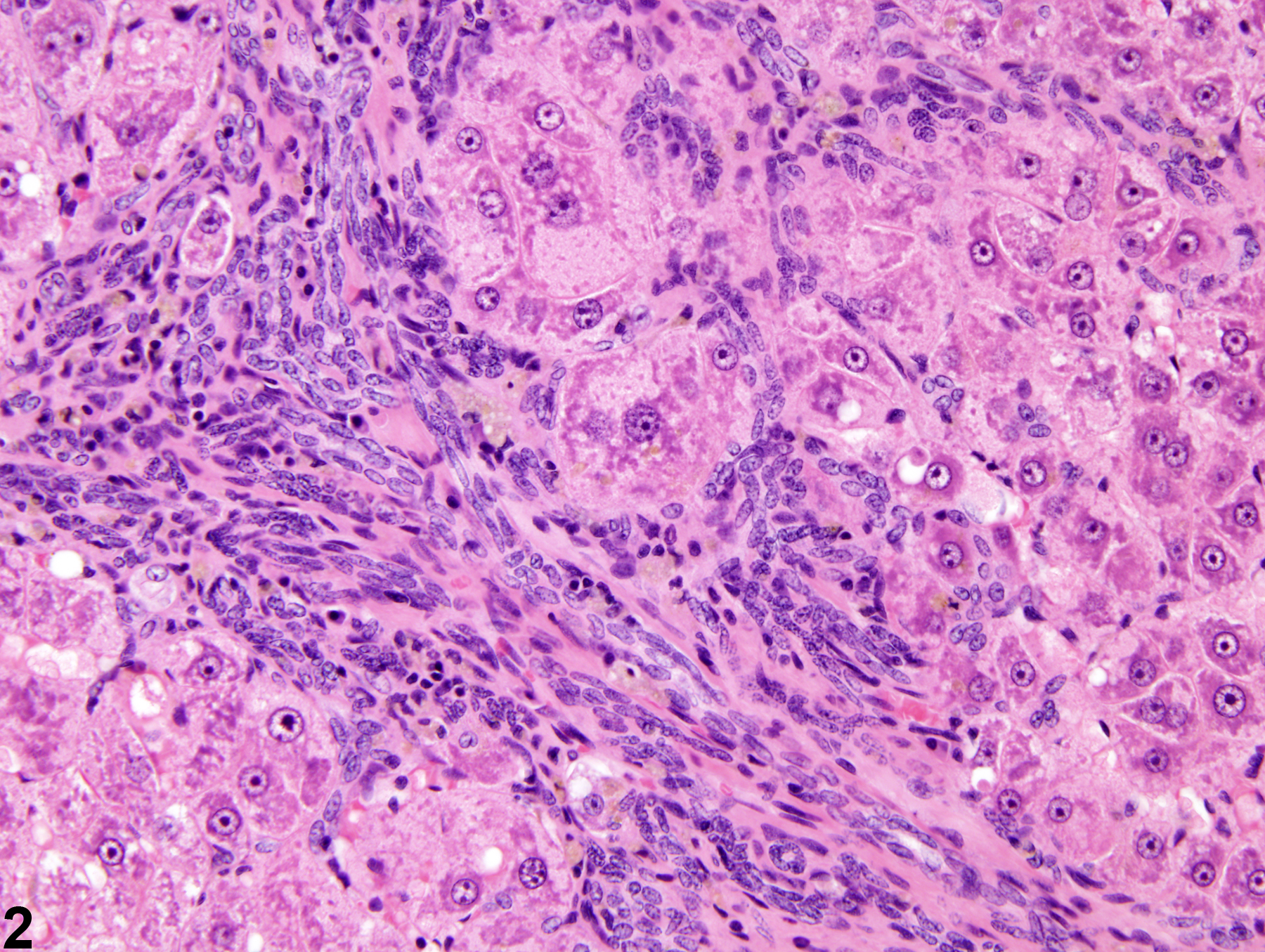
Hepatobiliary System
Liver, Hepatocyte – Hyperplasia, Nodular
Narrative
Eustis SL, Boorman GA, Harada T, Popp JA. 1990. Liver. In: Pathology of the Fischer Rat (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 71-94.
