Nervous System
Spinal Cord - Demyelination
Narrative
Demyelination may be primary of secondary. In primary demyelination, the axon is usually intact and may be preserved, especially if the myelin sheath is exclusively assaulted by the immune system with resulting phagocytosis (primary demyelination). In contrast, secondary demyelination is a common sequel to axonal injury and loss, such that axonopathy and demyelination often coexist as a result of axonal injury from classic axonopathic compounds such as organophosphate agents and acrylamide. In addition to demyelination resulting from multiple sclerosis in humans and the experimentally induced autoimmune encephalopathy of laboratory animals, rarely human immune-mediated demyelinating neuropathies occur during initial or maintenance treatment with immunomodulatory, immunosuppressive, or antineoplastic agents.
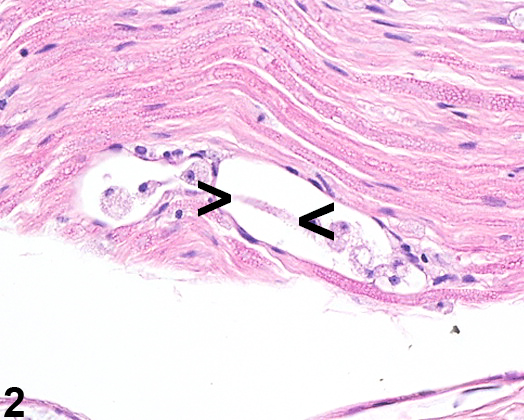
Krinke and Zurbriggen (1997) have described a demyelination in the thoracic spinal cord of aging mice that was attributed to a strain of Theiler’s murine encephalomyelitis virus (TMEMV) using immunohistochemical identification of the agent in their material. Prominent bilateral lesions of the spinal cord from their studies are shown in Figure 1 (arrows). However, TMEMV is uncommon in barrier-raised laboratory mice, particularly in North America (PB Little, personal communication with C Clifford, Charles River Laboratories).
Carozzi VA, Canta A, Oggioni N, Sala B, Chiorazzi A, Meregalli C, Bossi M, Marmiroli P, Cavaletti G. 2010. Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp Neurol 226:301-309.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/20832406Krinke GJ, Zurbriggen A. 1997. Spontaneous demyelinating myelopathy in aging laboratory mice. Exp Toxicol Pathol 49:501-503.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/9495654Kushlaf HA. 2011. Emerging toxic neuropathies and myopathies. Neurol Clin 29:679-687.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/21803218Schaumburg HH, Spencer PS. 1979. Toxic neuropathies. Neurology 29:429-431.
Stübgen JP. 2011. Drug-induced dysimmune demyelinating neuropathies. J Neurol Sci 307:1-8.
Abstract: http://www.ncbi.nlm.nih.gov/pubmed/21621795 Mouse spinal cord depicting bilateral demyelination of the lateral columns (arrows) in a mouse (age, strain, and sex unknown).
Mouse spinal cord depicting bilateral demyelination of the lateral columns (arrows) in a mouse (age, strain, and sex unknown).