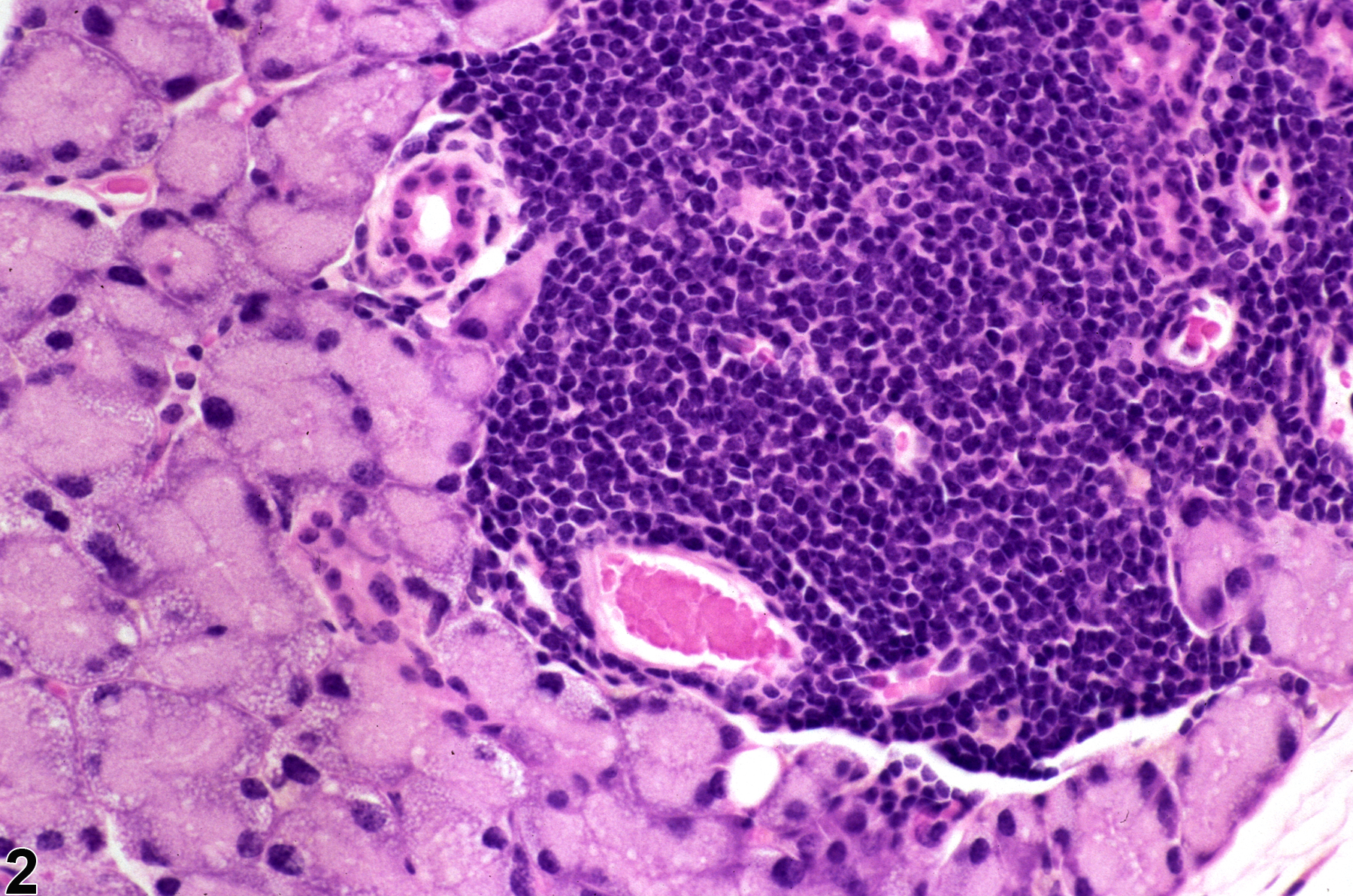
Special Senses System
Lacrimal Gland - Infiltration Cellular, Mononuclear Cell
Narrative
Botts S, Jokinen M, Gaillard ET, Elwell MR, Mann PC. 1999. Salivary, Harderian, and lacrimal glands. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 49-79.
