Respiratory System
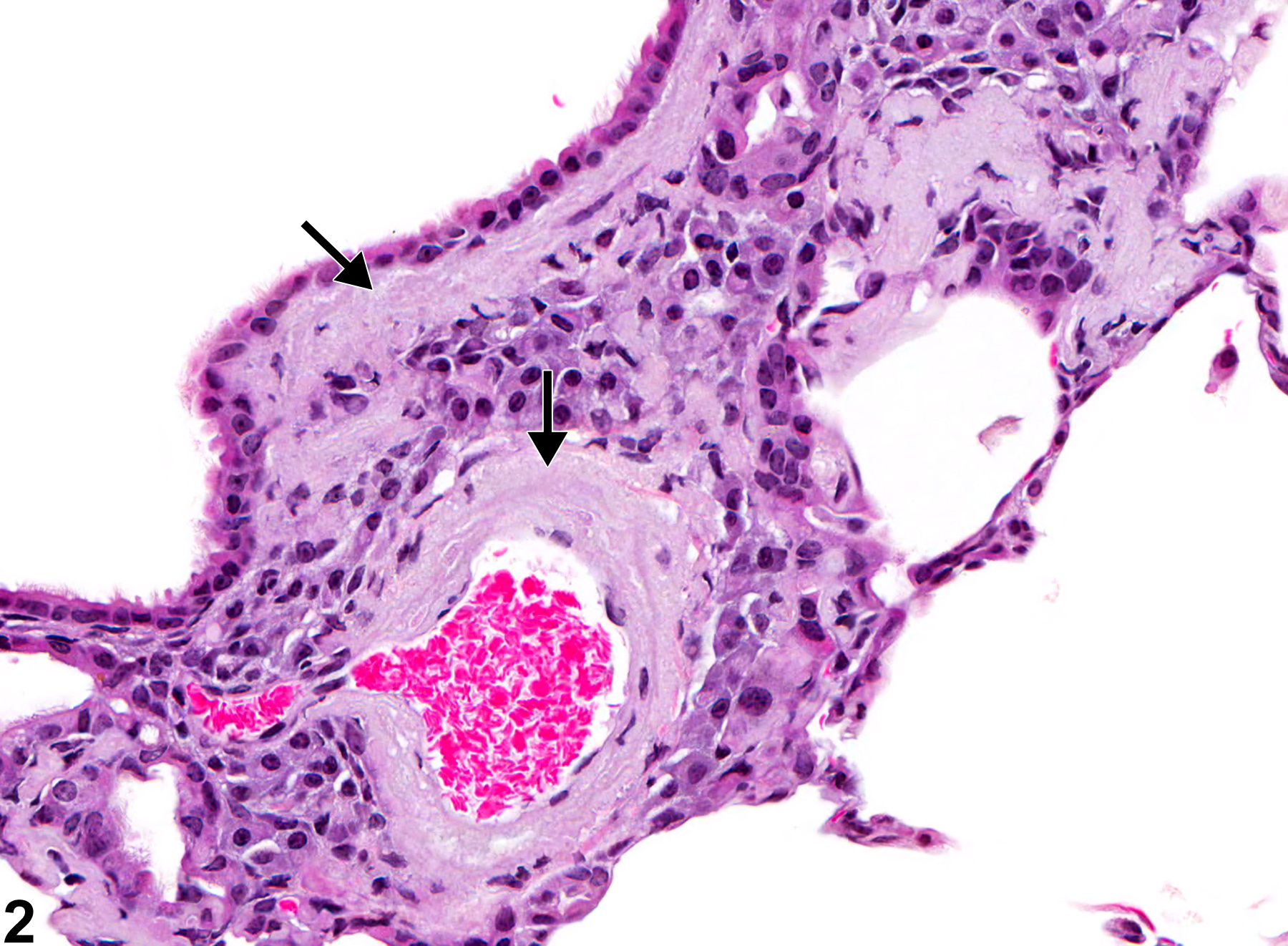
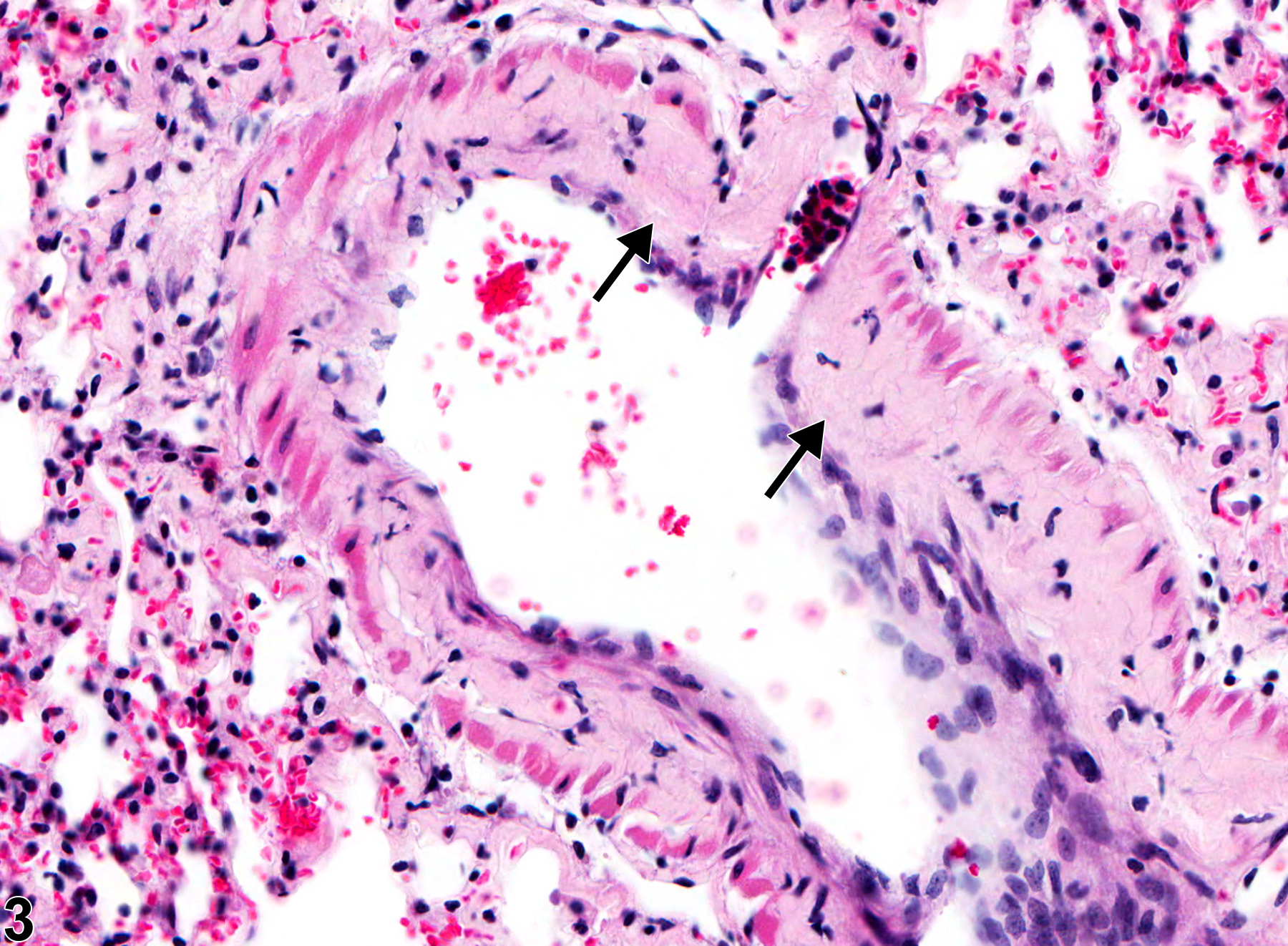
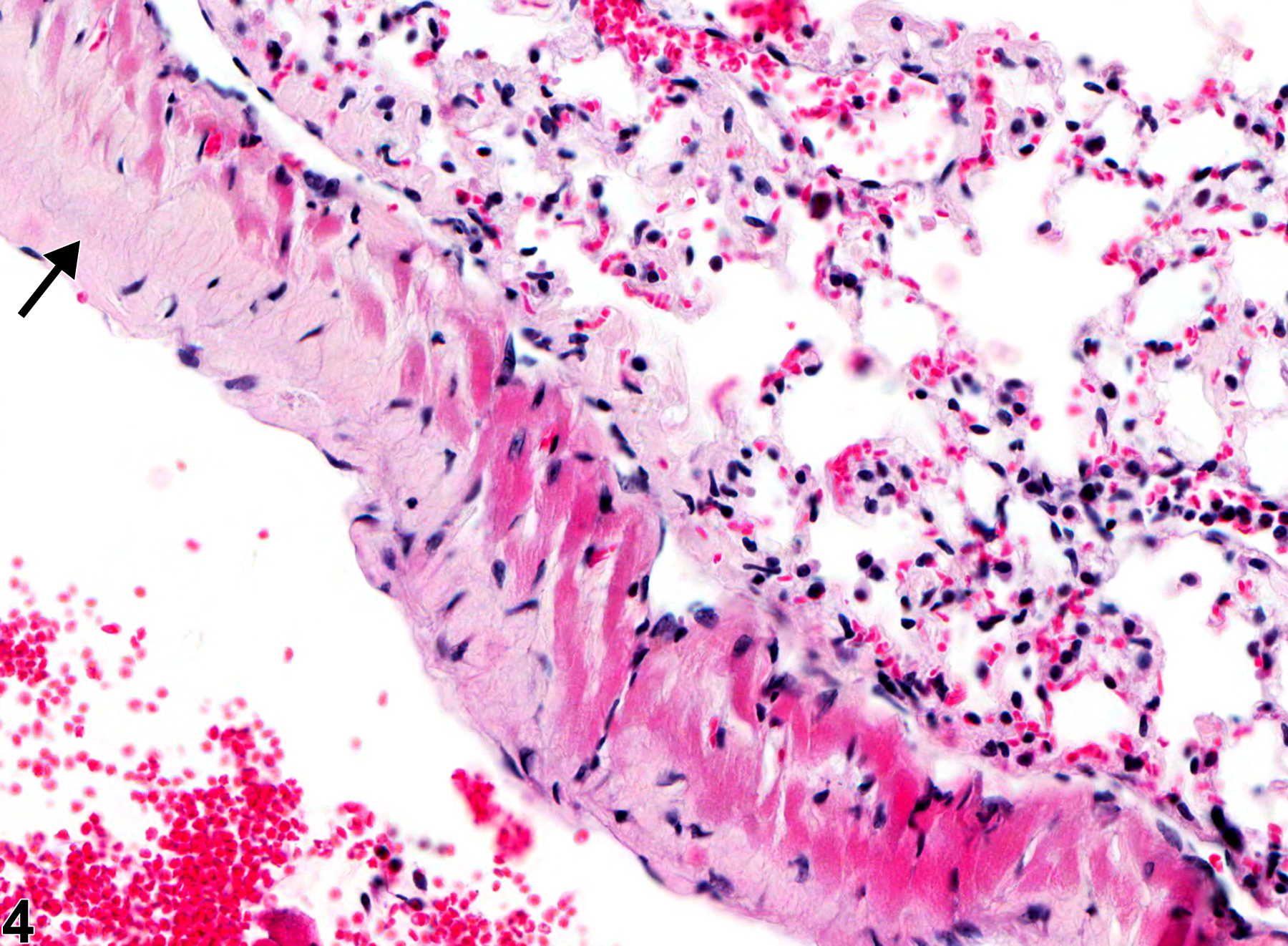
Lung - Amyloid
Narrative
Buxbaum JN. 2004. The systemic amyloidoses. Curr Opin Rheumatol 16:67-75.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/14673392Frith CH, Manik C. 1991. Incidence, distribution, and morphology of amyloidosis in Charles Rivers CD-1 mice. Toxicol Pathol 19:123-127.