Nervous System
Brain - Axonopathy
Narrative
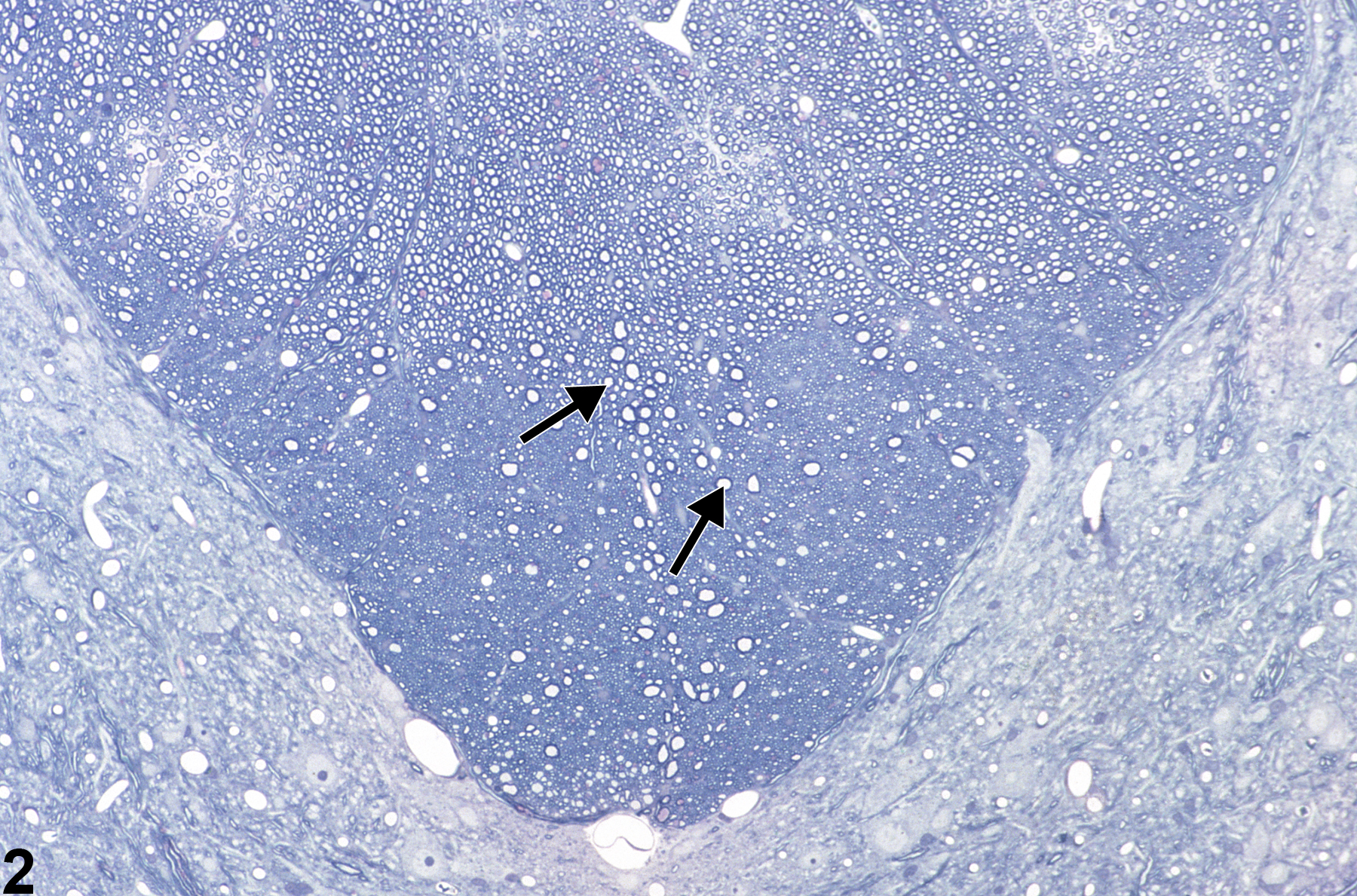
Figure 1 shows spinal cord at low magnification demonstrating the appearance of axonopathy in the dorsal proprioceptive funiculi, in this case the fasciculus gracilis. Note, at this magnification, that the lesion is represented by subtle vacuolation (arrow) of a focal area ordinarily populated by normal ascending axons. Damage to the axons in this zone leads to swelling of both the axon and its myelin sheath, thus creating a dilated space. This image uses toluidine blue stain. Figure 2 is a higher-magnification toluidine blue–stained section of spinal cord showing axonal swelling in the fasciculus gracilis in a subchronic toxicity study. Note the many distended axons (arrows), most still surrounded by their myelin sheath. There is a prominent reduction in myelinated fibers, particularly in the deepest parts of the funiculi, making the swollen myelinated axons in these regions stand out in relief as large ”vacuoles.”
