Immune System
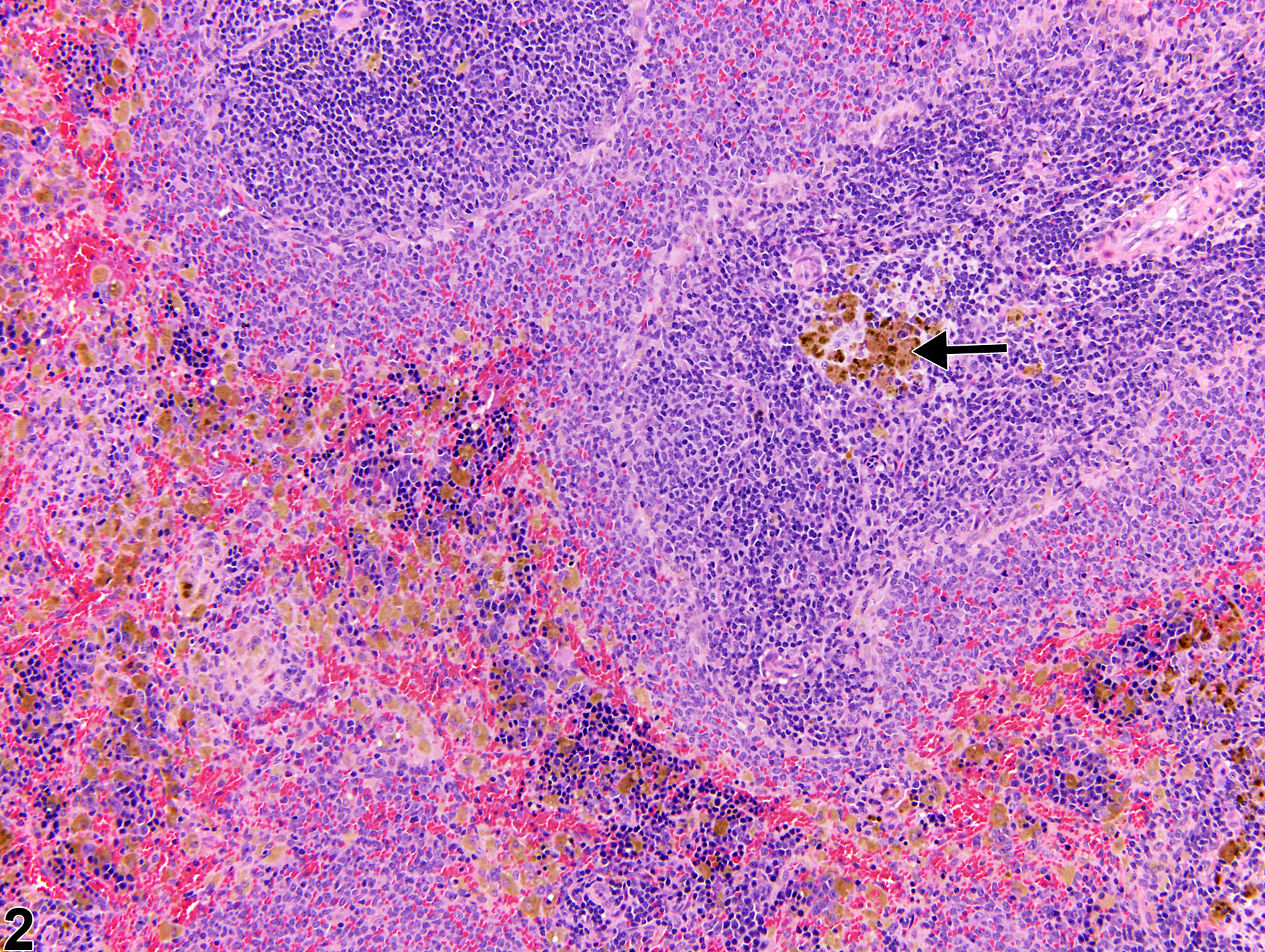
Spleen - Pigment
Narrative
National Toxicology Program. 2010. NTP TR-558. Toxicology and Carcinogenesis Studies of 3,3′,4,4′-Tetrachloroazobenzene (TCAB) [CAS No. 14047-09-7] in Harlan Sprague-Dawley Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/33564National Toxicology Program. 2011. NTP TR-570. Toxicology and Carcinogenesis Studies of α,β-Thujone (CAS No. 76231-76-0) in F344/N Rats and B6C3F1 Mice (Gavage Studies). NTP, Research Triangle Park, NC.
Abstract: https://ntp.niehs.nih.gov/go/36137Stefanski SA, Elwell MR, Stromberg PC. 1990. Spleen, lymph nodes, and thymus. In: Pathology of the Fischer Rat: Reference and Atlas (Boorman GA, Eustis SL, Elwell MR, Montgomery CA, MacKenzie WF, eds). Academic Press, San Diego, 369-394.
