Respiratory System
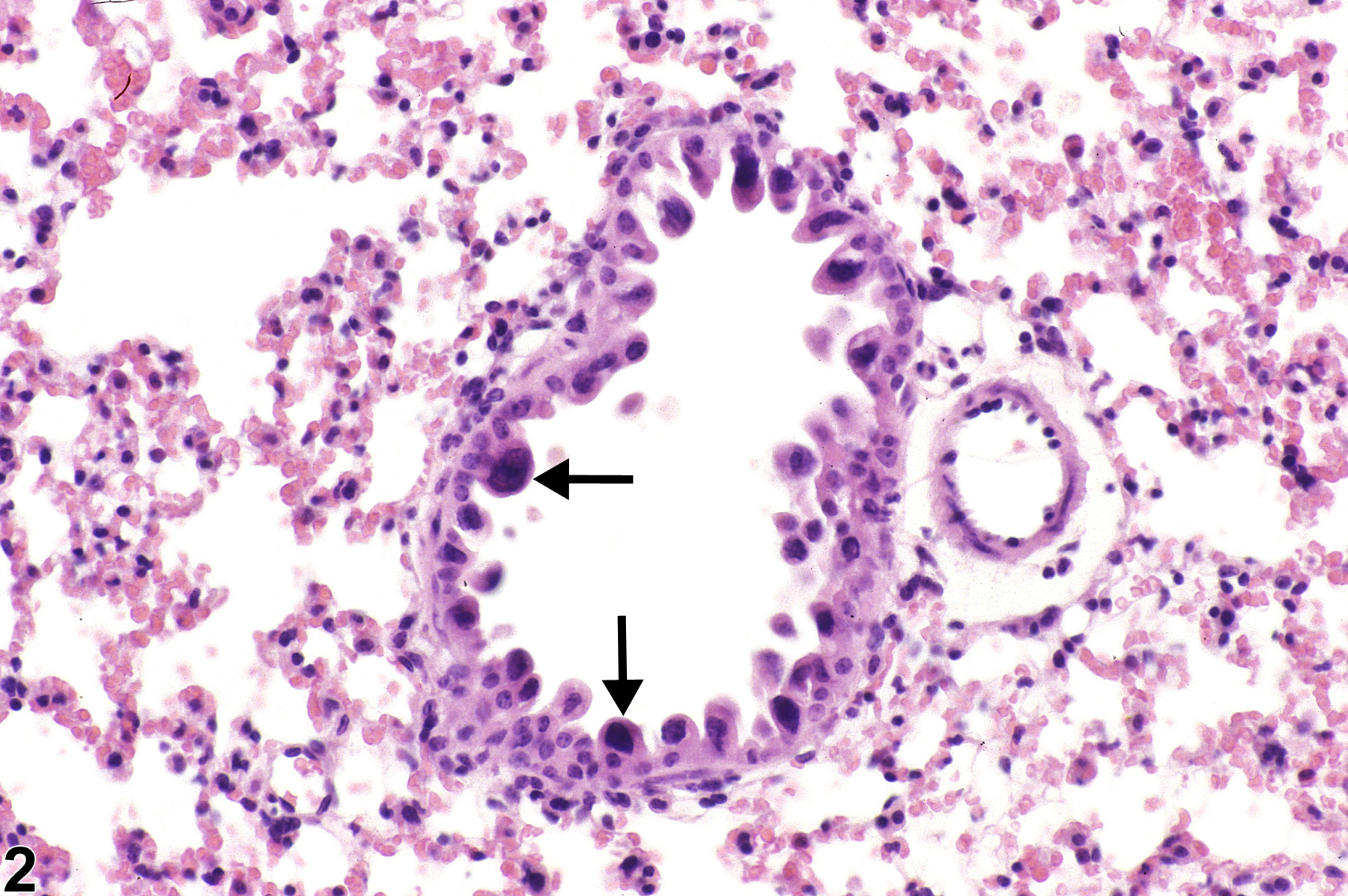
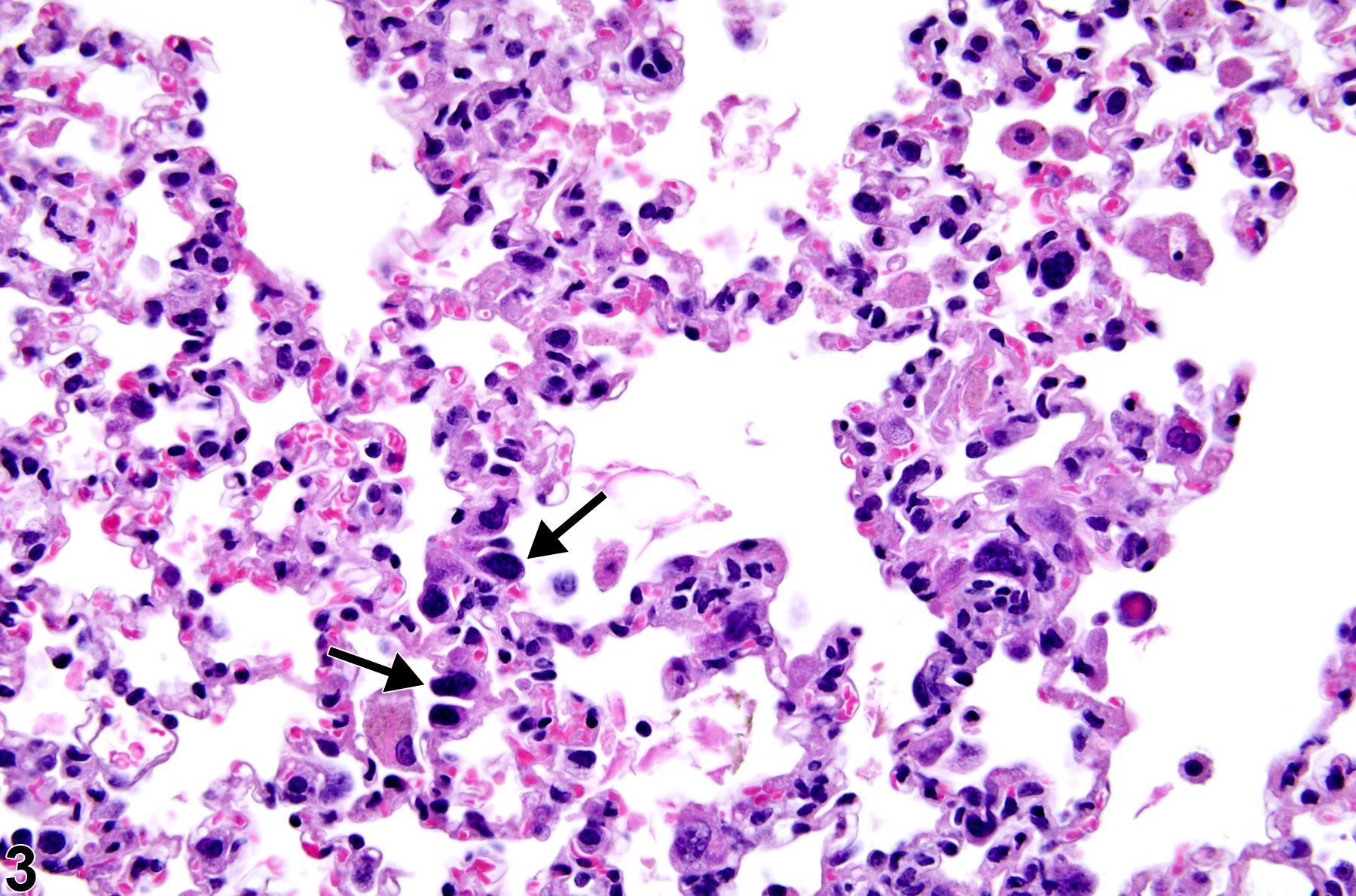
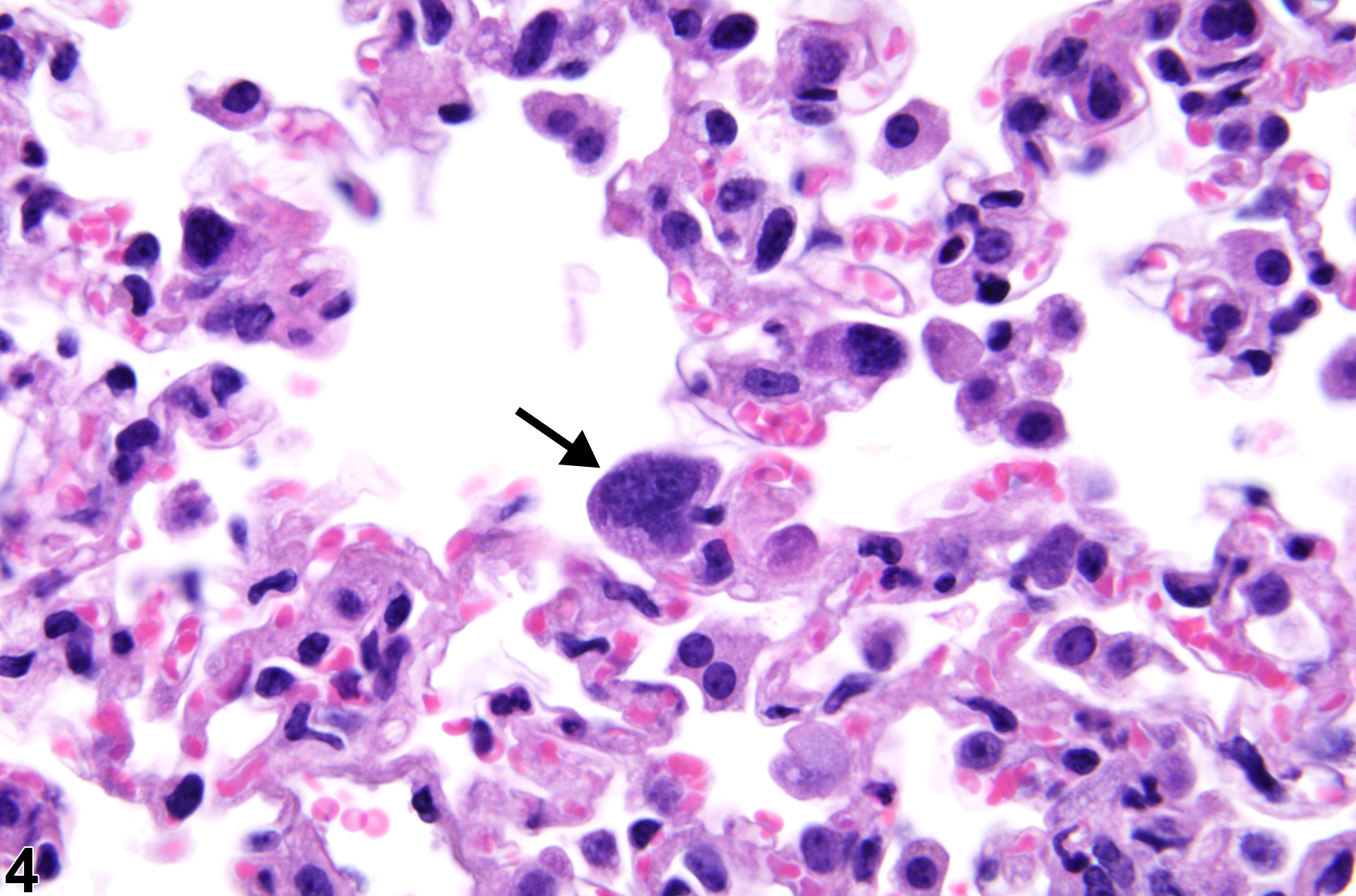
Lung - Atypia, Cellular
Narrative
Boorman GA, Eustis SL. 1990. Lung. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 339-367.
Knight JA, Rezuke WN, Gillies CG, Hopfer SM, Sunderman FW. 1988. Pulmonary histopathology of rats following parenteral injections of nickel chloride. Toxicol Pathol 16:350-359.