Greaves P. 2007. Nervous system and special sense organs. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 3rd ed. Academic Press, San Diego, CA, 861-933.
Abstract: http://www.sciencedirect.com/science/book/9780444527714Jiang G, Ke Y, Sun D, Li H, Ihnen M, Jumblatt MM, Foulks G, Wang Y, Bian Y, Kaplan HJ, Shao H. 2009. A new model of experimental autoimmune keratoconjunctivitis sicca (KCS) induced in Lewis rat by the autoantigen Klk1b22. Invest Ophthalmol Vis Sci 50:2245-2254.
Full Text: https://doi.org/10.1167/iovs.08-1949Kojima T, Wakamatsu TS, Dogru M, Ogawa Y, Igarishi A, Ibrahim OMA, Inaba T, Shimizu T, Noda S, Obata H, Nakmura S, Wakmatsu A, Shirasawa T, Simazaki J, Negishi K, Tsubota K. 2012. Age-related dysfunction of the lacrimal gland and oxidative stress: Evidence from the Cu,Zn,-superoxide dismutase (Sod1) knockout mice. Am J Pathol 180:1879-1896.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/22440255Krinke GJ, Schaetti PR, Krinke A. 1996. Nonneoplastic and neoplastic changes in the Harderian and lacrimal glands. In: Pathobiology of the Aging Mouse, Vol 2 (Mohr U, Dungworth DL, Capen CC, Carlton WW, Sundberg JP, Ward JM, eds). International Life Sciences Institute Press, Washington, DC, 139-152.
Mikulowska-Mennis A, Xu B, Berberian JM, Michie SA. 2001. Lymphocyte migration to inflamed lacrimal glands is mediated by vascular cell adhesion molecule-1/ α4 β1 integrin, peripheral node addressin/l-selectin, and lymphocyte function-associated antigen-1 adhesion pathways. Am J Pathol 159:671–681.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1850552/National Toxicology Program. 1990. NTP TR-386. Toxicology and Carcinogenesis Studies of Tetranitromethane (CAS No. 509-14-8) in F344/N Rats and B6C3F1 Mice (Inhalation Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/12223Rahimy E, Pitcher JD III, Pangelinan SB, Chen W, Farley WJ, Niederkorn JY, Stern ME, Li DQ, Pflugfelder SC, De Paiva CS. 2010. Spontaneous autoimmune dacryoadenitis in aged CD25KO mice. Am J Pathol 177:744-753.
Full Text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2913359/Rios JD, Horikawa Y, Chan L-L, Kubin CL, Hodges RR, Dartt DA, Zoukhri D. 2005. Age-dependent alterations in mouse exorbital lacrimal gland structure, innervation and secretory response. Exp Eye Res 80:477-491.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15781275Wu K, Joffre C, Li X, MacVeigh-Aloni M, Hom M, Hwang J, Ding C, Gregoire S, Bretillon L, Zhong JF, Hamm-Alvarez SF. 2009. Altered expression of genes functioning in lipid homeostasis is associated with lipid deposition in NOD mouse lacrimal gland. Exp Eye Res 89:319-332.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/19345210Yoshitomi K, Boorman GA. 1990. Eye and associated glands. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 239-260.
