Special Senses System
Eye - Inflammation
Narrative
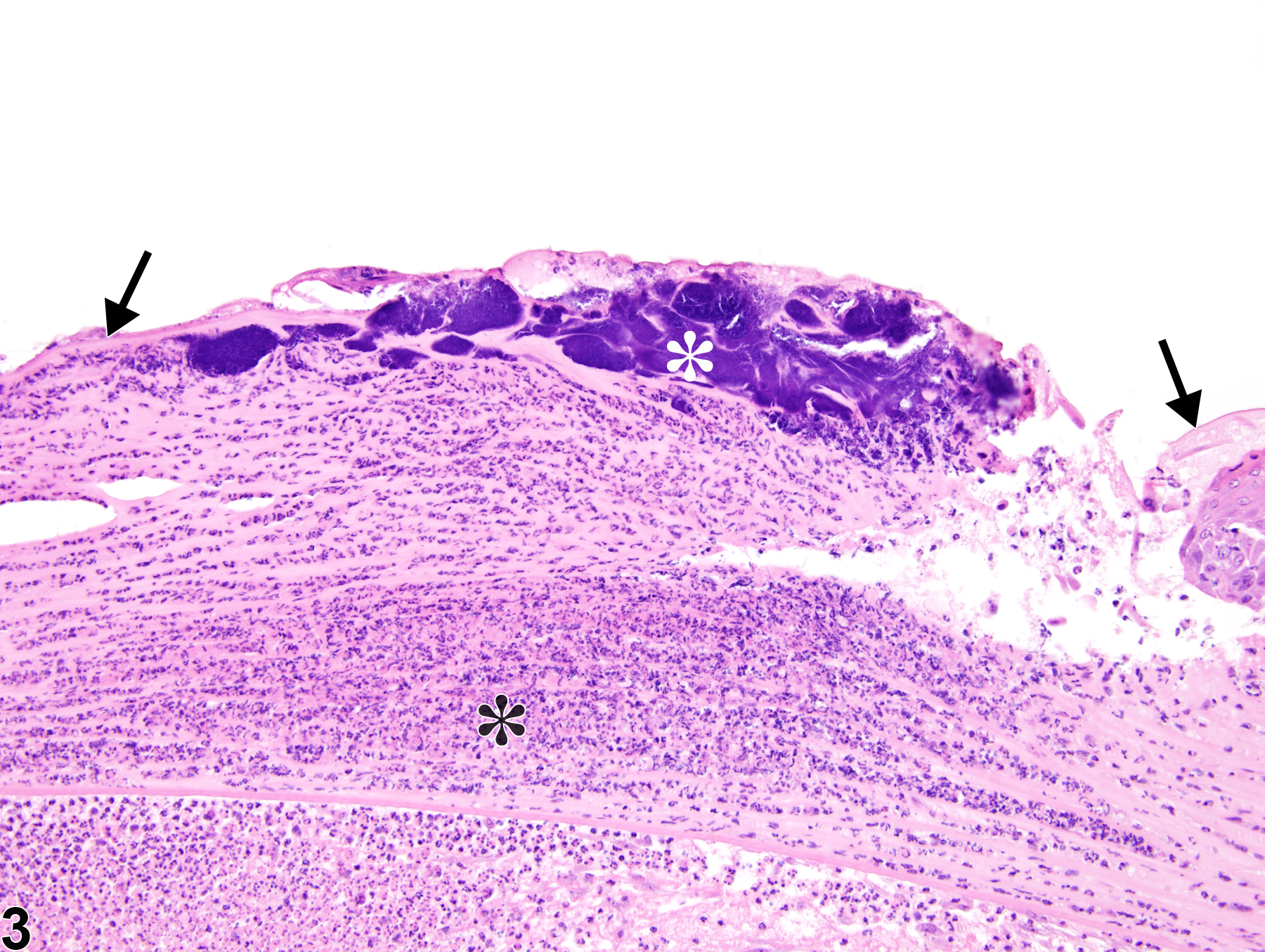
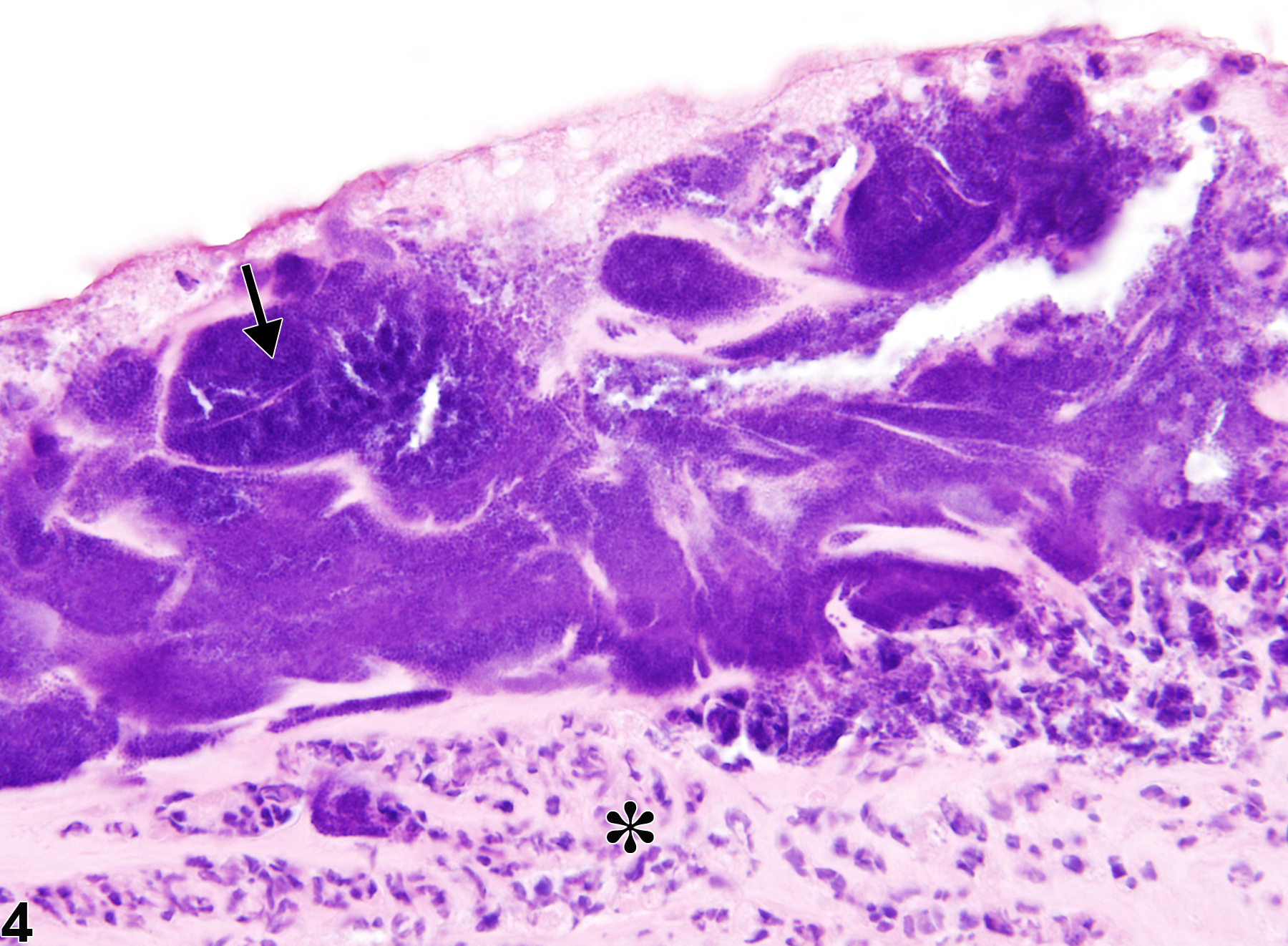
Corneal inflammation can be diffuse or localized in particular zones, such as the epithelium or stroma. In addition to the causes listed above, corneal inflammation can also be caused by insufficient tear production, nutritional deficiencies, and infectious agents, such as bacteria, fungi, and viruses. Since the cornea is normally an avascular tissue, stromal neovascularization is a common component of corneal inflammatory lesions. Hyperplasia of the epithelium is also a frequent finding (Figure 1 and Figure 2). In chronic lesions, stromal fibrosis may be seen. While the intact cornea is resistant to most bacterial pathogens, any breach in the epithelium (e.g., from abrasions, penetrating injury, or toxin-induced necrosis) can introduce bacteria, which incite an inflammatory response. Bacteria can also secondarily colonize preexisting areas of inflammation (Figure 3 and Figure 4) and/or ulceration. Anterior chamber inflammation can extend into the adjacent iris, ciliary body, and lens. Inflammatory cells in the anterior chamber can occlude the iridocorneal filtration angle channels and/or the adjacent trabecular meshwork, impeding normal drainage of aqueous humor with resultant elevations in intraocular pressure. Clinically, intracameral accumulations of neutrophils are referred to as hypopyon.
