Reproductive System, Female
Ovary - Inflammation
Narrative
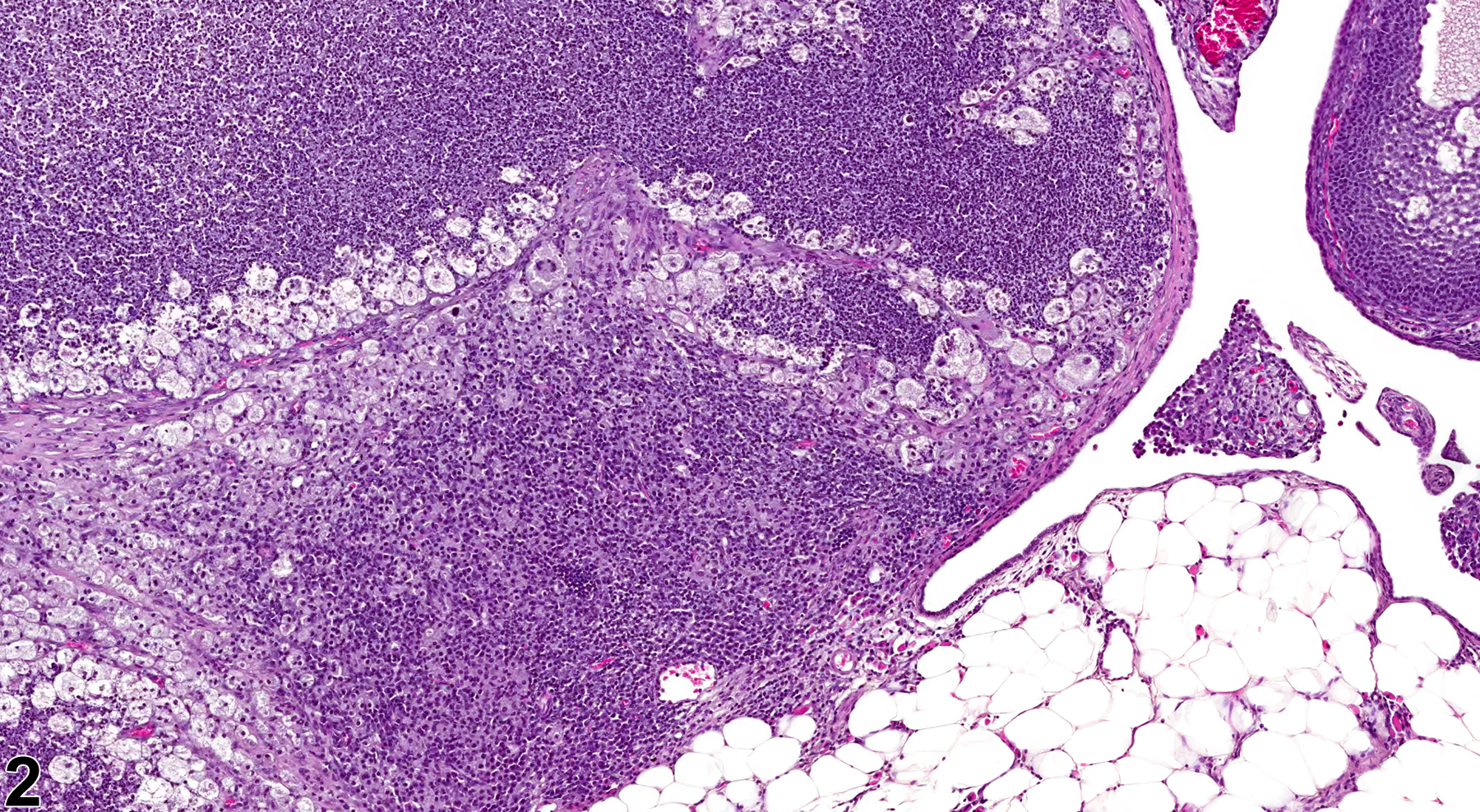
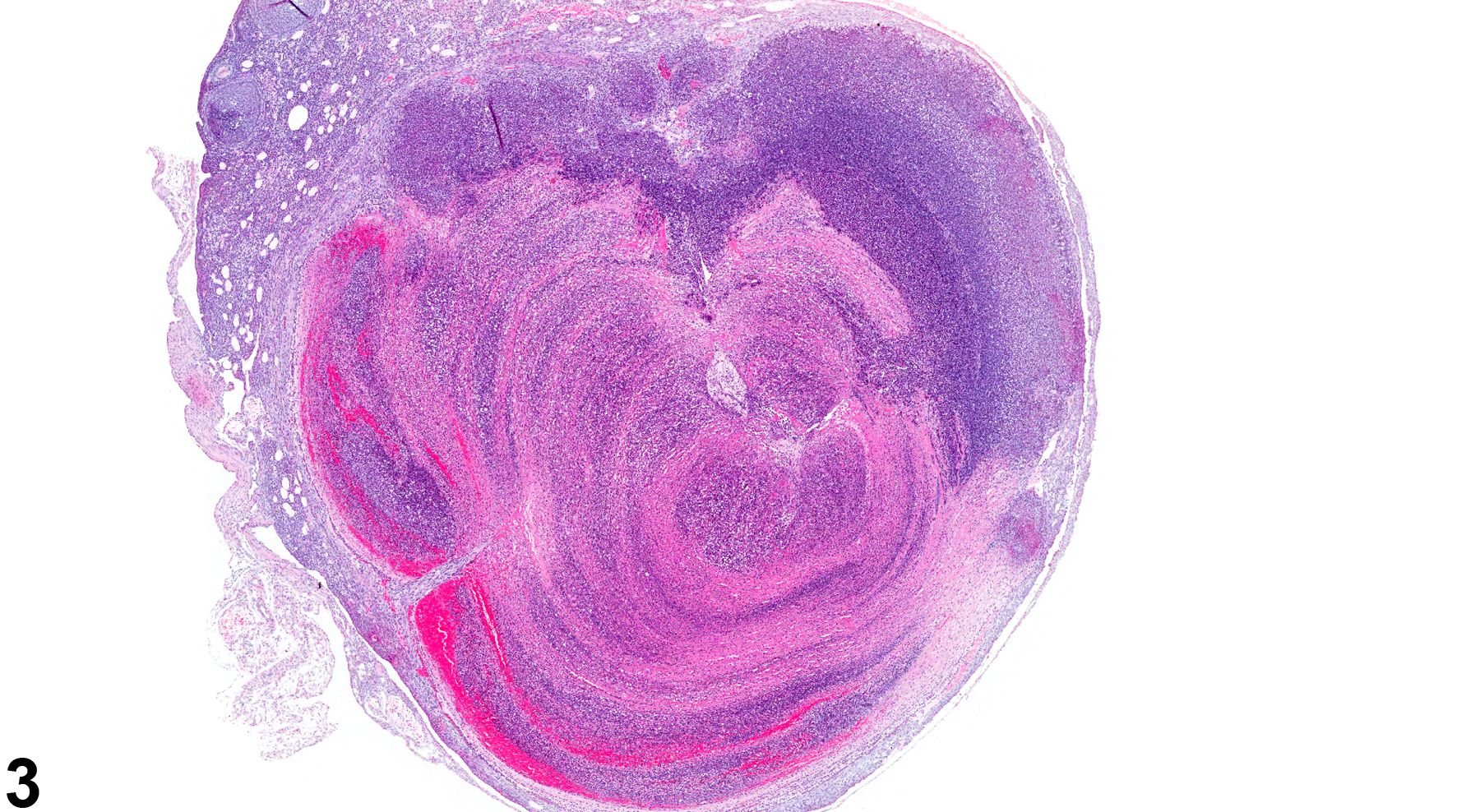
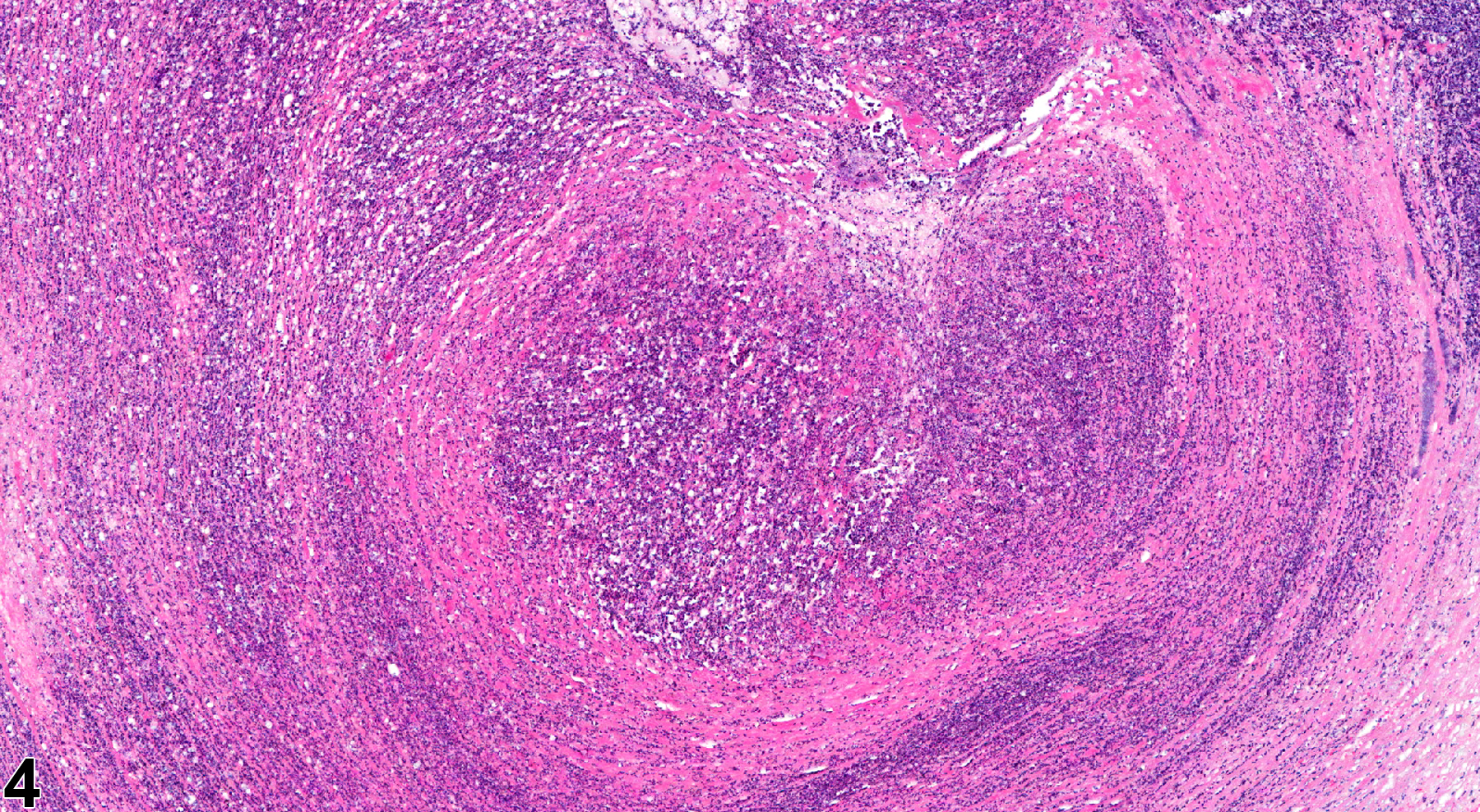
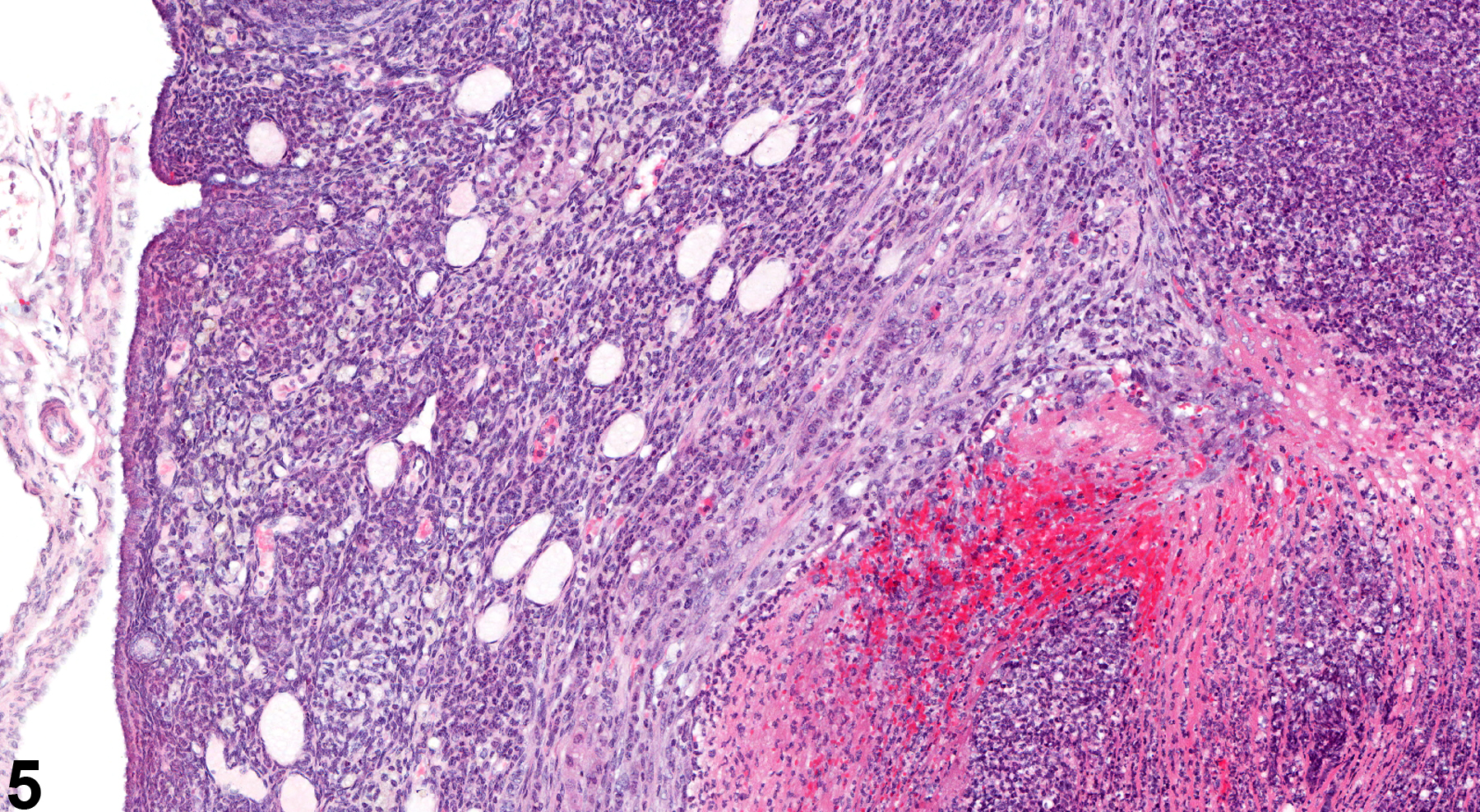
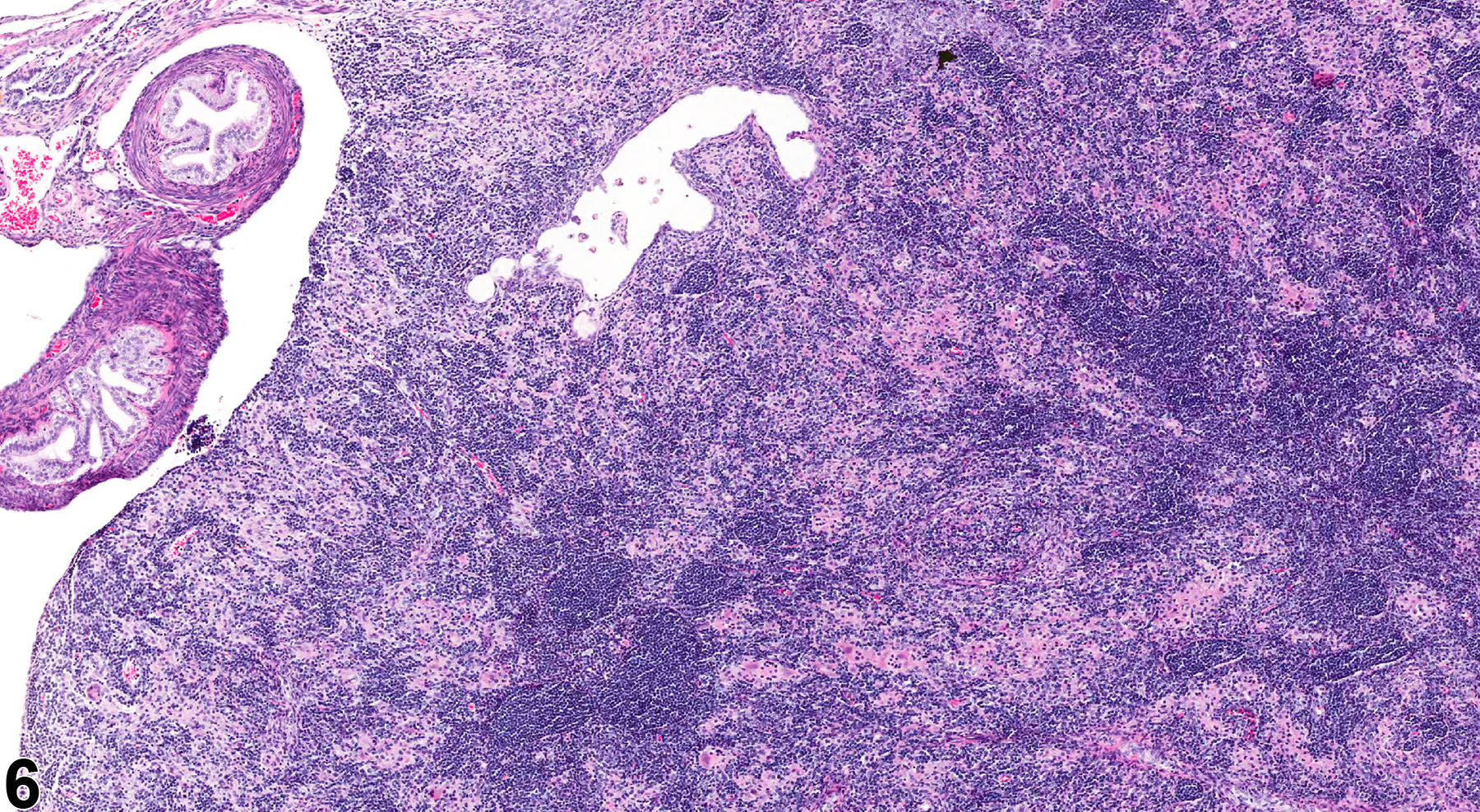
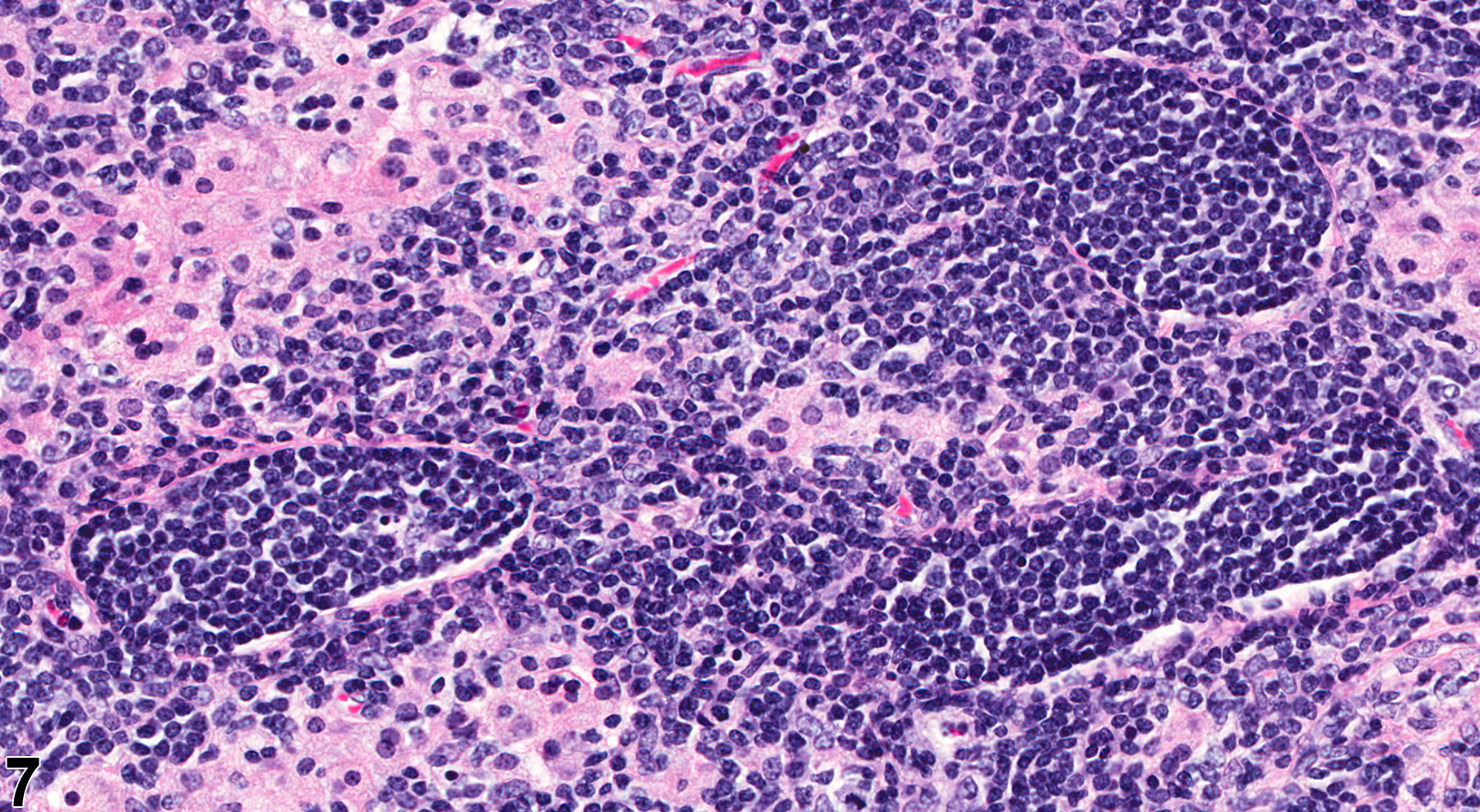
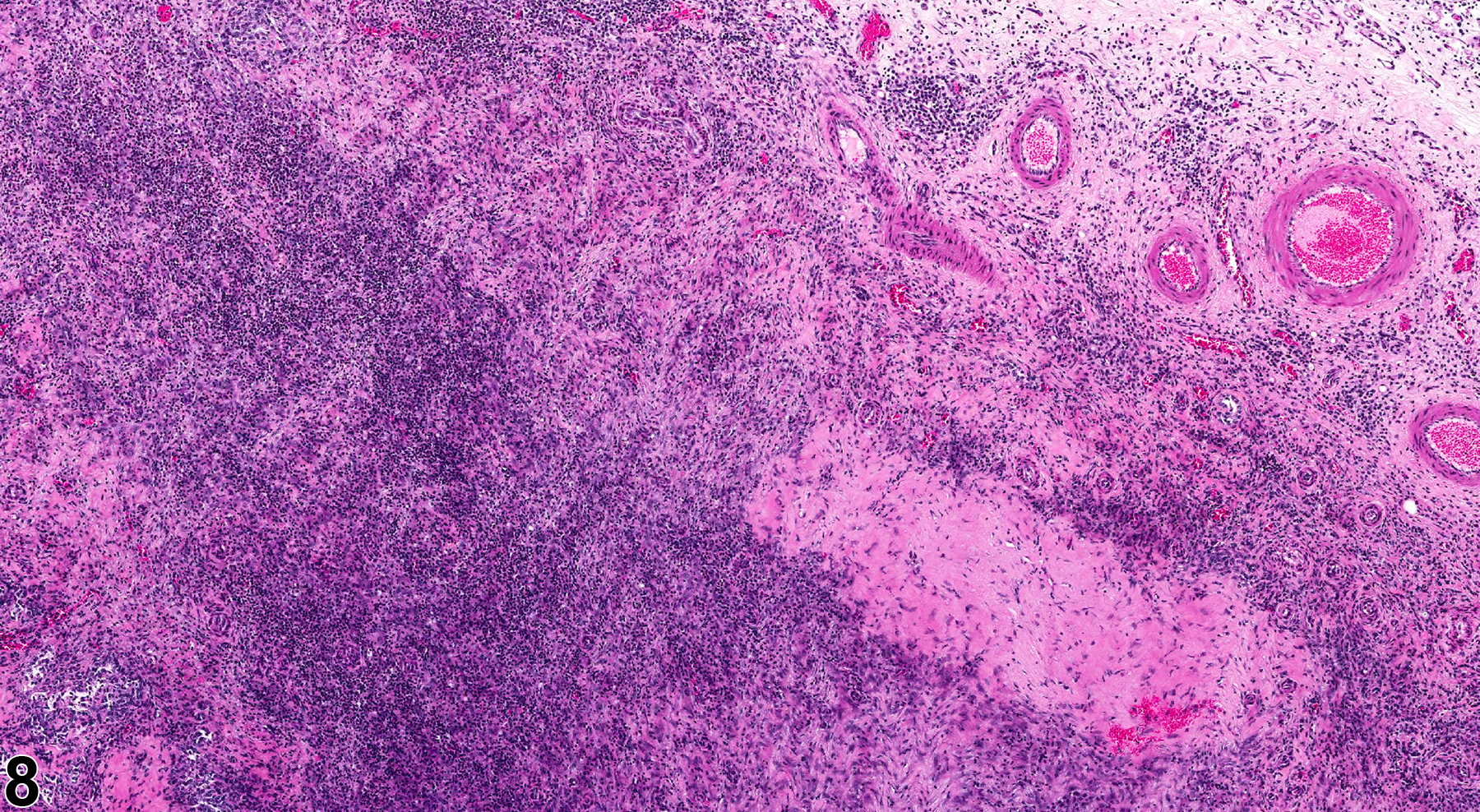
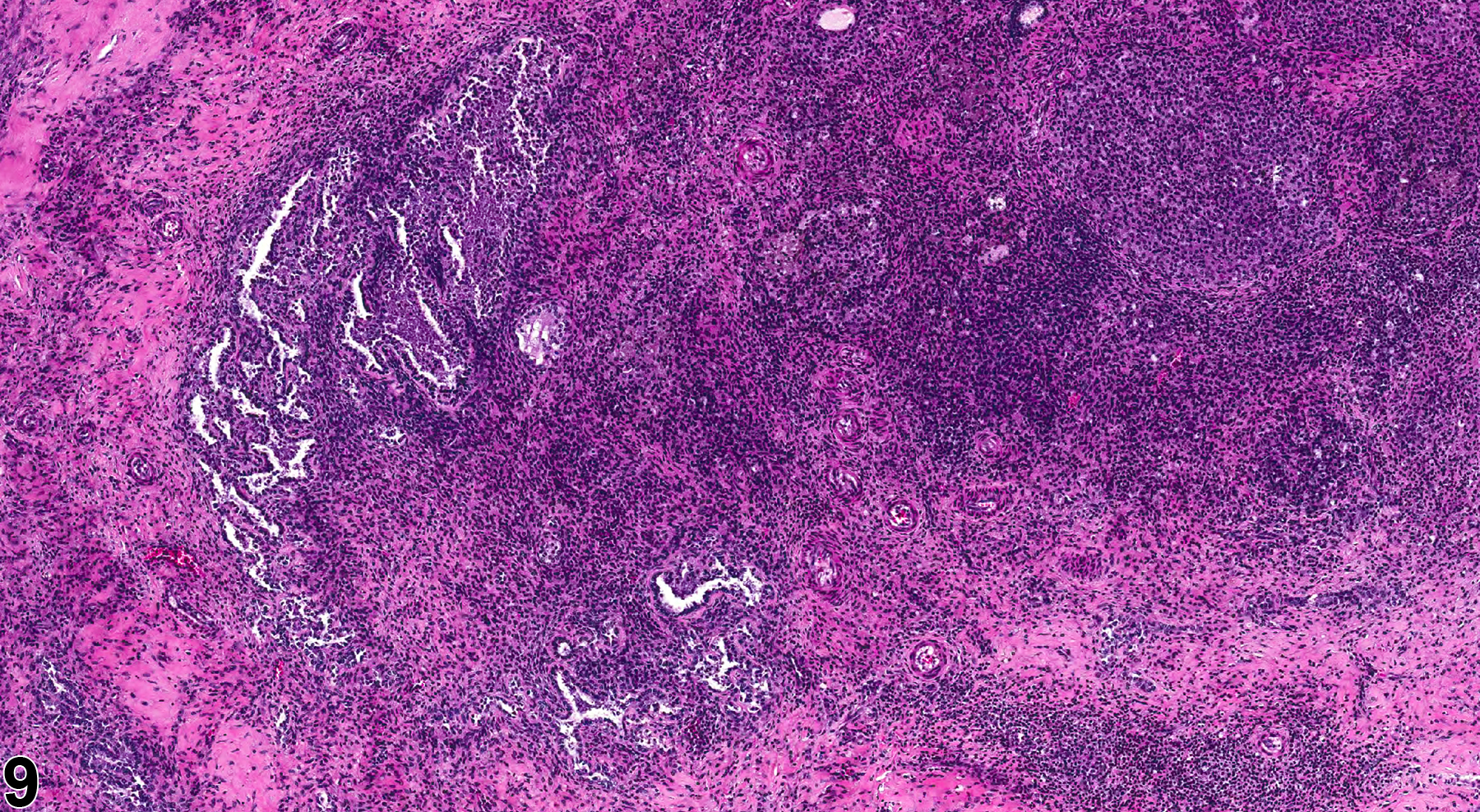
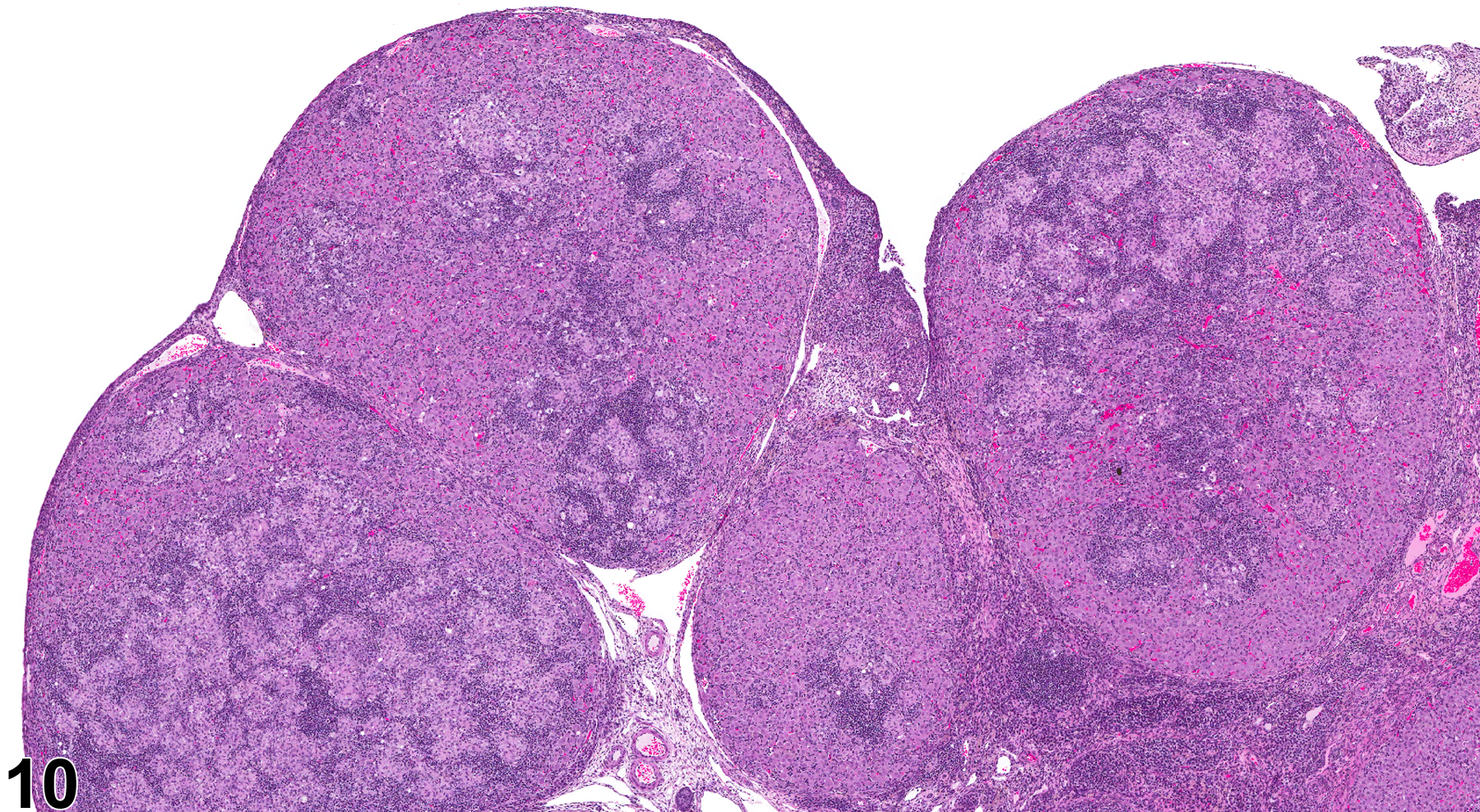
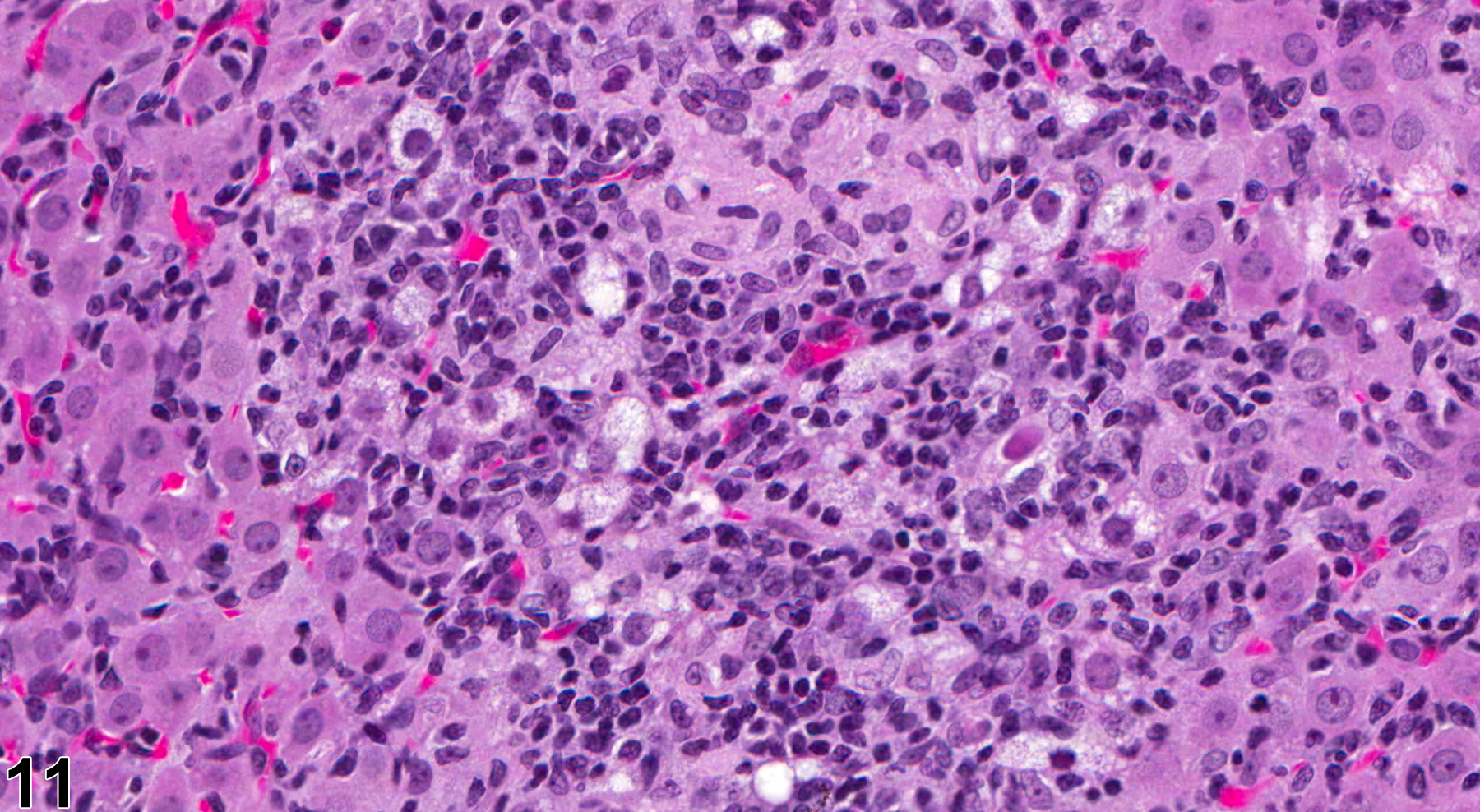
In NTP studies, there are five standard categories of inflammation: acute, suppurative, chronic, chronic active, and granulomatous. In