Nervous System
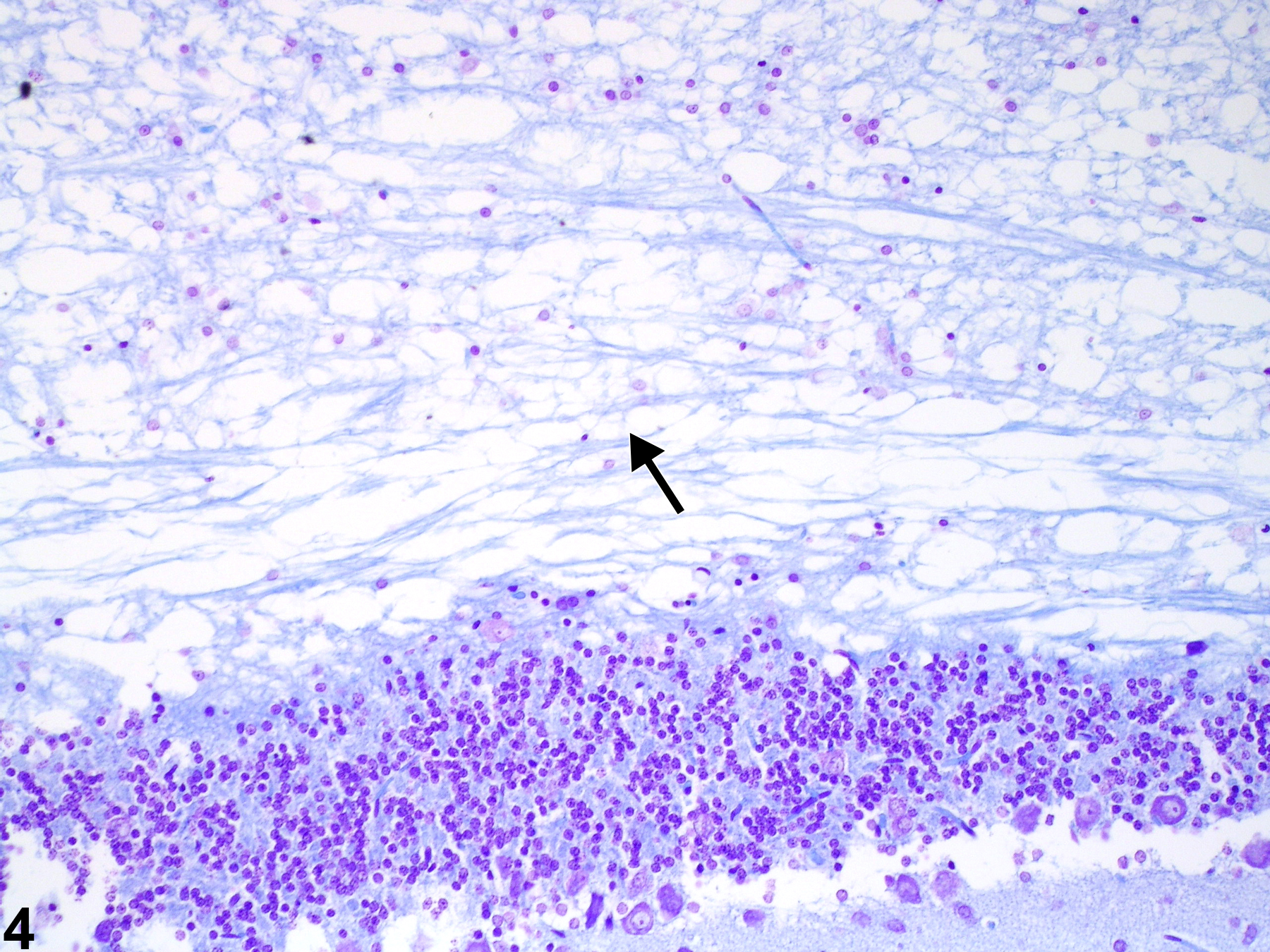
Brain - Intramyelinic Edema
Narrative
Kennedy GL Jr, Dressler IA, Richter WR, Keplinger ML, Calandra JC. 1976. Effects of hexachlorophene in the rat and their reversibility. Toxicol Appl Pharmacol 35:137-145.
