Respiratory System
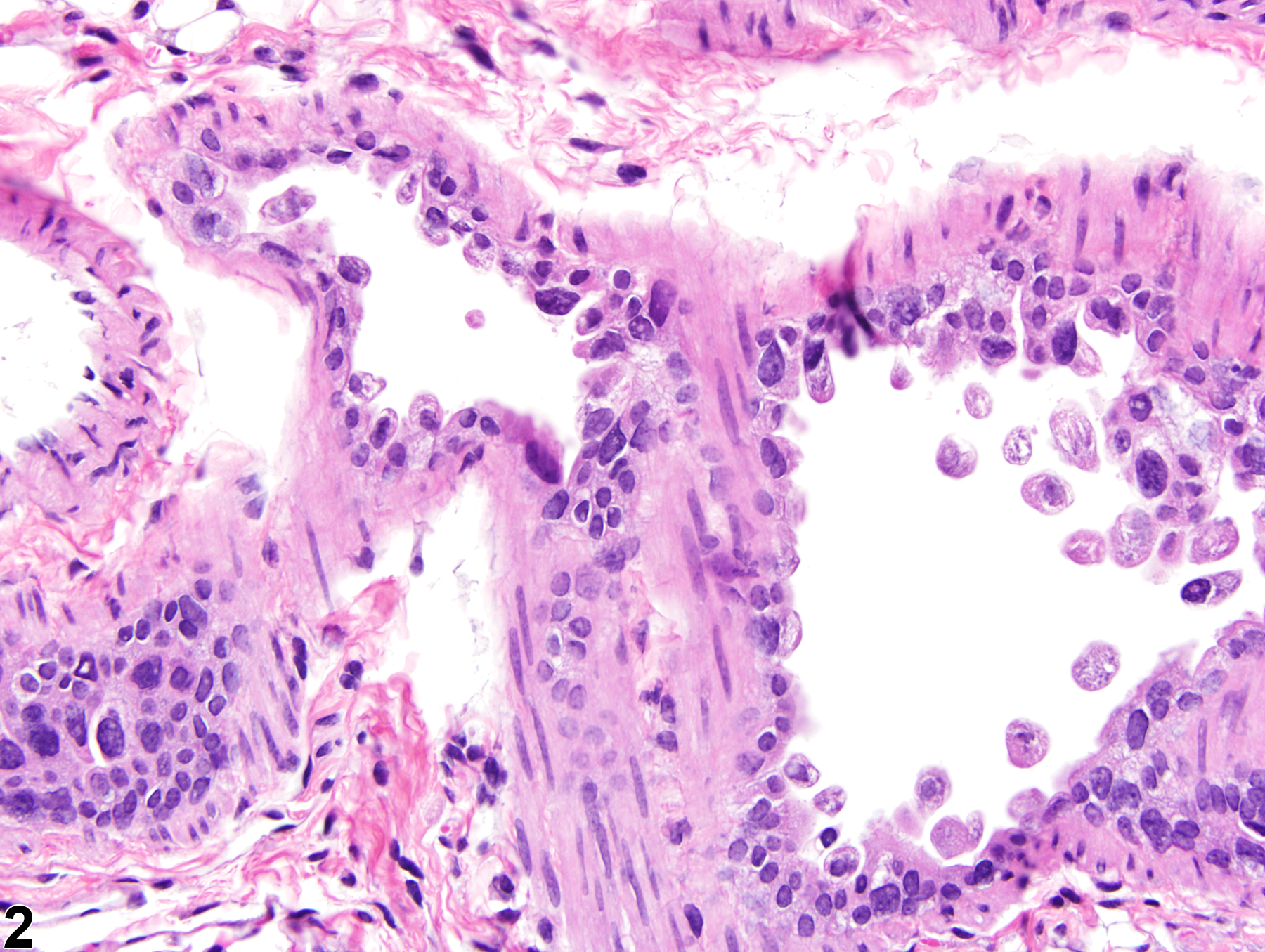
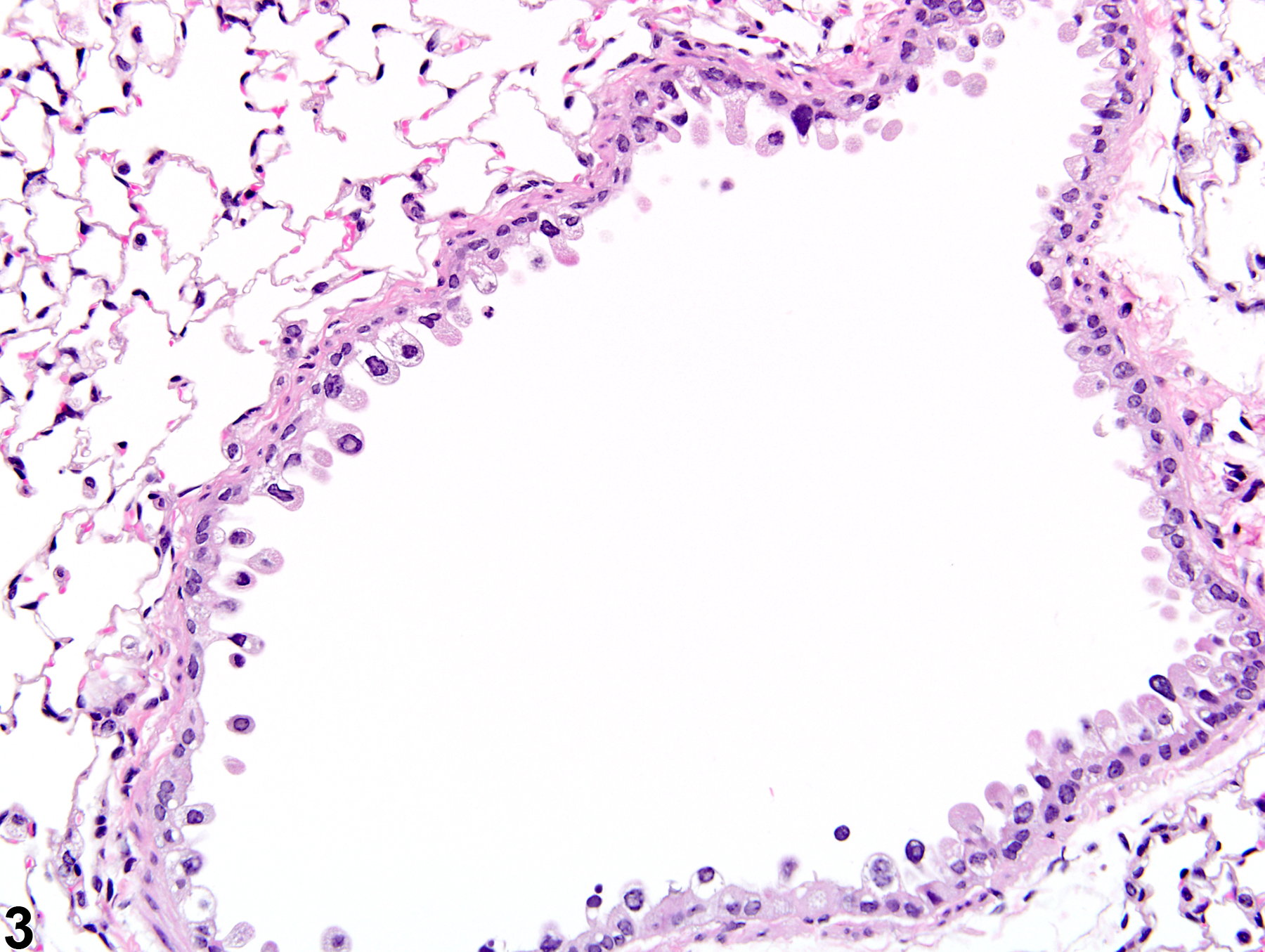
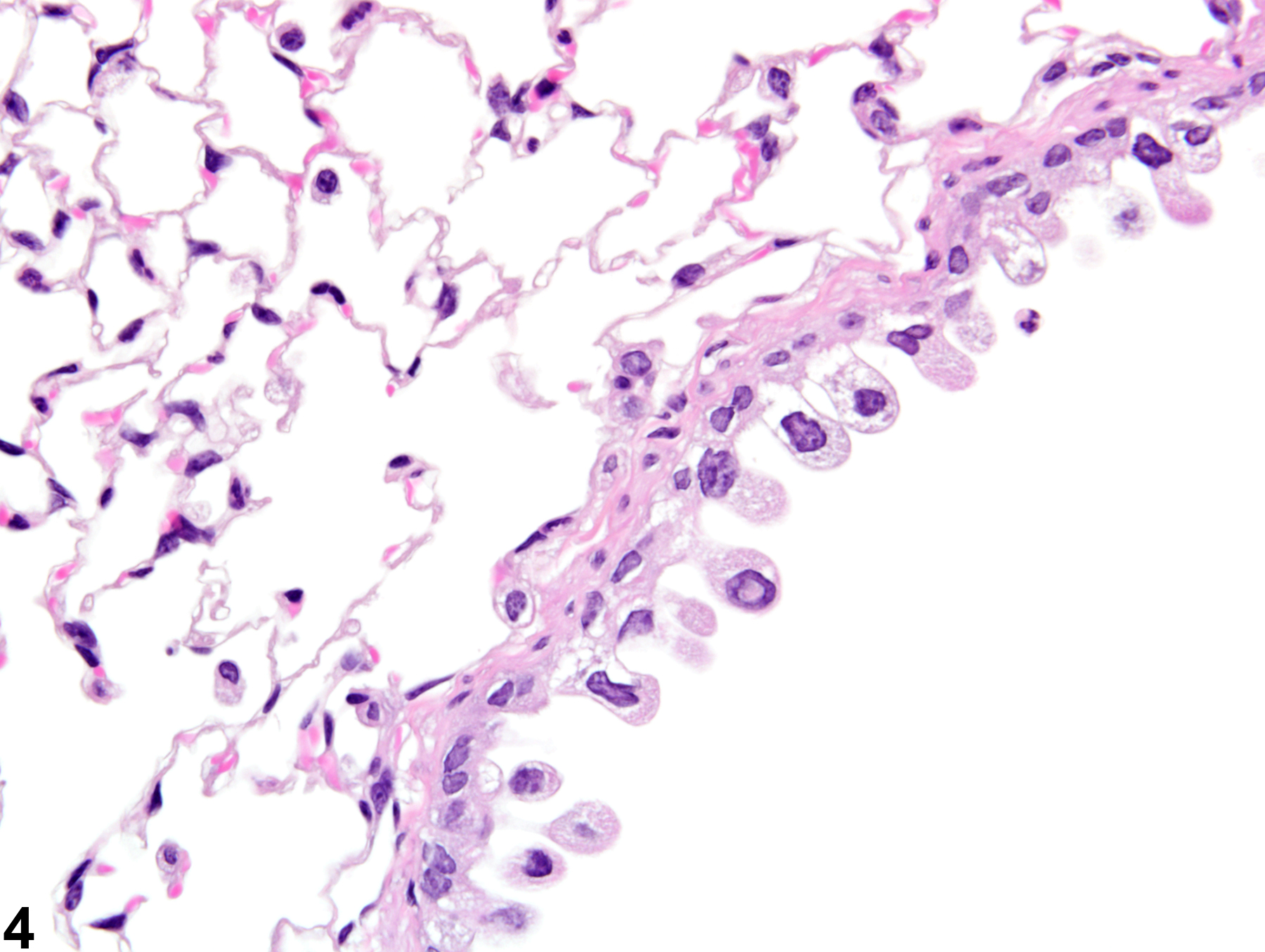
Lung, Epithelium - Degeneration
Narrative
The anatomic location of degenerative lesions may vary due to the physicochemical properties of the test agent or the susceptibility of a particular cell type to the test agent. The epithelium of the terminal bronchioles and alveolar ducts (i.e., the centriacinar region) and alveoli are particularly susceptible to injury due to the large surface area and fragility of the alveolar type I cells, the metabolic activity of P450 enzymes in Clara cells, and the generally thinner mucous layer.
Boorman GA, Eustis SL. 1990. Lung. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 339-367.
Haschek-Hock WM, Witschi HP. 1990. Respiratory system. In: Handbook of Toxicologic Pathology (Haschek-Hock WM, CG Rousseaux, eds). Academic Press, San Diego, CA, 761-827.
Kumar V, Abbas AK, Fausto N. 2005. Cellular adaptations, cell injury, and cell death. In: Robbins and Cotran Pathologic Basis of Disease, 7th ed. Elsevier Saunders, Philadelphia, 3-48.
Lopez A. 2007. Respiratory system. In: Pathologic Basis of Veterinary Disease, 4th ed (McGavin MD, Zachary JF, eds). Mosby, St Louis, 463-558.
Plopper CG, Dungworth DL. 197. Structure, function, cell injury and cell renewal of bronchiolar and alveolar epithelium. In: Lung Carcinomas (McDowell EM, ed). Churchill Livingstone, Edinburgh, 94-128.