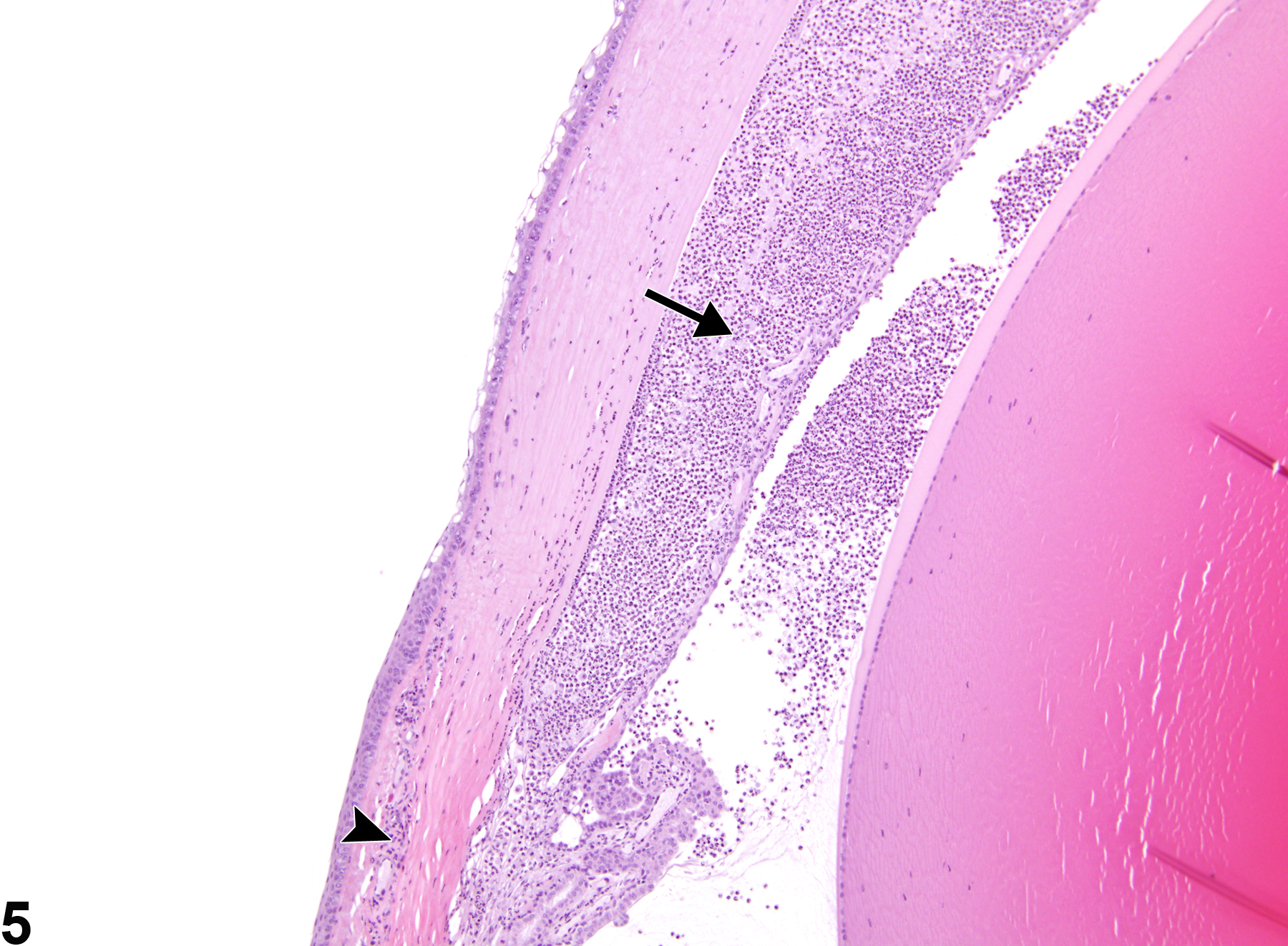
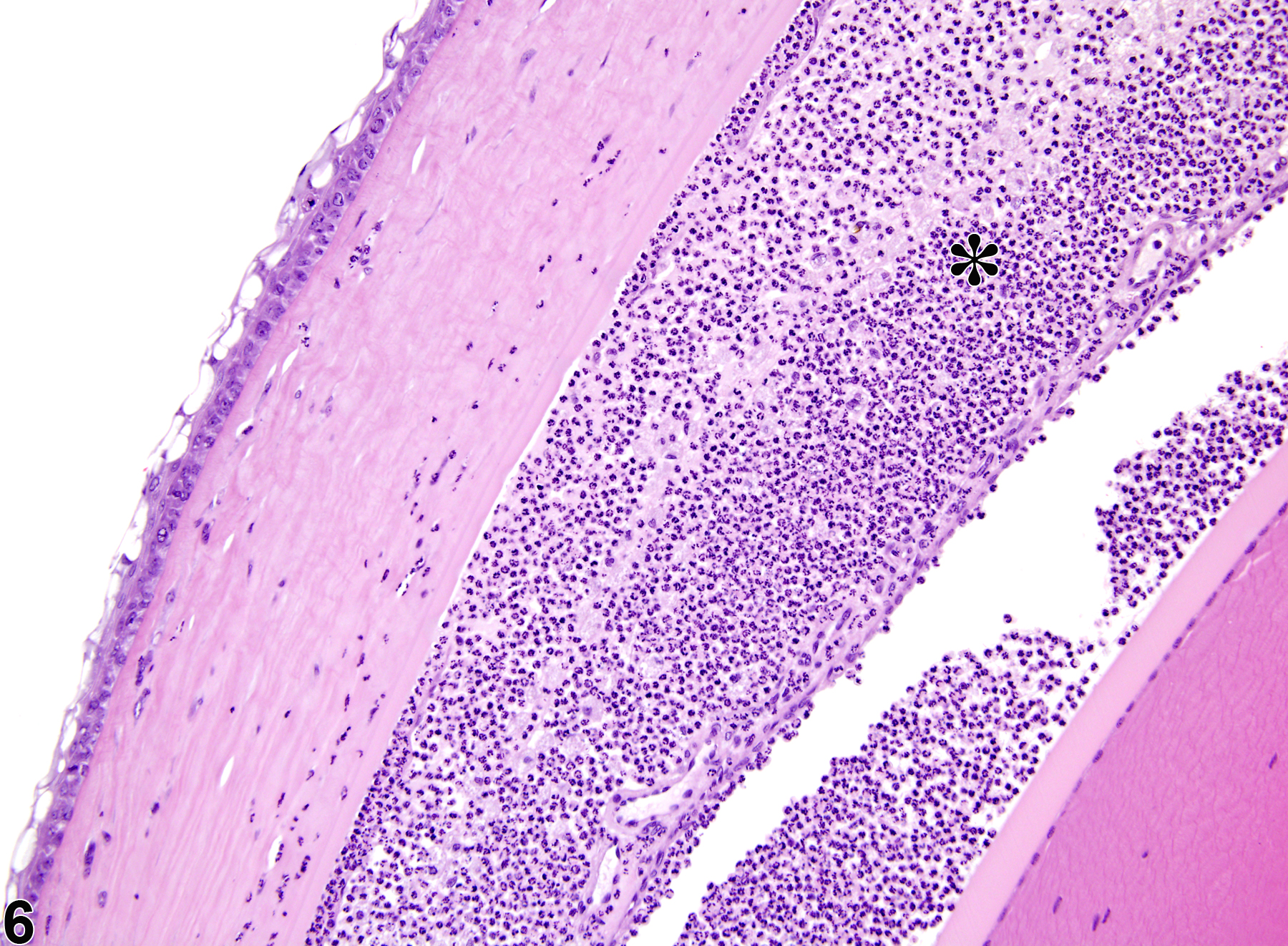
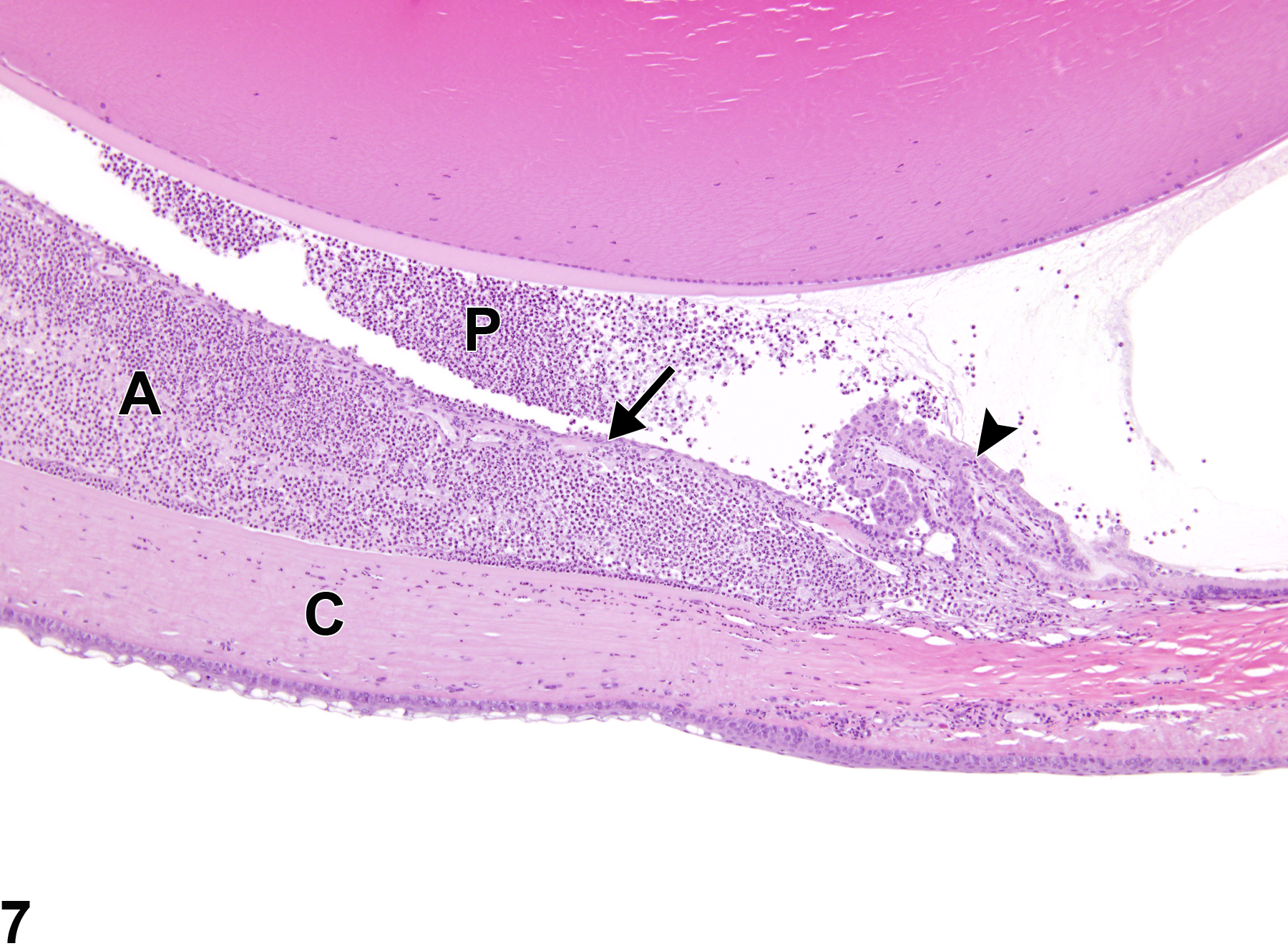
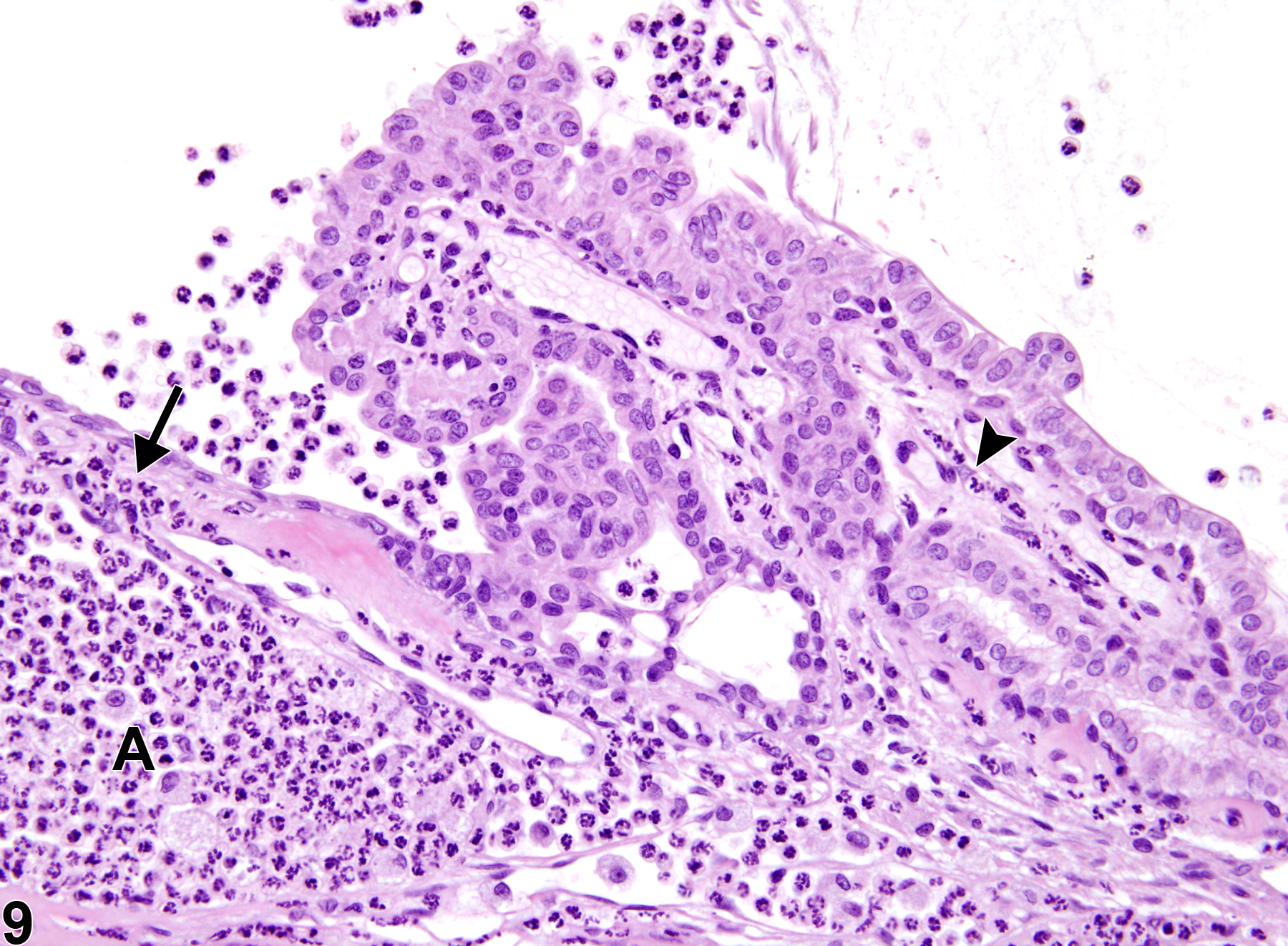
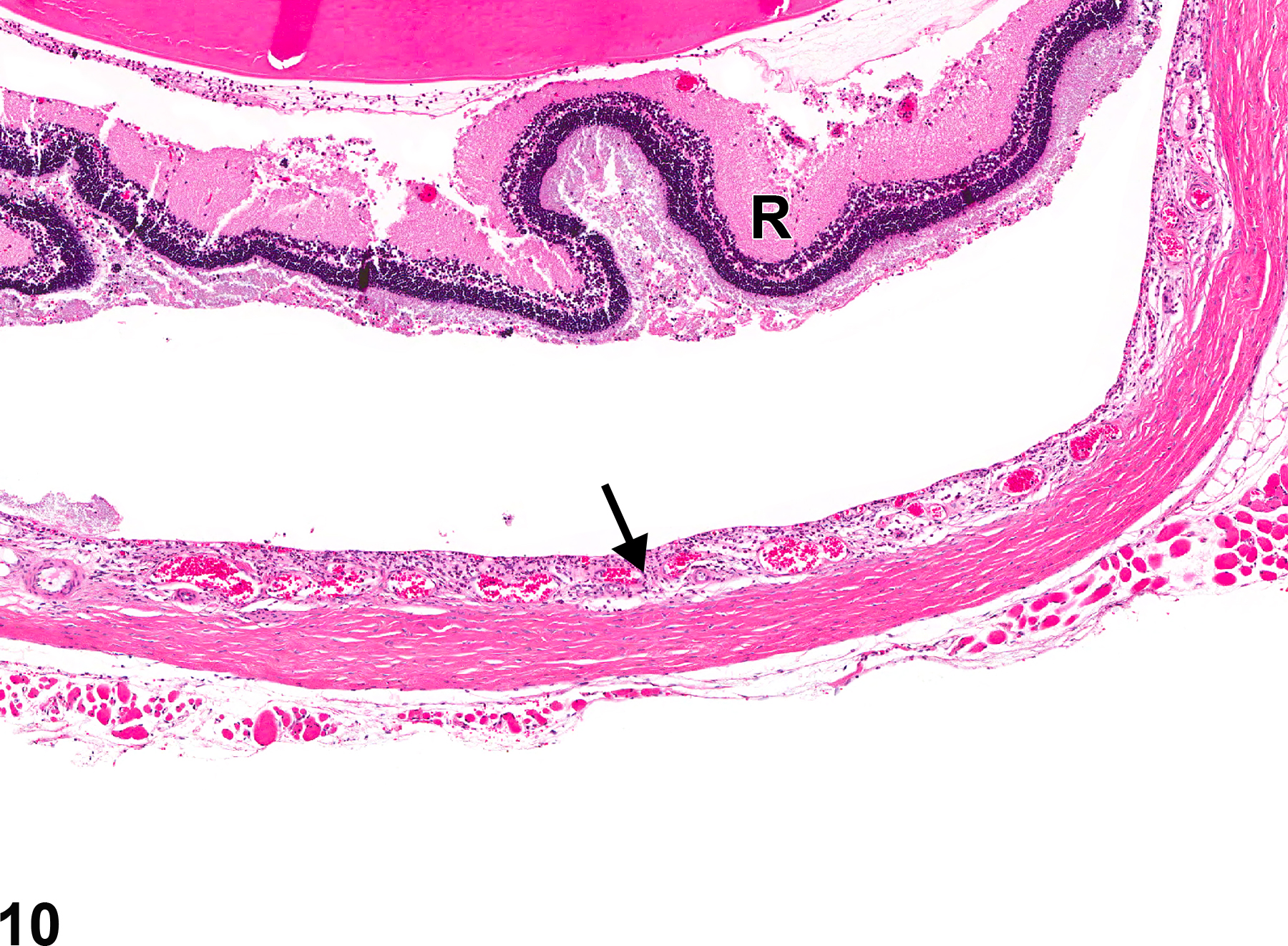
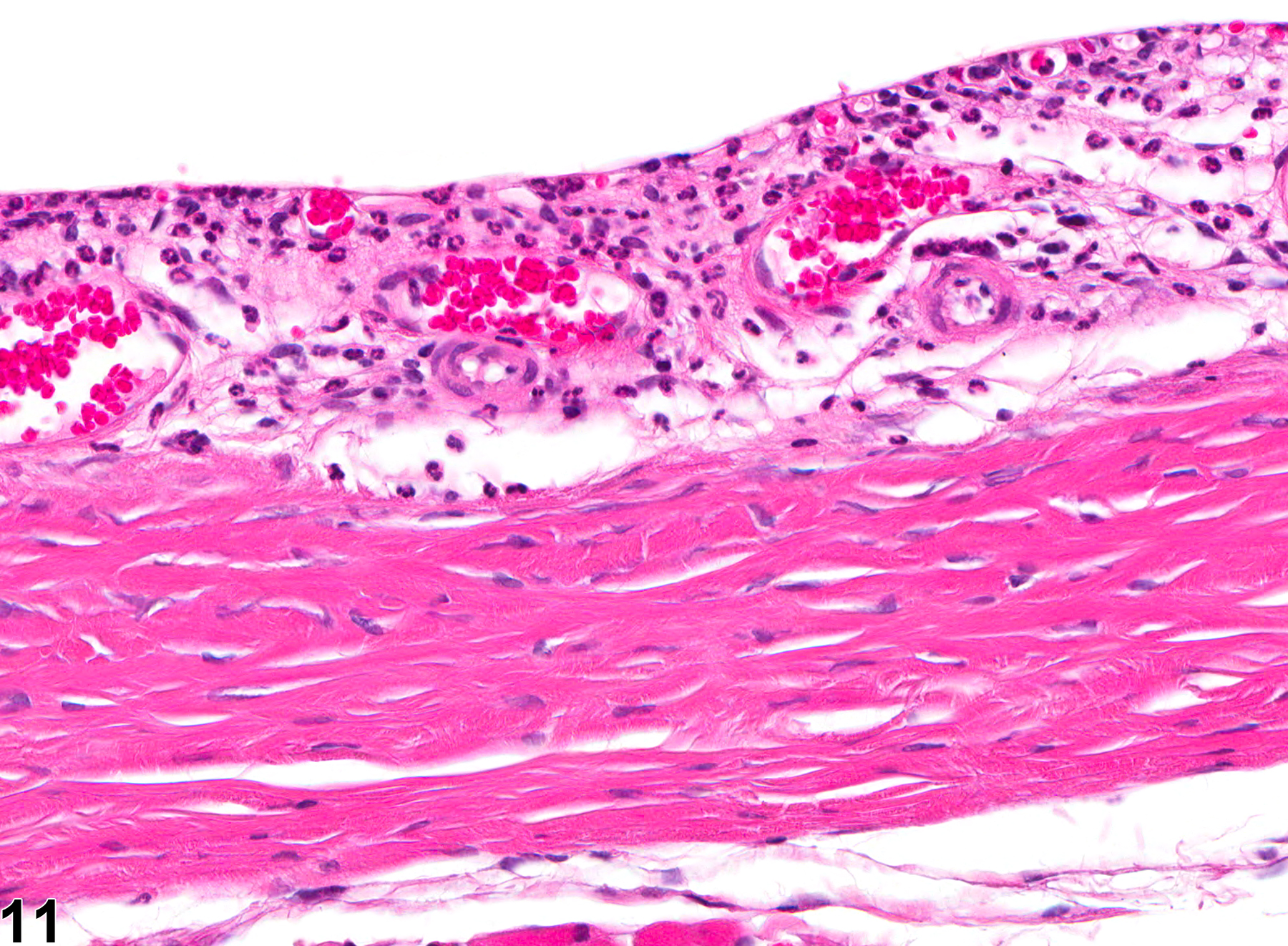
Anterior uveal tract (iris and ciliary body) inflammation can result from various causes, such as infectious agents such as bacteria and viruses, and immune-mediated (allergic) reactions. Iridial and ciliary body inflammation (Figure 7, Figure 8, and Figure 9) often extends into the adjacent anterior and posterior chambers, cornea, and/or choroid. Likewise, inflammation of the choroid, or posterior uvea (Figure 10 and Figure 11), often extends into the anterior uveal tract (ciliary body and iris) and/or into the adjacent retina.