Hailey JR, Walker NJ, Sells DM, Brix AE, Jokinen MP, Nyska A. 2005. Classification of proliferative hepatocellular lesions in Harlan Sprague-Dawley rats chronically exposed to dioxin-like compounds. Toxicol Pathol 33:165-174.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/15805068Thoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM. 2010. Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S-81S.
Full Text: http://tpx.sagepub.com/content/38/7_suppl/5S.full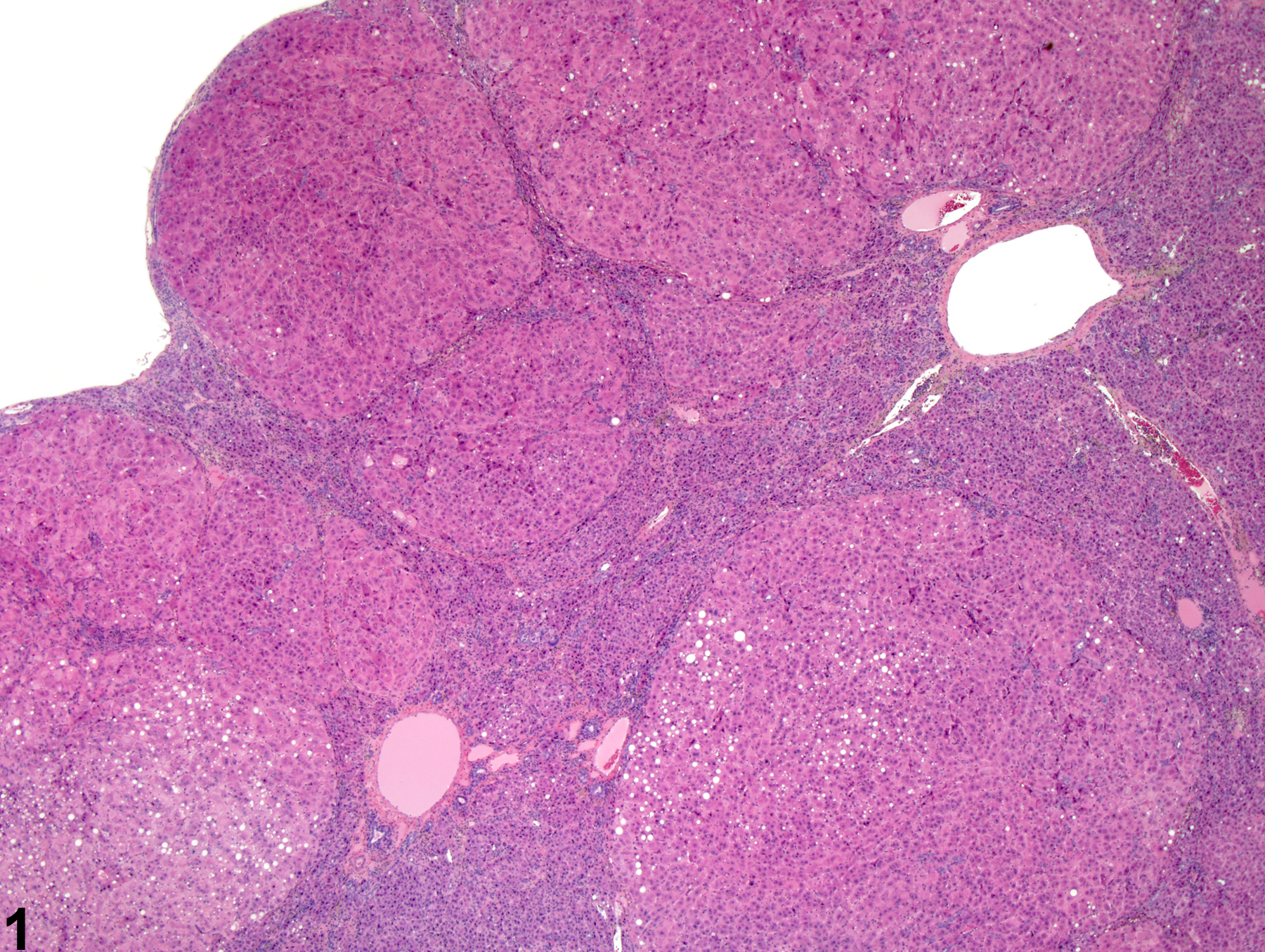
Nodular hyperplasia in a female Harlan Sprague-Dawley rat from a chronic study.