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/1771365Higuchi K, Matsumura A, Honma A, Takeshita S, Hashimoto K, Hosokawa M, Yasuhira K, Takeda T. 1983. Systemic senile amyloid in senescence-accelerated mice. A unique fibril protein demonstrated in tissues from various organs by the unlabeled immunoperoxidase method. Lab Invest 48:231-240.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/6337302Homburger F, Russfield AB, Weisburger JH, Lim S, Chak SP, Weisburger EK. 1975. Aging changes in CD-1 HaM/ICR mice reared under standard laboratory conditions. J Natl Cancer Inst 55:37-45.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/169366Takeshita S, Hosokawa M, Irino M, Higuchi K, Shimizu K, Yasuhira K, Takeda T. 1982. Spontaneous age-associated amyloidosis in senescence-accelerated mouse (SAM). Mech Ageing Dev 20:13-23.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/7176700Westermark P, Benson MD, Buxbaum JN, Cohen AS, Frangione B, Ikeda S, Masters CL, Merlini G, Saraiva MJ, Sipe JD. 2007. A primer of amyloid nomenclature. Amyloid 14:179-183.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/17701465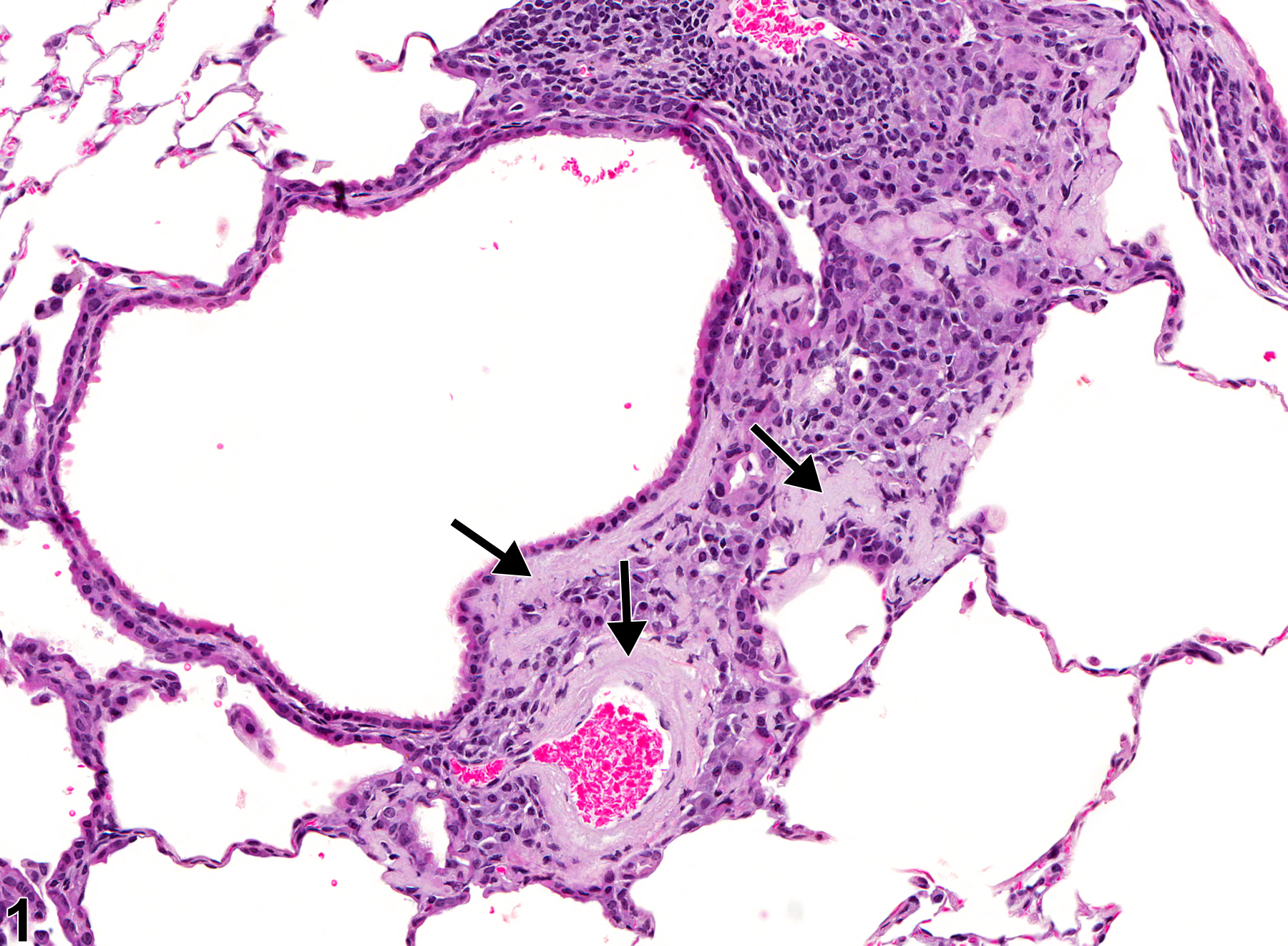
Lung - Amyloid in a female B6C3F1/N mouse from a chronic study. There is perivascular and peribronchiolar amyloid deposition (arrows).