Special Senses System
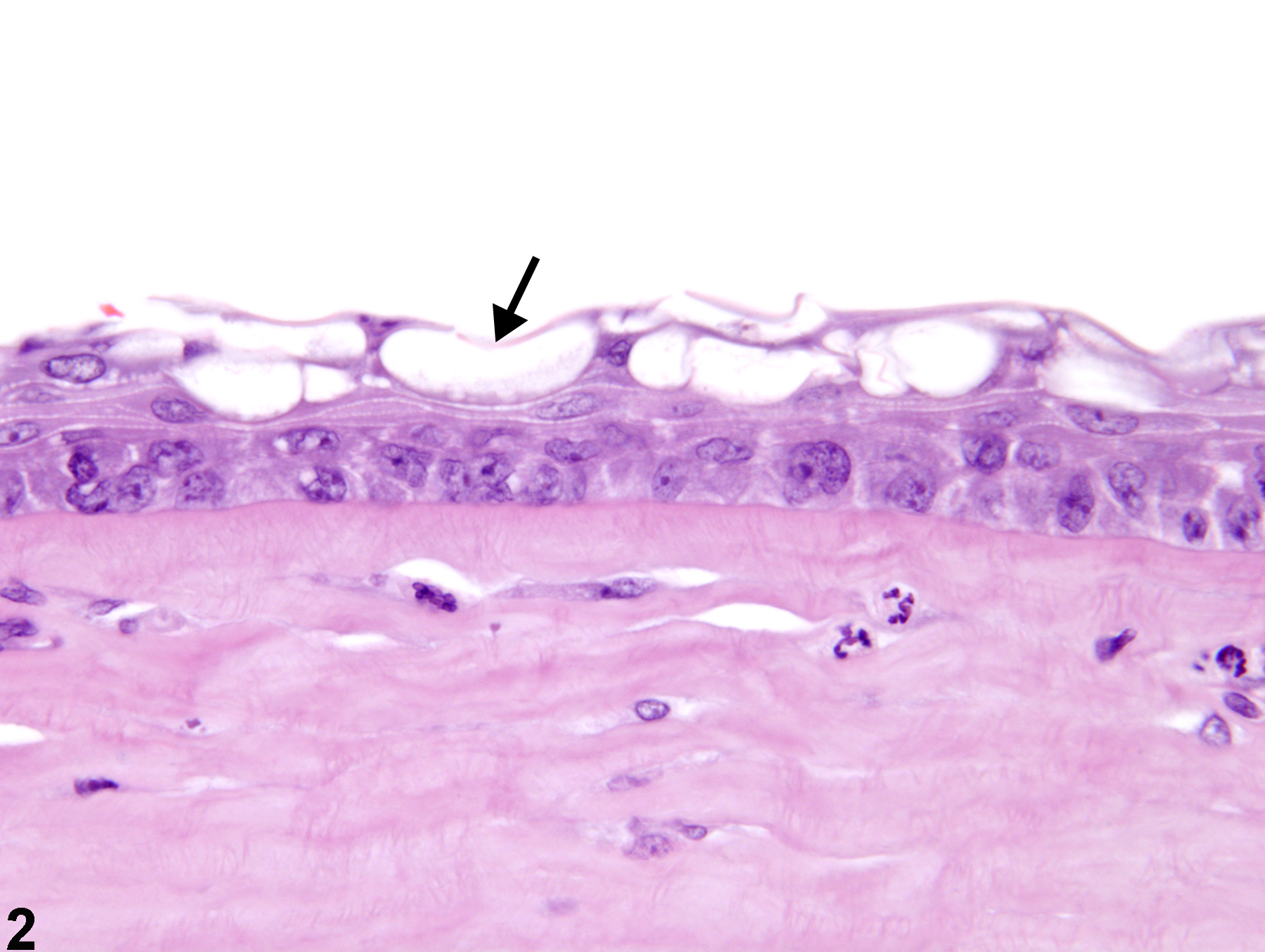
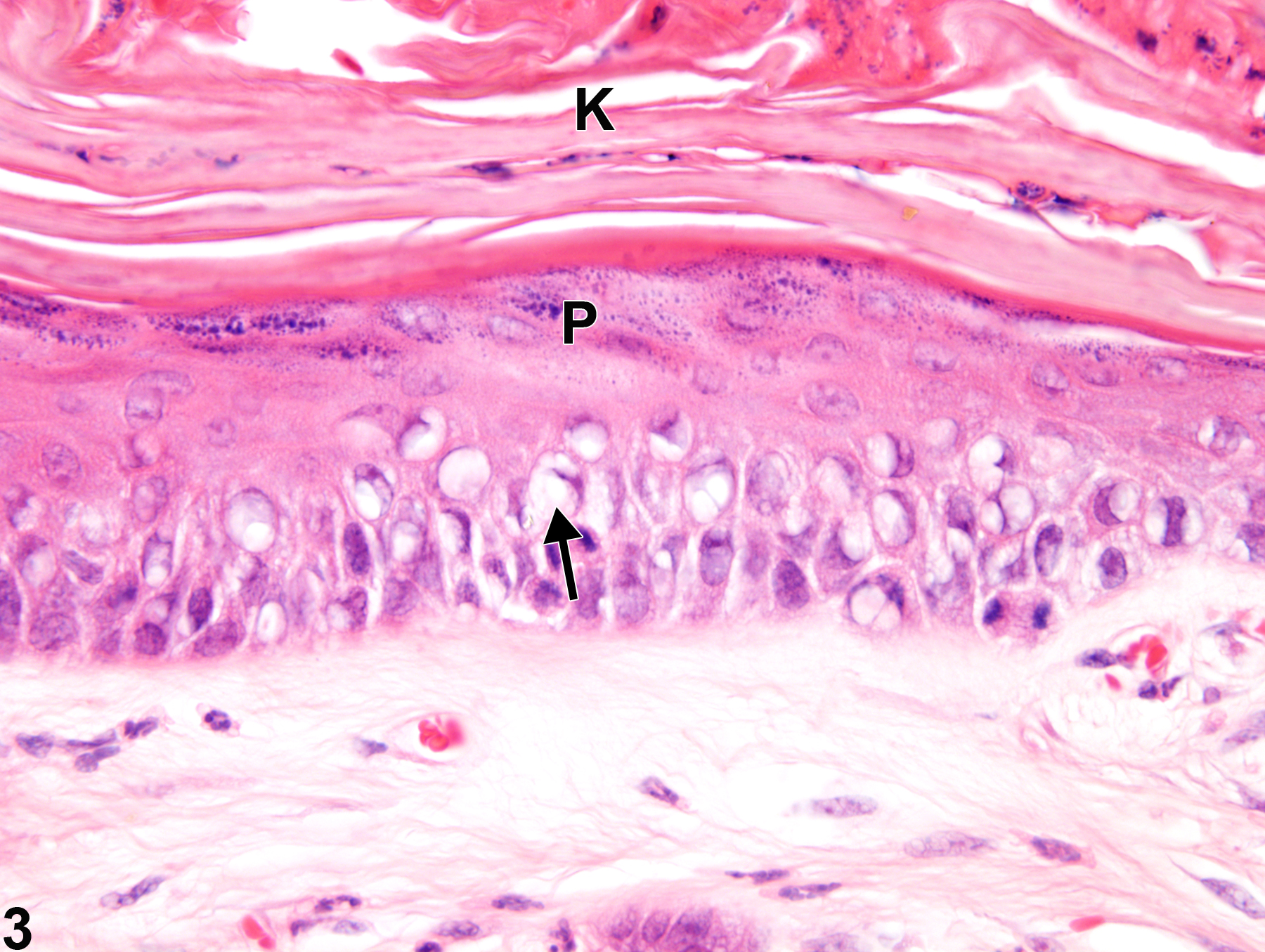
Eye, Cornea - Vacuolation, Cytoplasmic
Narrative
Geiss V, Yoshitomi K. 1991. Eyes. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 471-489.
Greaves P. 2007. Nervous system and special sense organs. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Academic Press, San Diego, CA, 861-933.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Guillet R, Wyatt J, Baggs RB, Kellogg CK. 1988. Anesthetic-induced corneal lesions in developmentally sensitive rats. Invest Ophthalmol Vis Sci 29:949-954.
Abstract: https://pubmed.ncbi.nlm.nih.gov/3372167/Kast A. 1991. Keratoconjunctivitis sicca and sequelae, mouse and rat. In: International Life Sciences Institute Monographs on the Pathology of Laboratory Animals, Vol 10, Eye and Ear (Jones TC, Mohr U, Hunt RD, eds). Springer, Berlin, 29-37.
Moore QA III, McCormick CC, Norcross EW, Onwubiko C, Sanders ME, Fratkin J, McDaniel LS, O’Calaghan RJ, Marquart ME. 2009. Development of a Streptococcus pneumoniae keratitis model in mice. Ophthalmol Res 42:141-146.
Full Text: http://www.karger.com/Article/Pdf/229028National Toxicology Program. 1994. NTP TR-435. Toxicology and Carcinogenesis Studies of 4,4’-Thiobis(6-t-butyl-m-cresol) (CAS No. 96-69-5) in F344/N Rats and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/6014National Toxicology Program. 2012. NTP TR-572. Toxicology and Carcinogenesis Studies of Methyl trans-Styryl Ketone (CAS No. 1896-62-4) in F344/N Rats and B6C3F1 Mice (Feed and Dermal Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/36154Smith RS, Sundberg JP, John SWM. 2002. The anterior segment. In: Systematic Evaluation of the Mouse Eye: Anatomy, Pathology, and Biomethods (Smith RS, John SWM, Nishina PM, Sundberg JP, eds). CRC Press, Boca Raton, FL, 111-159.
