Musculoskeletal System
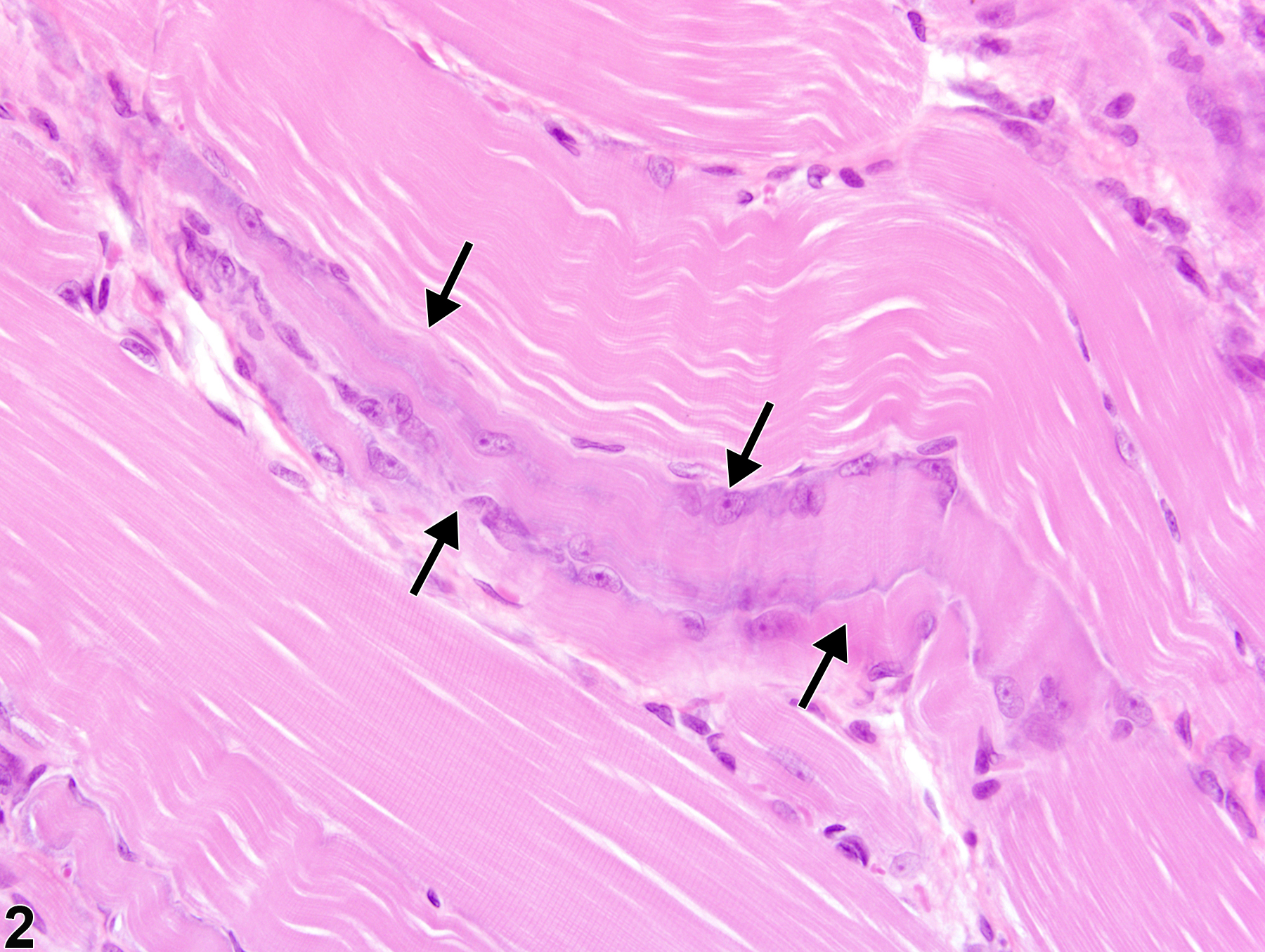
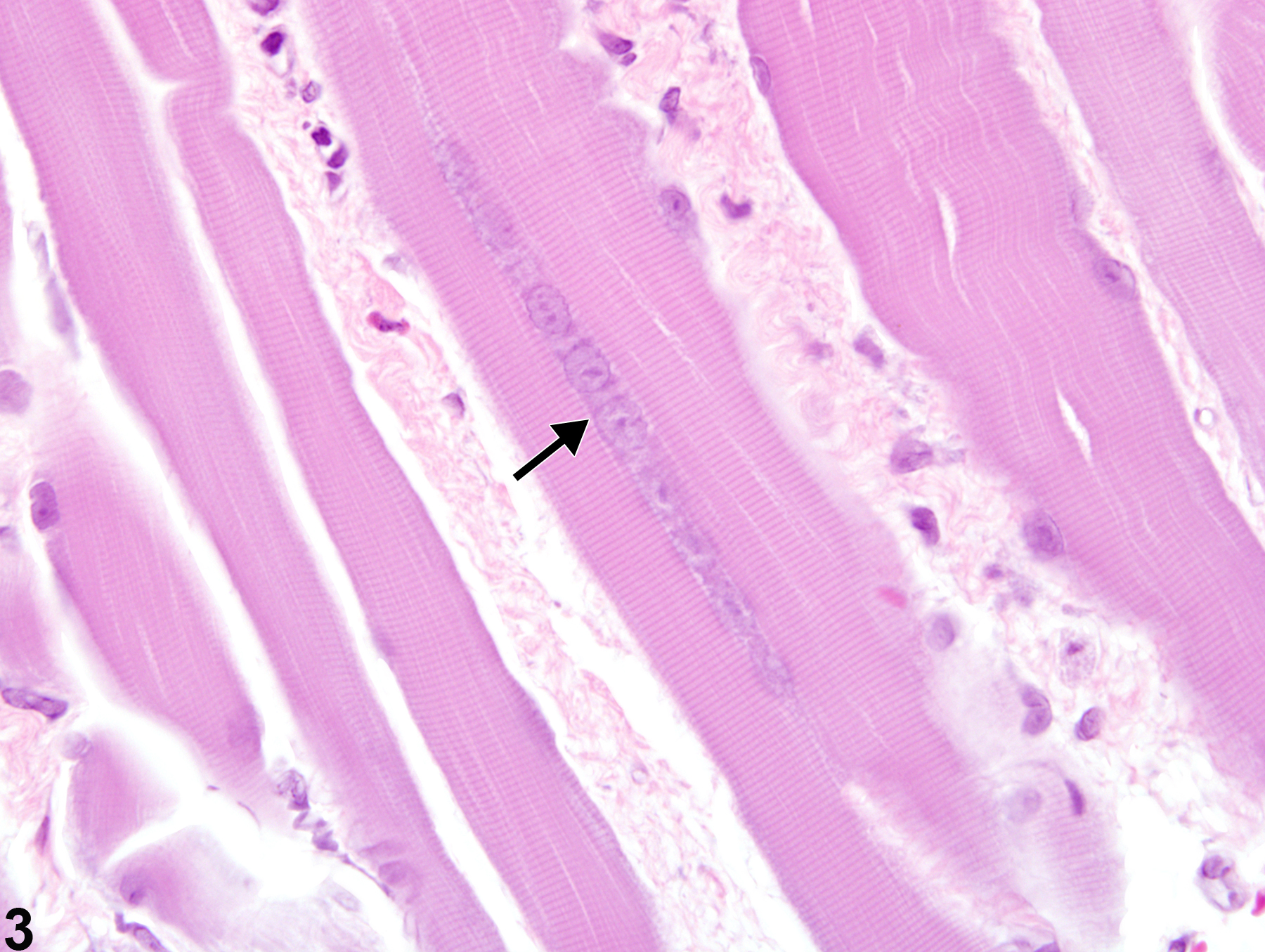
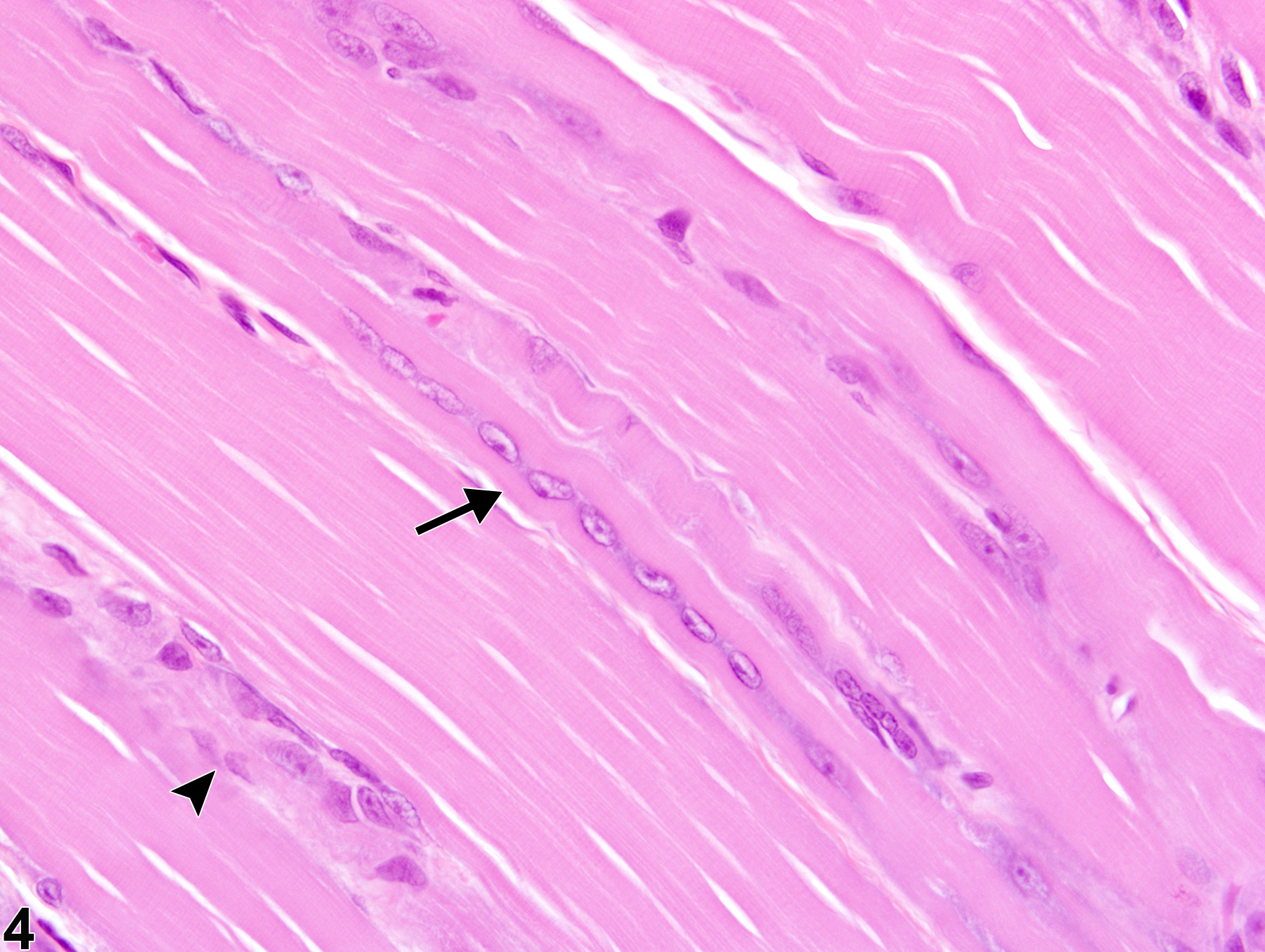
Skeletal Muscle - Regeneration
Narrative
Regenerating myofibers are commonly present within or near areas of active necrosis or degeneration, and the degree of regeneration present will be influenced by the amount of time that has passed between the injurious stimulus and sample collection. Occasional rowing of nuclei in the absence of prior or concurrent myotoxicity likely reflects background cellular turnover and repair and does not warrant a diagnosis.