acute inflammation, the predominant infiltrating cell is the neutrophil, though fewer macrophages and lymphocytes may also be present. There may also be evidence of edema or hyperemia. The neutrophil is also the predominant infiltrating cell type in suppurative inflammation (Figure 1, Figure 2, Figure 3, Figure 4 and Figure 5), but the neutrophils are aggregated, and many are degenerate (suppurative exudate). Cell debris, both from the resident cell populations and from infiltrating leukocytes, and proteinaceous fluid containing fibrin, fewer macrophages, occasional lymphocytes or plasma cells, and, possibly, an infectious agent may also be present within the exudate. Grossly, these lesions would be characterized by the presence of pus. The tissue surrounding the exudate may contain fibroblasts, fibrous connective tissue, and mixed inflammatory cells, depending on the chronicity of the lesion. Abscesses should be diagnosed histologically as suppurative inflammation. Lymphocytes predominate in chronic inflammation (Figure 6 and Figure 7). Lymphocytes also predominate in chronic active inflammation (Figure 8 and Figure 9), but there are also a significant number of neutrophils. Both lesions may contain macrophages. Granulomatous inflammation (Figure 10 and Figure 11) is another form of chronic inflammation, but this diagnosis requires the presence of a significant number of aggregated, large, activated macrophages, epithelioid macrophages, or multinucleated giant cells.
Inflammation is differentiated from cellular infiltrates by the presence of other changes, such as edema, hemorrhage, degeneration, necrosis, or other evidence of tissue damage. Granulomatous ovarian inflammation is characterized by multifocal, well-circumscribed, variably sized and irregularly