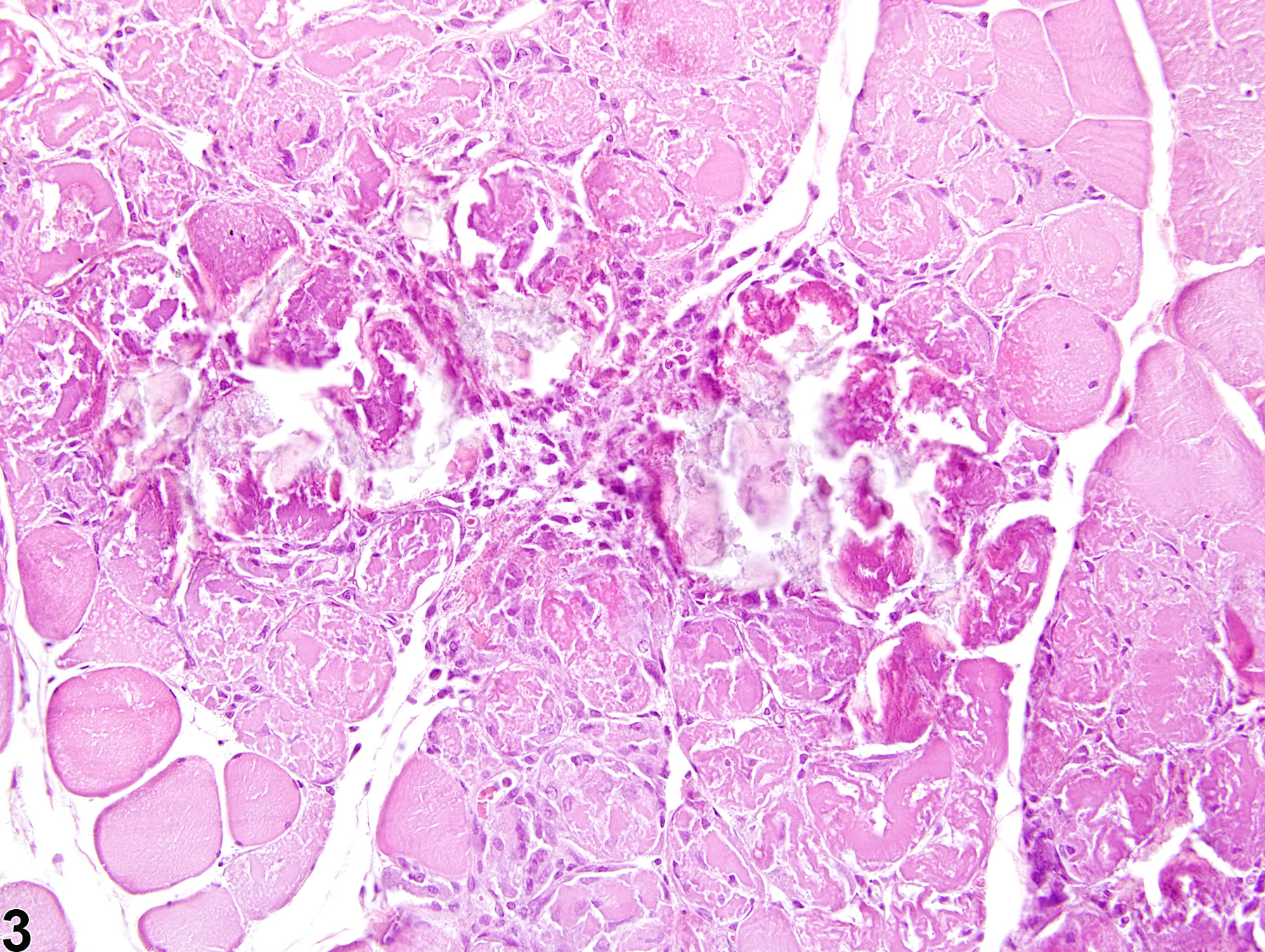
Skeletal Muscle - Necrosis
Narrative
Degeneration and necrosis represent a continuum of lesions and therefore are often both present within a given lesion. Due to the limited repertoire of skeletal muscle responses, they share similar morphologic features and can thus be difficult to differentiate histologically. This can create a diagnostic challenge. When evaluating a toxicity study, it is important for the pathologist to establish distinct criteria for both lesions and to be consistent and careful when applying them. Criteria should be described in the narrative. The term "myopathy" has commonly been used to describe disorders of skeletal muscle in which degeneration and necrosis are key features. However, since myopathy is a general term and one that typically encompasses a collection of lesions rather than one distinct lesion, its use is not recommended.
Berridge BR, Van Vleet JF, Herman E. 2013. Cardiac, vascular, and skeletal muscle systems. In: Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed (Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R, Mahler MW, eds). Elsevier, Amsterdam, 1635-1665.
Greaves P. 2007. Musculoskeletal system. In: Histopathology of Preclinical Toxicity Studies, 3rd ed. Elsevier, Oxford, 160-214.
Greaves P, Seely JC. 1996. Non-proliferative lesions of soft tissues and skeletal muscle in rats, MST-1. In: Guides for Toxicologic Pathology. STP/ARP/AFIP, Washington, DC.