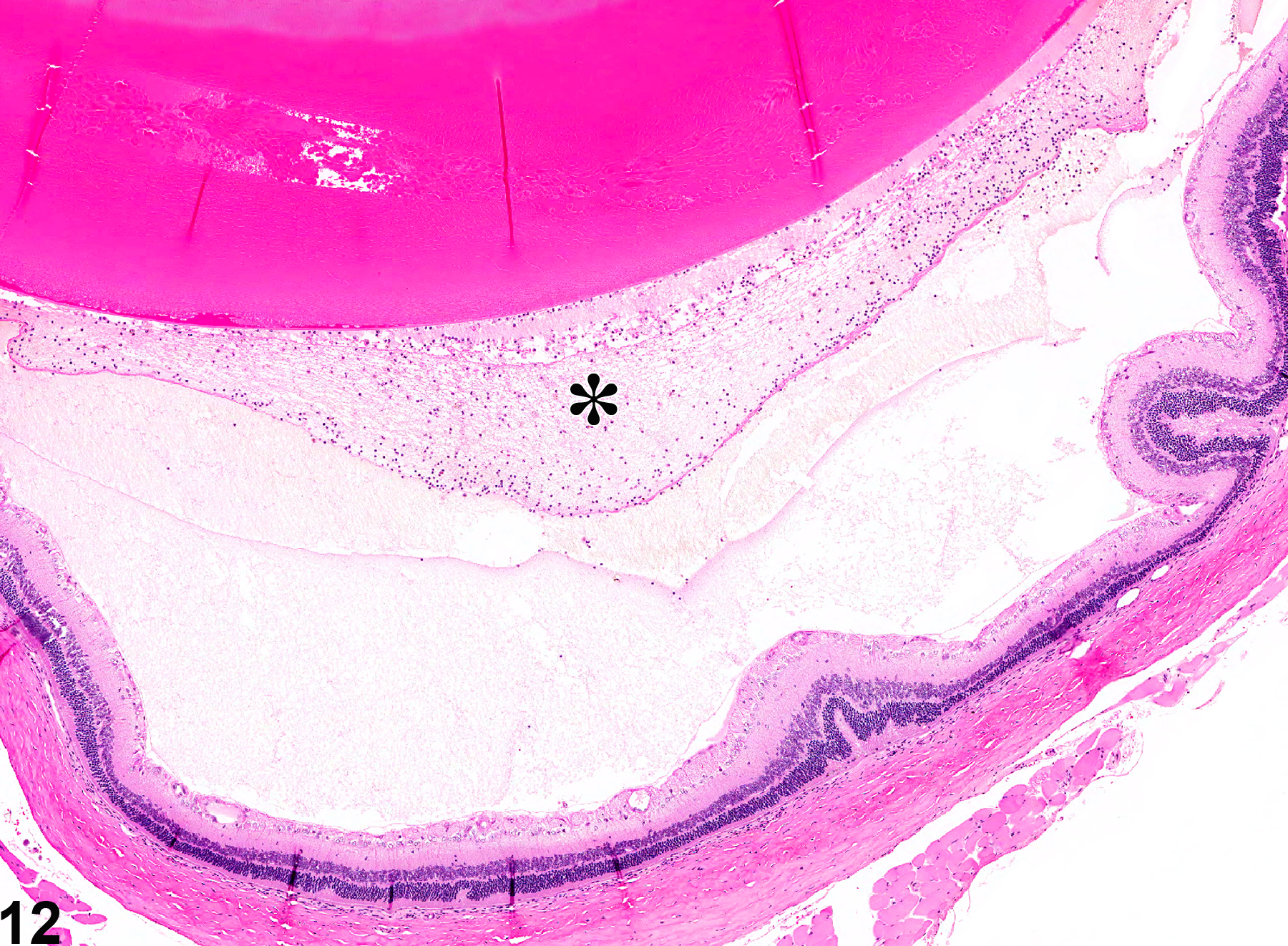
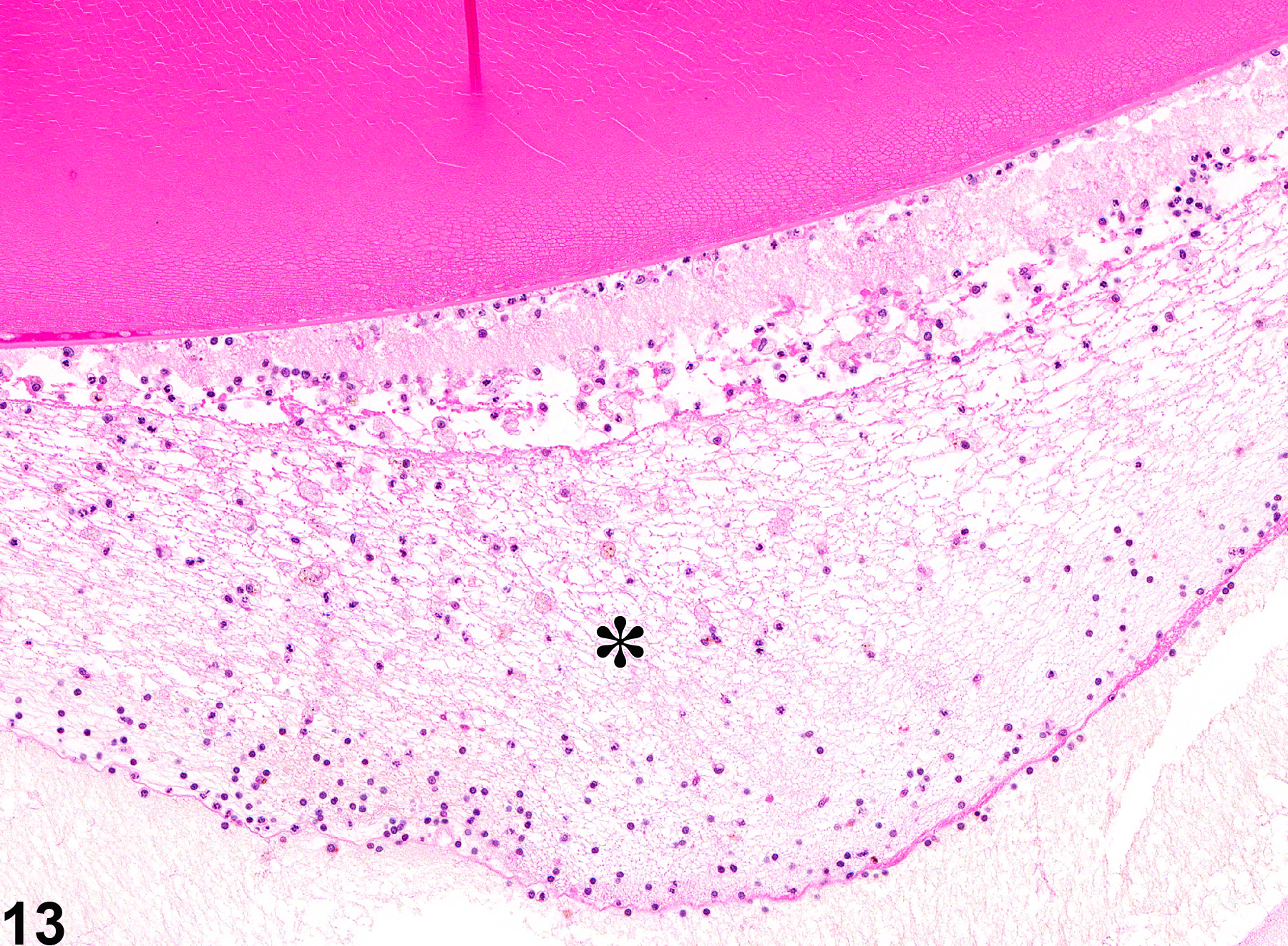
Vitreal inflammation (Figure 12 and Figure 13) is characterized by prominent accumulations of inflammatory cells in the vitreous. Vitreal inflammation can result from trauma (e.g., from intravitreal injections or other penetrating injury) or as a primary effect of intravitreally or systemically administered toxins. It can also occur secondary to other ocular lesions, such as cataracts and retinal inflammation. Chronic inflammatory infiltrates are often accompanied by vitreal fibrosis.
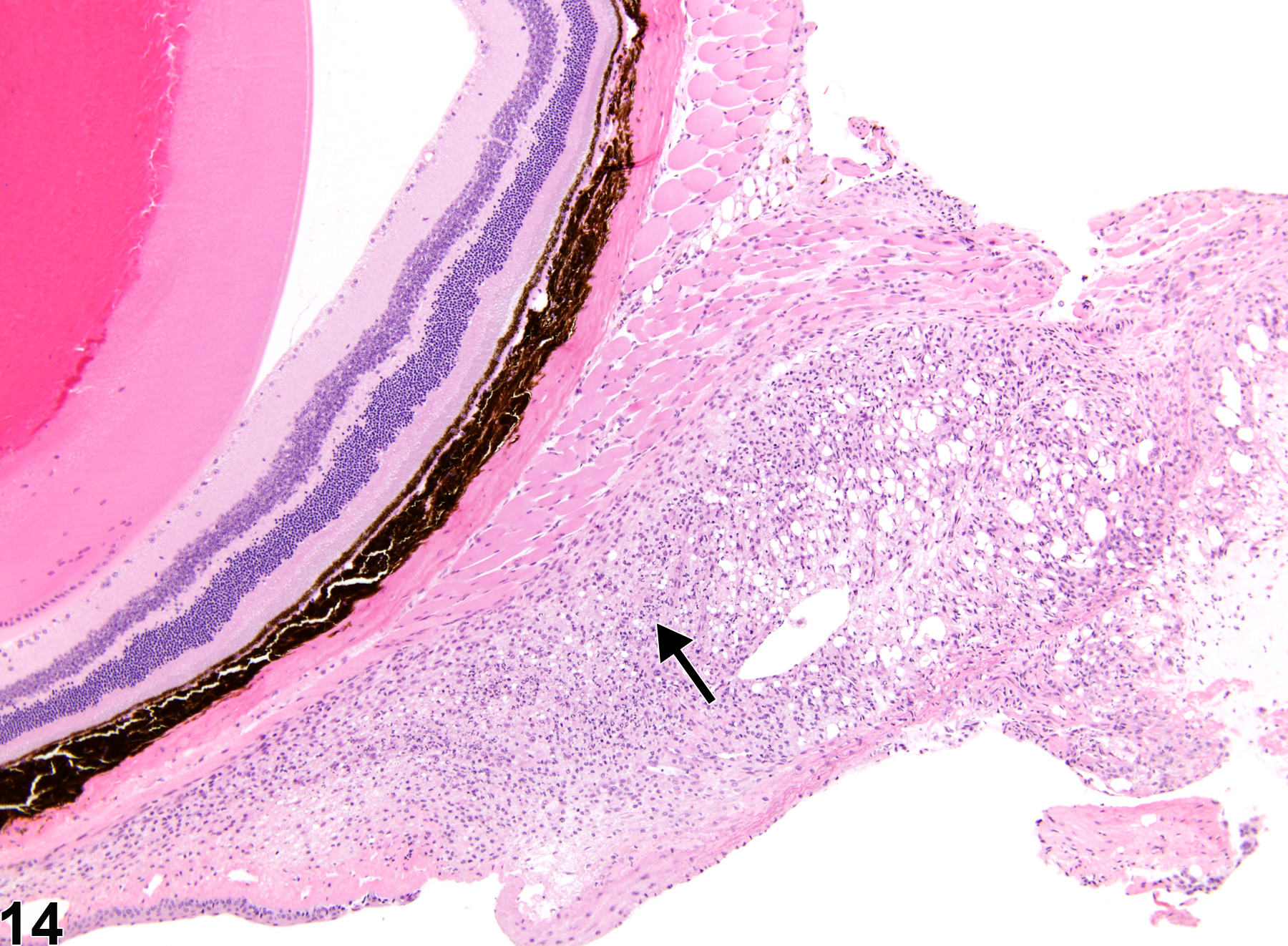
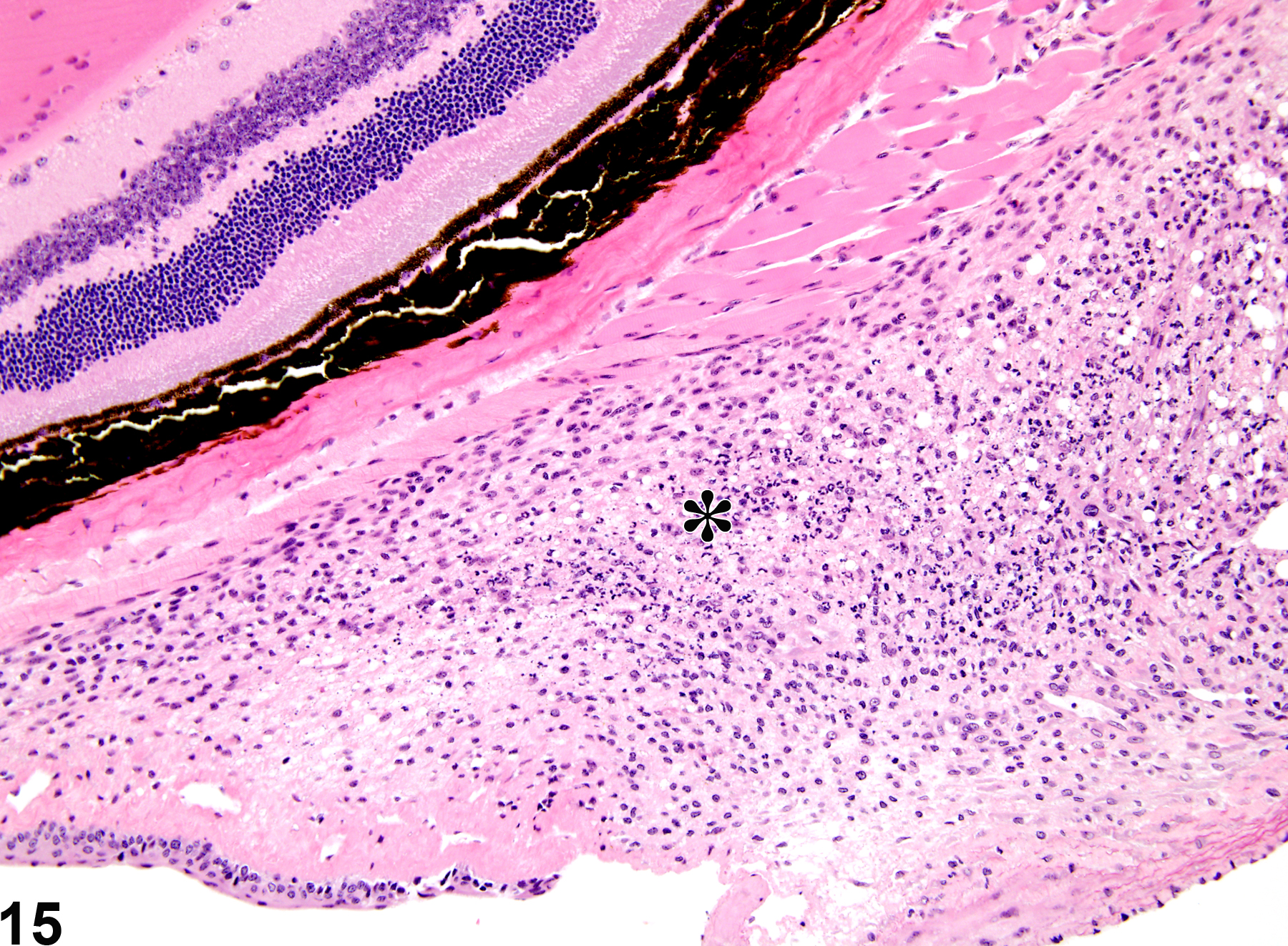
Retrobulbar inflammation (Figure 14 and Figure 15) is most frequently caused by trauma due to retrobulbar bleeding procedures and is typically acute. It is often accompanied by hemorrhage.
In NTP studies, there are five standard categories of inflammation: acute, suppurative, chronic, chronic-active, and granulomatous. In acute inflammation, the predominant infiltrating cell is the neutrophil, though fewer macrophages and lymphocytes may also be present. There may also be evidence of edema or hyperemia. The neutrophil is also the predominant infiltrating cell type in suppurative inflammation, however, in suppurative inflammation, the neutrophils are aggregated and many of them are degenerate (suppurative exudate). Cell debris, both from the resident cell populations and infiltrating leukocytes, proteinaceous fluid containing fibrin, fewer macrophages, occasional lymphocytes or plasma cells, and, possibly, an infectious agent may also be present in within the exudate. Grossly, these lesions would be characterized by the presence of pus. In the tissue surrounding the exudate, there may be fibroblasts, fibrous connective tissue, and mixed inflammatory cells, depending on the chronicity of the lesion. Lymphocytes predominate in chronic inflammation. Lymphocytes also predominate in chronic-active inflammation, but in chronic-active inflammation, there are also a significant number of neutrophils. Both lesions may contain macrophages. Granulomatous inflammation is another form of chronic inflammation, but this diagnosis requires the presence of a significant number of aggregated, large, activated macrophages, epithelioid macrophages, or multinucleated giant cells. Inflammation is differentiated from cellular infiltrates by the presence of other changes, such as edema, hemorrhage, degeneration, necrosis, or other evidence of tissue damage.