shaped eosinophilic clusters of foamy macrophages, surrounded by variable numbers of multinucleated giant cells and lymphocytes (Figure 10 and Figure 11). This type of inflammation can be seen in corpora lutea in control animals during proestrus, so this lesion should be diagnosed only if it is seen during other phases of the estrous cycle, if it appears to be outside of corpora lutea, or if it is more severe in the treated animals than in controls. Ovarian infections resulting in inflammation are generally ascending, through the vagina and uterine cavity. The cause may be related to environmental factors and cleanliness in the animal housing facility. Ovarian infections may be seen with systemic infections caused by a variety of bacteria. However, autoimmune ovarian inflammation has been demonstrated in thymectomized mice, possibly involving a T-lymphocyte-mediated mechanism.
Ovary - Inflammation should be diagnosed and graded whenever present. The type of inflammation should be indicated in the diagnosis by use of a qualifier. Lesions secondary to inflammation, such as necrosis, hemorrhage, and edema, should not be diagnosed separately unless warranted by severity but should be described in the pathology narrative. If the inflammation is secondary to another lesions (e.g., necrosis or neoplasia), it should not be diagnosed separately unless warranted by severity but should be described in the narrative as a component of the primary lesion.
Alison RH, Morgan KT, Montgomery CA. 1990. Ovary. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, CA, 429-442.
Davis BJ, Dixon D, Herbert RA. 1999. Ovary, oviduct, uterus, cervix and vagina. In: Pathology of the Mouse: Reference and Atlas (Maronpot RR, Boorman GA, Gaul BW, eds). Cache River Press, Vienna, IL, 409-444.