Yoshida M, Ikadai H, Maekawa, Takahashi M, Nagase S. 1996. Pathological characteristics of mucopolysaccharidosis VI in the rat. J Comp Path 109:141-155.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/8245230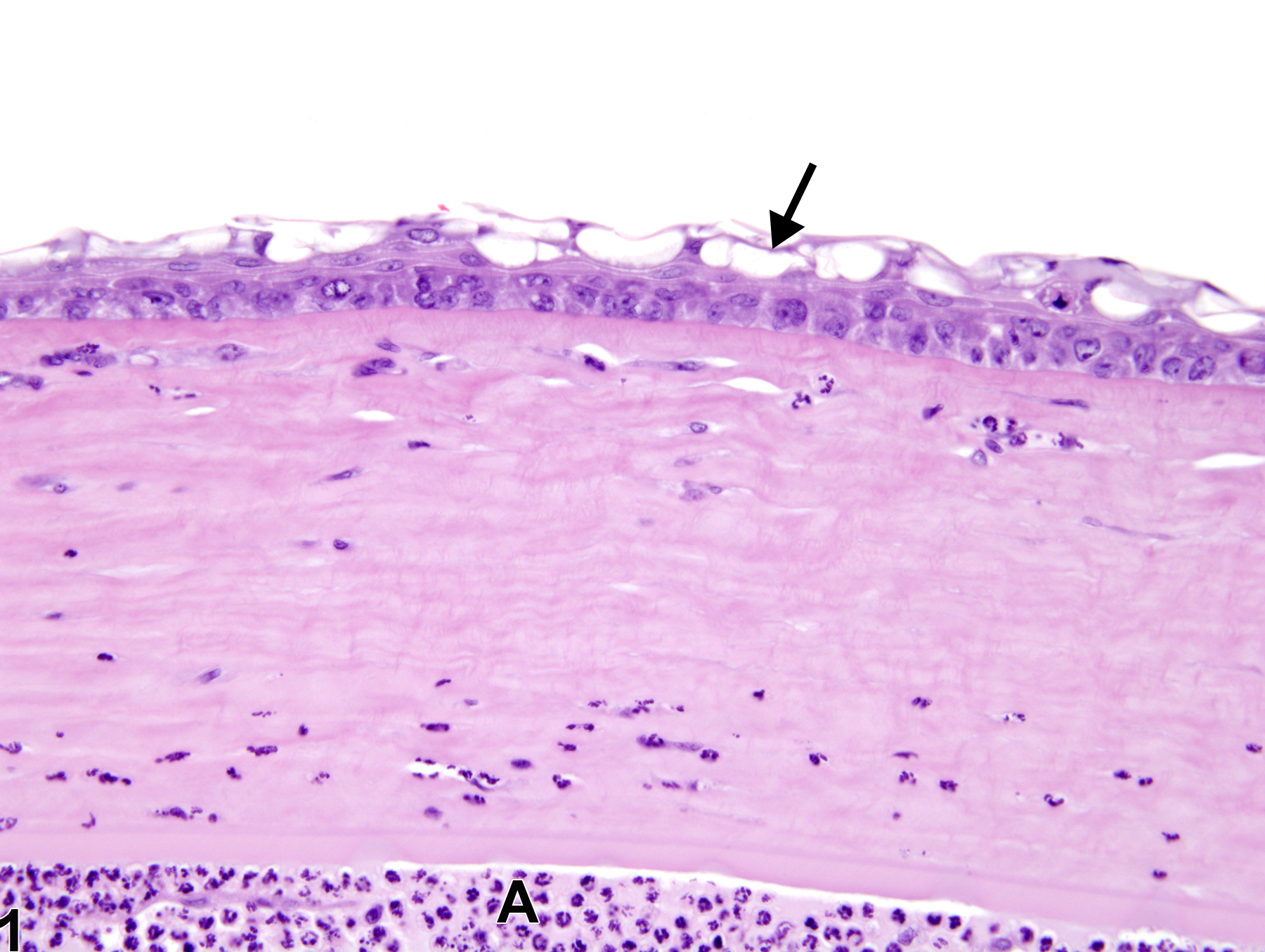
Eye, Cornea - Vacuolation, Cytoplasmic in a male F344/N rat from a chronic study. There are clear discrete vacuoles (arrow) in the corneal epithelial cells and inflammatory cells in the anterior chamber (A).