Atherton SS, Altman NH, Streilein JW. 1988. Histopathologic study of herpes virus-induced retinitis in athymic BALB/c mice: Evidence for an immunopathogenic process. Curr Eye Res 8:1179-1192.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/2558848Bora NS, Kim MC, Kabeer NH, Simpson SC, Tanhasetti MT, Cirrito TP, Kaplan AD, Kaplan HJ. 1995. Experimental autoimmune anterior uveitis: Induction with melanin-associated antigen from the iris and ciliary body. Invest Ophthalmol Vis Sci 36:1056-1066.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/7730015Chan C-C, Caspi RR, Ni M, Leake WC, Wiggert B, Chader G, Nussenblatt RB. 1990. Pathology of experimental autoimmune uveoretinitis in mice. J Autoimmunity 3:247-255.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/2397018De Kozak Y, Camelo S, Pla M. 2008. Pathological aspects of spontaneous uveitis and retinopathy in HLA-A29 transgenic mice and in animal models of retinal autoimmunity: Relevance to human pathologies. Ophthalmic Res 40:175-180.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/18421235Frame SR, Slone TW. 1966. Nonneoplastic and neoplastic changes in the eye. In: Pathobiology of the Aging Mouse, Vol 2 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). ILSI Press, Washington, DC, 97-103.
Geiss V, Yoshitomi K. 1991. Eyes. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 471-489.
Gelderman MP, Charukamnoetkanok P, Brady JP, Hung L, Zigler JS, Wawrousek EF, Vistica BP, Fortin E, Chan C-C, Gery I. 2003. A novel inflammatory eye disease induced by lymphocytes from knockout mice sensitized against the deleted ocular antigen. Clin Exp Immunol 133:177-181.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/12869022Greaves P. 2007. Nervous system and special sense organs. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Academic Press, San Diego, CA, 861-933.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Guillet R, Wyatt J, Baggs RB, Kellogg CK. 1988. Anesthetic-induced corneal lesions in developmentally sensitive rats. Invest Ophthalmol Vis Sci 29:949-954.
Abstract: https://pubmed.ncbi.nlm.nih.gov/3372167/Hu J, Wang Y, Xie L. 2009. Potential role of macrophages in experimental keratomycosis. Invest Ophthalmol Vis Sci 50:2087-2094.
Full Text: https://doi.org/10.1167/iovs.07-1237Jabs DA, Prendergast RA. 1991. Ocular inflammation in MRL/mp-lpr/lpr mice. Invest Ophthalmol Vis Sci 32:1944-1991.