Greaves P. 2012. Female genital tract. In: Histopathology of Preclinical Toxicity Studies: Interpretation and Relevance in Drug Safety Evaluation, 4th ed. Elsevier, Amsterdam, 667-724.
Montgomery CA, Alison RH. 1987. Non-neoplastic lesions of the ovary in Fischer 344 rats and B6C3F1 mice. Environ Health Perspect 73:53-75.
Abstract: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1474552/National Toxicology Program. 1989. NTP TR-354. Toxicology and Carcinogenesis Studies of Dimethoxane (CAS No. 828-00-2) (Commercial Grade) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/6969National Toxicology Program. 1994. NTP TR-424. Toxicology and Carcinogenesis Studies of o-Benzyl-p-chlorophenol (CAS No. 120-32-1) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/5992National Toxicology Program. 2000. NTP TR-500. Toxicology and Carcinogenesis Studies of Naphthalene (CAS No. 91-20-3) in F344/N Rats (Inhalation Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/14878National Toxicology Program. 2006. NTP TR-529. Toxicology and Carcinogenesis Studies of 2,2',4,4',5,5',-Hexachlorobiphenyl (PCB 153) (CAS No. 35065-27-1) in Female Harlan Sprague-Dawley Rats (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/20264Peluso JJ, Gordon LR. 1992. Nonneoplastic and neoplastic changes in the ovary. In: Pathobiology of the Aging Rat (Mohr U, Dungworth DL, Capen CC, eds). ILSI Press, Washington, DC, 351-364.
Rao GN, Hickman RL, Seilkop SK, Boorman GA. 1987. Utero-ovarian infection in aged B6C3F1 mice. Lab Anim Sci 37:153-158.
Yoshizawa K, Brix AE, Sells DM, Jokinen MP, Wyde M, Orzech DP, Kissling GE, Walker NJ, Nyska A. 2009. Reproductive lesions in female Harlan Sprague-Dawley rats following two-ear oral treatment with dioxin and dioxin-like compounds. Toxicol Pathol 37:921-937.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/19843953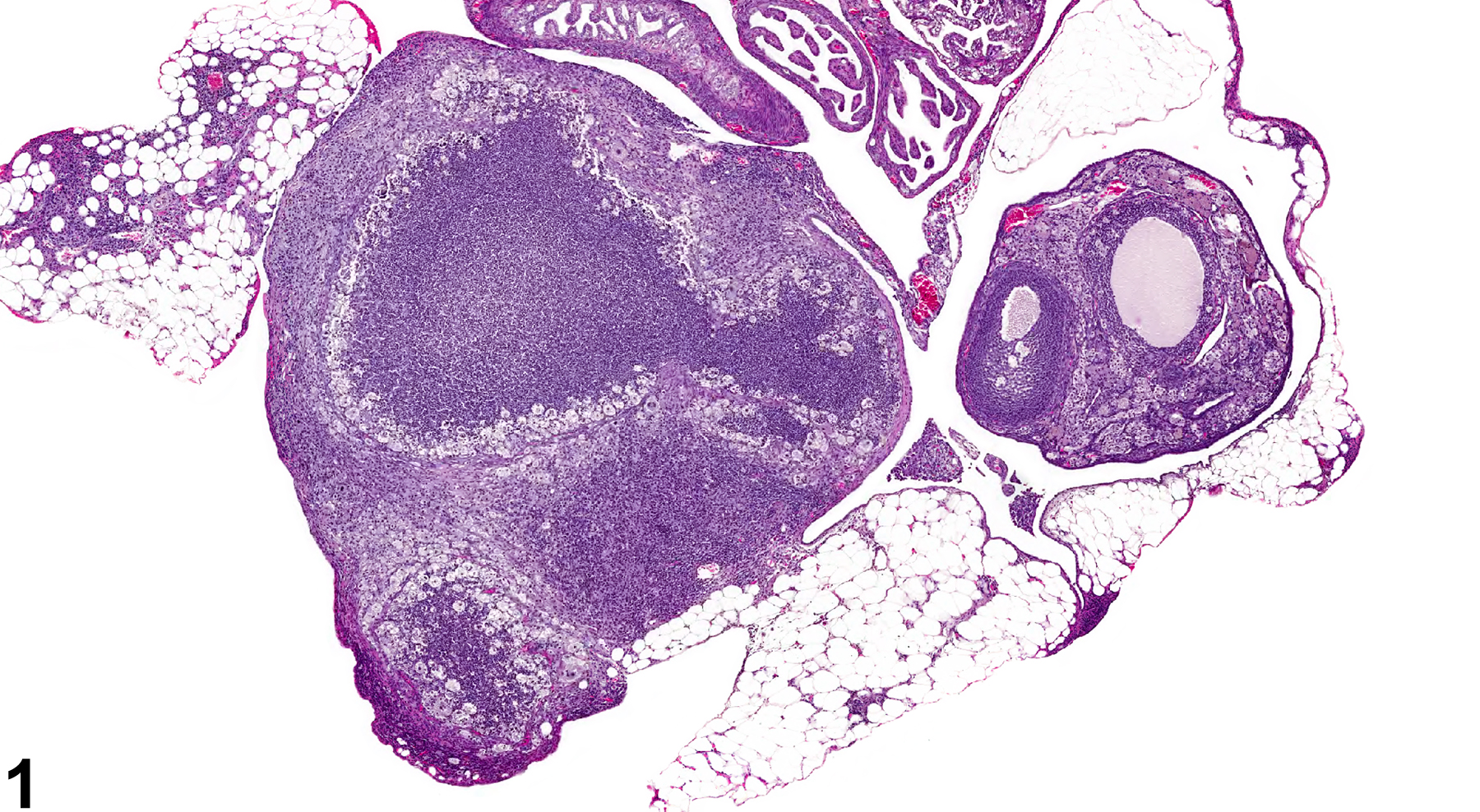
Ovary - Inflammation, Suppurative in a female B6C3F1/N mouse from a chronic study. Large focal accumulations of intact and degenerating neutrophils are present in the ovary.