Greaves P, Chouinard L, Ernst H, Mecklenburg L, Pruimboom-Brees IM, Rinke M, Rittinghausen S, Thibault S, von Erichsen J, Yoshida T. 2013. Proliferative and non-proliferative lesions of the rat and mouse soft tissue, skeletal muscle, and mesothelium. J Toxicol Pathol 26(3 suppl):1S-26S.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/25035576Haschek WM, Rousseaux CG, Wallig MA. 2010. Cardiovascular and skeletal muscle systems. In: Fundamentals of Toxicologic Pathology, 2nd ed. Academic Press, San Diego, 319-376.
Leininger JR. 1999. Skeletal muscle. In: Pathology of the Mouse (Maronpot R, Boorman G, Gaul BW, eds). Cache River Press, St Louis, 637-643.
Manor D, Sadeh M. 1989. Muscle fibre necrosis induced by intramuscular injection of drugs. Br J Exp Pathol 70:457-462.
Abstract: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2040563/McDonald MM, Hamilton BF. 1990. Bones, joints, and synovia. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman G, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 193-207.
Vahle JL, Leininger JR, Long PH, Hall DG, Ernst H. 2013. Bone, muscle, and tooth. In: Toxicologic Pathology: Nonclinical Safety Assessment (Sahota PS, Popp JA, Hardisty JF, Gopinath C, eds). CRC Press, Boca Raton, FL, 561-587.
Weller AH, Magliato SA, Bell KP, Rodenberg NL. 1997. Spontaneous myopathy in the SJL/J mouse: Pathology and strength loss. Muscle Nerve 20:72-82.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/8995586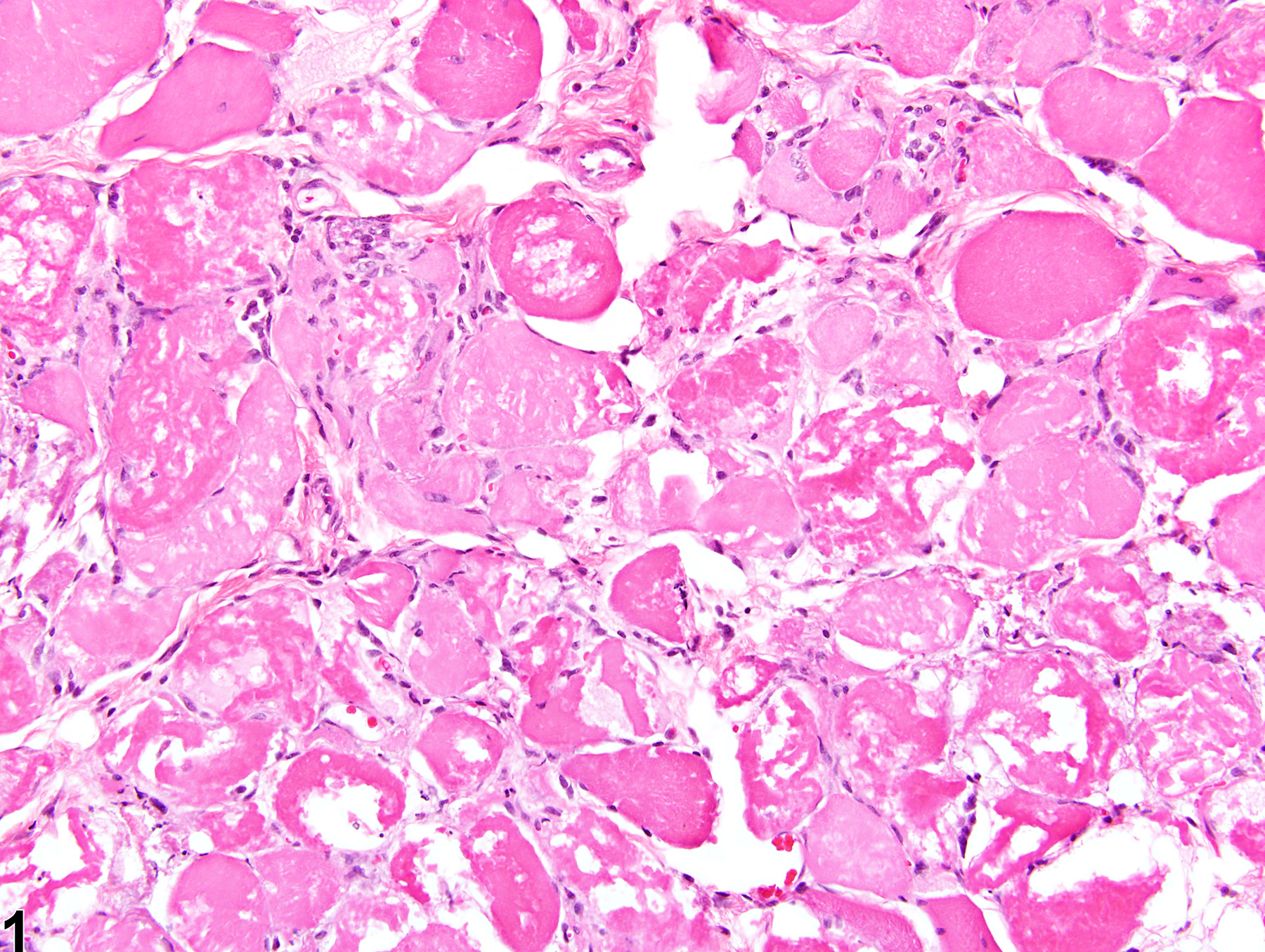
Skeletal muscle - Necrosis in a male F344/N rat from a chronic study. This cross section of skeletal muscle exhibits an extensive, highly fragmented and vacuolated collection of muscle fibers; some are hypereosinophilic, and others are pale.