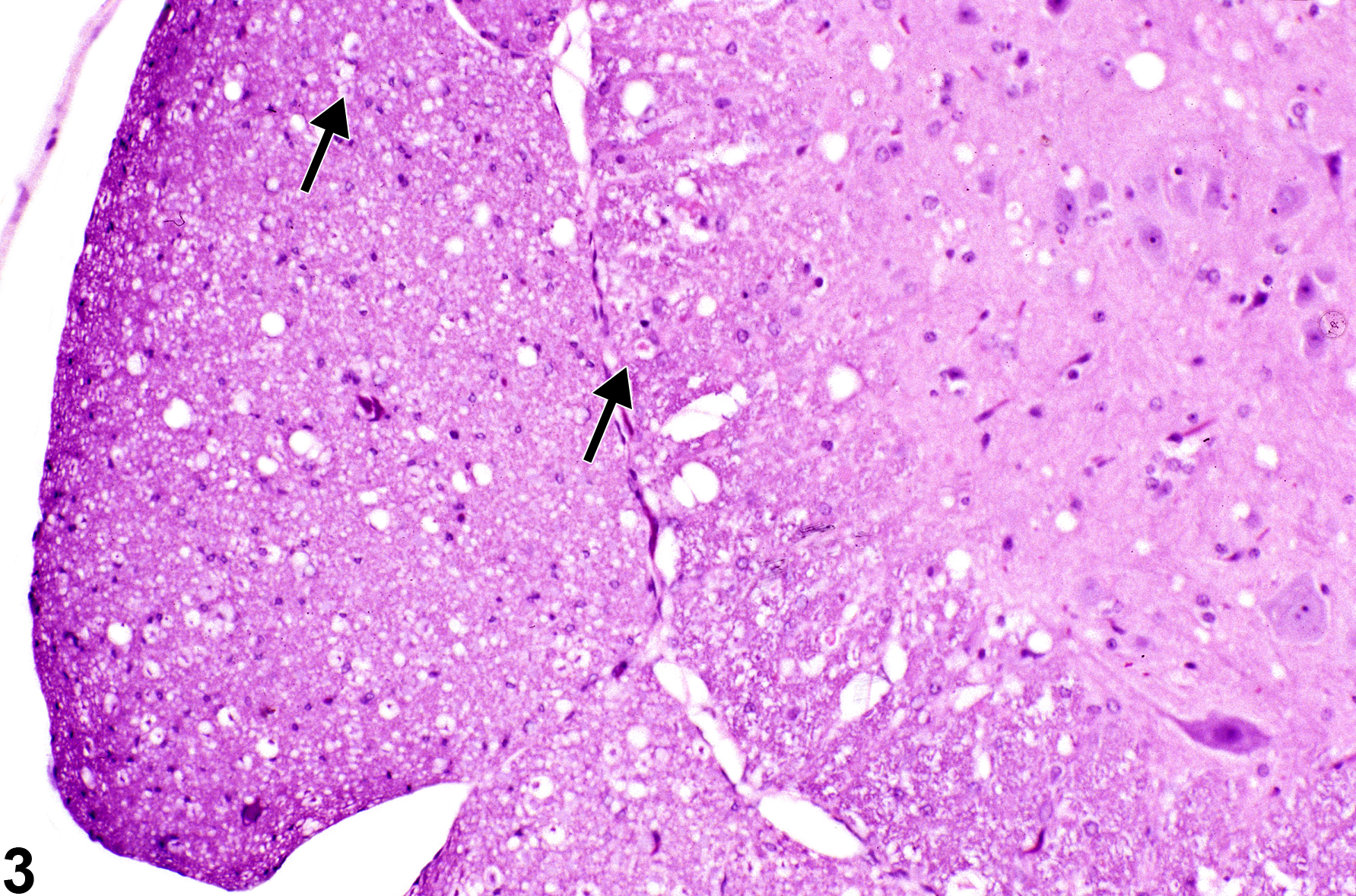
Figure 3, Figure 4, and Figure 5 depict swollen axons. In Figure 3, note the slightly enlarged occasional swollen axons (arrows) at the center of dilated spaces formerly occupied by a normal, compact lamellar myelin sheath. Many of the dilated spaces are empty because the axon has already undergone disintegration, lysis, and phagocytosis by macrophages. The space appears empty because the use of fat solvents in tissue processing has removed any remaining complex lipids, which made up the myelin sheath.