Suttie AW. 2006. Histopathology of the spleen. Toxicol Pathol 34:466-503.
Full Text: http://tpx.sagepub.com/content/34/5/466.full.pdfWard JM, Rehg JE, Morse HC III. 2012. Differentiation of rodent immune and hematopoietic system reactive lesions from neoplasias. Toxicol Pathol 40:425-434.
Abstract: https://www.ncbi.nlm.nih.gov/pubmed/22215512Ward JM, Mann PC, Morishima H, Frith CH. 1999. Thymus, spleen, and lymph nodes. In: Pathology of the Mouse (Maronpot RR, ed). Cache River Press, Vienna, IL, 333-360.
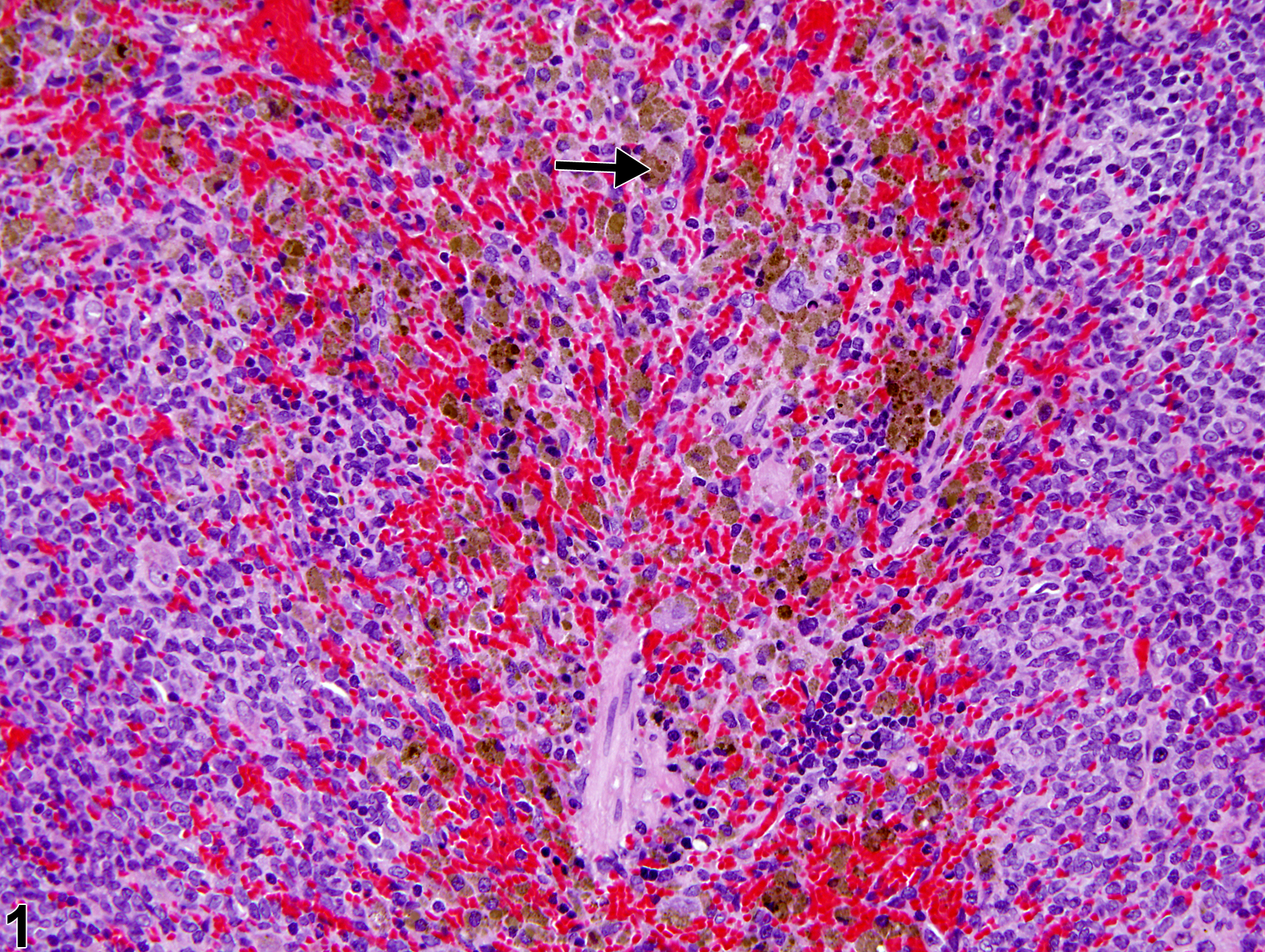
Spleen - Pigment in a male F344/N rat from a chronic study. Multifocal macrophages with intracytoplasmic dark-brown granular pigment (arrow) are present within the splenic red pulp.