Abstract: http://www.sciencedirect.com/science/article/pii/0041008X76901198Purves DC, Garrod IJ, Dayan AD. 1991. A comparison of spongiosis induced in the brain by hexachlorophene, cuprizone and triethyl tin in the Sprague-Dawley rat. Hum Exp Toxicol 10:439-444.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/1687857Tripier MF, Berard M, Toga M, Martin-Bouyer G, Le Breton R, Garat J. 1981. Experimental hexachlorophene encephalopathy in mice and baboons: Light and electron microscopic study. Acta Neuropathol Suppl 7:40-43.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/6939279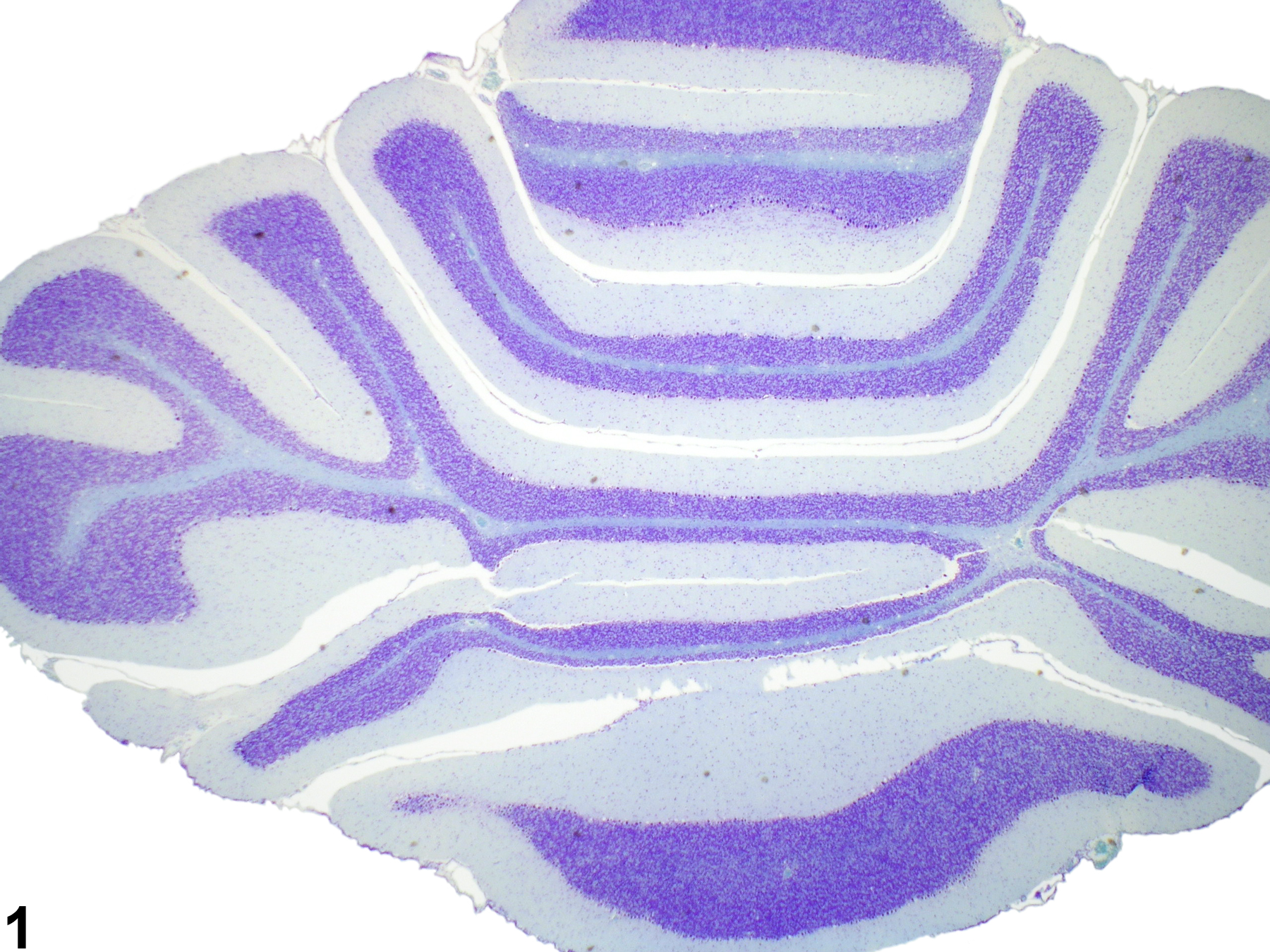
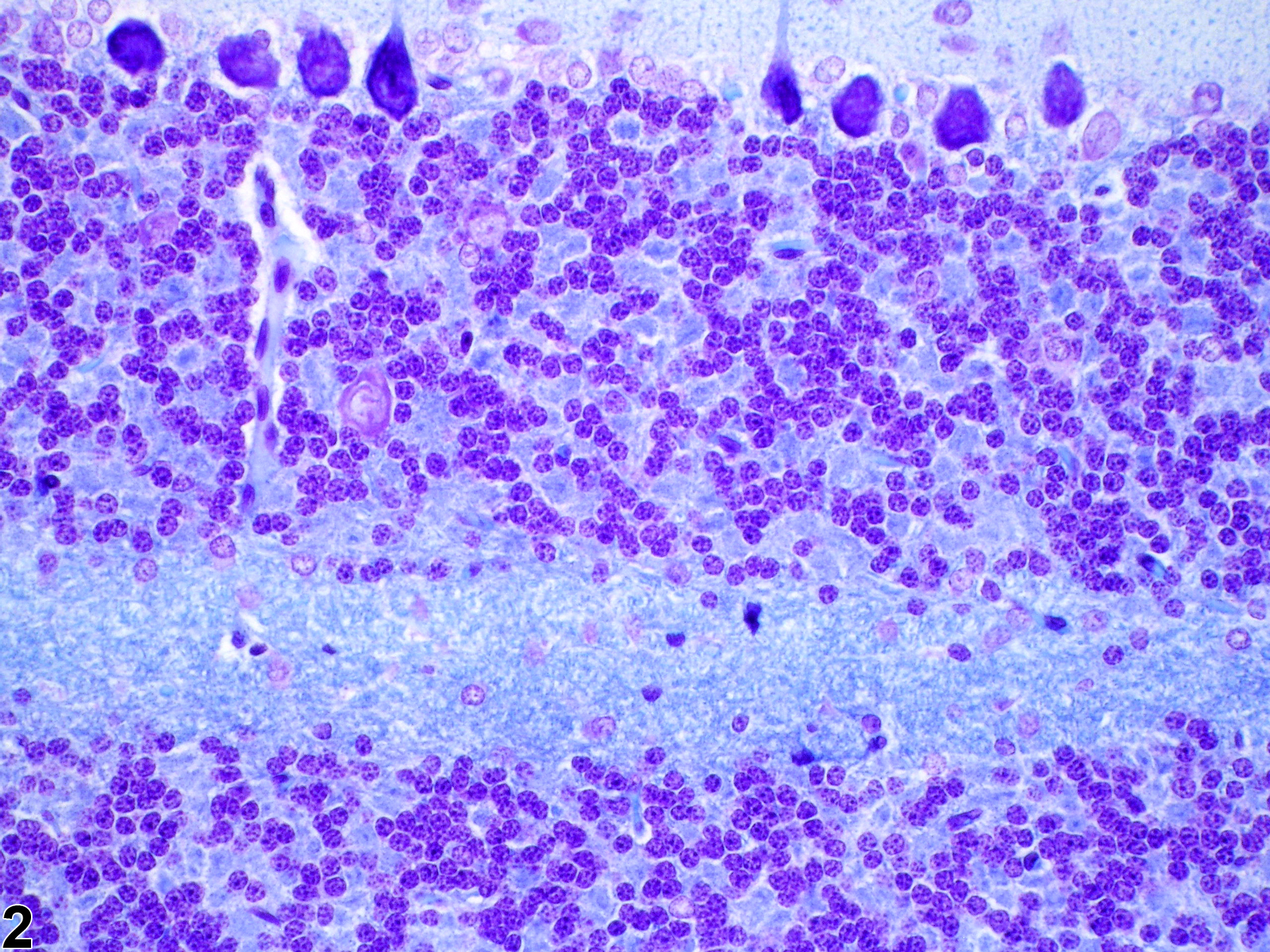
Normal appearance of myelin in cerebellar white matter stained with Luxol fast blue and cresyl violet in a control rat.