Nutritional, exertional, and toxic myopathies can result in extensive regions of muscle damage; however, repair is often extremely effective. In contrast, the reparative process following damage due to ischemia is often ineffective. This is likely due to the subsequent death of the satellite cells and endomysial cells. Extensive regions of fibrosis often result.
Berridge BR, Van Vleet JF, Herman E. 2013. Cardiac, vascular, and skeletal muscle systems. In: Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed (Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R, Mahler MW, eds). Elsevier, Amsterdam, 1635-1665.
Greaves P, Chouinard L, Ernst H, Mecklenburg L, Pruimboom-Brees IM, Rinke M, Rittinghausen S, Thibault S, von Erichsen J, Yoshida T. 2013. Proliferative and non-proliferative lesions of the rat and mouse soft tissue, skeletal muscle, and mesothelium. J Toxicol Pathol 26(3 suppl):1S-26S.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/25035576Leininger JR. 1999. Skeletal muscle. In: Pathology of the Mouse (Maronpot R, Boorman G, Gaul BW, eds). Cache River Press, St Louis, 637-643.
Mann CJ, Perdiguero E, Kharraz Y, Aguilar S, Pessina P, Serrano AL, Muñoz-Cánoves P. 2001. Aberrant repair and fibrosis development in skeletal muscle. Skelet Muscle 1:21.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/21798099McDonald MM, Hamilton BF. 1990. Bones, joints, and synovia. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman G, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 193-207.