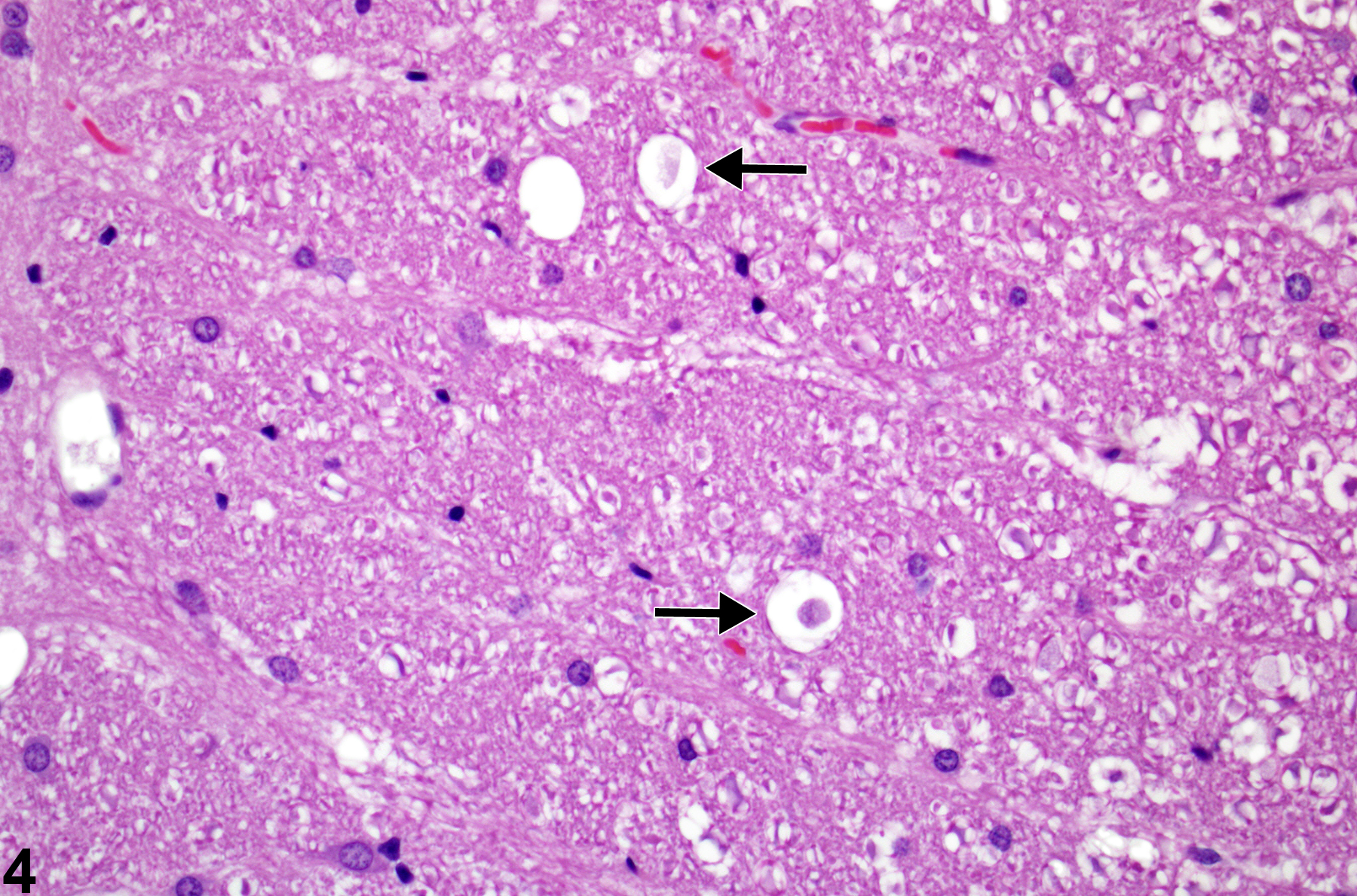
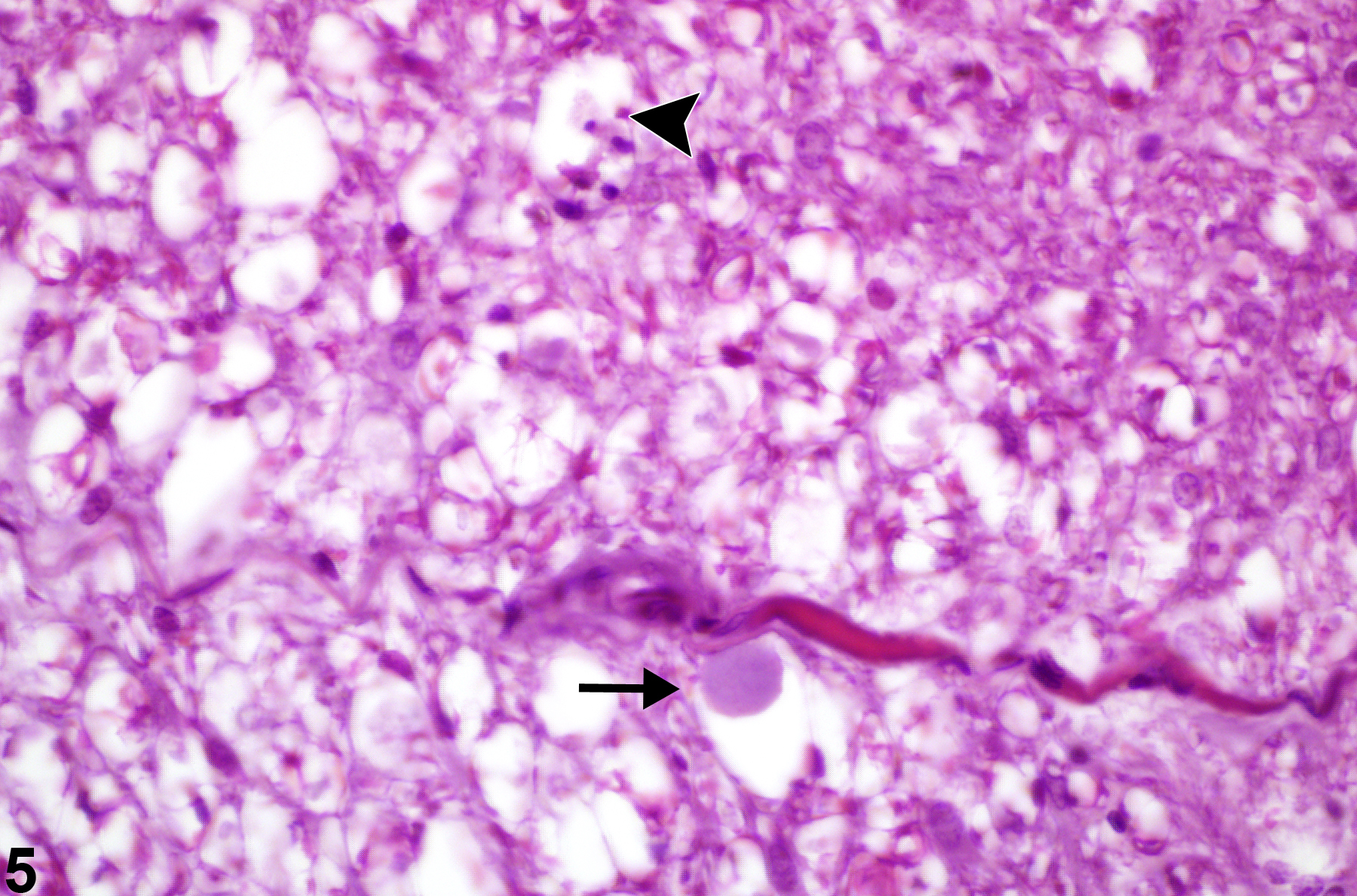
Figure 4 represents swollen degenerate axons at higher magnification (arrows), where the diameter of the distended space surrounding them is approximately three times larger than the space around adjacent, apparently normal axons. Figure 5 represents spinal funicular axonopathy with detail of a swollen axon, a so-called spheroid (arrow). Note the pyknotic macrophage in a vacuole representing a later stage of axonal degeneration characterized by axonal phagocytosis (arrowhead). In most cases of axonal degeneration, identifiable macrophages are usually necrotic, presumably from the effects of myelin lipid phagocytosis and cytokine release during the axonal degenerative process. Apoptosis of macrophages results when they are loaded with free cholesterol and triacylglycerol. In this high-magnification hematoxylin and eosin–stained image, the large swollen axon has an amphophilic appearance. This is a good example of a spheroid at an early stage of axonal swelling that precedes axonal disintegration.