Abstract: https://www.ncbi.nlm.nih.gov/nlmcatalog/9002563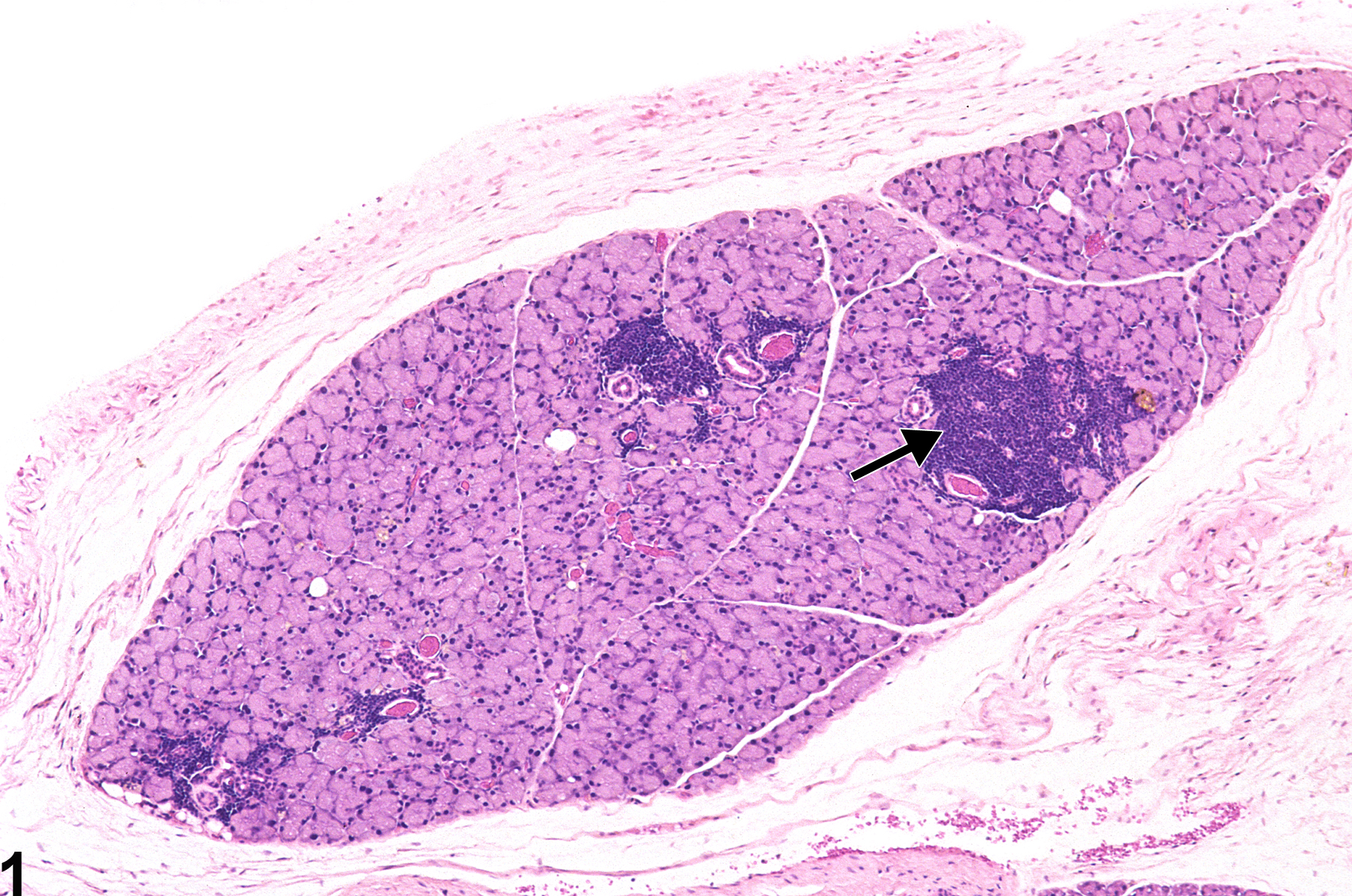
Lacrimal gland - Infiltration cellular, Mononuclear cell in a male B6C3F1 mouse from a chronic study. Variably sized interstitial foci of mononuclear cells (mainly lymphocytes) (arrow) are present in the lacrimal gland.