Vahle JL, Leininger JR, Long PH, Hall DG, Ernst H. 2013. Bone, muscle, and tooth. In: Toxicologic Pathology: Nonclinical Safety Assessment (Sahota PS, Popp JA, Hardisty JF, Gopinath C, eds). CRC Press, Boca Raton, FL, 561-587.
Van Vleet JF, Valentine BA. 2007. Muscle and tendon. In: Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, 5th ed, Vol 1 (Grant MG, ed). Elsevier, Edinburgh, 185-280.
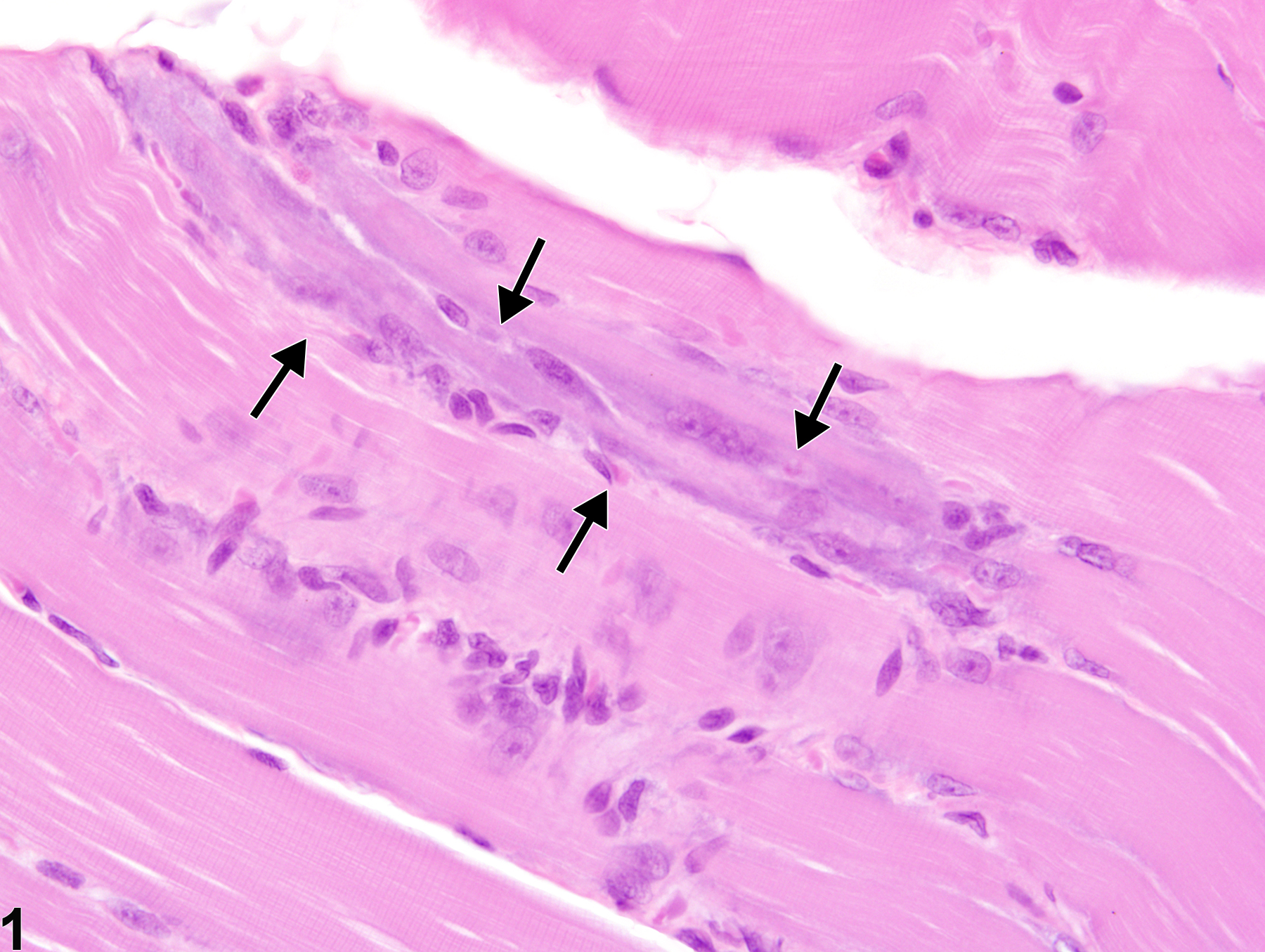
Skeletal muscle - Regeneration in a male Harlan Sprague-Dawley rat from a subchronic study. Myoblasts and cytoplasmic basophilia (arrows) characterize regeneration of a previously damaged muscle fiber.