Full Text: http://tpx.sagepub.com/content/16/3/350.full.pdfSolano-Lopez C, Zeidler-Erdely PC, Hubbs AF, Reynolds SH, Roberts JR, Taylor MD, Young SH, Castranova V, Antonini JM. 2006. Welding fume exposure and associated inflammatory and hyperplastic changes in the lungs of tumor susceptible a/j mice. Toxicol Pathol 34:364-372.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16844664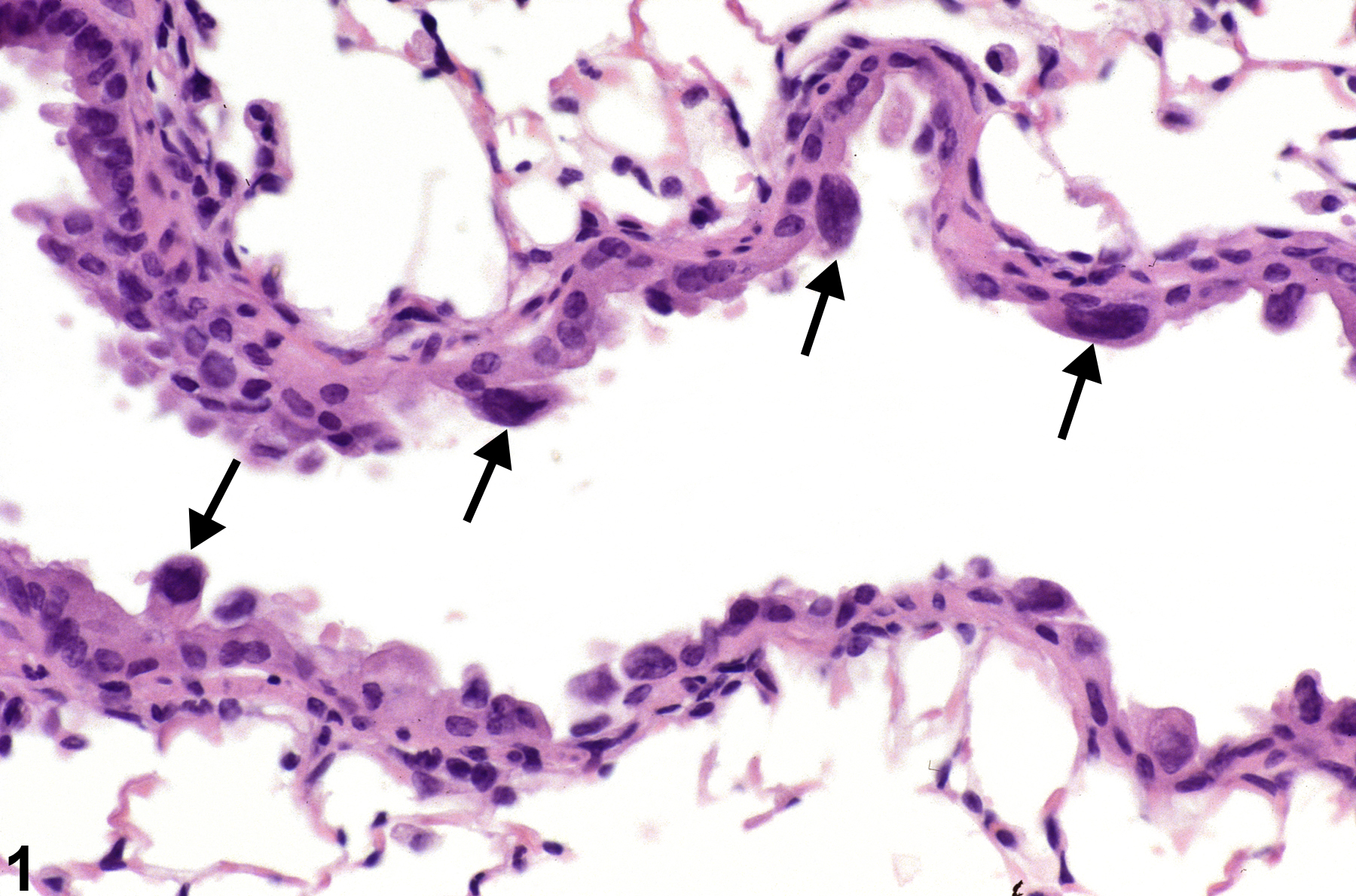
Lung, Bronchiole - Hyperplasia, Atypical in a female B6C3F1/N mouse from a subchronic study. The atypical cells have enlarged, hyperchromatic nuclei (arrows) and an increased nuclear to cytoplasmic ratio.