Abstract: https://pubmed.ncbi.nlm.nih.gov/2032814/Kozhich AT, Chan C-C, Gery I, Whitcup SM. 2000. Recurrent intraocular inflammation in endotoxin-induced uveitis. Invest Ophthalmol Vis Sci 41:1823-1826.
Abstract: https://pubmed.ncbi.nlm.nih.gov/10845604/Karicherla P, Hobden JA. 2009. Nona-D-arginine therapy for Pseudomonas aeruginosa keratitis. Invest Ophthalmol Vis Sci 50:256-262.
Abstract: https://pubmed.ncbi.nlm.nih.gov/18775859/Komatsu K, Miyazaki D, Morohoshi K, Kuo C-H, Kakimaru-Hasegawa A, Komatsu N, Namba S. 2008. Pathogenesis of herpetic stromal keratitis in CCR5- and/or CXCR3-deficient mice. Curr Eye Res 33:736-749.
Abstract: https://doi.org/10.1080/02713680802344716Lai Y-L, Jacoby RO, Bhatt PN, Jonas AM. 1976. Keratoconjunctivitis associated with sialodacryoadenitis Invest Ophthalmol Vis Sci 15:538-541.
Abstract: https://pubmed.ncbi.nlm.nih.gov/931700/Maurer JK, Parker RD, Carr GJ. 1998. Ocular irritation: Microscopic changes occurring over time in the rat with surfactants of known irritancy. Toxicol Pathol 26:217-225.
Abstract: http://tpx.sagepub.com/content/26/2/217.shortNational Toxicology Program. 1988. NTP TR-331. Toxicology and Carcinogenesis Studies of Malonaldehyde, Sodium Salt (3-Hydroxy-2-propenal, Sodium Salt) (CAS No. 24382-04-5) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/8898National Toxicology Program. 1992. NTP TR-407. Toxicology and Carcinogenesis Studies of C.I. Pigment Red 3 (CAS No. 2425-85-6) in F344/N Rats and B6C3F1 Mice (Feed Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/7694National Toxicology Program. 1998. NTP TR-468. Toxicology and Carcinogenesis Studies of Oxazepam (CAS No. 604-75-1) in F344/N Rats (Feed Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/6080National Toxicology Program. 2012. NTP TR-572. Toxicology and Carcinogenesis Studies of Methyl trans-Styryl Ketone (CAS No. 1896-62-4) in F344/N Rats and B6C3F1 Mice (Feed and Dermal Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/36154National Toxicology Program. 2012. NTP TR-579. Toxicology and Carcinogenesis Studies of N, N-Dimethyl-p-Toluidine (CAS No. 99-97-8) in F344/N Rats and B6C3F1/N Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/37162Pauly A, Brignole-Baudouin, Labbé A, Liang H, Warnet J-M, Baudouin C. 2007. New tools for the evaluation of toxic ocular surface changes in the rat. Invest Ophthalmol Vis Sci 48:5473–5463.
Full Text: https://doi.org/10.1167/iovs.06-0728Ravindranath RMH, Mondino BJ, Adamu SA, Pitchekian-Halabi H, Hasan SA, Glasgow BJ. 1995. Immunopathologic features of Staphylococcus aureus endophthalmitis in the rat. Invest Ophthalmol Vis Sci 36:2482-2491
Abstract: https://pubmed.ncbi.nlm.nih.gov/7591638/Smith RS. 2002. Choroid, lens, and vitreous. In: Systematic Evaluation of the Mouse Eye: Anatomy, Pathology, and Biomethods (Smith RS, John SWM, Nishina PM, Sundberg JP, eds). CRC Press Boca Raton, FL, 161-193.
Smith RS, Sundberg JP, John SWM. 2002. The anterior segment. In: Systematic Evaluation of the Mouse Eye: Anatomy, Pathology, and Biomethods (Smith RS, John SWM, Nishina PM, Sundberg JP, eds). CRC Press, Boca Raton, FL, 111-159.
Takase H, Yu C-R, Ham D-I, Chan C-C, Chen J, Vistica BP, Warousek EF, Durum SK, Egwuagu CE, Gery I. 2006. Inflammatory processes triggered by TCR engagement or by local cytokine expression: Differences in profiles of gene expression and infiltrating cell populations. J Leukocyte Biol 80:538-545.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/16793919Whittum JA, McCulley JP, Niederkorn JY, Streilein JW. 1984. Ocular disease induced in mice by anterior chamber inoculation of herpes simplex virus. Invest Ophthalmol Vis Sci 25:1065-1073.
Abstract: https://pubmed.ncbi.nlm.nih.gov/6469490/Yamamoto T, Goto H, Yamakawa N, Mori H, Okada S, Fujita K, Ishikawa A. 2010. Kinetics of polymorphonuclear leukocytes in an experimental hypopyon model. Exp Eye Res 91:685-690.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/20723542Yoshitomi K, Boorman GA. 1990. Eye and associated glands. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 239-260.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563Yudkin AM, Lambert RM. 1923. Pathogenesis of the ocular lesions produced by a deficiency of vitamin A. J Exp Med 38:17-24.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2128416/pdf/17.pdf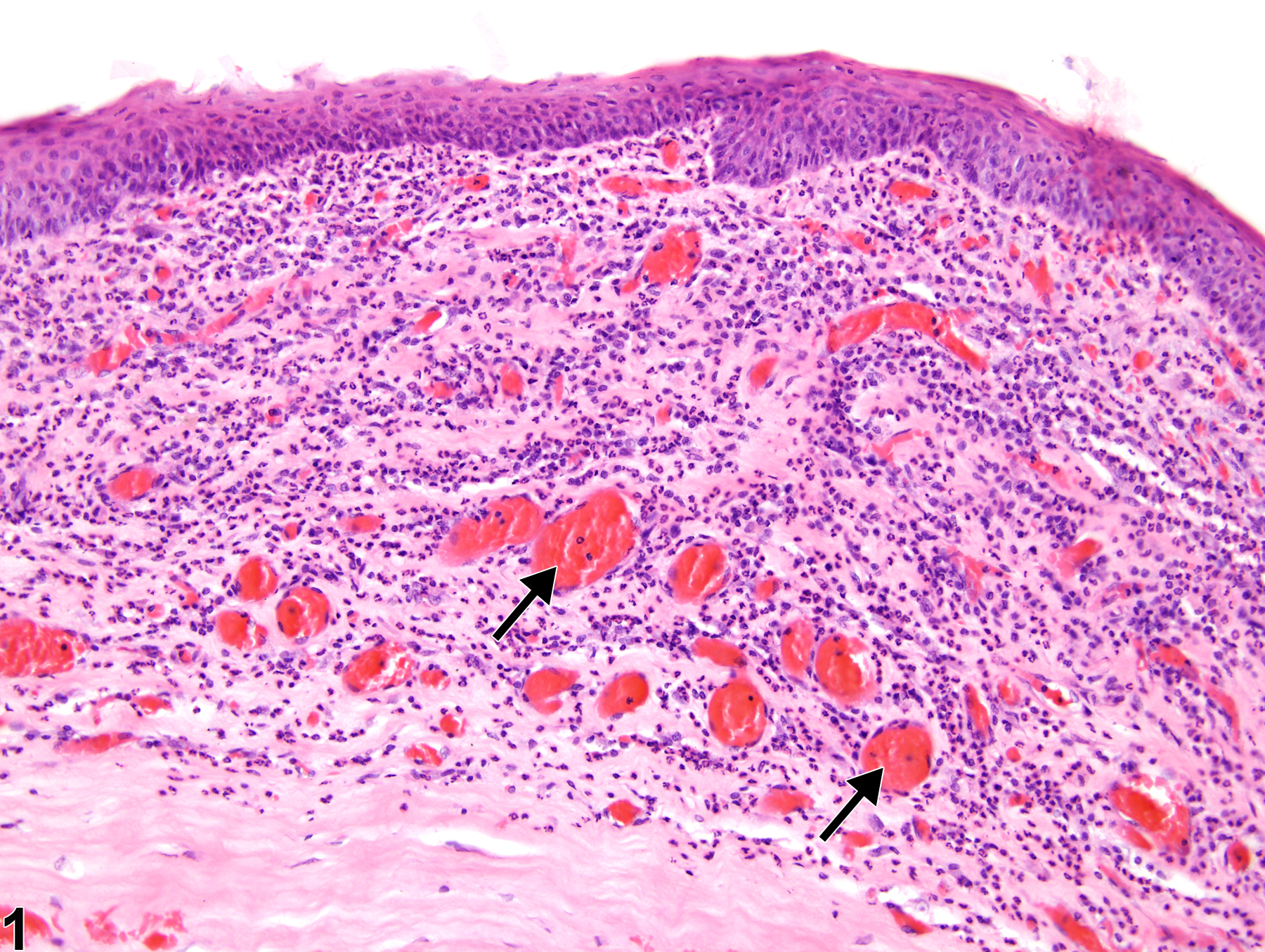
Eye, Cornea - Inflammation, Chronic active in a male F344/N rat from a chronic study. There are diffuse stromal inflammatory cell infiltrates (asterisk), stromal neovascularization (arrow), and fibrosis, and concurrent epithelial hyperplasia (arrowhead).