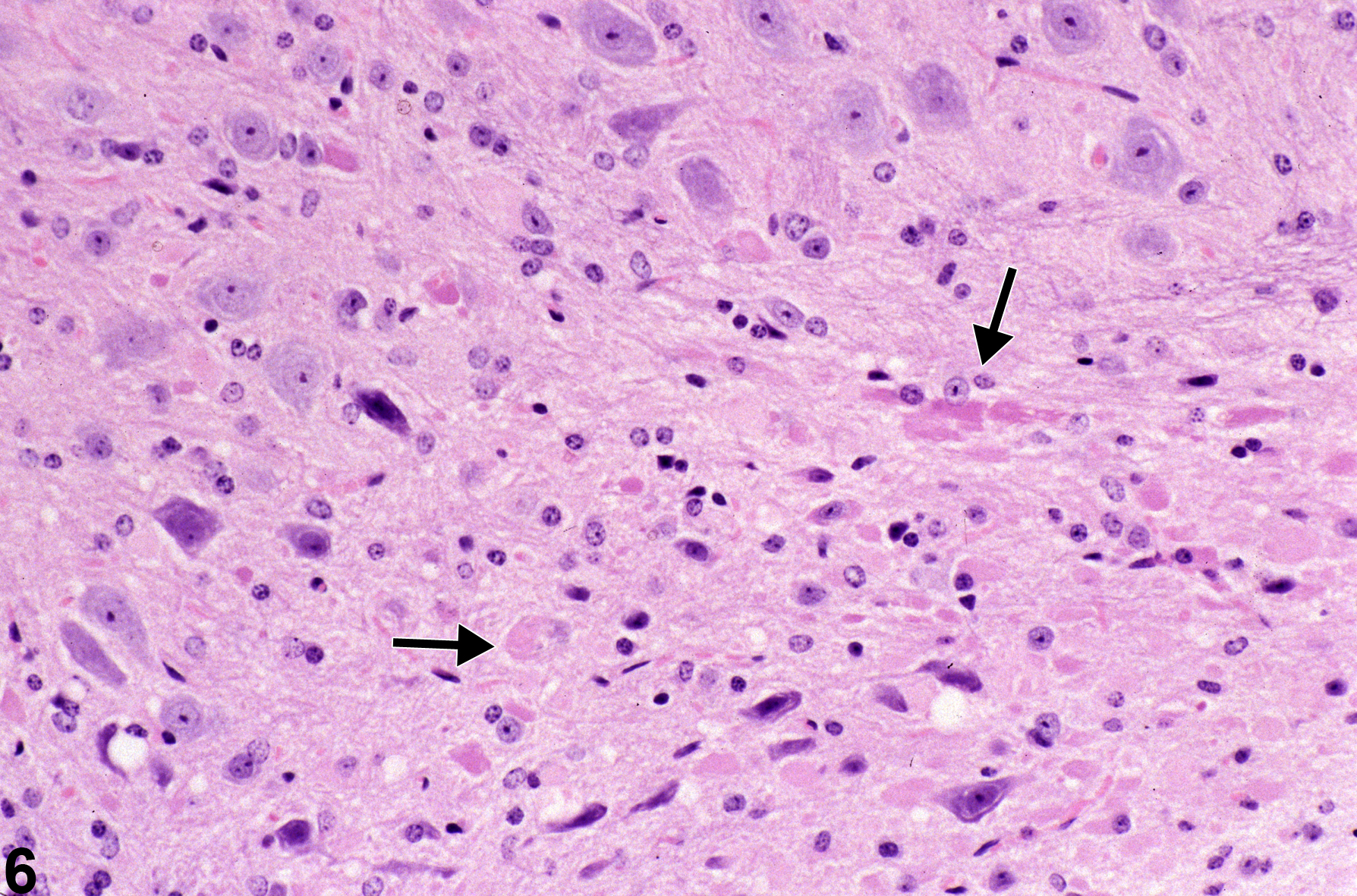
Figure 6 shows eosinophilic spheroids and oval to elongated structures (arrows) that represent injured axons in the brain sectioned in various planes, thus affecting their appearance. The presence of swollen axons in the brain is not generally accompanied by dramatic distension of the myelin sheath such as those seen in spinal cord and peripheral nerves. Note also that there is a mild increase in a mixed population of glial cells in the region, part of the associated inflammatory response.