Renne, R, Brix A, Harkema J, Herbert R, Kittle B, Lewis D, March T, Nagano K, Pino M, Rittinghausen S, Rosenbruch M, Tellier P, Wohrmann T. 2009. Proliferative and nonproliferative lesions of the rat and mouse respiratory tract. Toxicol Pathol 37(suppl):5S-73S.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/20032296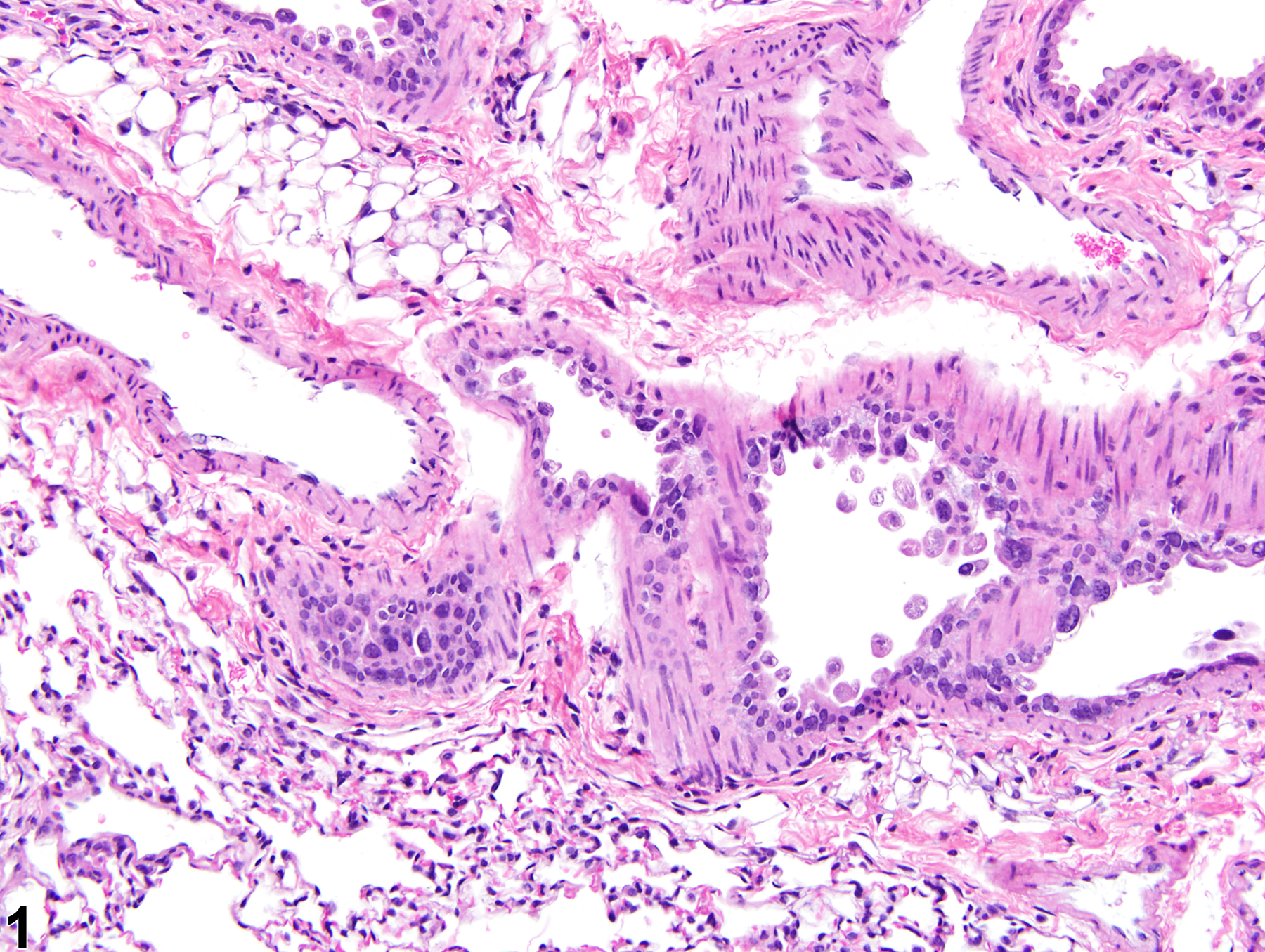
Lung, Epithelium, Bronchiole - Degeneration in a male B6C3F1/N mouse from a subchronic study. The epithelial cells in this bronchiole are sloughing into the lumen, but there is little inflammation.