All Images
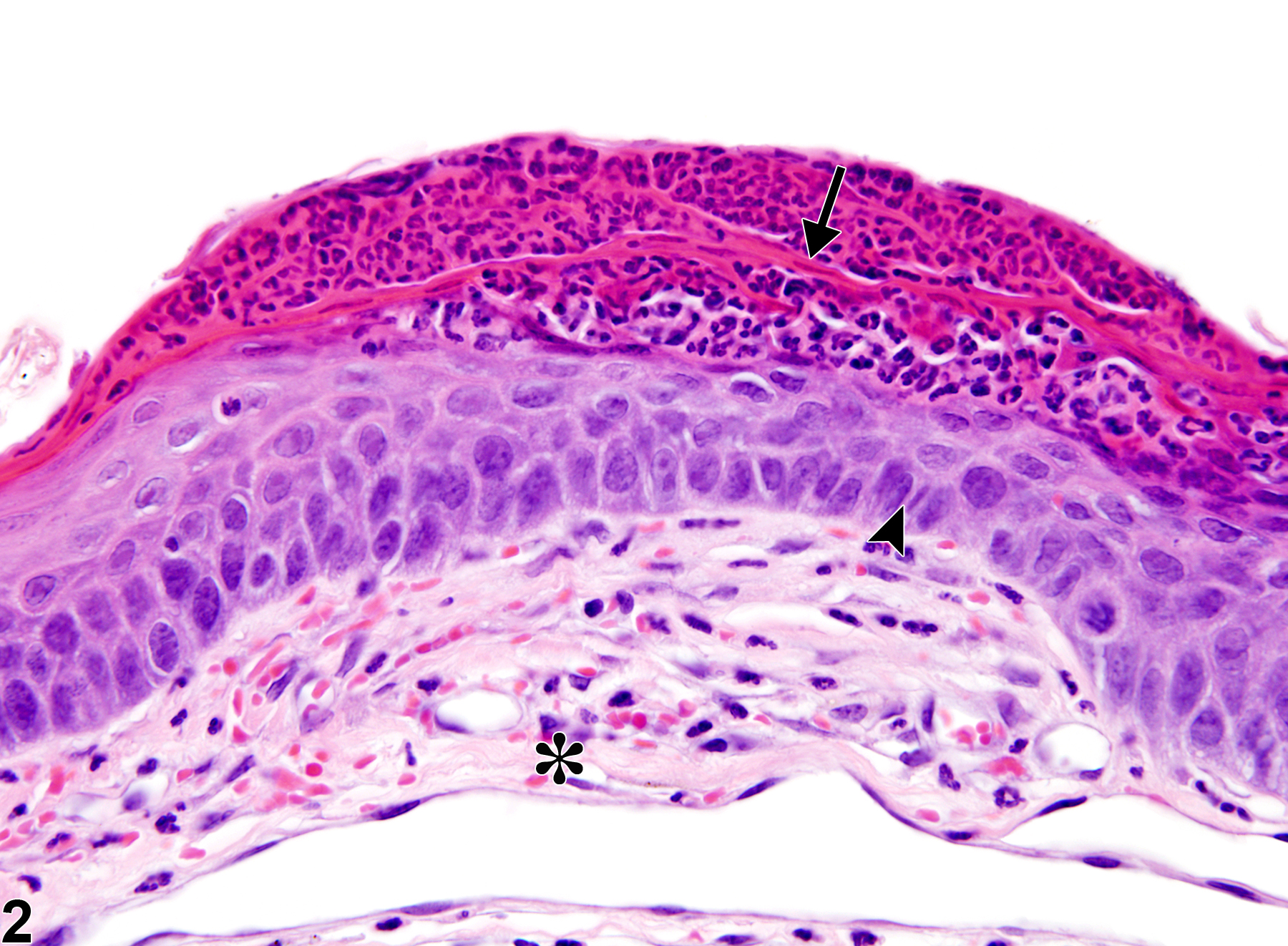
Eye, Cornea - Inflammation, Chronic active in a female B6C3F1 mouse from a chronic study. There is a focal accumulation of neutrophils and necrotic debris (arrow) within the concurrently hyperplastic epithelium (arrowhead); neovascularization and fibrosis are evident in the underlying stroma (asterisk).
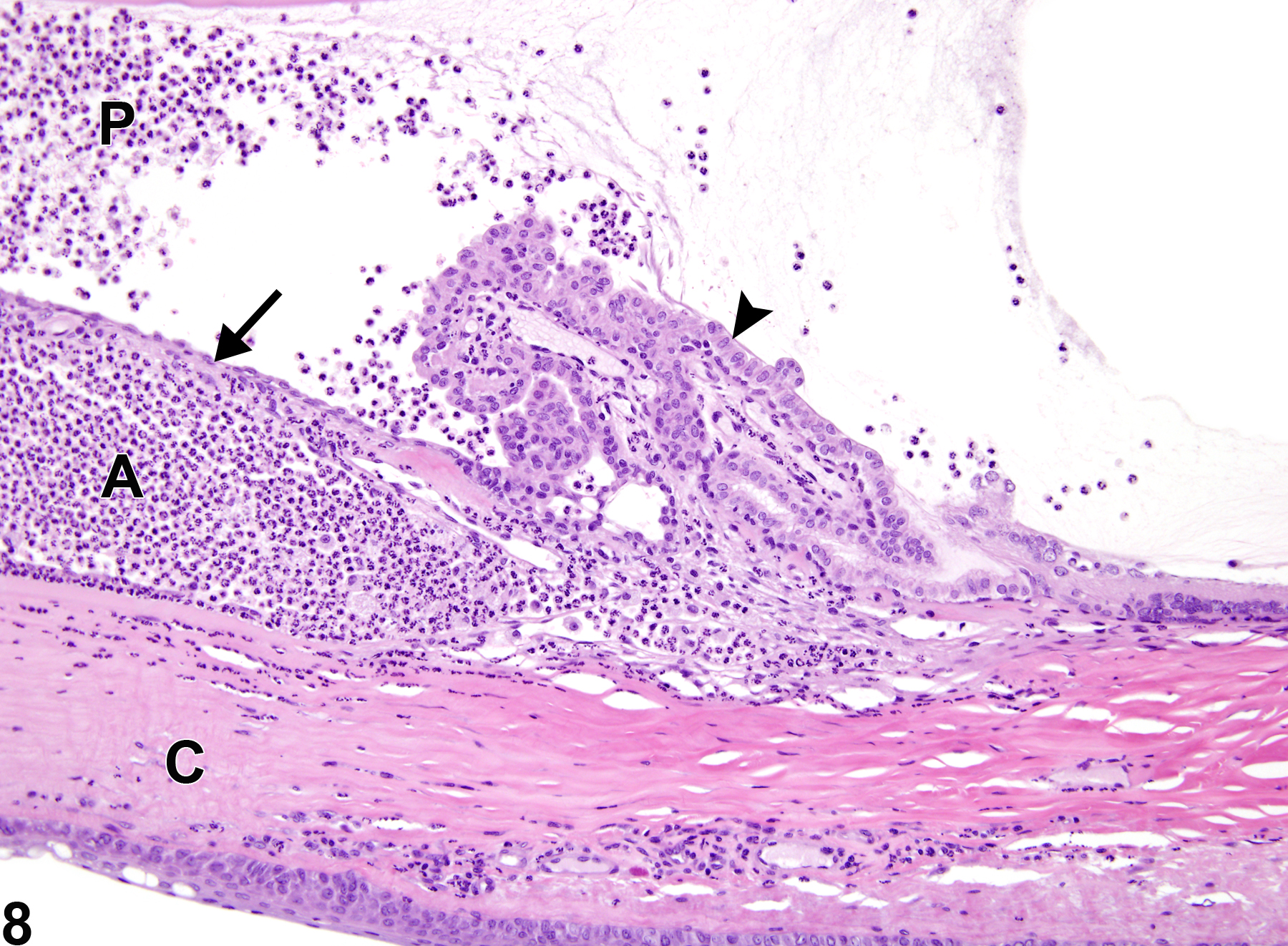
Eye, Iris - Inflammation, Acute and Eye, Ciliary body - Inflammation, Acute in a male F344/N rat from a chronic study (higher magnification of Figure 1). The inflammatory cells in the iris (arrow) and ciliary body (arrowhead) are neutrophils; those in the anterior chamber (A), posterior chamber (P), and cornea (C) are also neutrophils.