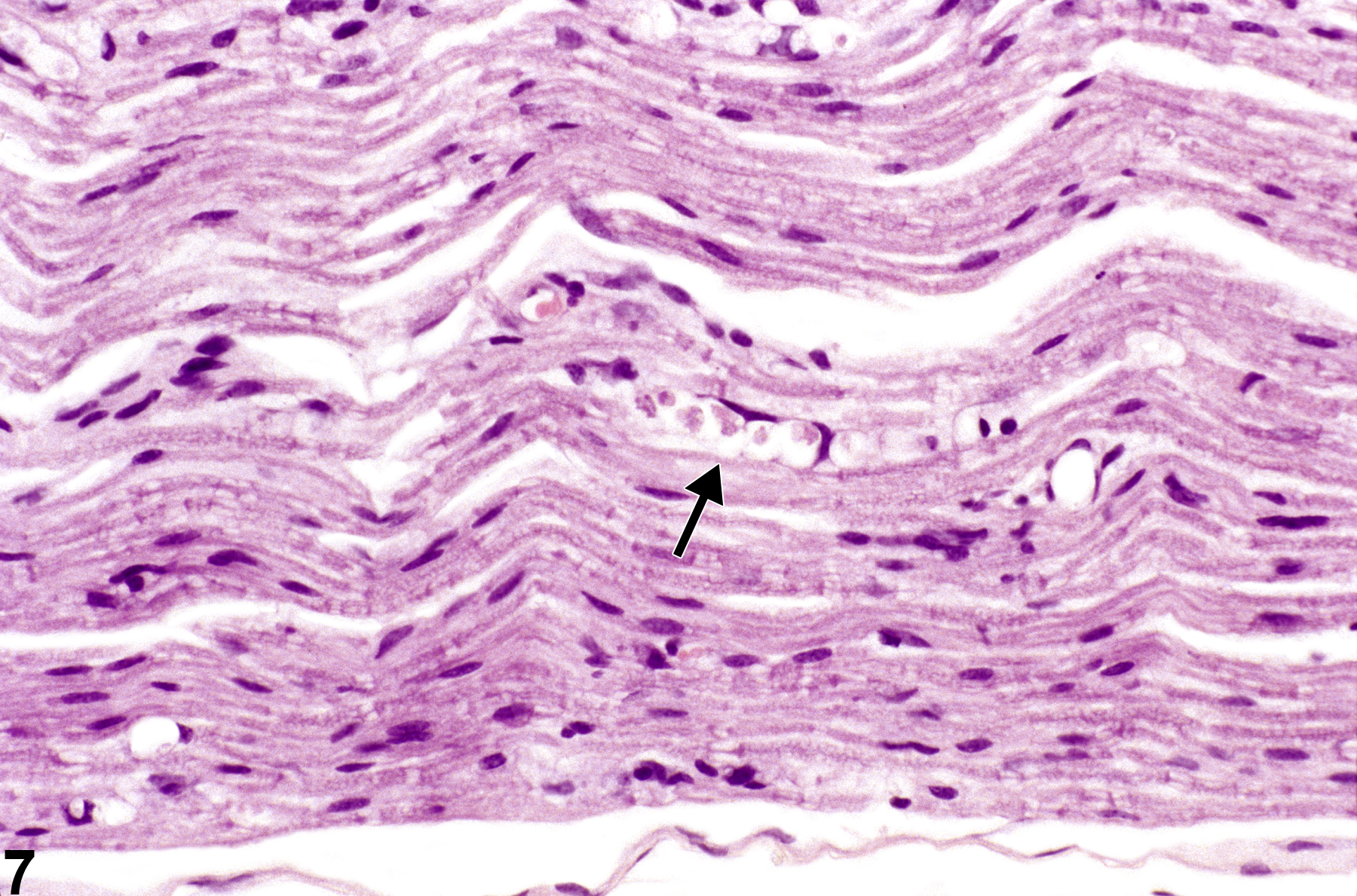
Figure 7 shows axon degeneration with a typical “digestion chamber,” containing axonal fragments (arrow). In this hematoxylin and eosin–stained image of a longitudinal section of the sciatic nerve, the typical appearance of axonal degeneration referred to as Wallerian degeneration is evident. The term “Wallerian degeneration” is best reserved to describe axonopathy in peripheral nerve; however, similar changes can be seen in spinal cord and brain. Augustus Waller, in 1850, introduced the criteria for axonopathy in peripheral nerve from his sequential studies of experimental nerve crush injury. The distal nerve, particularly after crush, is characterized by progressive axonal swelling and, approximately 36 hours after crush, by fragmentation of the fiber, Schwann cell nuclear hypertrophy, infiltration of macrophages, and the formation of so-called digestion chambers. In Figure 7 the digestion chamber contains several fragments of degenerate axon surrounded by a clear space, containing lipid breakdown products and macrophages. The enlarged nuclei of Schwann cells can be seen in adjacent nerve tissue.
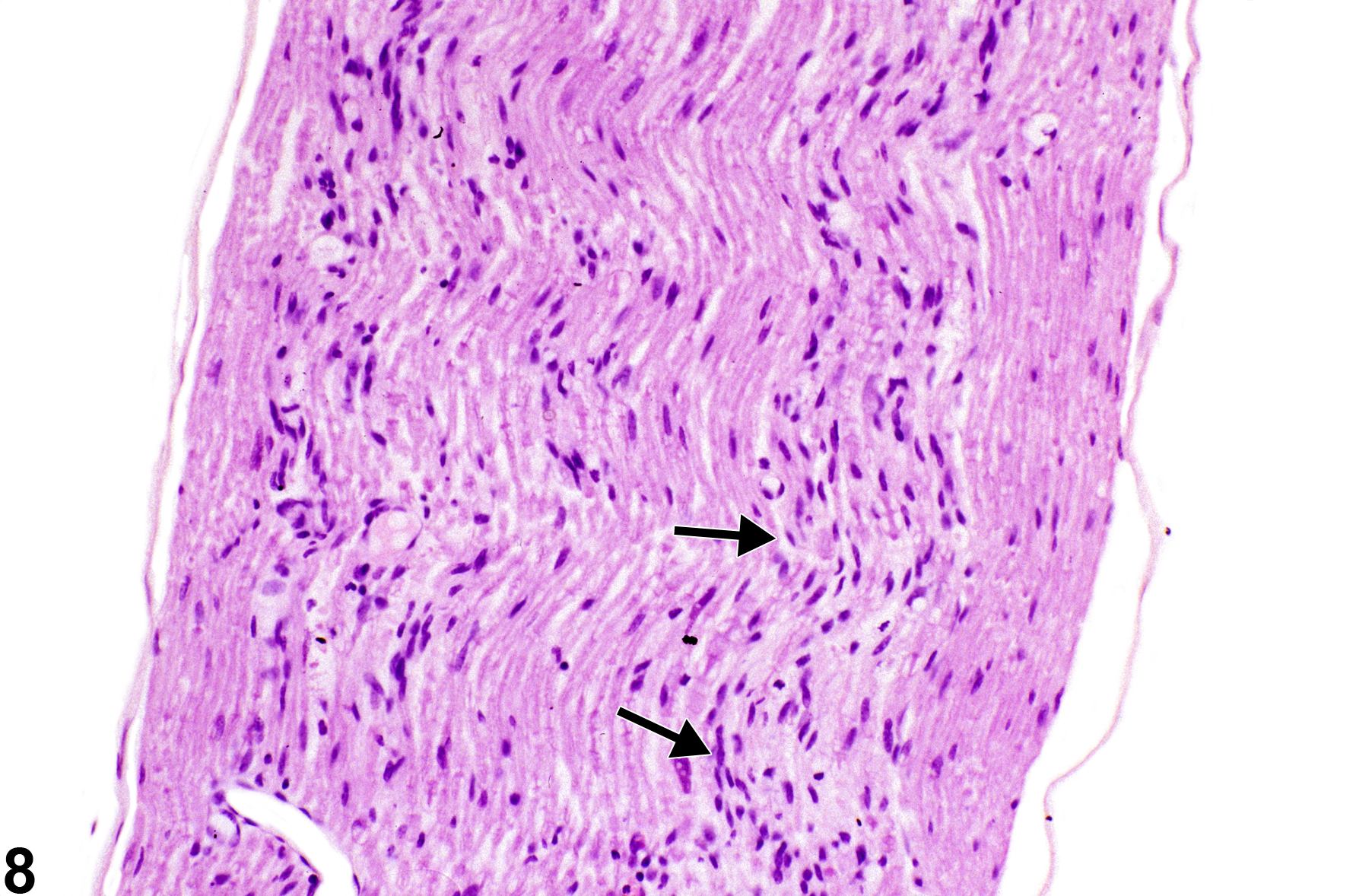
Figure 8 shows sciatic nerve axon degeneration with reactive hyperplasia and hypertrophic nuclei of Schwann cells (arrows). These linear band-like proliferative Schwann cells are known as bands of Büngner. They are formed as a response to axonal injury when Schwann cells lose their axonal complement. The remaining basement membrane forms endoneurial tubes, important in the guidance of regenerating proximal axonal sprouts. Schwann cells and blood-borne macrophages are important in phagocytosis of fragmented axons to clear endoneurial tubes for axonal regeneration. The bands of Büngner should not be interpreted as inflammatory fibrosis but, rather, as a part of the reinnervation process subsequent to significant axon degeneration in peripheral axons.
Baker HJ, Lindsey JR, Weisbroth SH, eds. 1979. The Laboratory Rat, Vol 1: Biology and Diseases. Academic Press, New York.
Beirowski B, Adalbert R, Wagner D, Grumme DS, Addicks K, Ribchester RR, Coleman MP. 2006. The progressive nature of Wallerian degeneration in wild-type and slow Wallerian degeneration (WldS) nerves. BMC Neurosci 6:6.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/15686598Lubinska L. 1977. Early course of Wallerian degeneration in myelinated fibres of the rat phrenic nerve. Brain Res 130:47-63.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/884520Stoll G, Griffin JW, Li CY, Trapp BD. 1989. Wallerian degeneration in the peripheral nervous system: Participation of both Schwann cells and macrophages in myelin degradation. J Neurocytol 18:671-683.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/2614485Tabas I. 1997. Free cholesterol-induced cytotoxicity. A possible contributing factor to macrophage foam cell necrosis in advanced atherosclerotic lesions. Trends Cardiovasc Med 7:256-263.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/21235894Tenkova TI, Goldberg MP. 2007. A modified silver technique (de Olmos stain) for assessment of neuronal and axonal degeneration. Methods Mol Biol 399:31-39.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/18309923Waller A. 1850. Experiments on the section of glossopharyngeal and hypoglossal nerves of the frog and observations of the alternatives produced thereby in the structure of their primitive fibers. Philos Trans R Soc Lond Biol 140:423-429.
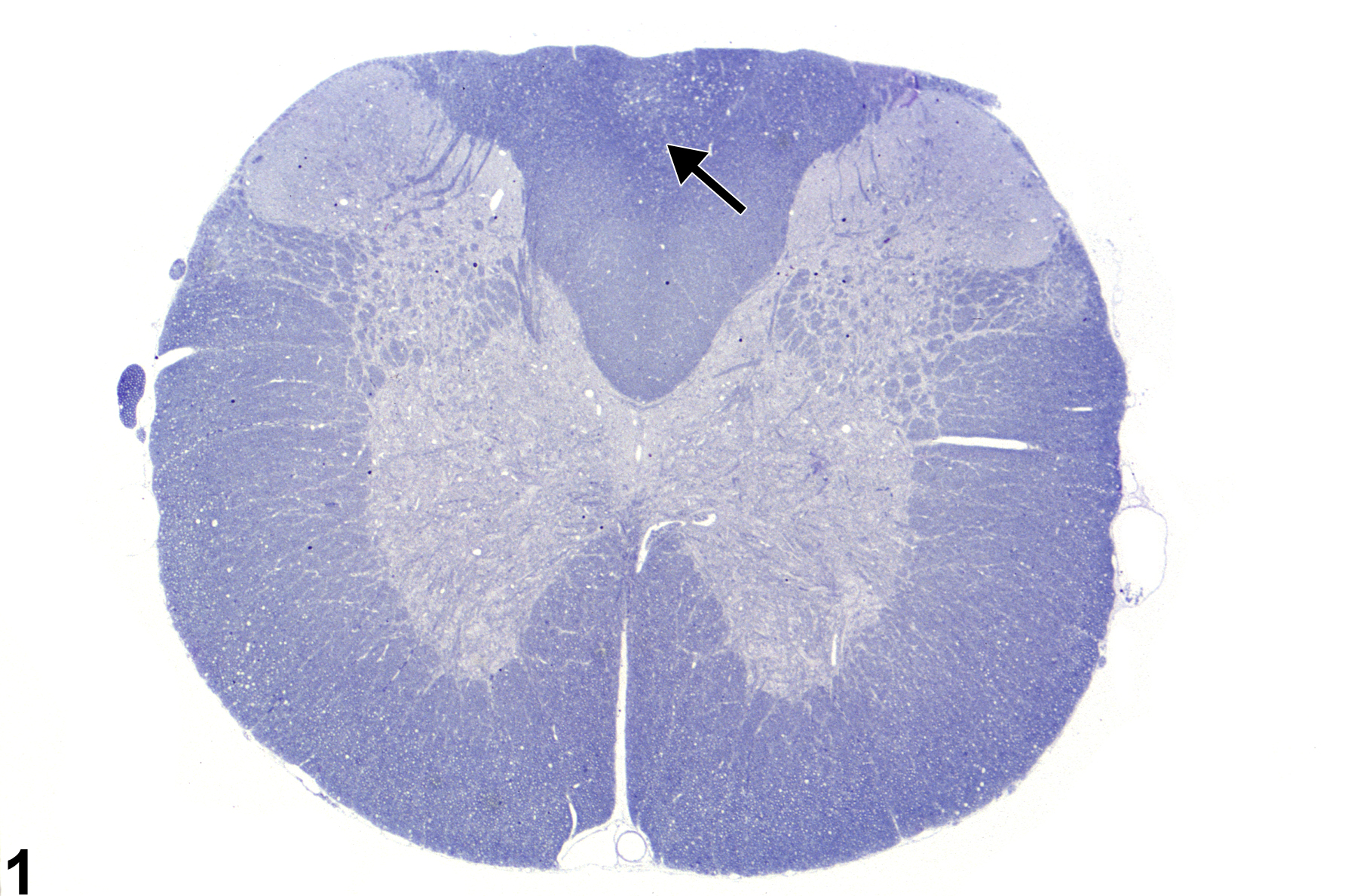
Spinal cord axonopathy in a male F344/N rat from a subchronic study. The arrow in this toluidine blue-stained section indicates the subtle pallor of the dorsal funiculus in which axonopathy is present.